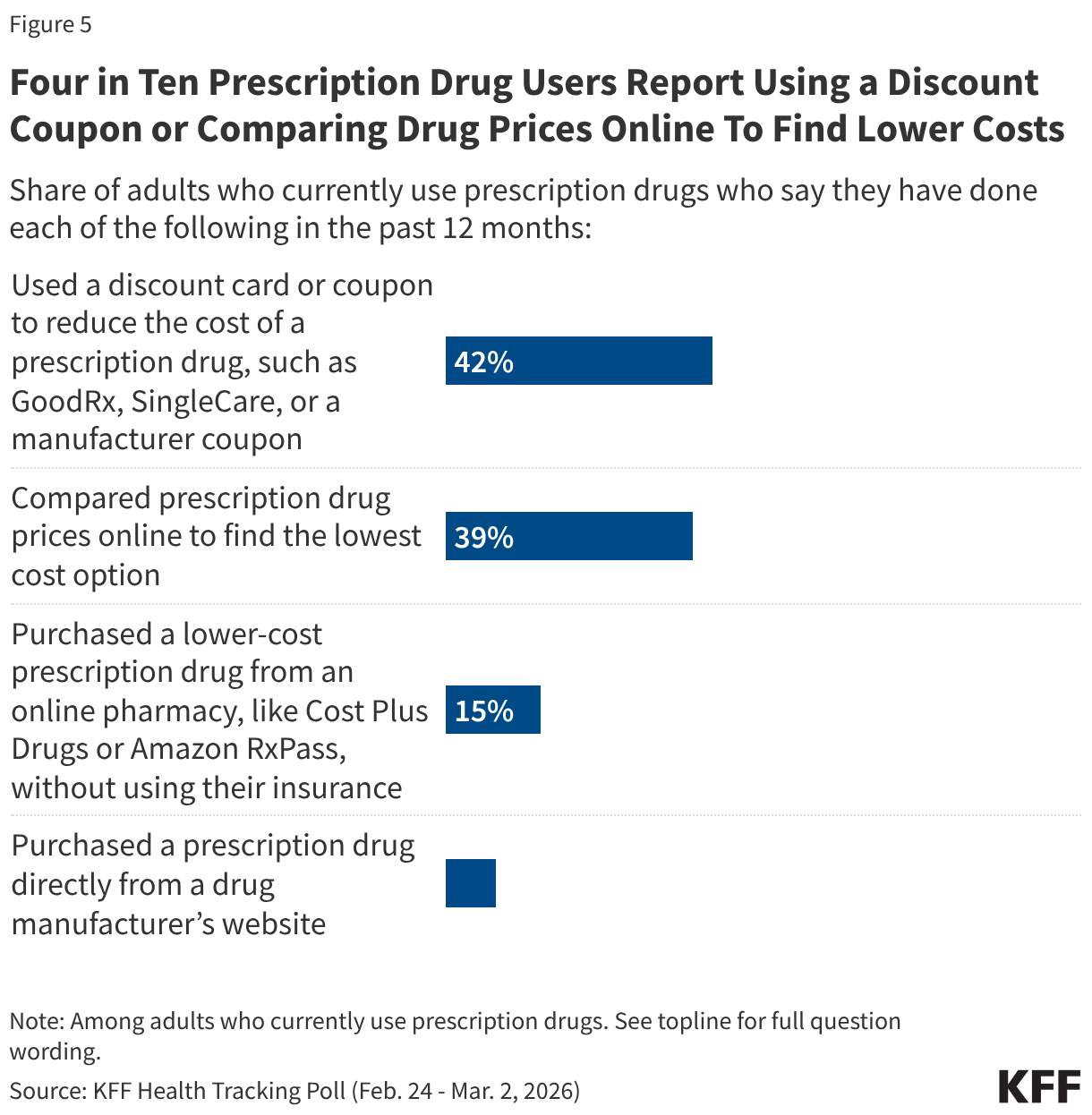
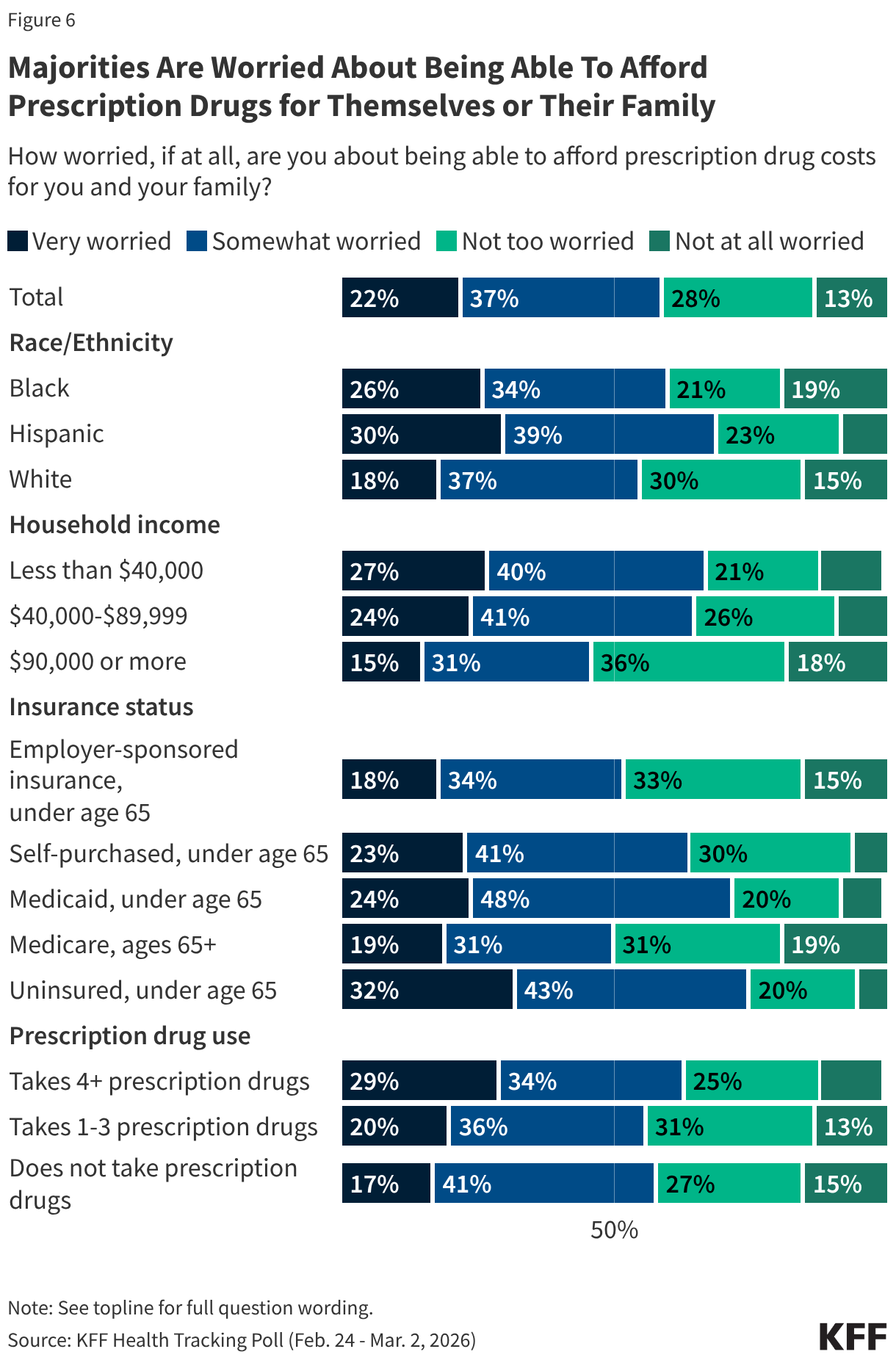
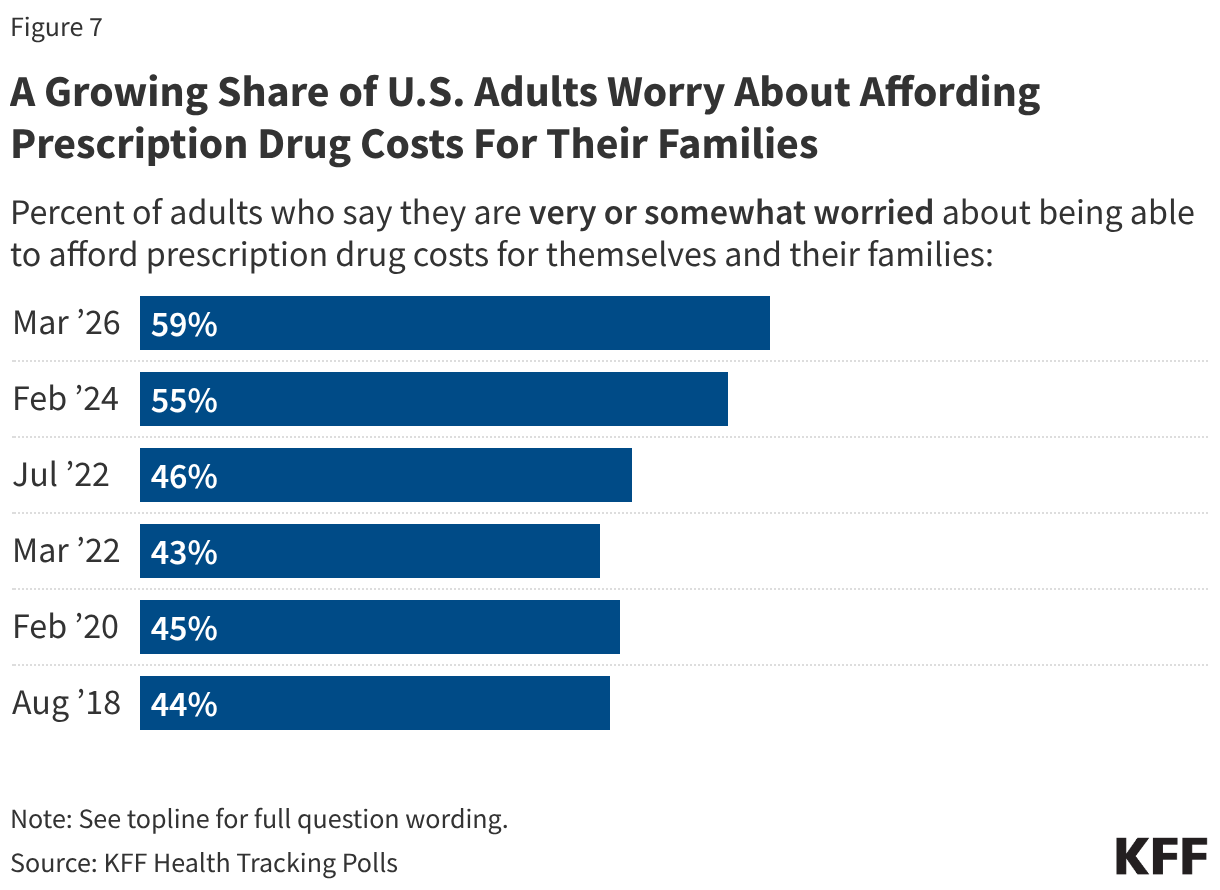
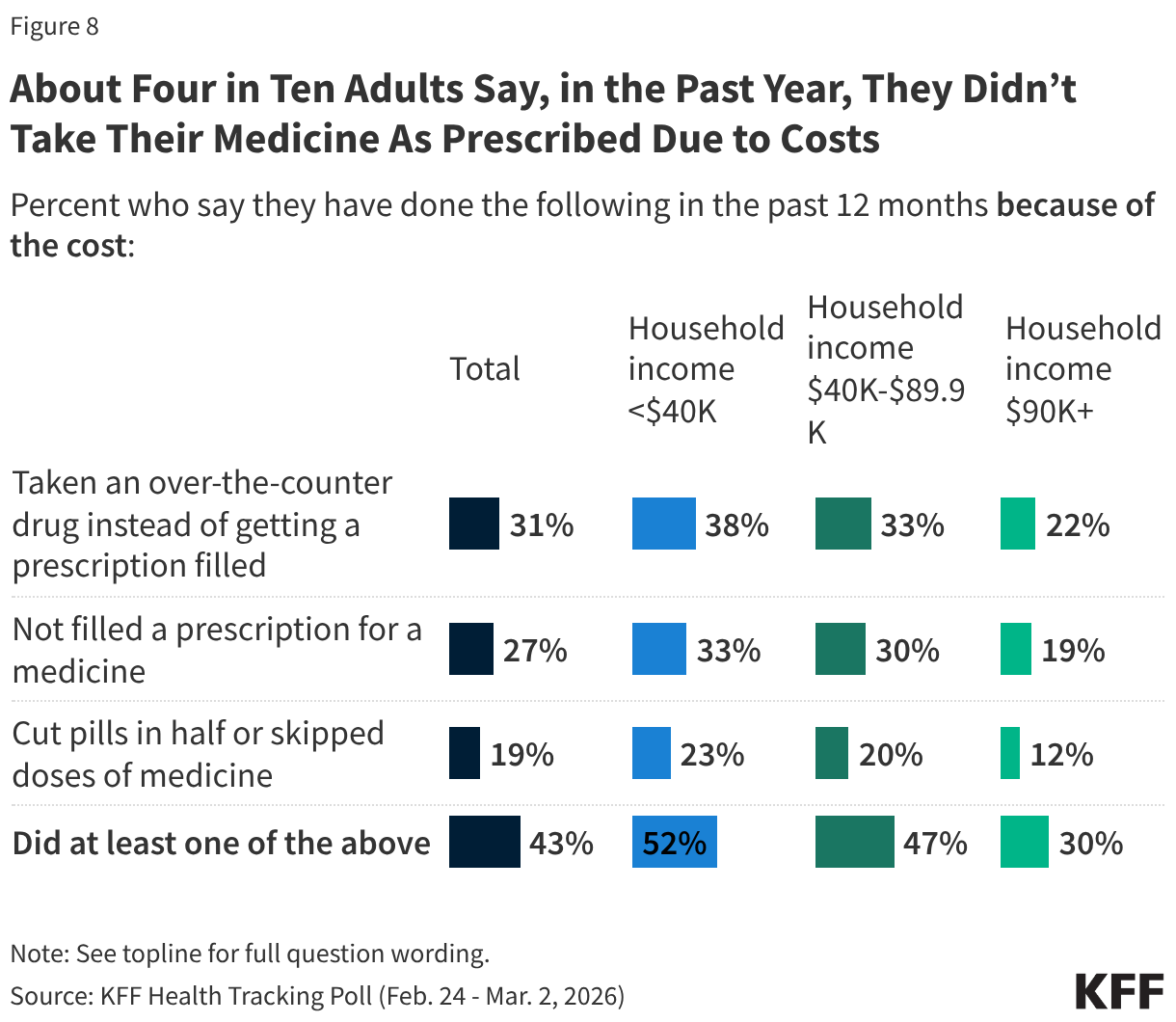
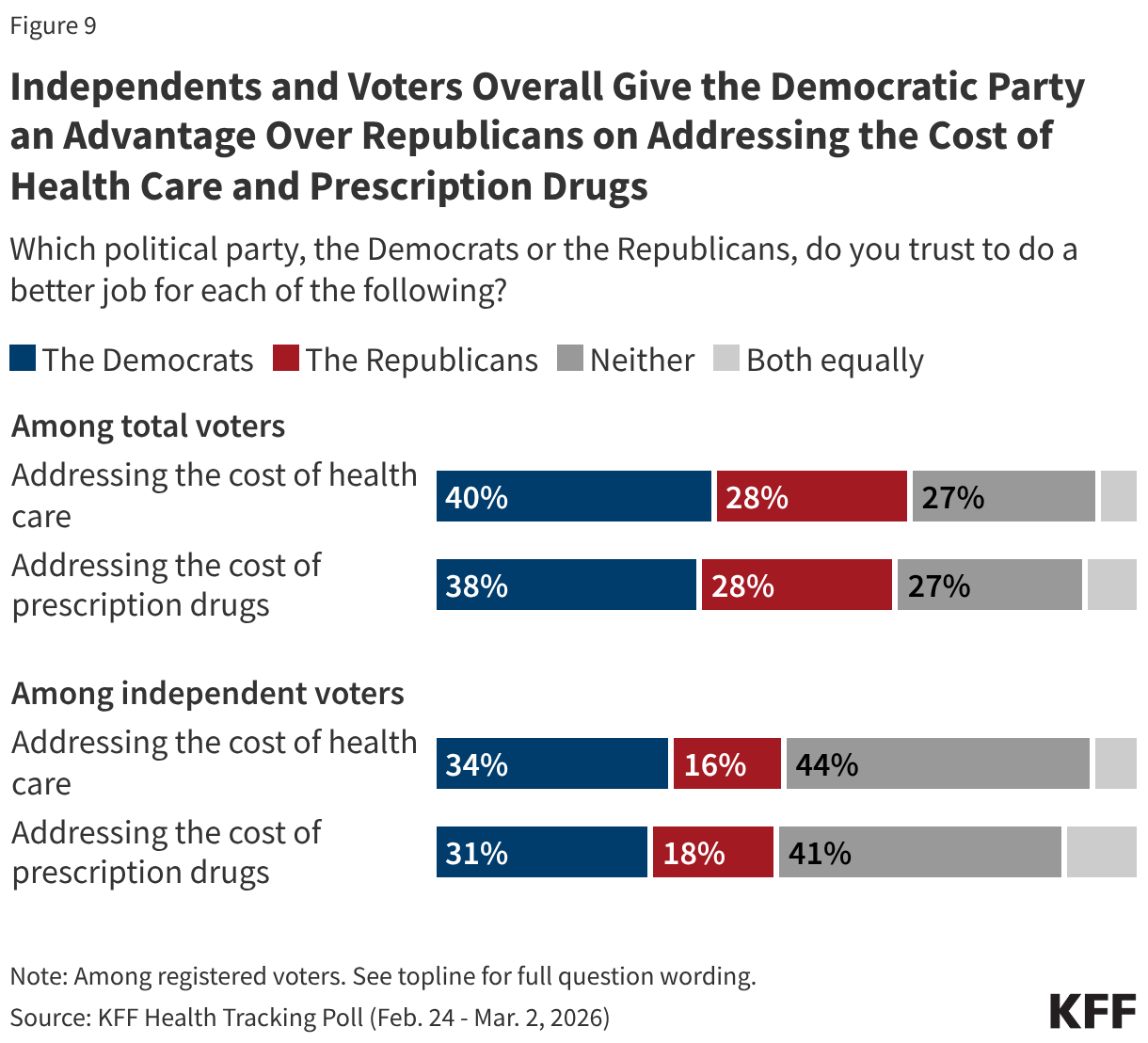
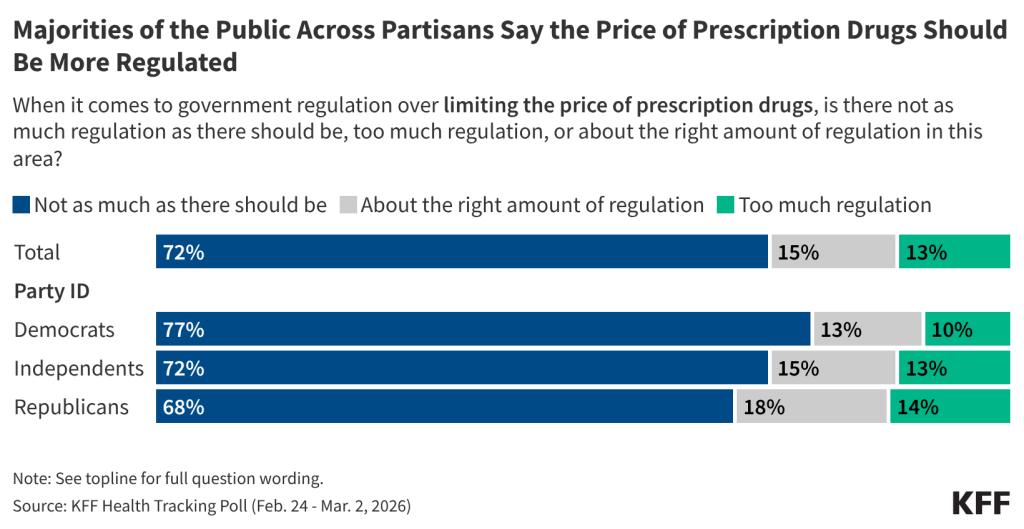
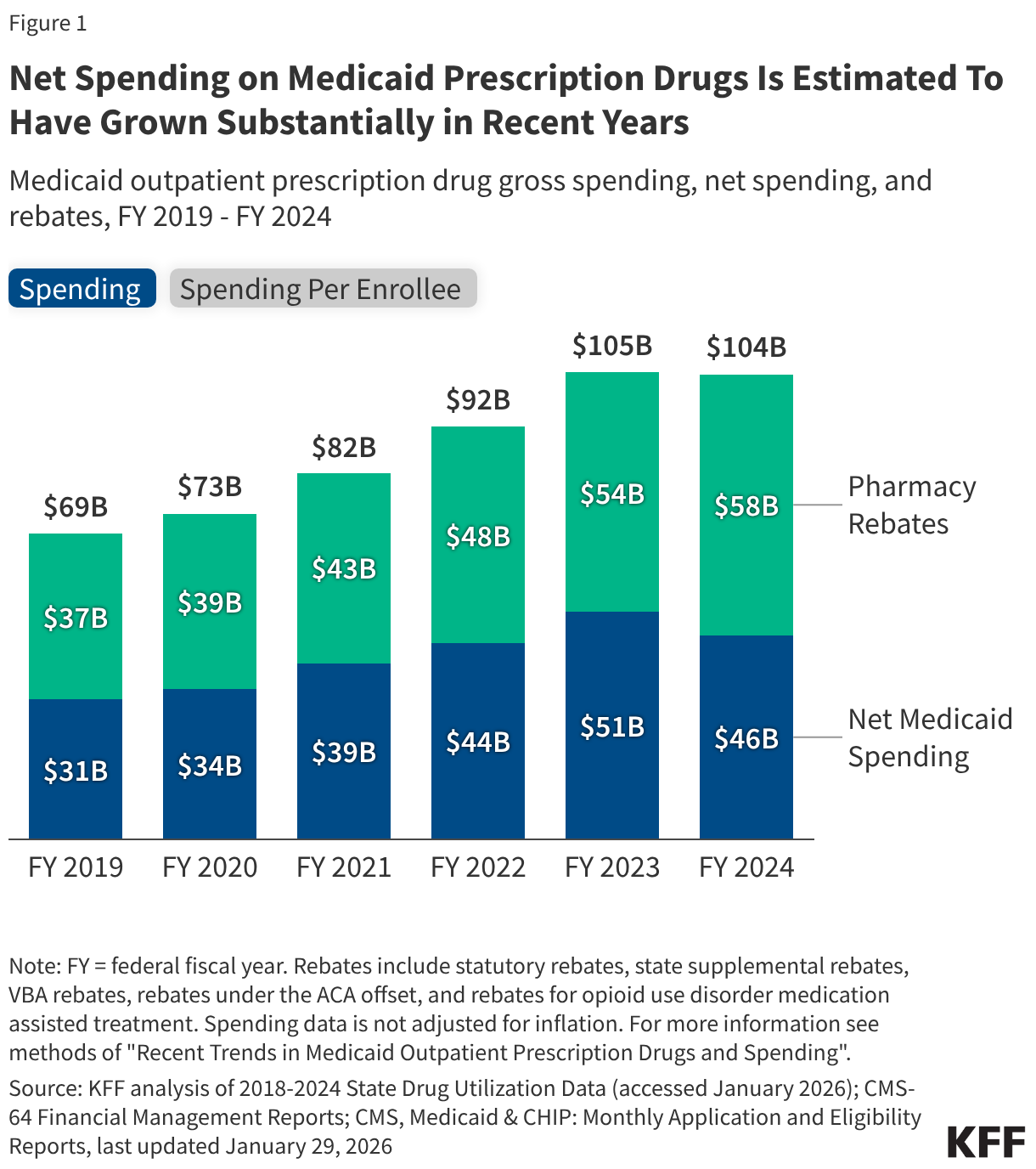
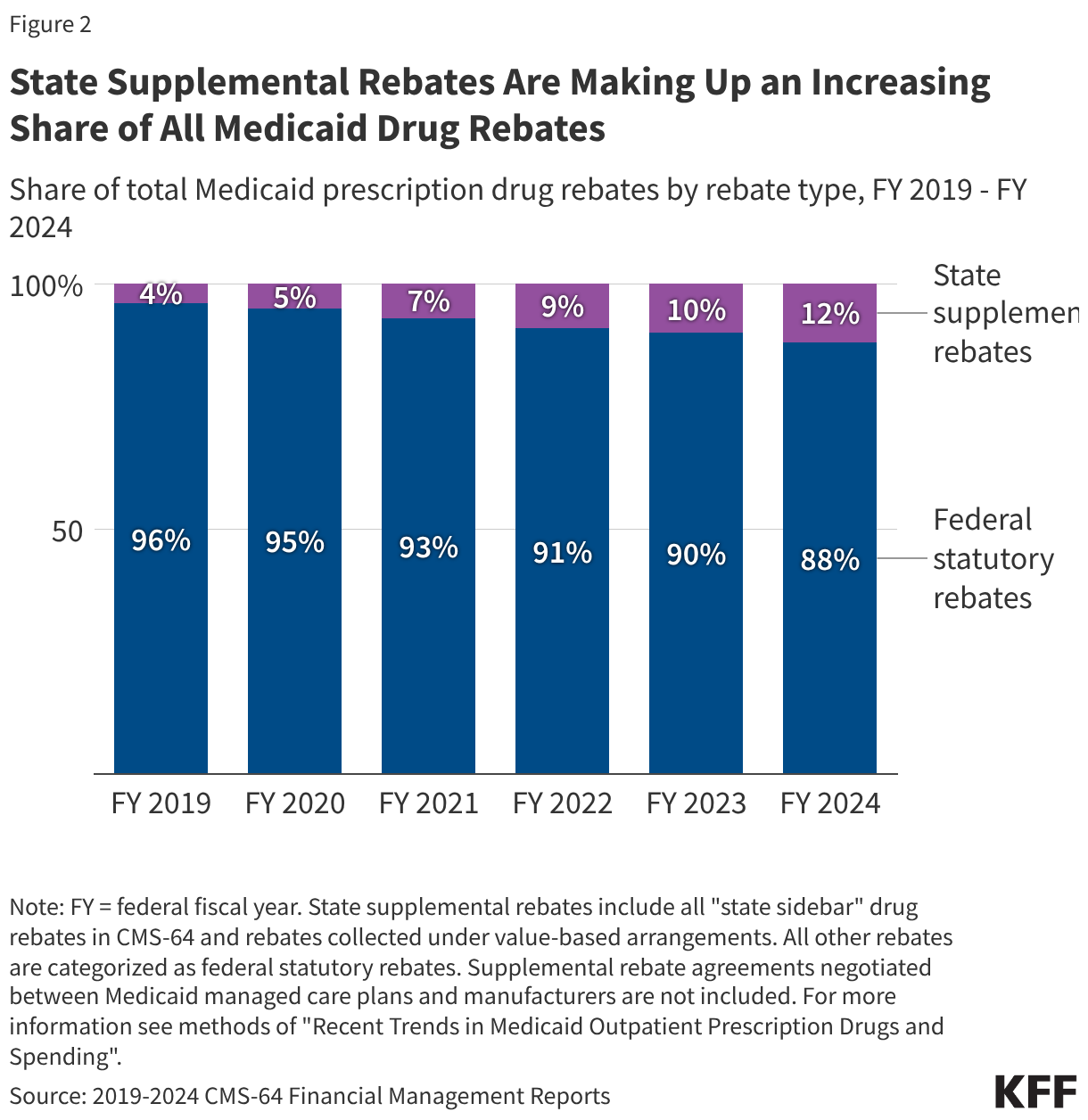
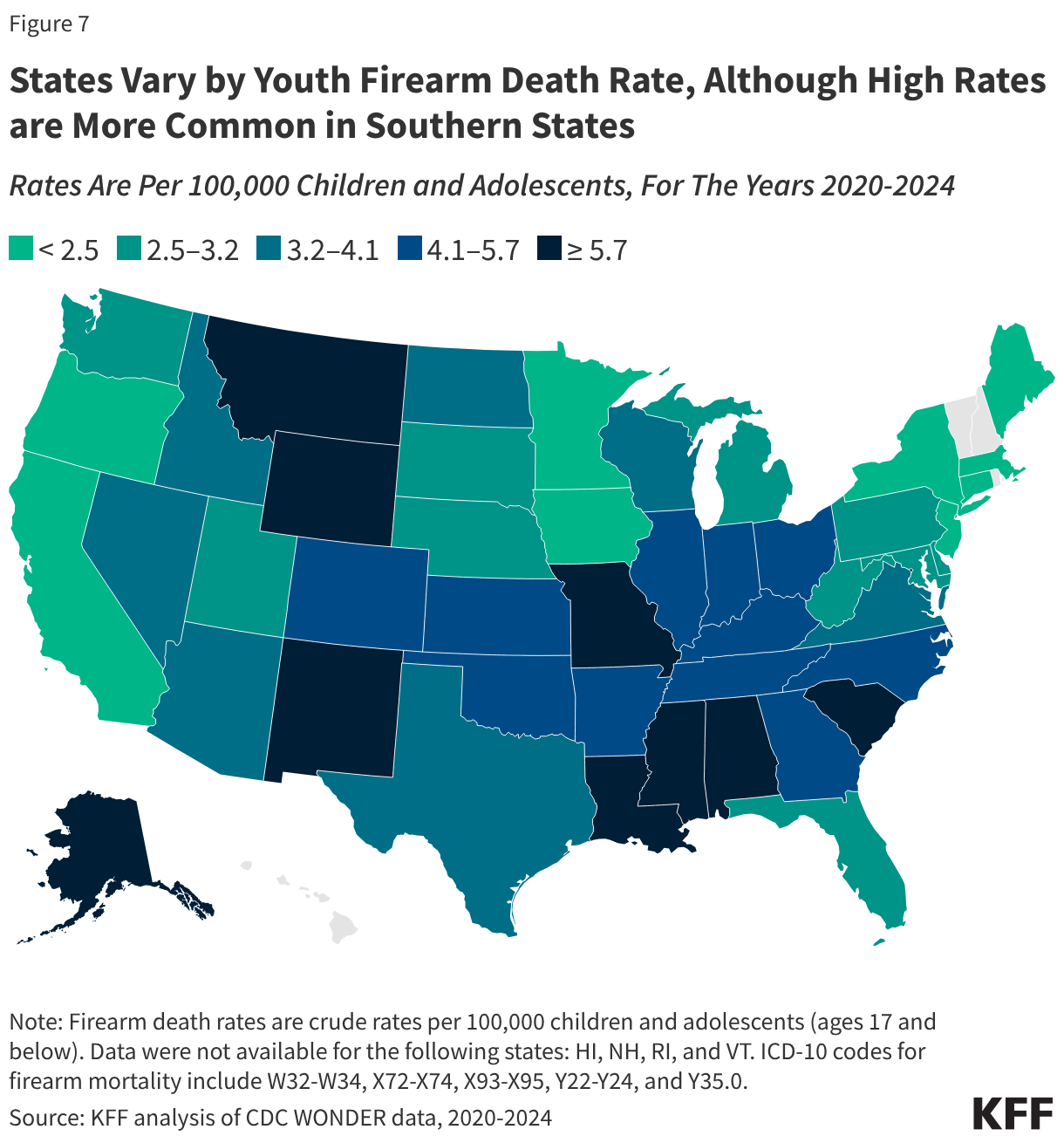
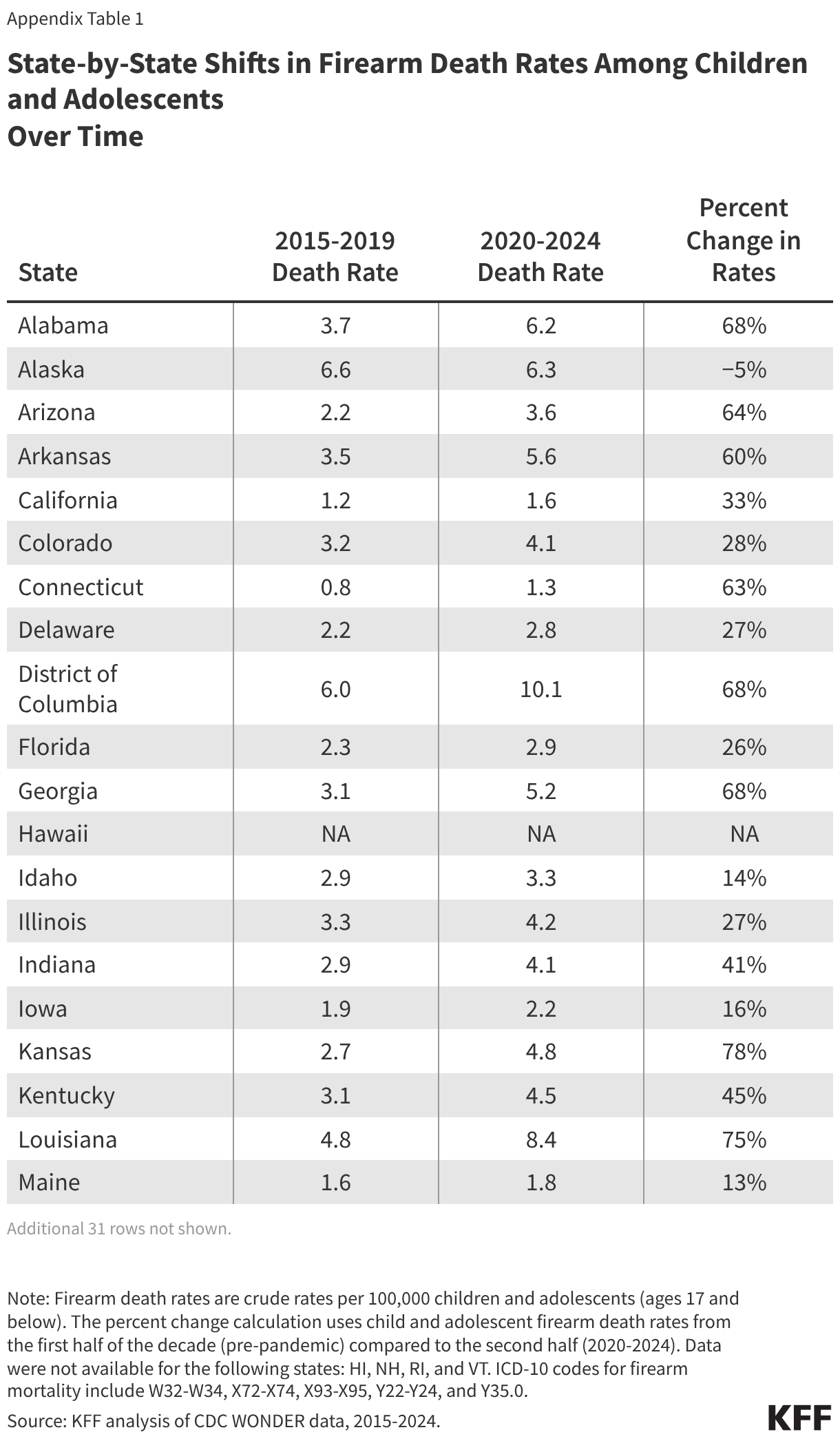
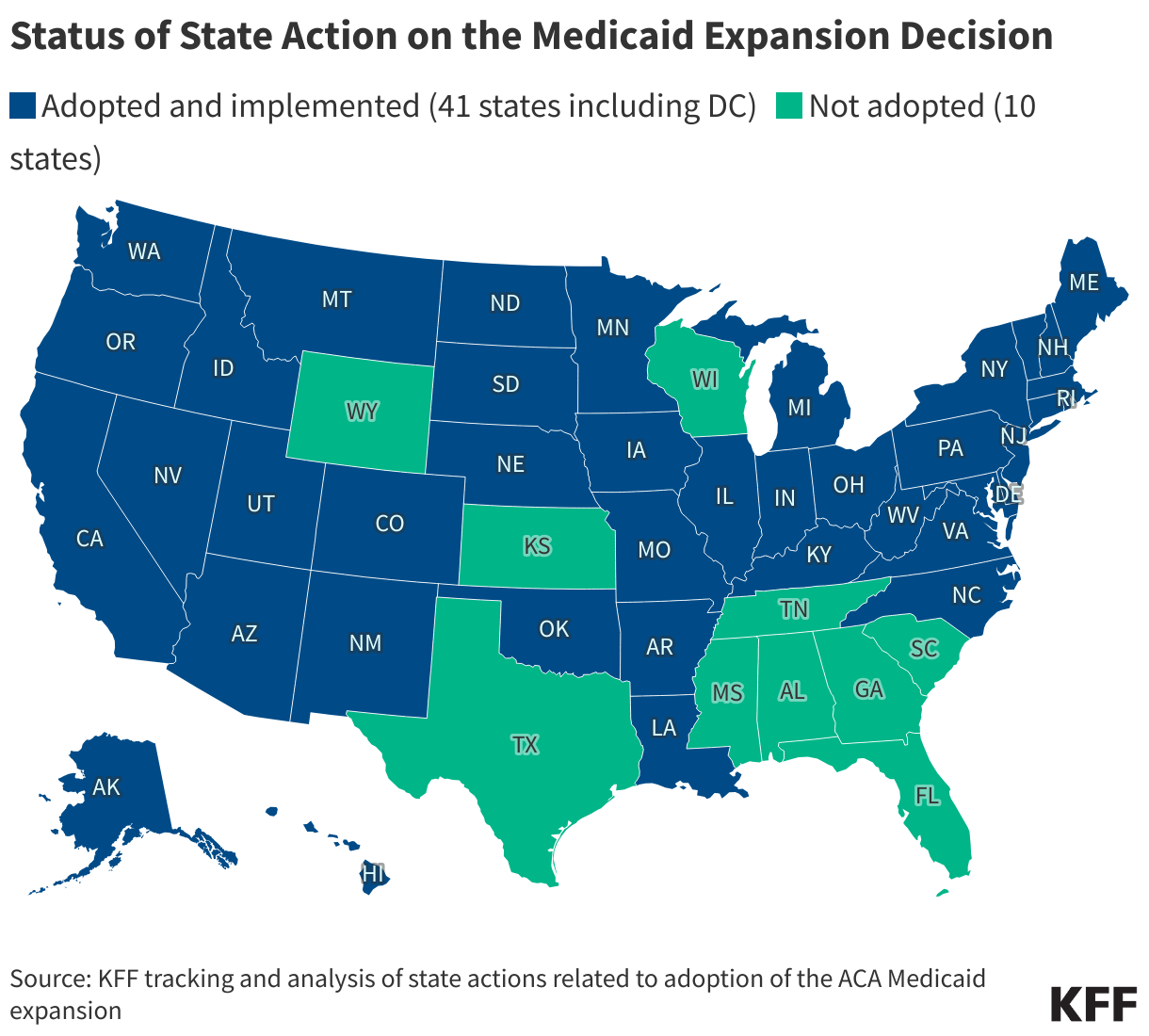
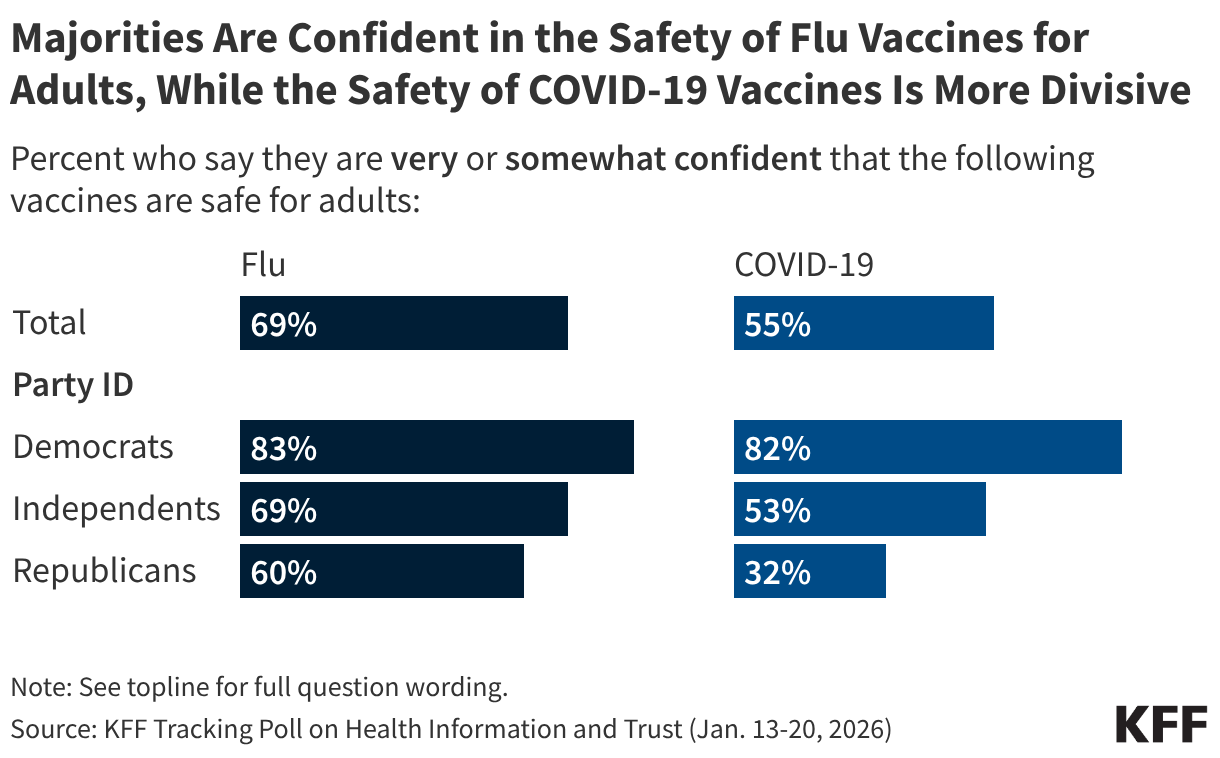
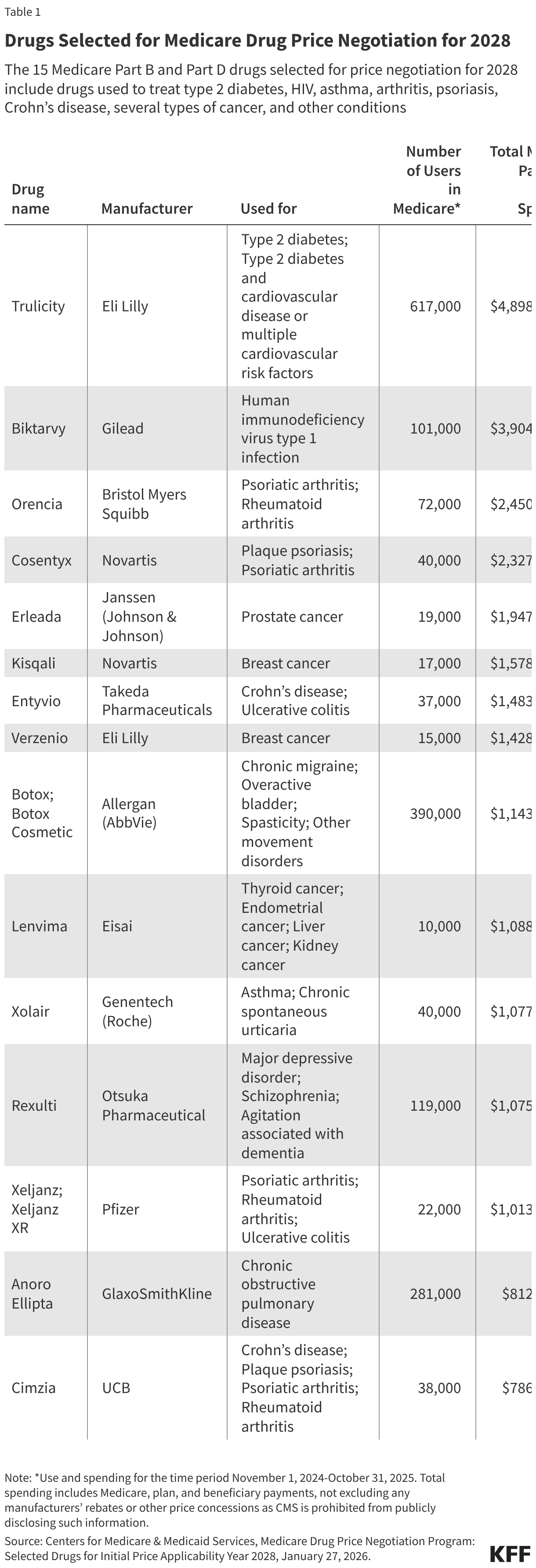
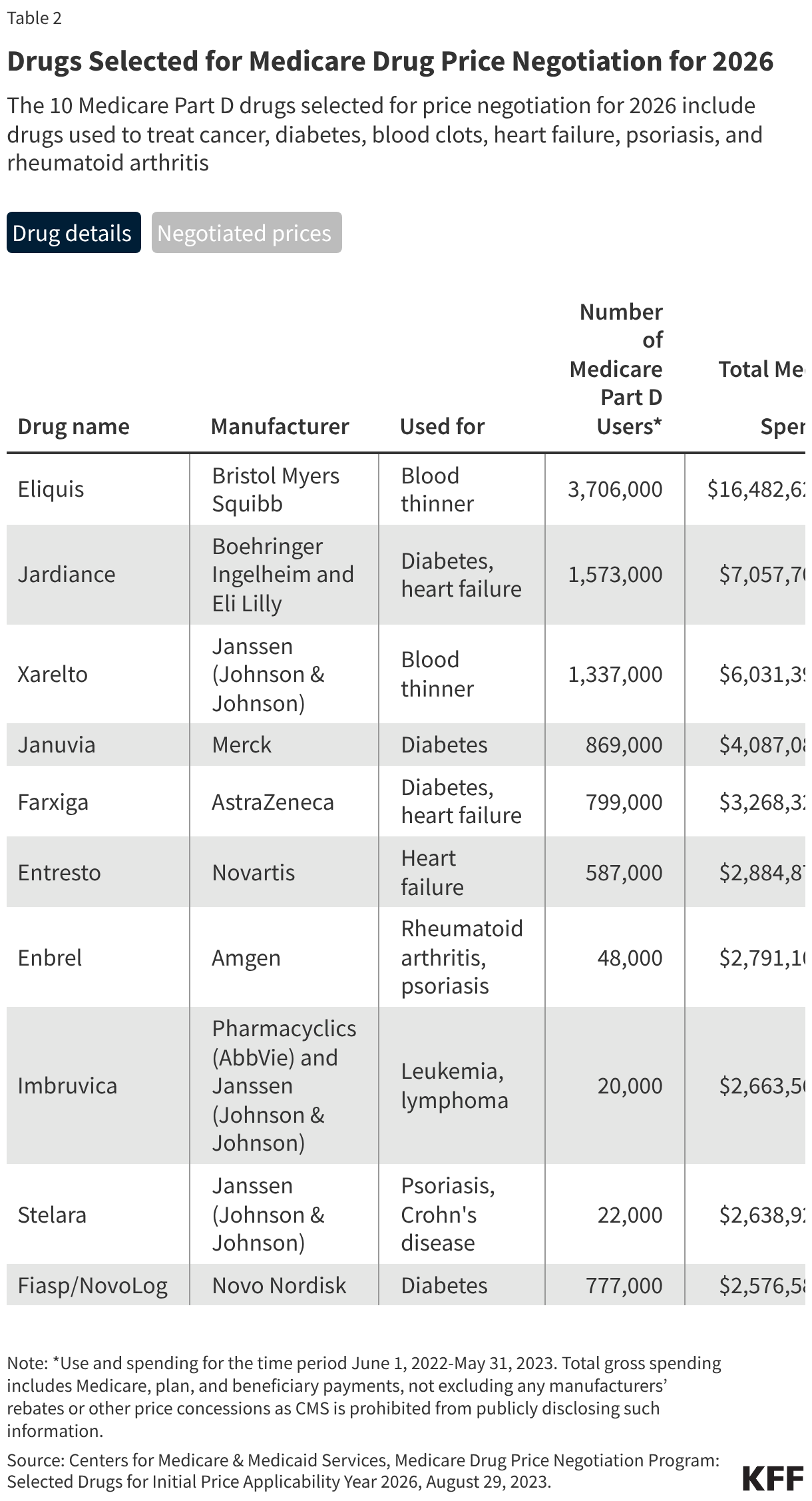
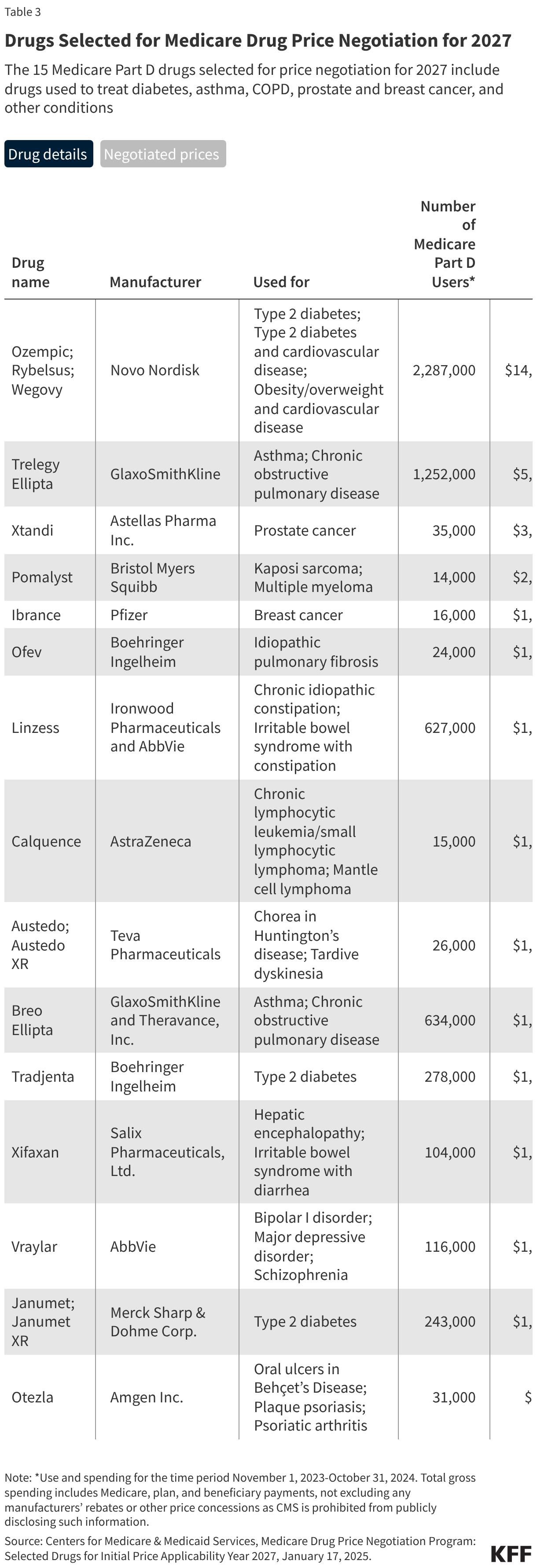
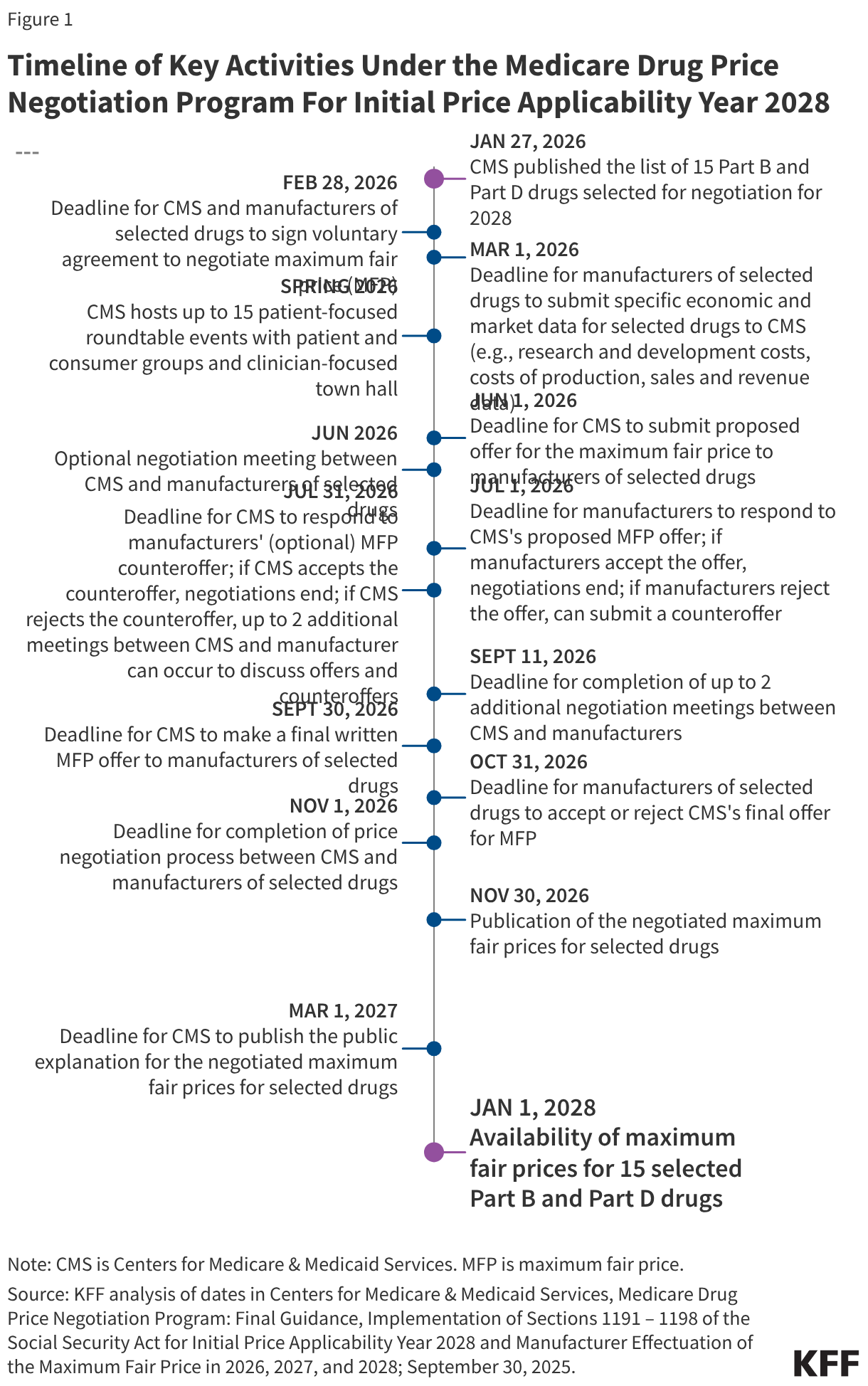
Fifteen drugs covered under Medicare Part B, which covers physician-administered drugs, or Medicare Part D, Medicare’s outpatient prescription drug benefit program, were selected for price negotiation in 2026, with Medicare’s negotiated prices for these drugs taking effect on January 1, 2028 (Table 1). These 15 drugs include treatments for type 2 diabetes, HIV, asthma, arthritis, psoriasis, Crohn’s disease, several types of cancer, and other conditions (See Table 1). Total gross Medicare spending on these 15 drugs between November 2024 and October 2025 was $27 billion, with 1.8 million Medicare beneficiaries using these medications during that time. Starting in 2027 and in each subsequent year, up to 20 additional drugs covered under Part B or Part D will be selected for negotiation. The number of drugs with negotiated prices available will accumulate over time.
Most Medicare Beneficiaries Affected by Plan Terminations in 2025 Have Robust Medicare Advantage Options in 2026
After years of rapid increases, Medicare Advantage enrollment growth slowed in 2025, a trend that continued in 2026. The number of Medicare Advantage prescription drug (MA-PD) plans available to the average Medicare beneficiary has declined from a peak of 36 in 2024 to 32 in 2026. Additionally, the trend of rapidly expanding extra benefits, spurred by sharp increases in rebate payments from the federal government, has stalled, with a smaller share of plans offering over-the-counter allowances and meals after hospital stays. Medicare Advantage insurers have warned that recent changes to the Medicare Advantage payment system have already hurt enrollees, leading to plan terminations, reduced benefits, and higher costs, and that these harmful effects will be exacerbated if the Trump Administration’s proposed payment rates for the 2027 plan year are finalized.
Despite concerns raised by the industry, the Medicare Advantage market remains robust in terms of enrollment, plan choice, and extra benefits. Enrollment in Medicare Advantage surpassed 35 million people in February of 2026, as more than half of eligible beneficiaries receive their Medicare coverage from a private plan. The number of plan options has ticked down in recent years, but remains higher in 2026 than in 2022 and every year before. Virtually all Medicare beneficiaries have at least one zero-premium plan with prescription drug coverage to choose from (excluding the Part B premium that all beneficiaries pay). Almost all plans (at least 98%) offer vision, dental, and hearing – benefits that are not covered by traditional Medicare. At the same time, rebate payments to plans from the Medicare program, which must be used to lower cost sharing, pay for extra benefits, and reduce premiums, are expected to reach their highest level ever, averaging more than $2,600 per enrollee in 2026.
Nevertheless, 2.6 million people who were covered by a MA-PD plan in 2025 had that coverage terminated at the end of the year as insurers decided to discontinue or reduce the service areas where certain plans were offered. Plan terminations affected 13% of all enrollees in individual MA-PDs in 2025, a substantially larger share than in previous years (6% of enrollees in individual MA-PDs in 2024 were affected by plan terminations). There are several reasons why plan terminations may have increased going into the 2026 plan year. Increases in the utilization of health care services, consistent with higher spending growth, and slower increases in the federal payments per enrollee to Medicare Advantage plans (stemming from changes to how payments are adjusted for the health status of enrollees) have somewhat reduced the relatively high gross margins private insurers realize on their Medicare Advantage business. This has led insurers to take a more careful look at the plans they offer and reduce the number of plans or withdraw from some markets in efforts to stabilize their margins. In some cases, insurers are also investing more in special needs plans (SNPs), which restrict enrollment to people with specialized health needs or who are covered by both Medicare and Medicaid (dual-eligible individuals). The number of SNPs offered has continued to increase, more than doubling since 2020.
This analysis examines the Medicare Advantage options in 2026 for Medicare beneficiaries who were covered by a MA-PD plan that was terminated at the end of 2025. It also examines the characteristics of plans that were terminated and the areas where the terminated plans were offered in 2025. The analysis excludes special needs plans, employer-and union-sponsored group plans, and individual plans that do not include prescription drug coverage (see Methods).
Key Findings
- Virtually all (98.9%) Medicare beneficiaries enrolled in a Medicare Advantage plan that terminated coverage at the end of 2025 (2.6 million beneficiaries) have at least one MA-PD plan available in 2026, with an average of 25 MA-PD options offered in their area in 2026. Most Medicare beneficiaries affected by a plan termination that had a zero-premium MA-PD option in 2025 also had a zero-premium MA-PD option in 2026.
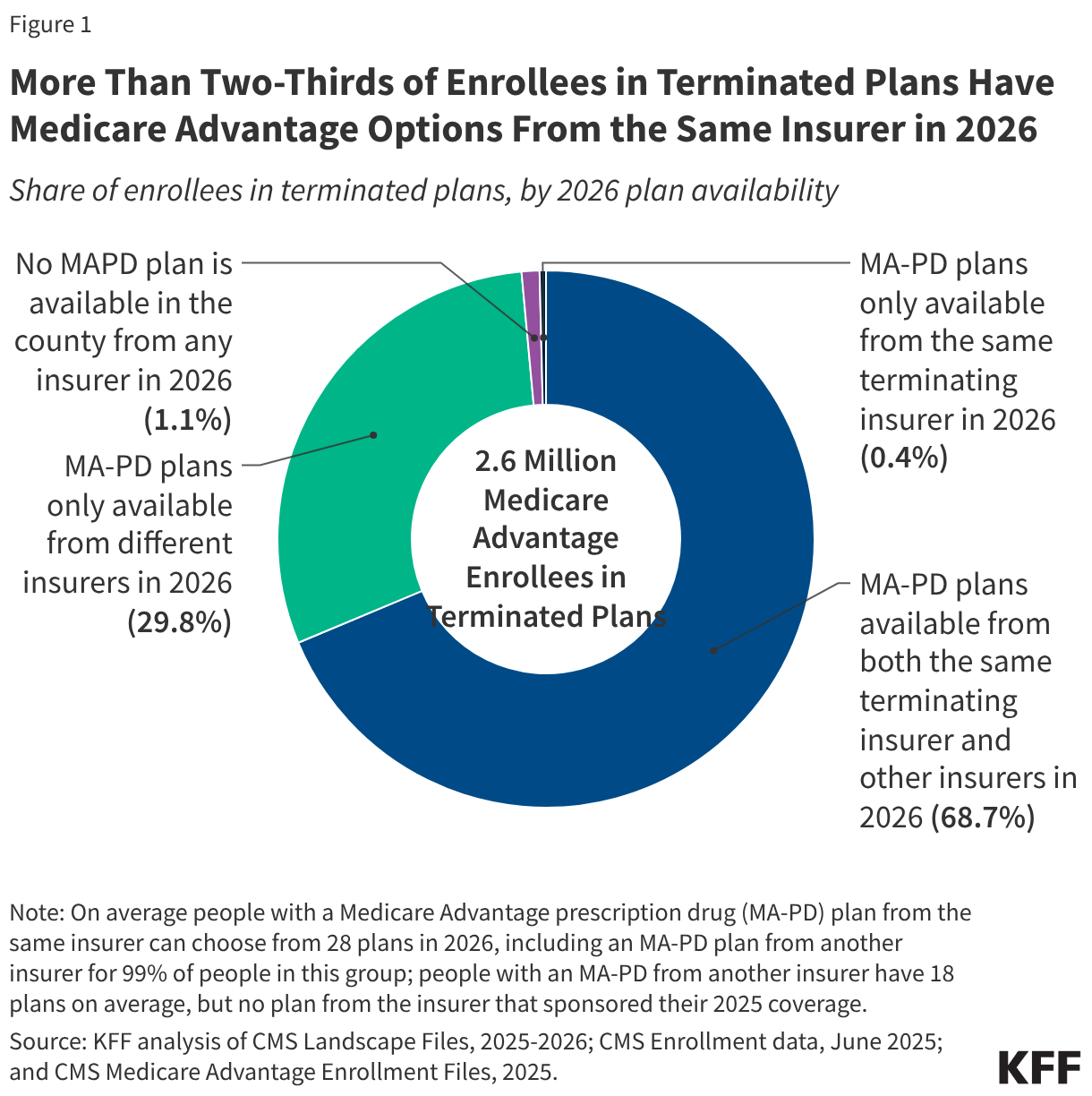
- More than two-thirds (68.7%) of Medicare beneficiaries enrolled in a plan that terminated coverage have at least one Medicare Advantage plan offered by the same insurer in 2026 in addition to MA-PDs from other insurers, while 29.8% have at least one option from another insurer in 2026 but none from the same insurer. Another 0.4% can choose an MA-PD from the same insurer in 2026 but have no MA-PD options from other insurers.
- Just 1.1% of people who were in terminated plans nationwide have no MA-PD options in 2026 (less than 30,000 people).
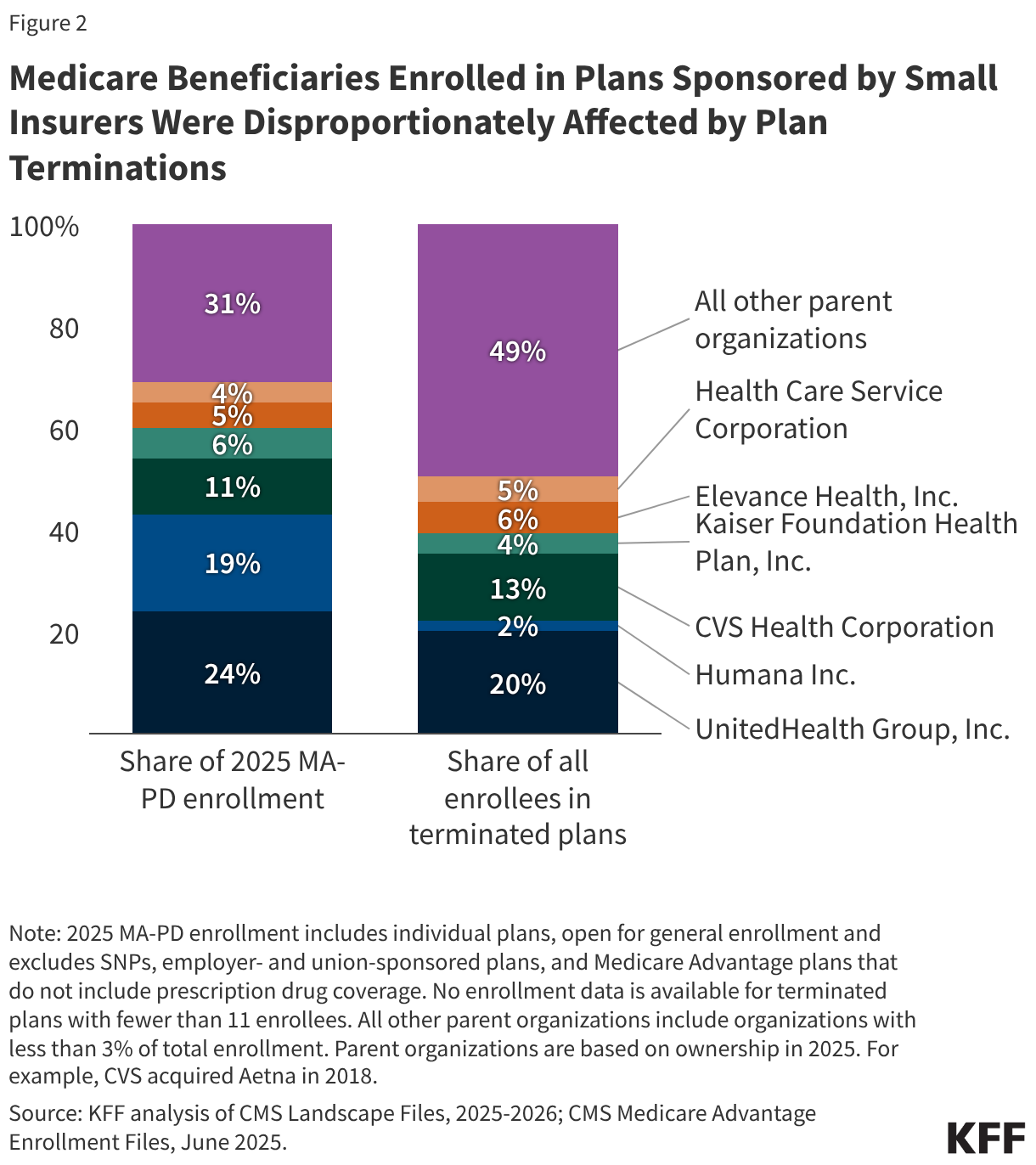
- About half (49%) of all enrollees in terminated plans were covered by small insurers; however, UnitedHealth Group, Inc. had the largest share (20%) of enrollees in plans that terminated coverage in 2025.
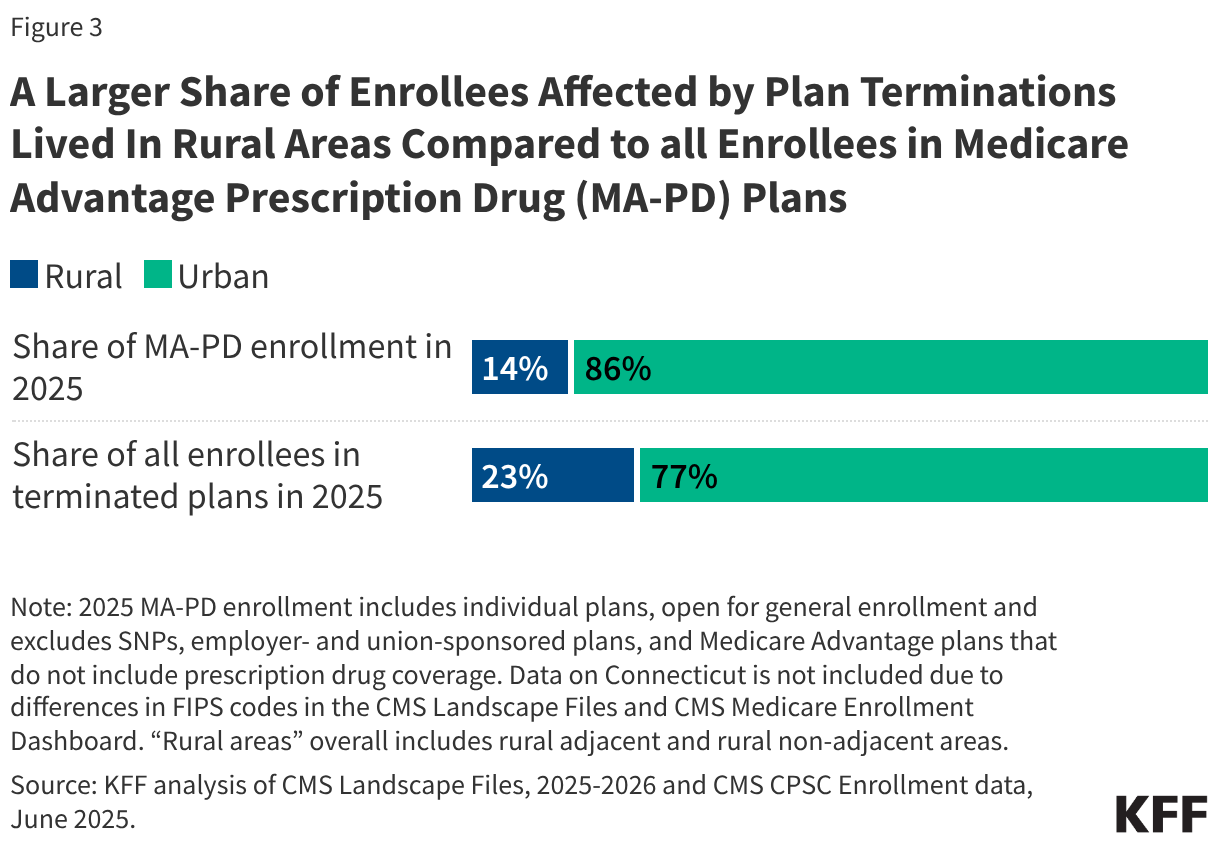
- Medicare Advantage enrollees living in rural areas were disproportionately affected by plan terminations. While 14% of 2025 MA-PD enrollees lived in a rural county, nearly one in four (23%) enrollees in a plan that terminated coverage at the end of 2025 live in a rural area. Plan terminations in rural areas were also more likely to lead to no MA-PD options in 2026.
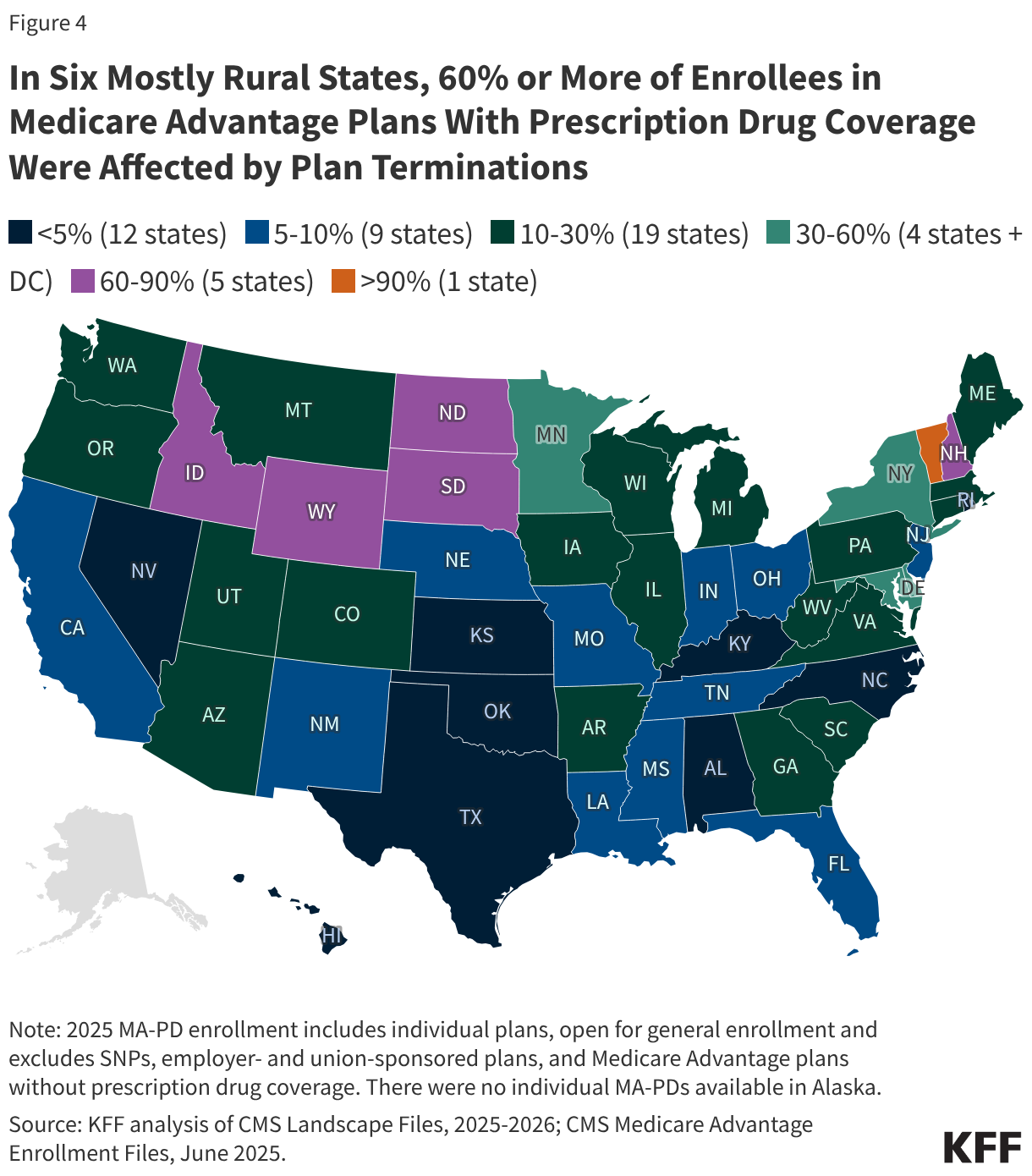
- The impact of Medicare Advantage plan terminations at the end of 2025 varied across states, ranging from less than 5% of enrollees in 12 states to 60% or more in 6 states, including Vermont where more than 90% of 2025 Medicare Advantage enrollees were in a plan that was terminated (the other states with 60% or more of enrollees affected are WY, SD, ID, NH, ND). The states where the largest shares of Medicare Advantage enrollees were impacted are mostly rural states that comprise a small share of Medicare Advantage enrollment (and a small share of enrollees affected by plan terminations).
More than two-thirds of enrollees in plans that terminated coverage at the end of 2025 have MA-PD options from the same insurer in 2026
Virtually all (98.9%) Medicare beneficiaries enrolled in a plan that was terminated at the end of 2025 have MA-PD plan options in 2026. On average, these Medicare beneficiaries can choose from 25 MA-PD plans offered by 7 firms, and for more than two-thirds of people (68.7%), those options include an MA-PD plan from the same insurer as their 2025 coverage, as well as a plan from another insurer (Figure 1). Just under a third (29.8%) of beneficiaries in a plan that was terminated at the end of 2025 have a MA-PD option from another insurer in 2026, but not the same insurer that sponsored their 2025 coverage. The vast majority (83%) of Medicare beneficiaries affected by plan terminations have at least one zero-premium MA-PD to choose from in 2026, similar to the share (86%) in 2025.
A small share of people in plans terminated at the end of 2025 (0.4%) only have the option of enrolling in a MA-PD plan from the same insurer that sponsored their 2025 coverage (no other insurers are offering a plan in their area). Just 1.1% (28,472) of Medicare beneficiaries in a plan terminated at the end of 2025 have no MA-PD plan available in 2026.
All Medicare beneficiaries enrolled in a plan that terminated coverage at the end of 2025 have the option to receive their Medicare coverage from traditional Medicare. Traditional Medicare offers broader access to providers and less utilization management than Medicare Advantage, but does not offer extra benefits, such as dental, vision, and hearing. Additionally, beneficiaries who want prescription drug coverage must purchase a standalone drug plan, and many traditional Medicare beneficiaries also purchase a supplemental Medigap policy. Beneficiaries who have had their coverage terminated by their Medicare Advantage insurer have a special guaranteed issue period to purchase a Medigap policy to supplement their coverage under traditional Medicare, meaning they cannot be denied coverage or charged a higher premium for a Medigap policy due to pre-existing conditions (something Medigap insurers are permitted to do in most states in most other cases outside of a person’s initial eligibility period for Medigap). Medigap policies require an additional monthly premium, which averaged more than $200 in 2023, though premiums vary both across and within states as well as by type of Medigap policy.
That additional cost is one reason many of the beneficiaries affected by plan terminations may choose a different Medicare Advantage plan for 2026 rather than switch to traditional Medicare. Enrolling in a different Medicare Advantage plan could lead to changes in benefits and cost sharing, as well as other plan characteristics. For example, previous KFF work has shown that Medicare Advantage provider networks vary substantially, even across plans offered by the same insurer, so having to switch plans, even if to another plan from the same insurer, could require a change in providers to stay in-network, unless the individual chooses to switch to traditional Medicare.
About half (49%) of the 2.6 million Medicare Advantage enrollees in plans that terminated coverage in 2025 were covered by small insurers
Medicare Advantage enrollment is highly concentrated among a small number of relatively large firms. Two insurers, UnitedHealth Group, Inc. and Humana Inc., together comprised just under half of all enrollment in individual MA-PDs in 2025, another four insurers comprise between 4% and 11% of enrollment each, while more than 100 small insurers (that represent less than 3% of MA-PD enrollment each) together enrolled just less than one-third (31%) of Medicare beneficiaries in an individual MA-PD in 2025. Those smaller firms, however, accounted for about half (49%) of Medicare beneficiaries enrolled in plans that were terminated at the end of 2025 (Figure 2).
UnitedHealth Group had the largest number of enrollees (532,869) in terminated plans, which represents a slightly smaller share (20%) of people affected by plan terminations than the firm’s share of individual MA-PD enrollment (24%) in 2025. Fewer than 2% of UnitedHealth Group enrollees (8,500 people) affected by plan terminations have no MA-PD options in 2026. More than half of this group lives in Vermont, where UnitedHealth Group pulled out completely.
In contrast, the second largest insurer, Humana, had a relatively small number of people affected by terminations, comprising just 2% of all terminated enrollees compared with the firm’s share of individual MA-PD enrollment (19%).
Among the smaller insurers, four firms terminated plans that affected at least 100,000 enrollees. Two of the firms, UCare Minnesota and Blue Cross Blue Shield (BCBS) of Michigan Mutual Ins. Co., were among the largest insurers in a single state, Minnesota and Vermont, respectively. (Note, while an insurer’s name may include a specific state, those insurers may operate in multiple states). UCare Minnesota, the second largest insurer in the state of Minnesota in 2025 (which also offered plans in some Wisconsin counties), terminated all of its individual MA-PD plans, which enrolled nearly 150,000 Medicare beneficiaries, although it is still offering D-SNPs. Fewer than 1% of former UCare Minnesota enrollees have no MA-PD options in 2026.
BCBS of Michigan Mutual Ins. Co., which offered MA-PDs in five states in 2025 (IA, MI, ND, SD, and VT), terminated plans that affected just over one-third of their 2025 enrollees and was one of two insurers to pull out of Vermont completely (leaving Humana as the only insurer in the market in 2026). Of the almost 110,000 enrollees in a BCBS of Michigan Mutual Ins. Co. sponsored plan in 2025 that was terminated, 17,000 (all in VT) have no MA-PD option in 2026.
Two other insurers terminated plans affecting over 100,000 enrollees and all of the Medicare beneficiaries in these two plans have other MA-PD options in 2026. Those insurers are Highmark Health, which terminated plans affecting 148,000 enrollees (44% of 2025 the firm’s individual MA-PD enrollment) in four states (DE, NY, PA, WV), and Lifetime Healthcare, Inc., which terminated plans affecting 106,000 enrollees (45% of the firm’s individual MA-PD enrollment) in New York.
The disproportionate share of Medicare beneficiaries in plans sponsored by small insurers affected by plan terminations could raise questions about the impact on competition and market concentration. However, recently released 2026 Medicare Advantage enrollment data suggests the impact of plan terminations is more mixed, as some small insurers have increased enrollment substantially year-over-year. For example, Devoted Health, Inc., which had 0.8% of enrollment in 2025, added more than 160,000 enrollees to its individual MA-PD plans between February 2025 and February 2026, more than doubling enrollment and expanding its market share to 1.6%. Overall, enrollment in individual MA-PDs sponsored by small insurers grew by more than 300,000 enrollees between February 2025 and February 2026, reflecting increases in enrollment for over half of all small insurers.
Additionally, enrollment in Medicare Advantage plans fluctuates and an insurer can lose enrollees beyond the number that were in a terminated plan or gain enrollees that offset losses from discontinuing plans or shrinking service areas. It is possible that higher levels of disruption in plan offerings from one year to the next may lead enrollees to switch plans, even if not directly affected by a plan termination. For example, enrollment in UnitedHealth Group plans declined by more than the enrollment in terminated plans, while the net increase in Humana MA-PDs between February 2025 and February 2026 was 831,000 enrollees.
Just under one-quarter (23%) of Medicare Advantage enrollees in a plan that terminated coverage live in a rural area
Medicare beneficiaries living in a rural county were disproportionately affected by plan terminations. Medicare Advantage enrollees living in a rural county comprised 23% of those who lost their coverage at the end of 2025 but just 14% of all individual MA-PD enrollment in 2025 (Figure 3). On average, enrollment in terminated plans was lower in rural counties than in urban counties. Consistent with the larger impact in rural counties, terminated plans had relatively low enrollment, on average. The median county-level enrollment per terminated plan was just 22 people.
Additionally, plan terminations in rural counties were more likely lead to no MA-PD options in 2026 than plan terminations in urban areas, though only a small number of enrollees overall were left with no options. Nearly two-thirds (65%) of the approximately 30,000 enrollees in terminated plans with no MA-PD options in 2026 live in rural counties (these counties are in just 8 states: CA, CO, MN, MT, NE, OR, SD, and VT), while the remaining 35% live in urban counties (all in VT). Among all enrollees in rural areas affected by plan terminations, 3% have no MA-PD option in 2026 compared to less than 1% in urban areas.
In some, mostly rural states, at least 60% of Medicare Advantage enrollees were affected by plan terminations
Just under 13% of enrollees in individual MA-PD plans in 2025 nationwide were affected by plan terminations, but in a handful of mostly rural states the impacts were much larger. In Vermont, more than nine-in-ten (93%) Medicare beneficiaries enrolled in an individual MA-PD in 2025 were in a plan that terminated at the end of the year. In five other states, at least 60% of enrollees were affected: Wyoming (65%), South Dakota (64%), Idaho (63%), New Hampshire (61%), and North Dakota (60%) (Figure 4). Altogether, these six states comprised 9% of the 2.6 million MA-PD enrollees who were in terminated plans, and just 2% of individual MA-PD enrollment in 2025.
In contrast, in 12 states fewer than 5% of Medicare Advantage enrollees were affected by a plan termination in 2025. These states represent less than one-tenth (9%) of people enrolled in terminated plans but nearly one-third (30%) of individual MA-PD enrollment.
In Vermont, plan terminations were significantly more likely to leave Medicare beneficiaries without any Medicare Advantage option than in other states. More than two-thirds (68%) of enrollees in terminated plans in Vermont have no MA-PD options in 2026. In contrast, less than 5% of enrollees in terminated plans in California, Colorado, Minnesota, Montana, Nebraska, and South Dakota have no MA-PD options in 2026. In all other states, every Medicare Advantage enrollee affected by plan terminations has MA-PD options in 2026.
Methods
This analysis examined the Medicare Advantage prescription drug (MA-PD) plans that were terminated at the end of 2025 and MA-PD plan availability in 2026, including by firm and rurality. KFF uses the term “plan terminations” to apply to all county-level plan offerings that are no longer available in 2026. Those include plans that did not have their contract renewed and other types of situations, such as service area reductions where a subset of the plan’s 2025 enrollees will no longer have the option of continuing to receive coverage through the same plan. Medicare Advantage plans without prescription drug coverage, Special Needs Plans, and employer- and union-sponsored plans are excluded from this analysis. Cost plans, PACE plans, HCPPS, and MMPs are also excluded from this analysis.
Data on Medicare Advantage plan terminations, enrollment, and availability were collected from a set of data files released by the Centers for Medicare & Medicaid Services (CMS): 2025 and 2026 Medicare Advantage plan landscape files, released each fall prior to the annual enrollment period and the Medicare Advantage contract/plan/state/county level enrollment file for February 2025, June 2025, and February 2026.
Enrollment data is only provided for plan-county combinations that have at least 11 beneficiaries; thus, this analysis excludes enrollees who reside in a county where county-wide plan enrollment does not meet this threshold.
This analysis determines urban and rural analysis based on the 2024 Urban Influence Codes (UIC) published by the U.S. Department of Agriculture (USDA) Economic Research Service. See Methods of KFF, “Key Facts About Medicare Beneficiaries in Rural Areas” (June 2025) for more details. Connecticut is excluded from the analysis by rurality because of differences in FIPS codes in the CMS Medicare Advantage data and the USDA 2024 UIC.
This work was supported in part by Arnold Ventures and AARP. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.