Emergency Contraception
Note: This brief was updated on January 29, 2025 to incorporate new data and policy changes on emergency contraception.
Emergency contraception (EC) is a form of backup birth control that can be used up to several days after unprotected intercourse or contraceptive failure and still prevent a pregnancy. In 1999, Plan B was the first oral product approved for use in the U.S. as an EC by the Food and Drug Administration (FDA). Since then, more EC products have been approved. Many have confused EC pills with mifepristone, the “abortion pill,” but EC pills do not cause abortion. EC pills prevent pregnancy by delaying or inhibiting ovulation and will not work if the pregnancy is established. This fact sheet reviews the methods of EC, known mechanisms of action, use of EC, and current national and state policies affecting EC access.
What is Emergency Contraception?
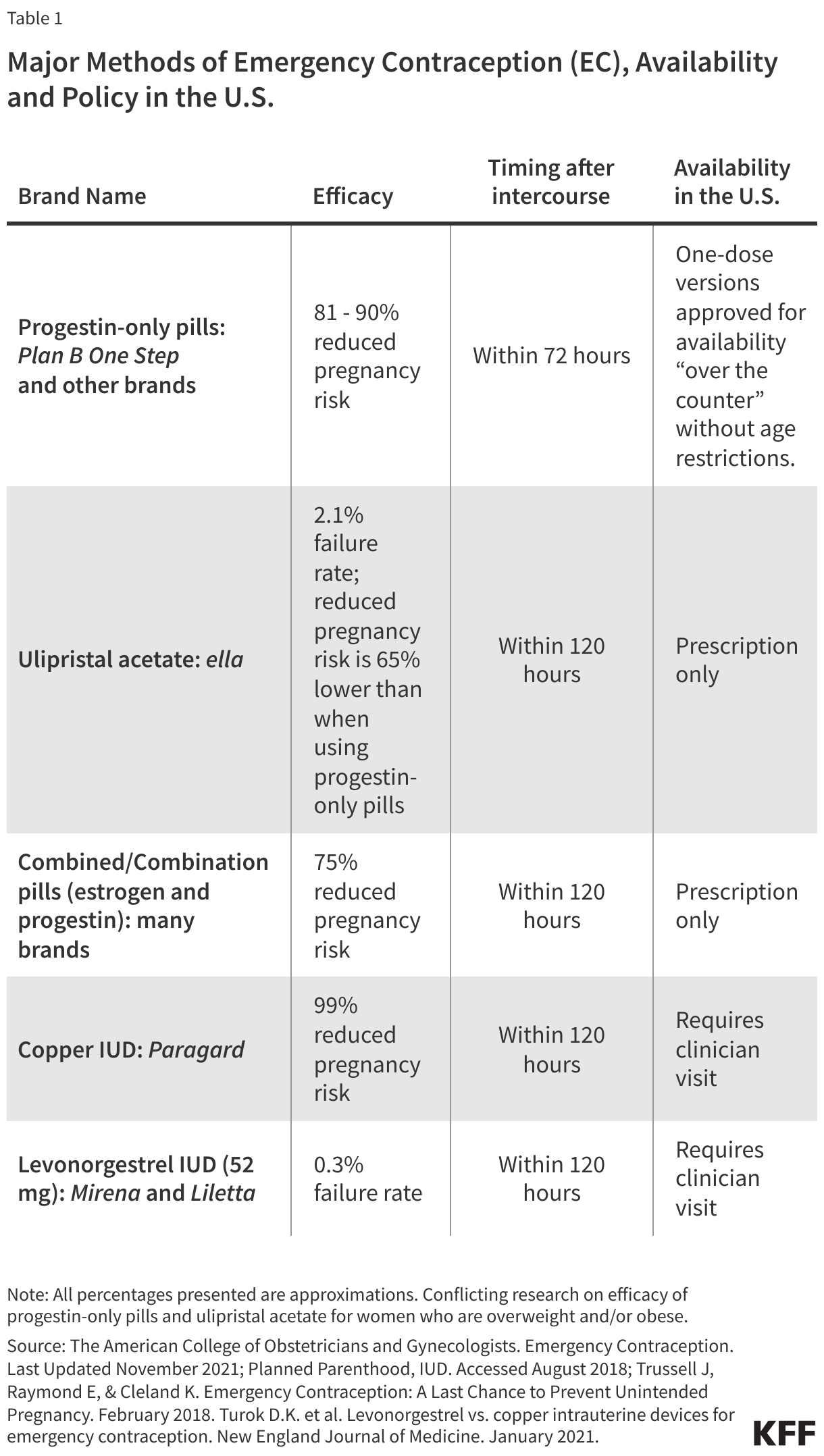
EC is used as a back-up birth control method to prevent pregnancy in the event of unprotected sex, sexual assault, or a contraceptive failure, such as a condom breaking. ECs do not terminate a pregnancy, stop the implantation of a fertilized egg, or affect a developing embryo. There are several methods of EC that are available in the U.S. including progestin-based pills, ulipristal acetate, and intrauterine devices (IUDs) (Table 1). The copper and levonorgestrel IUDs can be used after unprotected sex and as ongoing regular contraceptive methods. EC pills are only to be used after unprotected sex and are not intended for use as a regular method.
Progestin-Based Pills
- Plan B was the first oral form of EC to be made available in the U.S. as a pre-packaged dose of pills containing the progestin, levonorgestrel. Now, there are generic alternatives available as well. Progestin-based EC pills use the same hormones found in daily oral contraceptives and are the most widely used form of EC. EC pills are marketed today under the brand name Plan B One-Step and generic names and are available over-the-counter, without a prescription/ (Table 1).
- Progestin-based EC pills do not interrupt or adversely impact an established pregnancy, nor are they medical abortion drugs like mifepristone or methotrexate that end an established pregnancy. Plan B One-Step and the generic versions prevent pregnancy by inhibiting or delaying ovulation or by making it harder for sperm to reach an egg.
- Progestin-based EC is to be taken within 72 hours of unprotected sex in order to be most effective and reduce the likelihood of pregnancy by 81% to 90% when taken in this timeframe.
- There are no known serious side effects associated with progestin-based EC; side effects may include bleeding or spotting, headaches, nausea, and breast tenderness.
- Some research has suggested that efficacy of progestin-based EC is lower among women with Body Mass Index (BMI) levels greater than 25. However, in May 2016 the FDA announced that it had reviewed the available scientific data regarding the effectiveness of EC pills in overweight and obese women, and that the data are inconclusive and did not recommend a labeling change.
- Progestin-based pills can be stored for several years, with Plan B One Step having a shelf-life of four years when stored as directed. Consumers should always check the expiration date on the packaging prior to taking the pill.
Ulipristal acetate: ella
- Ulipristal acetate, marketed as ella, was approved by the FDA in 2010 for sale and use in the U.S.
- ella is a single-dose (30 mg) pill that is effective in preventing pregnancy up to five days after unprotected intercourse, giving women a longer timeframe to prevent unintended pregnancy than Plan B. Its mechanism of action is similar to that of progestin-based EC.
- Study findings show that side effects for ella are comparable to those for Plan B, and some research suggests that its effectiveness appears to diminish at BMI thresholds above 35.
- A 2025 study based on a small sample of women in Mexico suggests that when paired with misoprostol, one of the two drugs used in the FDA’s approved abortion medication regimen, higher doses of ulipristal acetate (60 mg) can terminate a pregnancy. It has already been demonstrated that misoprostol alone terminates early pregnancies. Prior research has shown that a 30 mg dose of ulipristal acetate alone, the dose that is used for emergency contraception in the U.S., does not disrupt an established pregnancy.
Combined/Combination Pills
- Certain daily oral contraceptive pills can also act as EC when taken in doses four or five times higher than the daily dose, although they are not specifically sold as emergency contraception. Oral contraceptive pills contain progestin and estrogen and are taken in two doses 12 hours apart to be effective as EC.
- Combined pills have been found to be safe and effective for preventing pregnancy within 5 days of intercourse.
Copper-T IUD: Paragard
- Available to women since the 1970s, copper-T IUDs are the most effective forms of EC, reducing the risk of pregnancy by more than 99% when inserted within 5 days of unprotected intercourse. IUDs are inserted into the uterus by a health care provider and require a visit to a clinic or provider’s office. They also can be used to effectively prevent subsequent pregnancy for up to 10 years.
- The hormone-free copper-T IUD works by interfering with egg fertilization by preventing sperm from reaching the egg. Previous research suggests the copper IUD inhibits implantation of a fertilized egg, but this mechanism of action has not been conclusively proven.
- Efficacy of copper IUDs does not diminish in women who are overweight or obese.
Levonorgestrel IUD: Mirena and Liletta
- Levonorgestrel IUDs are a type of hormonal IUD that contain the progestin levonorgestrel. To date, only levonorgestrel IUDs that contain 52 mg of levonorgestrel (LNG 52 mg IUDs) have been studied as a form of EC. LNG 52 mg IUDs are marketed under the brand names Mirena and Liletta.
- Like copper-T IUDs, LNG IUDs are inserted into the uterus by a clinician and require a visit to a clinic. LNG IUDs can subsequently be used as a regular form of contraception for up to seven years.
- Levonorgestrel IUDs recently started being used as a method of EC, and research has found that LNG 52 mg IUDs can be as effective as copper-T IUDs when inserted within five days after unprotected intercourse or contraceptive failure. The hormonal IUD prevents egg fertilization by making the cervical mucus impenetrable to sperm.
- Efficacy of levonorgestrel IUDs does not diminish in women who are overweight or obese.
Women’s Use of EC Pills
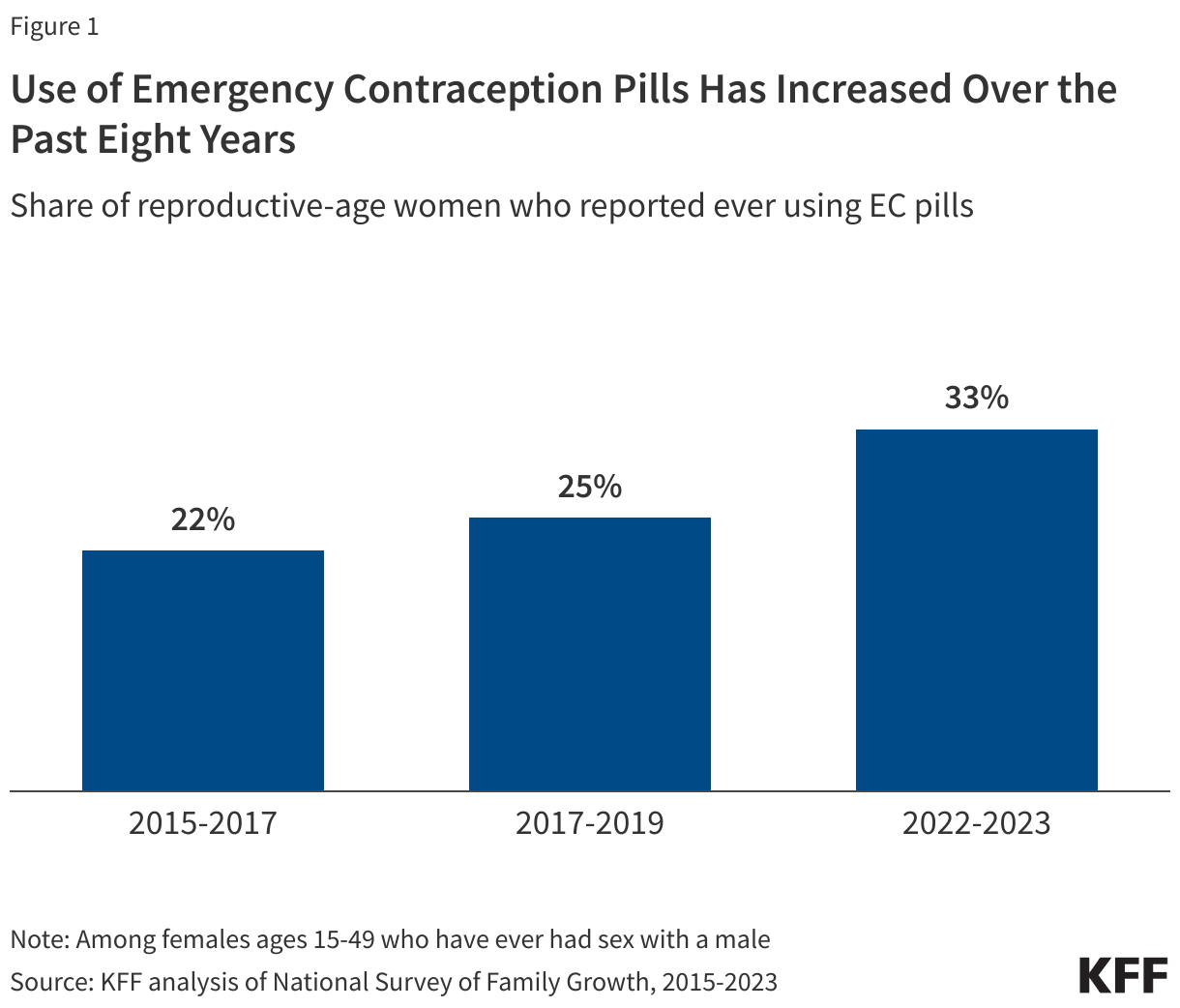
There have been numerous public health and educational initiatives to increase use of EC. Use of EC pills has increased over the past 20 years. Between 2022 and 2023, the most recent years for which data are currently available, 33% of women ages 15 to 49 who have ever had sex with a male reported they had used EC pills at least once in their lives, an increase from 22% in 2015-2017 (Figure 1).
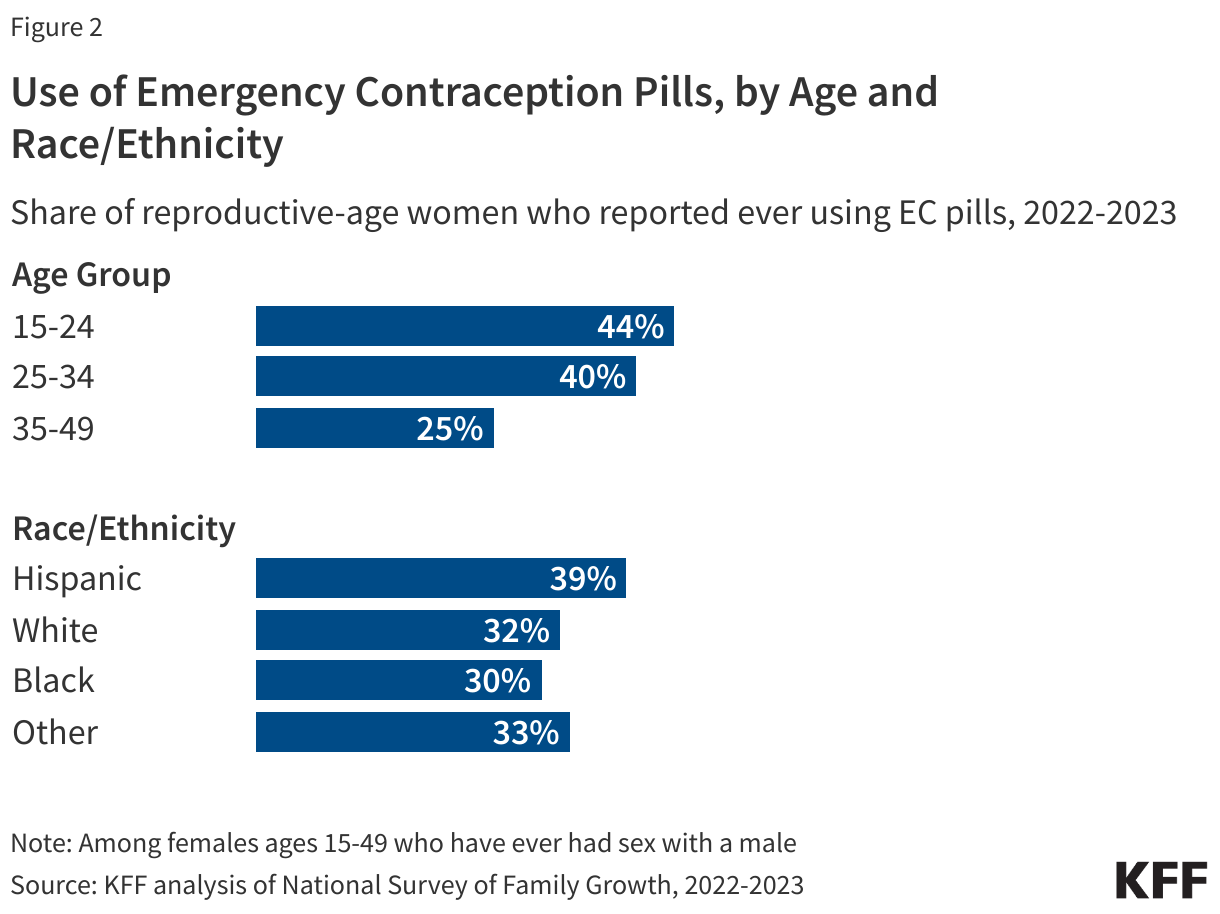
Younger women are more likely to report that they have ever used EC (Figure 2). More than four in ten women ages 15-24 (44%) and women ages 25-34 (40%) say they have taken EC pills, compared to 25% of women ages 35-49. One in four Hispanic women (40%) and approximately one in three Black and White women (30% and 33%, respectively) report ever taking EC.
Access and Availability
At least one form of oral EC has been available in the U.S. for over a decade and there have been several efforts to broaden women’s access to EC, particularly since its effectiveness window is time-limited.
Over the Counter Access of EC Pills
- Prior to 2006, a prescription was needed for all individuals seeking EC pills. Between 2006 and February 2014, Plan B and its generic equivalent were available without a prescription for men and women 17 and older, but adolescents under 17 needed a prescription.
- In 2014, the FDA removed point-of-sale age requirements for Plan B obtained over the counter (OTC) and generic versions began to enter the OTC market. Currently, several generic brands of EC pills, including Next Choice One Dose, My Way, Fall Back Solo, Take Action, levonorgestrel tablet, and Aftera are available OTC to women of all ages.
- A prescription is still required for ella for women of all ages.
Cost and Coverage
- The Affordable Care Act (ACA) requires most new private health plans (individual and group) to cover without cost-sharing all FDA-approved contraceptive drugs and devices as prescribed, including emergency contraception. This means that although OTC formulations are available without a prescription, the pills are only required to be covered by insurance if they were prescribed. Private insurance plans must also cover the cost of IUDs, as well as services related to insertion, follow up and removal, without cost sharing. However, there have been numerous reports of individuals having to pay out-of-pocket for contraception that they believe should have been fully covered, including for services related to the insertion and removal of IUDs.
- Seven states require private health insurance plans to cover some over-the-counter contraceptives, without any cost-sharing, including EC. For employer-sponsored plans, state coverage requirements generally apply only to fully-insured plans and not to self-funded plans.
- Family planning services are a required benefit under Medicaid. The coverage requirements under Medicaid are different for states that have expanded eligibility under the ACA. These programs must cover all prescribed, FDA-approved contraceptives, meaning that they must cover EC pills if a woman has a prescription. States have discretion in deciding whether they include EC in their traditional full-scope Medicaid programs or family planning expansion programs. A 2021 survey of state Medicaid programs found that while all responding states cover at least one form of EC in their traditional Medicaid programs, some states impose quantity limits. To increase access to contraception, eight states have opted to use state-only funds to cover at least some methods of OTC contraception for their Medicaid enrollees, primarily emergency contraception, without a prescription.
- Without a prescription or insurance, women in most states accessing EC OTC must pay the retail price. Plan B pills and the generic versions average can range from $10 to $50 when purchased OTC. Without insurance, the cost of IUDs can range from $500 to $1,300.
- Individuals without health insurance or who are ineligible for Medicaid may be able to obtain free or low-cost EC (in addition to other reproductive health care services) through a clinic or community health center participating in the federal Title X family planning program. The Title X program may also be an option for lower-income individuals covered by employer-sponsored health insurance plans that are not required to cover contraception because they existed prior to the ACA (known as “grandfathered” plans) or because their employer excludes coverage of emergency contraception based on a religious or moral objection to it. Regardless of the pathway, eligibility for the program is based on the individual’s household income and costs are based on a sliding fee scale.
Online Contraceptive Platforms
- New markets have emerged, particularly since the start of the COVID-19 pandemic, that allow people to obtain health care services without the need for an in-person visit, including several online reproductive health services platforms and smartphone apps where an individual can obtain hormonal contraceptives, including EC pills, outside of traditional health care settings. A 2023 KFF study on telecontraception found that 9% of survey respondents report getting a prescription for emergency contraception.
- These online platforms and apps typically employ licensed medical professionals to determine a client’s eligibility and to prescribe contraception in a similar manner to a clinic. The patient is then either mailed their contraceptives or their prescription is sent to a nearby pharmacy for pick-up.
- Some, but not all, of these platforms accept private insurance and/or Medicaid, and there is considerable variation in out-of-pocket costs for those who self-pay.
Provision of EC in Health Care Settings
- Several major medical and public health organizations, such as the American Academy of Pediatrics, American College of Obstetricians and Gynecologists, and the American Public Health Association, endorse the use of EC and advocate for broader access to EC.
- Counseling and coverage of EC is included as a standard of care in the federal requirements for providing Quality Family Planning Services (QFP). Providers are encouraged to discuss EC with their patients, inform them of its availability, and provide them with an advanced supply of EC pills if the patient requests them. Advance provision of EC can increase the chances that someone will have EC on hand and use it when needed, but research has found that few clinicians regularly provide advance provision of EC pills.
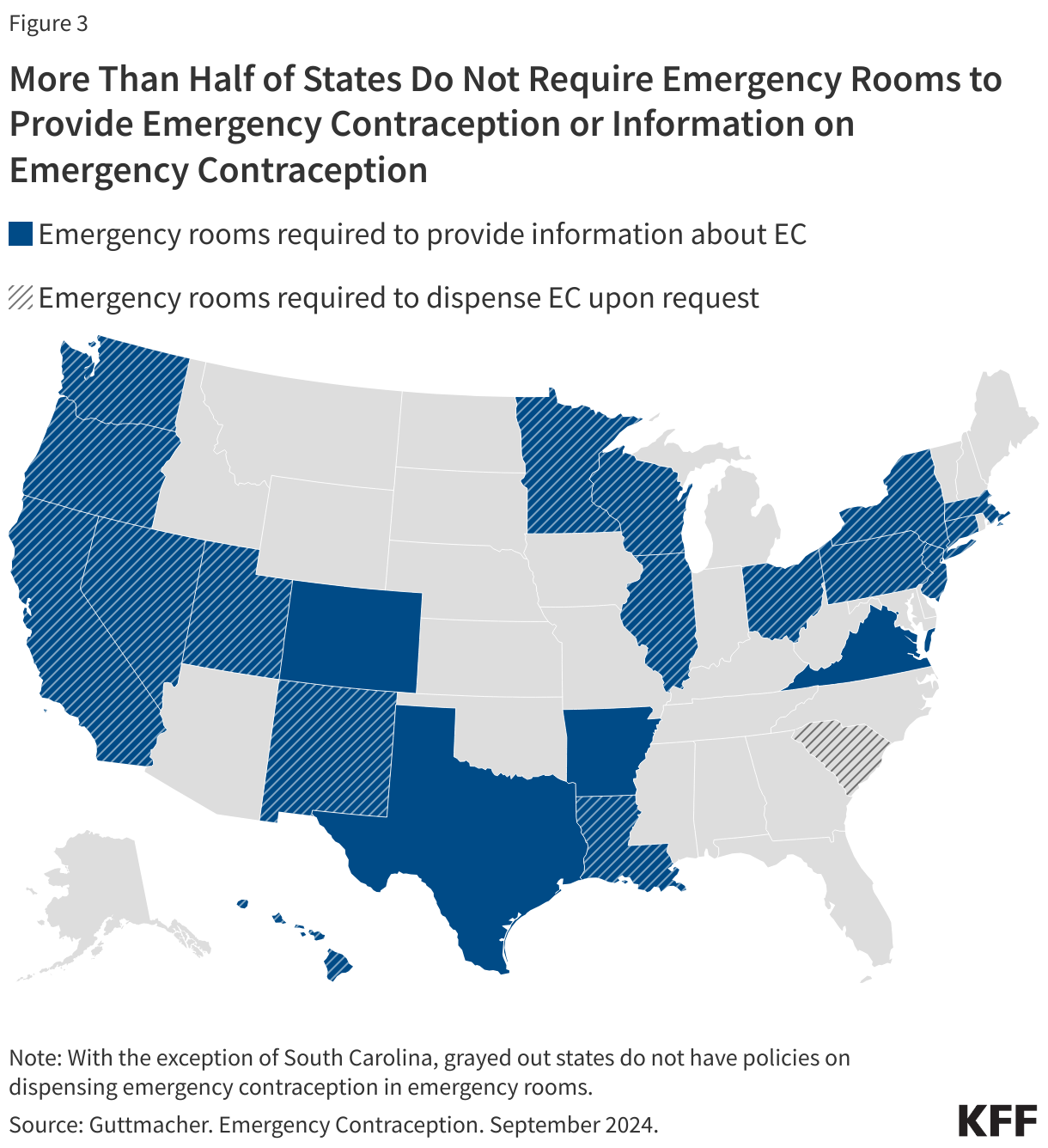
- There have been ongoing efforts to make EC more readily available to survivors of sexual assault. Currently, 21 states and the District of Columbia require that emergency room staff provide EC to women after sexual assault (Figure 3). However, some studies have documented that a sizable share of clinicians are not fully informed about EC options, safety, and efficacy. Additionally, some community health centers have reported challenges stocking IUDs due to high costs.
- In 2015, Indian Health Service (IHS) clarified its policy on access to the OC pill for Native American women, including that a prescription or age verification to access Plan B is not required.
Availability and Access in Pharmacies
- Nine states have laws that allow pharmacists to directly prescribe and dispense EC to women of all ages without obtaining a clinician’s prescription.
- Four states have measures that require pharmacies or pharmacists to fill all valid prescriptions. These policies have been enacted, in part, in response to reports of pharmacists refusing to fill prescriptions for EC pills because they oppose its use on moral or religious grounds.
- Ten states have laws allowing pharmacies or pharmacists to refuse to dispense EC pills on the basis of moral or religious objections.
- Pharmacies are not required to stock EC pills and some studies show that EC pills that may be sold OTC are not consistently stocked on store shelves and are sometimes kept behind the counter or in a locked display due to the high cost of the product. This report also documented misinformation regrading age and ID requirements among pharmacy staff and consumers.