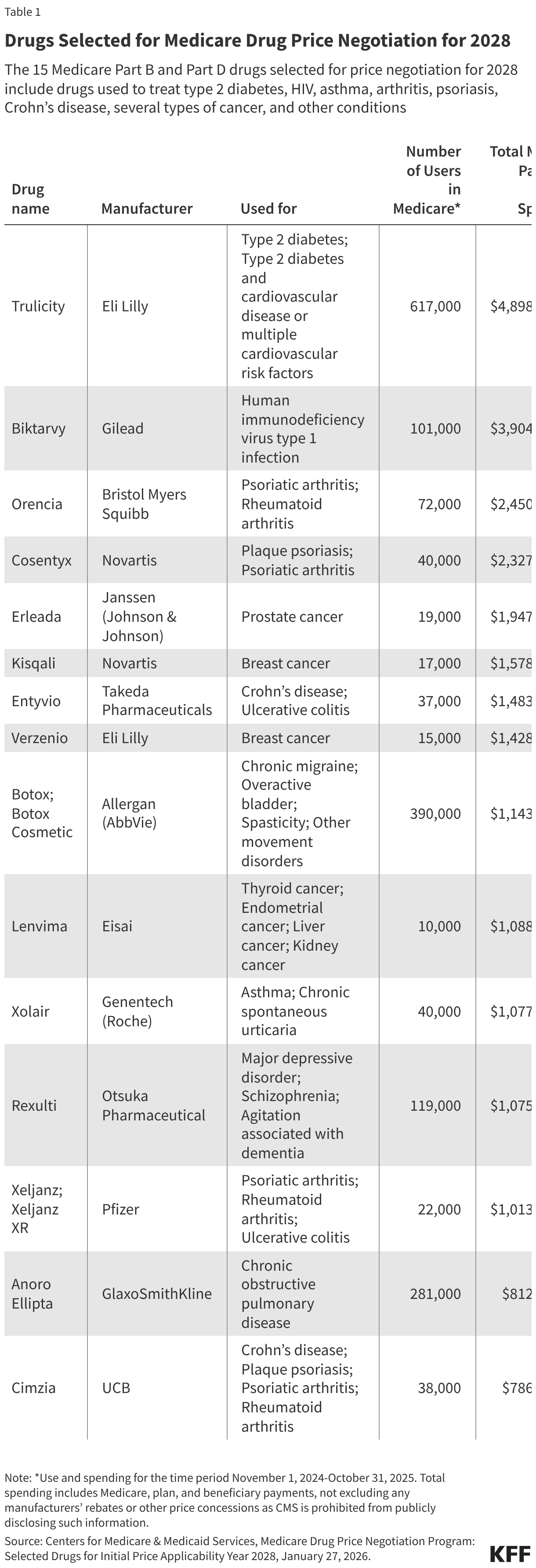
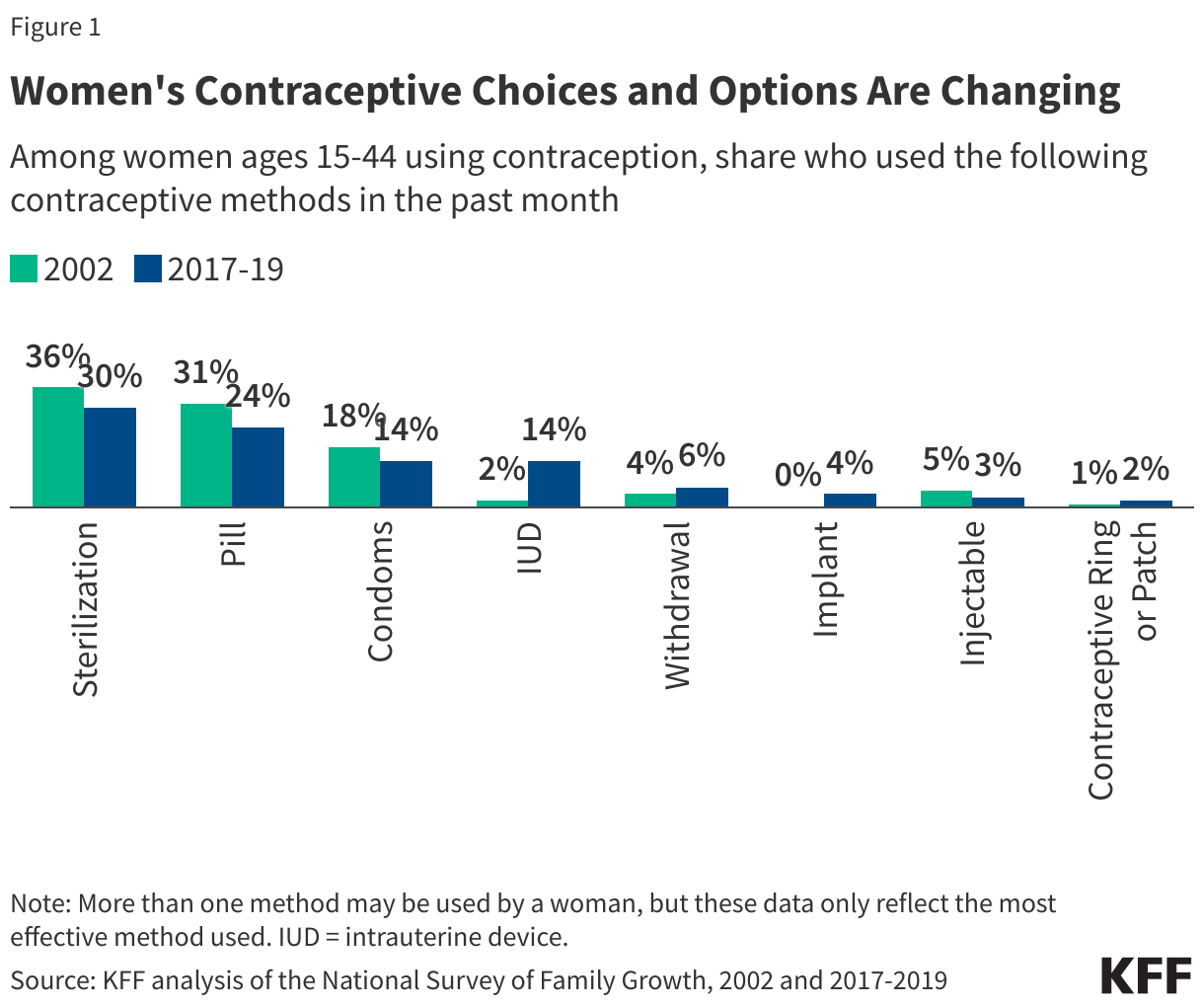
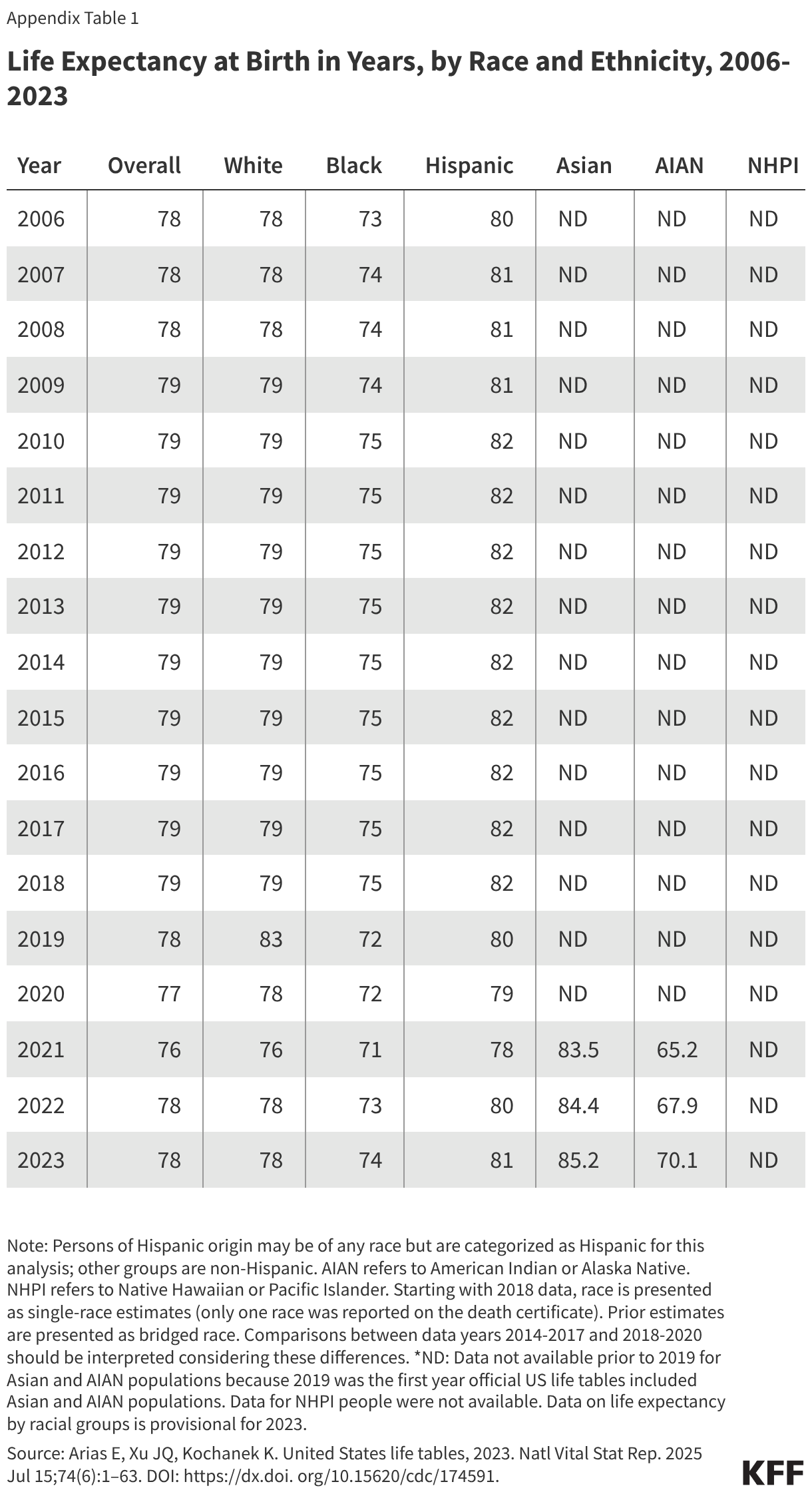
Fifteen drugs covered under Medicare Part B, which covers physician-administered drugs, or Medicare Part D, Medicare’s outpatient prescription drug benefit program, were selected for price negotiation in 2026, with Medicare’s negotiated prices for these drugs taking effect on January 1, 2028 (Table 1). These 15 drugs include treatments for type 2 diabetes, HIV, asthma, arthritis, psoriasis, Crohn’s disease, several types of cancer, and other conditions (See Table 1). Total gross Medicare spending on these 15 drugs between November 2024 and October 2025 was $27 billion, with 1.8 million Medicare beneficiaries using these medications during that time. Starting in 2027 and in each subsequent year, up to 20 additional drugs covered under Part B or Part D will be selected for negotiation. The number of drugs with negotiated prices available will accumulate over time.
Key Facts About Medicare Drug Price Negotiation
Editorial Note: This brief was updated in March 2026 to reflect the results of the second round of Medicare drug price negotiation and details about the third cycle of the negotiation program, including the list of drugs selected for negotiation in early 2026.
Under the Medicare Drug Price Negotiation Program, the Secretary of Health and Human Services (HHS) is required to negotiate prices with drug companies for certain high-cost drugs covered under Medicare. This requirement, a provision of the Inflation Reduction Act of 2022 (IRA), was the culmination of years of debate among lawmakers over whether to grant the federal government the authority to negotiate drug prices in Medicare.
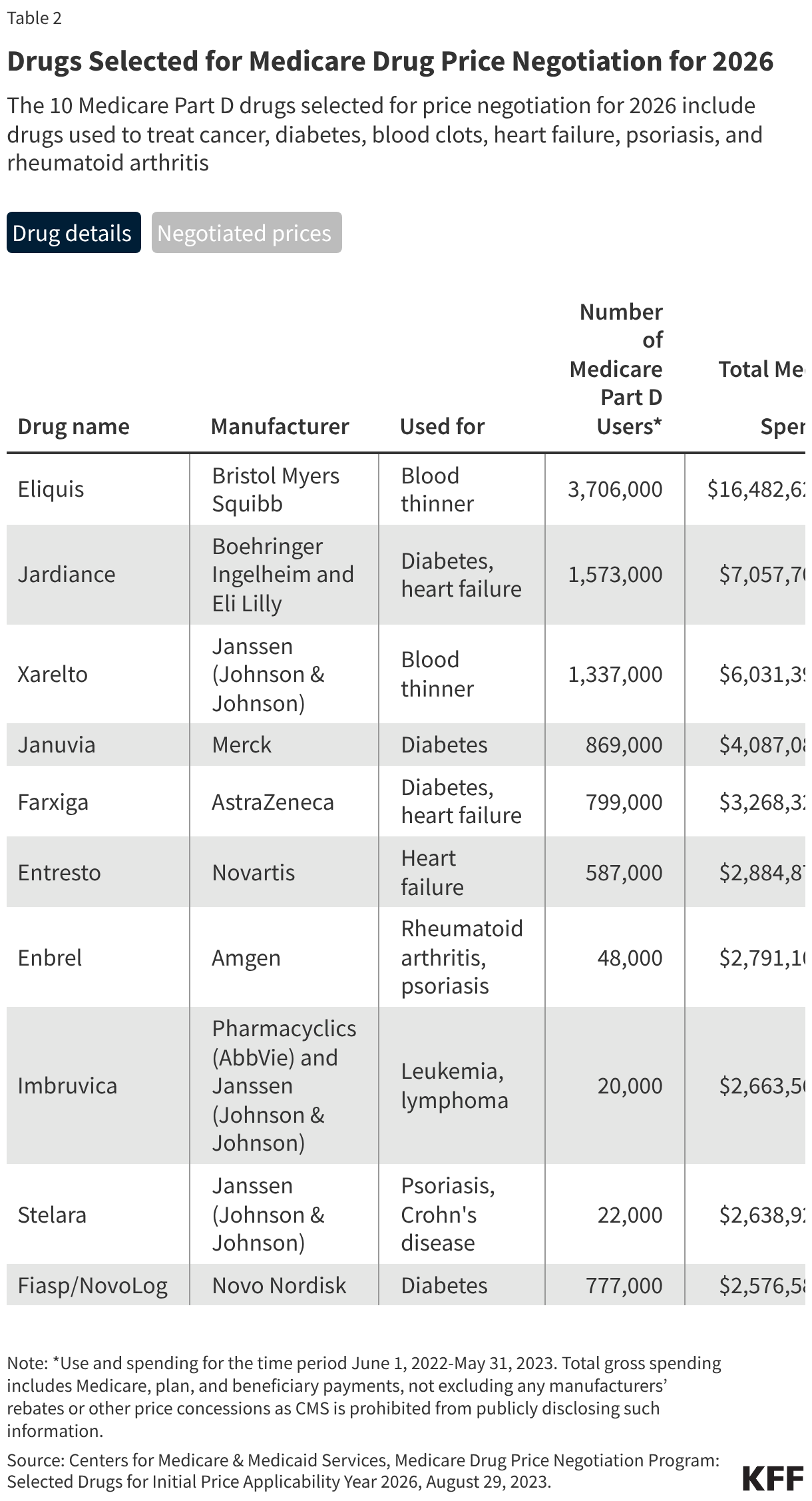
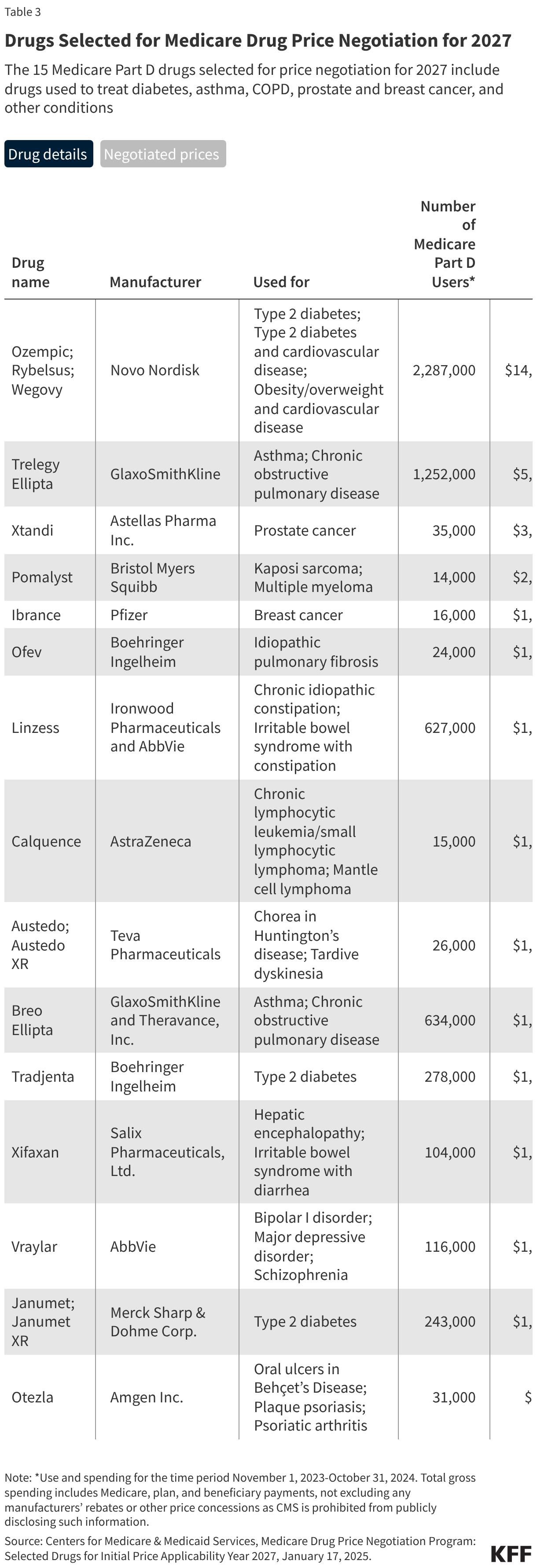
Medicare’s drug price negotiation program is now in its third cycle, with CMS having concluded two rounds of drug price negotiation to date. Negotiated prices for the first set of 10 Medicare Part D drugs selected for negotiation went into effect on January 1, 2026, while negotiated prices for the second set of 15 Part D selected drugs (including the popular GLP-1 diabetes and obesity drugs Ozempic and Wegovy) will take effect in 2027. In January 2026, CMS announced an additional 15 Part D and Part B drugs selected for negotiation, with negotiated prices effective in 2028. Total Medicare spending on the 40 drug products that have been selected for negotiation to date accounted for more than one-third (36%) of total Medicare spending on all drugs covered under Part B and Part D in 2024, or $125 billion out of $350 billion (not accounting for rebates in Part D and excluding spending on Part B drugs under Medicare Advantage since data are unavailable). The Centers for Medicare & Medicaid Services (CMS) has estimated several billion dollars in net savings to Medicare for round one and round two Part D drugs based on Medicare’s negotiated prices relative to existing net prices paid by Part D plans.
This brief provides information about several key aspects of the Medicare drug price negotiation program, with a focus on the 2028 implementation year, drawing on guidance from the Centers for Medicare & Medicaid Services (CMS). This is the first year CMS is required to negotiate physician-administered drugs covered under Medicare Part B. It is also the first year that a change to the IRA’s orphan drug exclusion is in effect, based on a provision in the 2025 reconciliation law that broadened this exclusion and exempted more drugs from negotiation.
Table of Contents
In 2026, CMS selected 15 Medicare Part B and D drugs for price negotiation, with negotiated prices taking effect in 2028
CMS has concluded two rounds of drug price negotiation to date, with estimated savings of several billion dollars based on Medicare’s negotiated prices relative to existing net prices
Based on the negotiated prices for first 10 Part D drugs selected for negotiation, CMS estimated that Medicare would have saved $6 billion if the prices that CMS negotiated had been in effect in 2023, amounting to net savings of 22% on these drugs. CMS also estimated that Medicare beneficiaries will save $1.5 billion when these negotiated prices take effect in 2026. Based on negotiated prices for the next 15 Part D drugs selected for negotiation, CMS estimated even greater savings of $12 billion relative to existing net prices in 2024, amounting to net savings of 44% on these medications – higher due in part to the larger number of drugs subject to negotiation. CMS has also estimated that Medicare beneficiaries will save $685 million when these negotiated prices take effect in 2027.
Selected Drugs for Implementation Year 2026: The first 10 Medicare Part D drugs that were selected for negotiation include treatments for several medical conditions, including diabetes (Farxiga, Fiasp/NovoLog, Januvia, Jardiance), blood clots (Eliquis, Xarelto), heart failure (Entresto, Farxiga), psoriasis (Stelara, Enbrel), rheumatoid arthritis (Enbrel), Crohn’s disease (Stelara), and blood cancers (Imbruvica) (Table 2). Medicare’s negotiated prices took effect on January 1, 2026. CMS published explanations of its negotiated prices and the factors that were considered in the negotiation process, including manufacturer-specific financial data about the selected drugs and evidence about the clinical benefits of selected drugs compared to alternative treatments.
Selected Drugs for Implementation Year 2027: The 15 Medicare Part D drugs selected for price negotiation in round two included the popular GLP-1 diabetes and obesity drugs Ozempic and Wegovy, along with other drugs used to treat asthma and chronic obstructive pulmonary disease, type 2 diabetes, prostate and breast cancer, and other conditions (Table 3). Negotiated prices for these drugs will take effect on January 1, 2027.
Drugs qualify for price negotiations if they have high total Medicare spending, no generic or biosimilar equivalents, and are several years past FDA approval
Drugs qualify for price negotiation if they are single source brand-name drugs or biological products without therapeutically-equivalent generic or biosimilar alternatives that are approved or licensed and marketed on a “bona fide” basis – a determination to be made by CMS using FDA reference sources to determine whether a generic or biosimilar has been approved, along with claims and pricing data to assess utilization and sales of generics or biosimilars and other public information related to product launch and distribution. In addition, a drug product must be at least 7 years (for small-molecule drugs) or 11 years (for biologics) past its FDA approval or licensure date, as of the date that the list of drugs selected for negotiation is published, giving manufacturers several years to market their products before being eligible for negotiation. This means that for a single source drug to be eligible for negotiation for 2028, a drug product must have been approved on or before February 1, 2019, and a biological product must have been licensed on or before February 1, 2015. For drugs with multiple FDA approvals, CMS uses the earliest approval date to determine the number of years that have elapsed.
The definition of ‘qualifying single source drug’ excludes certain types of drugs:
- Orphan-drugs – drugs that are designated for rare diseases or conditions and approved only for those diseases or conditions (known as the orphan drug exclusion, which was modified by the 2025 reconciliation law, as described below),
- Low-spending drugs – drugs with combined total spending under Part B and Part D of less than an inflation-adjusted threshold amount (originally set at $200 million, inflation adjusted by the growth in the consumer price index for all urban consumers (CPI-U) to $207 million for negotiation year 2027),
- Plasma-derived products, and
- For 2026 to 2028, “small biotech” drugs (i.e., drugs that account for 1% or less of total Medicare drug spending on all qualifying single source drugs under either Part B or Part D but 80% or more of Part B or Part D spending for a given manufacturer’s Part B or Part D covered qualifying single source drugs).
Policy for Fixed Combination Drugs: To identify potential qualifying single source drugs, the IRA requires aggregating data across dosage forms (such as tablets and capsules) and strengths of a given drug product. However, the law does not address how to handle fixed combination drugs (that is, a drug that includes two or more active ingredients). According to CMS guidance for 2028, data for a fixed combination drug will not be aggregated with data for a drug that includes only one of those active ingredients. Instead, CMS will treat these as distinct drug products for the purposes of identifying potential qualifying single source drugs. CMS has acknowledged the possibility that manufacturers might try to avoid or delay having a drug selected for negotiation by modifying the formulation to add an active ingredient, a “program integrity risk” that CMS stated it plans to address in future rulemaking around the negotiation program for 2029.
The 2025 reconciliation law changed the orphan drug exclusion to delay or make more drugs ineligible for negotiation
The IRA excluded orphan drugs from Medicare drug price negotiation if they were designated for only one rare disease or condition with approvals under that one designation. The 2025 reconciliation law broadened the orphan drug exclusion in two ways: 1) making orphan drugs that are designated for multiple rare diseases or conditions, not just a single rare disease, ineligible for Medicare drug price negotiation; and 2) delaying the start of the 7- or 11-year period before a drug can be selected for price negotiation for orphan drugs that subsequently receive FDA approval for a non-orphan indication.
These changes likely had an impact on which drugs were selected for Medicare price negotiation for 2028 by delaying the selection of the biologic drugs Keytruda and Opdivo, which were likely to have been selected for negotiation in 2026 based on their total Medicare spending levels and meeting other statutory criteria. In 2023, Medicare and beneficiaries spent $5.6 billion on Keytruda and $2.0 billion on Opdivo. Both drugs were initially approved as orphan drugs in 2014 and subsequently approved for non-orphan indications beginning in 2015. However, due to the change in law, the time that they were on the market as orphan-only drugs no longer counts towards the 11-year period following FDA licensure that biologics become eligible for the negotiation program. Therefore, selection of Keytruda and Opdivo for Medicare drug price negotiation has been delayed beyond 2026. Overall, the provision in the reconciliation law that delays or excludes additional orphan drugs from Medicare drug price negotiation will cost the federal government $8.8 billion over the coming decade, according to CBO, and will also mean higher out-of-pocket costs for Medicare beneficiaries who use these medications.
Drugs selected for negotiation are chosen from the top-ranking qualifying negotiation eligible drugs based on total Medicare Part B and Part D spending
The 15 Part B and Part D drugs that were selected for price negotiation for 2028 were chosen from the top 50 negotiation-eligible Part D drugs with the highest total Medicare Part D expenditures and the top 50 negotiation-eligible Part B drugs with the highest total Medicare Part B expenditures. For this purpose, total Part D expenditures are defined as total gross covered prescription drug costs from Part D prescription drug event (PDE) data, and total Part B expenditures are for separately payable Part B covered drugs (that is, not bundled or packaged into the payment for another service, such as anesthesia drugs, or most drugs used in treatment of end-stage renal disease) and include traditional Medicare claims data and Medicare Advantage encounter data. (Since encounter data do not include spending amounts, CMS estimates spending on Part B drugs in Medicare Advantage based on what would have been paid in traditional Medicare.)
To derive these lists, CMS first identified the qualifying single source drugs among all covered Part B and Part D drugs, applying the relevant statutory exclusions (as described above). CMS then calculated total expenditures for each qualifying drug separately under Part B and Part D, based on spending data for the 12-month period from November 1, 2024 to October 31, 2025. The top 50 Part B drugs and the top 50 Part D drugs with the highest total expenditures for this 12-month period were the negotiation-eligible drugs for 2028. CMS combined total expenditures under Part B and Part D for each negotiation-eligible drug (where applicable), ranked negotiation-eligible drugs by total spending, and selected the 15 highest-ranked drugs. (Drugs already selected for negotiation in previous rounds are removed from the list of qualifying single source drugs prior to ranking.)
The IRA provides for a delay in selecting drugs for negotiation if they are biological products where there is a “high likelihood” of biosimilar market entry within two years of the publication date of the selected drug list. The rationale for this delay is to not create financial incentives that could deter biosimilars from entering the market if, for example, a reference product (the original biological product approved by FDA against which a proposed biosimilar product is compared) is selected for negotiation and ultimately priced lower than potential competitor biosimilar products. CMS announced that for 2028, when selecting the 15 highest-ranked Part B and Part D drugs, no products qualified for delayed selection based on a high likelihood of biosimilar market entry.
Drugs that were previously selected for negotiation can be selected for renegotiation under certain circumstances
In addition to the 15 drugs selected for negotiation for 2028, CMS announced that one drug (Tradjenta) has been selected for renegotiation. This drug was initially included on the list of drugs selected for price negotiation in January 2025 (see Table 3 below). CMS identifies renegotiation-eligible drugs from the list of drugs previously selected for negotiation based on: (1) a change in status to “long-monopoly” (that is, at least 16 years have passed since the date of FDA approval or licensure, which would affect the ceiling price that applied during the negotiation process; such drugs are automatically selected for renegotiation); (2) the availability of new indications; or (3) a material change in the factors that CMS uses in negotiating drug prices that could meaningfully affect the outcome of renegotiation relative to the original negotiation process. In determining whether a renegotiation-eligible drug will be selected for renegotiation, CMS will evaluate whether a new indication or a material change in negotiation factors will result in a 15% or greater change in the negotiated price (higher or lower) and whether that would have a significant impact on the Medicare program.
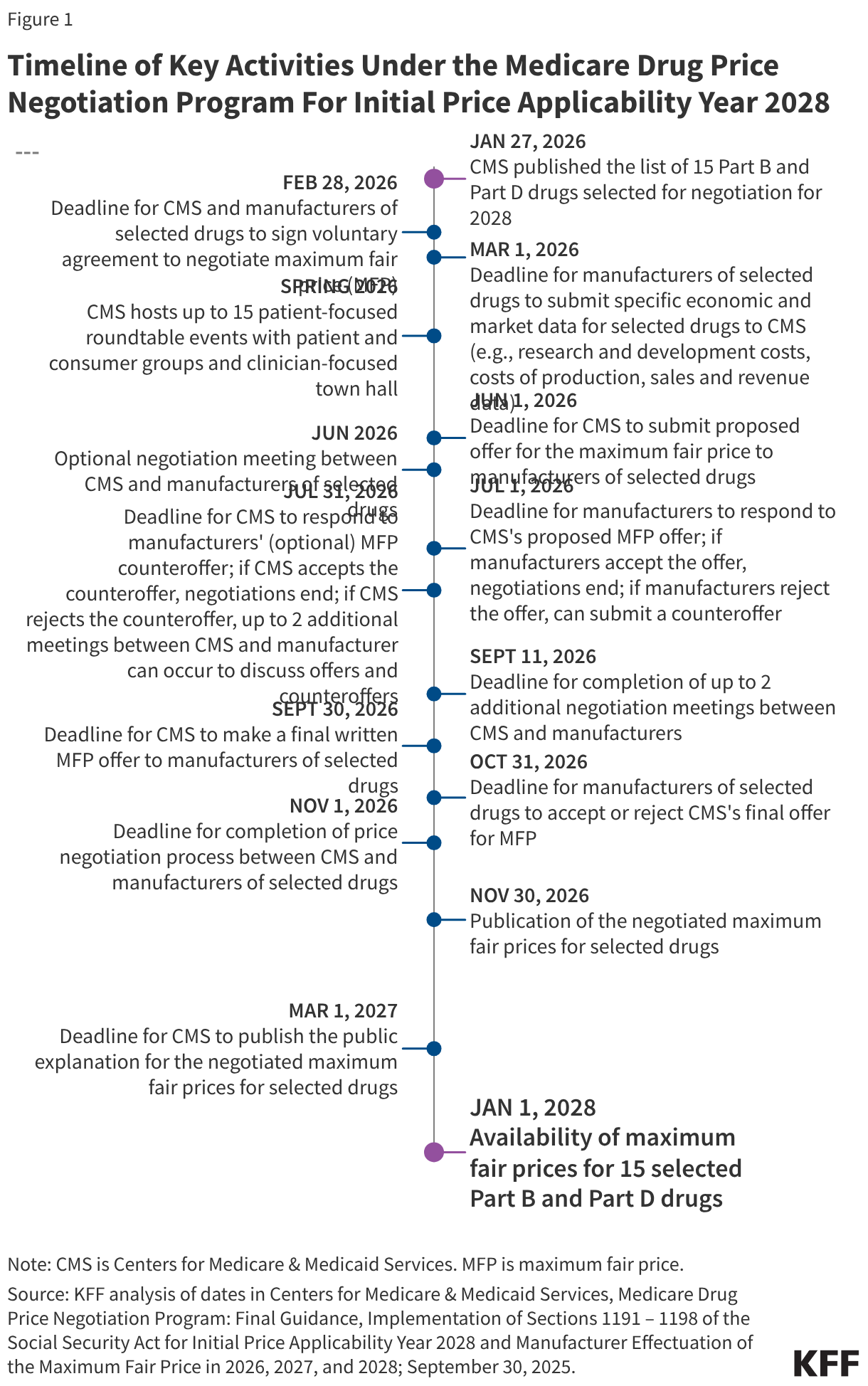
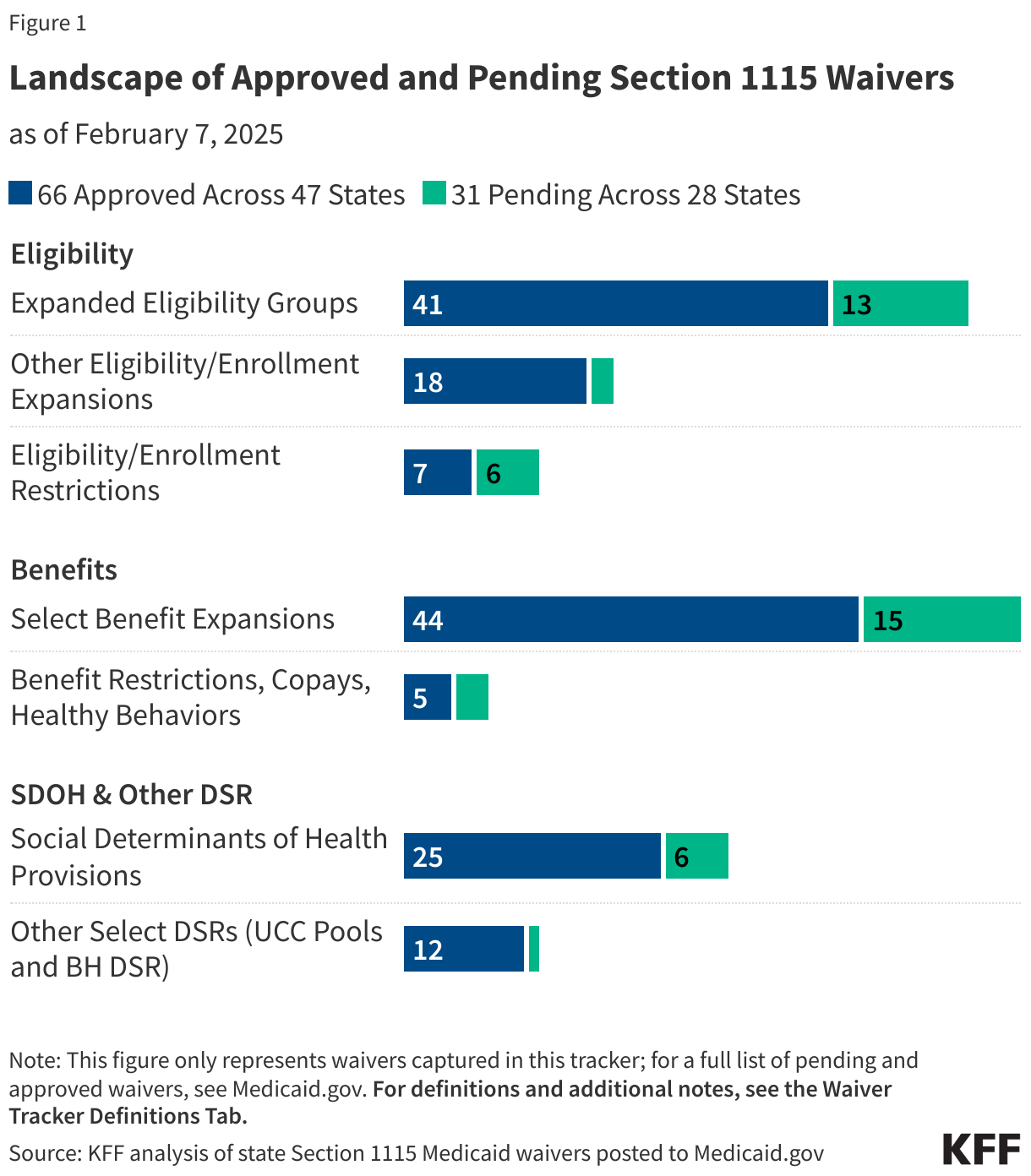
The annual timeline for Medicare drug price negotiation begins with the announcement of selected drugs by February 1 and ends with the announcement of negotiated prices by November 30
The annual timeline of key activities in the negotiation process begins no later than February 1 of a given year when the list of selected drugs is announced and ends no later than November 30, the statutory deadline for CMS to announce negotiated prices for selected drugs (Figure 1). The price negotiation process between CMS and drug manufacturers spans several months between spring and fall.
CMS is required to consider certain manufacturer-specific factors and information about therapeutic alternatives in its price negotiations
The IRA requires CMS to consider certain manufacturer-specific factors and information about therapeutic alternatives to selected drugs in negotiating the “maximum fair price” (MFP) for selected drugs, although the law does not specify how CMS should weigh these different elements in the process of developing its offer for the maximum fair price.
The manufacturer-specific factors related to selected drugs include:
- The manufacturer’s research and development costs and the extent to which the manufacturer has recouped these costs.
- The current unit costs of production and distribution.
- Federal financial support for novel therapeutic discovery and development related to the drug.
- Data on pending and approved patent applications, exclusivities, and certain other applications and approvals.
- Market data and revenue and sales volume data in the U.S.
For the manufacturers of the 15 selected drugs for 2028, these data elements are required to be reported to CMS by March 1, 2026.
Information about therapeutic alternatives includes:
- The extent to which the selected drug represents a therapeutic advance compared to existing therapeutic alternatives and the costs of these alternatives.
- Prescribing information for the selected drug and its therapeutic alternatives, which may include generics or biosimilars.
- Comparative effectiveness of the selected drug and its therapeutic alternatives, taking into account their effects on specific populations, such as individuals with disabilities, the elderly, the terminally ill, children, and other patient populations.
- The extent to which the selected drug and its therapeutic alternatives address unmet needs for a condition that is not adequately addressed by available therapy.
According to CMS guidance, information on these factors may be submitted by several entities, including the manufacturer of the selected drug, other drug manufacturers, people with Medicare, academic experts, clinicians, and others. Submissions are due by March 1, 2026 for the selected drugs for 2028. In addition to evaluating the information in these submissions, CMS will review the literature and real-world evidence, conduct internal analysis, and consult with experts regarding evidence of the clinical benefits of the selected drugs and their therapeutic alternatives.
The IRA explicitly directs that the HHS Secretary “shall not use evidence from comparative clinical effectiveness research in a manner that treats extending the life of an elderly, disabled, or terminally ill individual as of lower value than extending the life of an individual who is younger, non-disabled, or not terminally ill.” In other words, the use of health outcomes evidence based on quality-adjusted life years (QALYs) in the process of negotiating a maximum fair price is not permitted.
CMS determines its initial offer for the negotiated price of a selected drug based in part on the price of therapeutic alternatives, information about clinical benefits, and manufacturer-specific data
To determine its initial offer for a maximum fair price for a selected drug, CMS: (1) identifies therapeutic alternative(s) for the selected drug; (2) determines pricing information about the therapeutic alternatives to determine the starting point for the initial offer; (3) adjusts the initial offer based on information about the clinical benefit of the selected drug compared to its therapeutic alternatives; and (4) makes further adjustments to the offer price as needed based on manufacturer-specific data to determine the initial offer price. (The IRA does not include international drug price data as a benchmark to be used in CMS’s initial pricing decisions or the negotiation process overall.)
CMS uses the price of therapeutic alternative(s) as the starting point for determining the initial offer for the maximum fair price for a given selected drug. Specifically, for the 2028 negotiation year, CMS will use the lower of: for Part D drugs, the net Part D plan payment and beneficiary liability, which excludes both rebates as well as Manufacturer Discount Program payments, the wholesale acquisition cost (WAC), or the maximum fair price negotiated for previously selected drugs if any are therapeutic alternatives for 2028 selected drugs; and for Part B drugs, the average sales price (ASP) or WAC. If there is more than one therapeutic alternative for a selected drug, CMS will determine the starting point within the range of prices for those products.
For selected drugs with no therapeutic alternative or where the price of the alternative(s) is above the ceiling price, CMS will use the Federal Supply Schedule (FSS) or “Big Four Agency” price as the starting point, whichever is lower. (Drug prices listed on the FSS, which establishes prices available to all direct federal purchasers, are determined through both statutory rules and negotiation. A statutory cap on drug prices for the Big Four agencies (the Department of Veterans Affairs, the Department of Defense, the Public Health Service, and the Coast Guard) means the prices they pay are generally lower than prices paid by other direct federal purchasers.) If the FSS or Big Four prices are above the statutory ceiling, CMS will use the statutory ceiling as the starting point for its initial offer.
CMS will adjust the starting point for the initial offer based on a broad evaluation of evidence, including that which is submitted by manufacturers and the public, about the clinical benefit the selected drug provides relative to its therapeutic alternatives, including information about potential safety concerns and side effects, whether the selected drug represents a therapeutic advance as measured by improvements in clinical outcomes, and information about the effects of the selected drug and its therapeutic alternatives on specific populations, including people with disabilities and older adults. CMS will also consider comparative effectiveness data on patient-centered outcomes and patient experiences.
If a selected drug has no therapeutic alternatives, CMS will evaluate evidence about the selected drug’s clinical benefit, including improvements in outcomes, and also will consider the extent to which the selected drug fills an unmet medical need, meaning the drug treats a disease or condition where there are very limited or no other treatment options, or the existing treatments do not adequately address the disease or condition. This consideration will be made separately for each indication of a selected drug, where applicable.
After considering information about clinical benefit, CMS will adjust its starting point for the initial offer price to arrive at a “preliminary price.” After determining the preliminary price, CMS will take into account manufacturer-specific data elements. These data, and their illustrative effect on the preliminary price as described in CMS’s guidance, are:
- Research and development (R&D) costs: if a manufacturer has recouped its R&D costs, CMS could adjust the preliminary price downward, or upward if such costs have not been recouped.
- Current unit costs of production and distribution: if lower than the preliminary price, CMS could adjust the price downward, or upward if such costs are higher than the preliminary price.
- Prior federal financial support: if discovery and development of the selected drug was supported by federal funding, CMS could adjust the preliminary price downward.
- Patent information: this data will support CMS’s evaluation of whether a selected drug represents a therapeutic advance or meets an unmet medical need, particularly in light of any exclusivities which mean that a selected drug is the only available therapy.
- Market data and revenue and sales volume data for the drug in the U.S.: depending on how CMS’s preliminary price compares to other market pricing data for the selected drug, CMS could, for example, revise downward the preliminary price if the average commercial net price is lower, or upward if the average commercial net price is higher.
After making any necessary adjustments to the preliminary price based on a review of manufacturer-specific data, CMS will arrive at its initial offer for the maximum fair price.
The law establishes a ceiling on the negotiated price that Medicare will pay for selected drugs, based on existing price benchmarks
The IRA establishes an upper limit for the maximum fair price for a given drug, which varies depending on whether the drugs is covered under Part B only, Part D only, or both parts. The upper limit is the lower of the drug’s enrollment-weighted negotiated price (net of all price concessions, including rebates) for a Part D-only drug, the average sales price (the average price to all non-federal purchasers in the U.S., inclusive of rebates, other than rebates paid under the Medicaid program) or a percentage of a drug’s average non-federal average manufacturer price (non-FAMP) (the average price wholesalers pay manufacturers for drugs distributed to non-federal purchasers) for a Part B-only drug, or a weighted average of these amounts for drugs covered under both Part B and Part D. The percentage of non-FAMP varies depending on the number of years that have elapsed since FDA approval or licensure: 75% for small-molecule drugs and vaccines more than 9 years but less than 12 years beyond approval; 65% for drugs between 12 and 16 years beyond approval or licensure; and 40% for drugs more than 16 years beyond approval or licensure. This approach means that the longer a drug has been on the market, the lower the ceiling on the maximum fair price.
The negotiation process between CMS and drug manufacturers spans several months and allows for multiple opportunities to exchange price offers
CMS’s guidance outlines several steps in the negotiation process (Figure 1). These steps, and the relevant dates for selected drugs for 2028, are:
- CMS and manufacturers of selected drugs enter into a written agreement to negotiate to determine the maximum fair price for selected drugs by February 28, 2026.
- Submission of economic and market data from manufacturers of selected drugs to CMS and information about therapeutic alternatives is due on March 1, 2026.
- CMS will host one meeting with manufacturers of selected drugs in Spring 2026 after the submission of manufacturer-specific data elements so that manufacturers can provide additional context for their data submission and share new information, if applicable.
- CMS will host up to 15 patient-focused roundtable events with consumer and patient organizations (with selected drugs aggregated by condition, as appropriate) and one clinician-focused town hall event in Spring 2026 to solicit patient-focused and clinical information on therapeutic alternatives and other information for CMS to consider in developing its initial offer for selected drugs.
- CMS will make a written offer to the manufacturer of a selected drug with its initial offer of the maximum fair price by June 1, 2026. This written offer will include a justification for CMS’s initial offer based on the methodology used, including how CMS evaluated various data submitted by manufacturers and evidence about alternative therapies.
- An optional negotiation meeting between CMS and manufacturers of selected drugs could take place between the date of CMS’s initial offer and the deadline for manufacturers to respond.
- Manufacturers respond to CMS’s initial offer in writing either accepting the offer or making a counteroffer within 30 days of receiving the initial offer (e.g., July 1, 2026, for initial offers made by CMS on June 1, 2026). The written counteroffer should include the manufacturer’s proposed maximum fair price, along with a justification for that amount and a response to CMS’s justification for its initial offer. If the manufacturer does not accept CMS’s initial offer, a written counteroffer must be submitted, If the manufacturer accepts CMS’s initial offer, the negotiation process ends.
- CMS will provide a written response to the manufacturer in response to an optional written counteroffer, either accepting or rejecting the counteroffer, within 30 days (e.g., July 31, 2026, if the manufacturer’s counteroffer is made on July 1, 2026). If CMS accepts the manufacturer’s counteroffer, the negotiation process ends.
- If CMS rejects the manufacturer’s counteroffer, up to 2 additional in-person or virtual meetings could occur between CMS and the manufacturer to discuss offers and counteroffers. The meetings would focus on manufacturer-submitted data and information about therapeutic alternatives, and how that information should factor into the maximum fair price. The timeframe for negotiation meetings would end no later than September 11, 2026. Additional written offers and counteroffers could be exchanged after CMS’s rejection of the manufacturer’s counteroffer and final agreement on the maximum fair price (up to one week prior to CMS submitting a final written offer).
- After any negotiation meetings between CMS and the manufacturer, CMS makes a final written offer for the maximum fair price (no later than September 30, 2026 for the 2028 negotiation cycle).
- Manufacturers consider CMS’s final offer and either accept or reject the offer in writing (by October 31, 2026 for the 2028 negotiation cycle).
- The negotiation process ends when CMS and manufacturers of selected drugs reach agreement on the maximum fair price, but no later than the statutorily defined deadline for the negotiation process to end (October 31, 2026 for the 2028 negotiation cycle) and the end of the negotiation period (November 1).
If an agreement on the maximum fair price is not reached by the October 31 deadline, manufacturers may be subject to an excise tax, which is being administered by the IRS, as specified in the Inflation Reduction Act. CMS has outlined an expedited process manufacturers can follow if they choose to not participate in the negotiation program, which would enable them to withdraw their drugs from coverage under Medicare and Medicaid to avoid paying the excise tax.
According to CMS, manufacturers may disclose information related to the negotiation process with CMS if they choose to do so. CMS will not publicly discuss the specifics of the negotiation process related to any manufacturer but reserves the right to do so if manufacturers themselves choose to disclose this information.
The marketing of a generic or biosimilar version of a drug previously selected for negotiation affects the availability of the negotiated price for that product
Drugs are not eligible to be selected for negotiation if there is a generic or biosimilar using that drug as the reference product approved or licensed by the FDA and being marketed. (Authorized generics do not count for this purpose, since they are not technically generic drugs as that term is commonly used, but rather the same drug product as the brand-name drug with a different label.) If a drug has already been selected for negotiation and CMS determines that a generic or biosimilar drug has been approved or licensed and is being “bona fide” marketed (as described above) – either before or during the negotiation process – the negotiation process will not start or will be suspended. The drug will continue to be a selected drug (not replaced by another drug), but no maximum fair price will be negotiated. To be removed from the list of selected drugs for 2028, CMS will need to make this determination between February 1, 2026 and November 1, 2026 (between the selected drug publication date and the end of the negotiation period.)
If CMS determines that a generic or biosimilar drug has been approved and marketed after a drug has been selected for negotiation and after a maximum fair price has been established, the maximum fair price will take effect, but depending on when the determination is made, that drug will no longer be a selected drug and the maximum fair price will not apply in subsequent years. For selected drugs for 2028, if the determination of generic drug availability is made between November 2, 2026 and March 31, 2028, the maximum fair price will only apply in 2028 and the drug will no longer be a selected drug for 2029; if the determination is made between April 1, 2028 and March 31, 2029, the maximum fair price will apply in 2028 and 2029 and the drug will no longer be a selected drug for 2030.
Potential savings for Medicare beneficiaries from Medicare’s negotiated drug prices depend on several factors
There is uncertainty about how many Medicare beneficiaries will see lower out-of-pocket drug costs in any given year under the Medicare price negotiation program and the magnitude of potential savings, since both will depend on which drugs are subject to the negotiation process and the price reductions achieved through the negotiation process relative to what prices would otherwise be. In addition, whether Part D enrollees pay lower out-of-pocket costs for a given Part D selected drug will depend in part on whether they pay flat copayment amounts or a coinsurance rate for the drug in their chosen Part D plan. If they pay coinsurance, they could see savings from a Medicare-negotiated price that is lower than their plan’s negotiated price. This applies for Part B selected drugs as well, where beneficiaries in both traditional Medicare and Medicare Advantage typically face a coinsurance rate of 20% (although many beneficiaries in traditional Medicare have supplemental coverage that helps with Medicare cost-sharing requirements).
Aside from the potential for out-of-pocket cost savings, the drug price negotiation program could improve Medicare Part D enrollees’ access to Part D drugs that are selected for negotiation, since the IRA requires Part D plans to cover all selected drugs with negotiated maximum fair prices, including all dosage forms and strengths. (Part D plans generally can choose which drugs to cover and not cover on their formularies, subject to CMS’s formulary guidelines and requirements, except for drugs in six called “protected classes,” where all or substantially all drugs must be covered.) KFF analysis of 2026 Medicare Part D formulary coverage of drugs selected for negotiation shows that the IRA’s coverage requirement for selected drugs led to improved coverage of the Part D drugs with negotiated prices available in 2026.
CMS will use the annual formulary review process to ensure that all Part D plans cover all dosages and formulations of selected drugs. CMS will also review whether Part D plan sponsors place selected drugs on non-preferred tiers; place selected drugs on a higher tier than non-selected drugs in the same class; require utilization of an alternative brand prior to a selected drug; or impose more restrictive utilization management tools on a selected drug relative to a non-selected drug in the same class. In any such instances, CMS expects Part D plan sponsors to provide a clinical justification for these practices and will only approve those formularies that adhere to all statutory and regulatory guidelines and requirements.
This work was supported in part by Arnold Ventures. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.