Insurance Coverage of OTC Oral Contraceptives: Lessons from the Field
Key Takeaways
In July 2023, the U.S. Food and Drug Administration (FDA) approved the first over-the-counter (OTC) daily oral contraceptive pill, Opill. Despite years of OTC access to contraception such as Plan B and condoms, there has been little research on how private insurance plans or Medicaid programs cover non-prescribed OTC contraception and whether they do so without cost-sharing. Currently, federal law requires most private health insurance plans and Medicaid expansion programs to cover, without cost-sharing, the full range of FDA-approved contraceptive methods. Health plans usually require a prescription to indicate medical necessity and trigger coverage, including for methods that do not require a prescription to purchase. Traditional Medicaid programs are required to cover family planning services without cost-sharing, though states have flexibility in which contraceptive methods they cover.
Earlier this year, the Biden administration issued an executive order directing the U.S. Departments of Labor, Health and Human Services, and Treasury to consider new actions to improve access to affordable contraception, which includes promoting increased access to and coverage of OTC contraception at no costs to consumers. Currently, the guidance from these three agencies “encourages” private plans to cover OTC contraceptives without cost-sharing but does not require it.
Six states, however, have passed laws requiring state-regulated health plans to cover, without cost-sharing, certain OTC contraception without a prescription and seven states use state-only funds to provide this coverage for Medicaid enrollees. Twenty-seven states and D.C. have expanded pharmacist prescribing of at least some contraceptive methods, which could facilitate coverage of OTC methods but few pharmacists have training or time, and those that do, are not compensated for counseling patients.
This report is based on 35 structured interviews conducted from January to August 2023, with nearly 80 experts and key players such as pharmacists, health plans, and state Medicaid officials involved in the coverage and provision of OTC contraception in seven states with one or more of these coverage approaches (IL, NJ, NM, NY, OR, UT, and WA). It discusses the challenges and opportunities identified by the interviewees that they have experienced regarding coverage under private health insurance and Medicaid and reviews potential options for operationalizing insurance coverage of non-prescribed OTC contraception such as Opill. These experiences are also informed by recent policies that required plans to pay for OTC COVID tests and more recently by interest in expanding access to Narcan, now available without a prescription.
With the imminent availability of Opill and the possibility of a new OTC oral contraceptive pill in the near future, the issue of coverage has been raised by many stakeholders. While some states have moved forward with coverage requirements for OTC contraceptives, operational issues and concerns such as pharmacy and pharmacists’ capacity to submit claims for OTC products, a lack of uniformity and oversight of health plans’ billing protocols, and low awareness of these policies remain as implementation challenges. While required coverage of OTC contraception without a prescription and without patient cost-sharing would increase access, it could also create a precedent for coverage of other OTC treatments, raising issues of cost for insurers and state Medicaid programs.
Key Takeaways
- Across the spectrum of stakeholders interviewed, there is interest and engagement about the potential of an over-the-counter (OTC) contraceptive pill to broaden access to contraceptive options, but many raised concerns about challenges related to affordability, implementation, and coverage options based on experiences in several states.
- In states where OTC methods are currently covered without a prescription by Medicaid or private plans, consumers generally need to obtain OTC contraception at the pharmacy counter, where they can show evidence of coverage and get their pharmacy claim processed. In some private health plans, consumers can pay for OTC contraception up front and then seek reimbursement from the plan, but that could be financially and administratively burdensome for consumers and is rarely used.
- Interviewees indicated that there has been little specific outreach about this covered benefit to pharmacies in states where OTC contraception, such as emergency contraception and condoms, is covered without a prescription in private insurance or for Medicaid enrollees. Health plan interviewees reported that they receive few claims for non-prescribed OTC contraception, which could be due to low awareness of the benefit and how to bill for it. Few plans provide information about the benefit in their enrollee-facing information.
- State Medicaid programs need to submit a State Plan Amendment (SPA) to the Centers for Medicare & Medicaid Services (CMS) to cover OTC drugs and products. After obtaining this authorization, states can determine which OTC drugs and products their Medicaid programs will cover. However, federal Medicaid law requires a prescription to cover all drugs, even those that are available without a prescription. A few states have chosen to use their own funds, without federal matching dollars, to pay for OTC contraceptives for Medicaid enrollees without a prescription. Federal funds will remain unavailable to cover OTC drugs without a prescription unless Congress amends the federal Medicaid law.
- In states where coverage for OTC contraception is provided without a prescription, interviewees noted that billing protocols for OTC contraception vary widely by health insurance plan and even within state Medicaid programs, leading to confusion for some pharmacists. States/state agencies do not usually determine the billing mechanism to be used.
- Some interviewees raised the importance of addressing quantity limits for OTC contraception. Interviewed health plans and a national PBM suggested that quantity limits have the potential to control fraud, waste, and abuse, which they cited as leading to higher costs for insurers.
- There has been limited communication about billing for non-prescribed OTC contraception between pharmacies, PBMs, health plans, and state insurance departments. Many interviewees from these sectors expressed that the mechanics of these state laws and how to operationalize them are unclear.
- While several interviewees expressed confidence that their current billing process for other non-prescribed OTC contraception can easily accommodate, most state-level discussions on insurance coverage of this product are in the preliminary stages.
- In some states, pharmacist prescribing plays an integral intermediary role in access to contraception where a prescription is required for coverage by removing the need to obtain a prescription from a physician or other prescriber. However, challenges and shortcomings with this approach persist, such as pharmacist time constraints, training requirements, and low or no payment from health plans for pharmacists’ services.
- When interviewees were asked what general suggestions they had for how to best implement coverage for Opill without a prescription, many stressed the importance of having a standardized billing process to help facilitate the transition to covering OTC contraception such as without a prescription as well as the role of clearer federal guidance regarding what plans are required to cover.
- While state actions to increase access to non-prescribed OTC contraception without cost-sharing can be meaningful for people with private insurance, the reach of these actions is limited, in large part because the majority of those with private health insurance are enrolled in self-funded employer plans, which are not subject to state insurance requirements.
- The extent to which OTC contraceptive pills can broaden the availability of effective contraceptives to those who seek them will depend on many factors including state and federal policies, pharmacy engagement, pharmacy stocking and signage, religious refusals, affordability, and insurance coverage, as well as public awareness and education.
This work was supported by Arnold Ventures. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.
Report
This report is based on 35 structured interviews conducted from January to August 2023, with nearly 80 experts and key players such as pharmacists, health plans, and state Medicaid officials involved in the coverage and provision of OTC contraception in seven states with one or more of these coverage approaches (IL, NJ, NM, NY, OR, UT, and WA). It discusses the challenges and opportunities identified by the interviewees that they have experienced regarding coverage under private health insurance and Medicaid and reviews potential options for operationalizing insurance coverage of non-prescribed OTC contraception such as Opill. These experiences are also informed by recent policies that required plans to pay for OTC COVID tests and more recently by interest in expanding access to Narcan, now available without a prescription:
Background
Oral contraceptives are the most commonly used method of reversible contraception in the U.S. Oral contraceptive pills were first approved for prescription use by the U.S. Food and Drug Administration (FDA) in 1960. In July 2023, the FDA approved Opill, the first daily oral contraceptive pill available over-the-counter (OTC) without a clinician or provider’s prescription. It is expected to be available in early 2024.
OTC status is an FDA designation meaning that a drug or product is available without needing a prescription from a health care provider. The ability to access oral contraceptives without a prescription from a clinician can save time spent on travel, at a clinician’s office, and off work or school. Studies suggest that OTC access to oral contraceptives would increase the use of contraception, facilitate continuity of use, and reduce the risk of unintended pregnancy.
Most drugs available OTC today were initially labeled and approved as prescription-only medications. Other commonly known OTC drugs that were previously only available with a prescription include levonorgestrel emergency contraceptive pills; smoking cessation aids such as Nicorette; several brands of allergy medications such as Claritin D, Allegra, and Zyrtec; heartburn medication such as Prilosec; and Narcan nasal spray, the most recent product to switch from prescription to OTC status before Opill, which is used to treat opioid overdoses.
Levonorgestrel emergency contraceptive (EC) pills, marketed as Plan B One-Step and other brands, are a form of backup birth control intended to be taken within three days after unprotected sex or contraceptive failure to prevent pregnancy. They were the first contraceptive pills to have switched from prescription to OTC status, in 2006. While EC pills were originally FDA-approved for OTC use by people ages 17 and older, the FDA removed the point-of-sale age requirement in 2014. Unlike daily oral contraceptives, EC pills are not intended for daily use.
Other contraceptive products that are available over the counter without a prescription include external condoms, spermicides, and contraceptive sponges. These non-hormonal contraceptive methods are less effective than oral contraception at preventing pregnancy. The FDA’s approval of Opill will make it the most effective method of contraception available over the counter intended for regular use.
FDA Prescription-to-OTC Switch Process
The FDA requires applicants to conduct extensive testing, often over several years, of products and labeling before they can be approved for OTC use. Opill’s application was approved through the “RX-to-OTC switch” pathway, which is available for drugs that have already been approved by the FDA, like Opill, by submitting a New Drug Application (NDA). Medications may be eligible for OTC status if the FDA determines that they can be used appropriately by consumers for self-diagnosed conditions; they do not require a clinician for safe and effective use; and they have a low potential for misuse and abuse. Applicants typically must conduct studies to assess whether consumers are able to comprehend the product’s labeling and use the product safely and appropriately without the supervision of a clinician.
Opill, a daily progestin-only oral contraceptive, was initially approved for prescription use by the FDA in 1973 (manufactured by Pfizer under the brand name Ovrette). HRA Pharma acquired the rights to Opill in 2015 and began the research process required to apply to the FDA for an Rx-to-OTC switch, including label comprehension studies and self-selection actual use trials. HRA Pharma, subsequently acquired by Perrigo in April 2022, submitted its application to move Opill over the counter in July 2022.
In addition to review by the FDA itself, the process often also entails review by independent advisory committees comprised of scientific experts and consumer representatives. In May 2023, after reviewing evidence and hearing public testimony, FDA advisory committees unanimously voted in favor of the FDA moving Opill over the counter. Although the FDA is not required to follow advisory committees’ recommendations, they typically do. The FDA approved Opill for OTC use on July 13, 2023, without age restrictions, and the manufacturer reports that it will be available in stores and online in early 2024. Consumers will be able to purchase a pack of one, two, three, or six months’ worth of pills, depending on availability.
Although it is farther behind in the process, another pharmaceutical company, Cadence, is working toward FDA approval of an OTC version of its combined (progestin and estrogen) oral contraceptive pill, Zena. Cadence is reported to be considering a different FDA pathway to OTC status that has been proposed by the Biden administration.
Federal and State Laws on Coverage of OTC Contraception
MEDICAID
Medicaid is the joint federal/state health insurance program for people with low incomes. Federal law requires state Medicaid programs to cover family planning services and supplies without cost-sharing, but they are not required to cover OTC products (with a few exceptions) or to cover OTC contraception without a prescription. States cannot obtain federal matching dollars for covering a medication without a prescription, but they may opt to use state-only funds to cover OTC drug benefits without a prescription.
Seven states (CA (effective in 2024), IL, MD, MI, NJ, NY, and WA) cover, with state funds, at least some OTC contraceptive methods without a prescription for Medicaid enrollees. Note that New Jersey Medicaid’s coverage of non-prescribed OTC contraception applies to fee-for-service (FFS) enrollees only, which represent 2% of enrollees in the state. With the exception of California, coverage is specific to emergency contraception or condoms in these states, so these programs may not include Opill without a change in policy. (See Over-the-Counter Oral Contraceptive Pills for the coverage details of these and other state Medicaid programs.)
PRIVATE INSURANCE
The Affordable Care Act (ACA) requires most private health plans (individual, small group, and large group) to cover recommended preventive services without cost-sharing. The ACA tasks the Health Resources and Services Administration (HRSA) with recommending coverage requirements for a range of preventive services for women, which initially consisted of eight recommendations that included contraceptive services and supplies, identified by a committee of the Institute of Medicine in 2011. The initial HRSA 2011 recommendation included the language “as prescribed” in reference to the coverage requirement for contraception. Today, the Women’s Preventive Services Initiative (WPSI) is the expert body currently commissioned by HRSA to issue and update preventive clinical recommendations for women. WPSI updated its contraceptive coverage recommendation in 2021 and it does not include a prescription requirement for coverage of contraception. HRSA dropped the prescription requirement in its language when the preventive services guidelines were updated and posted. Currently, “as prescribed” is only referenced in the U.S. Departments of Labor, HHS, and Treasury (“tri-agency”) federal FAQs.
In addition to the federal ACA requirements, six states (CA (effective in 2024), MD, NJ, NM, NY, and WA) have laws or regulations requiring state-regulated private health insurance plans (individual and fully-insured employer-sponsored plans) to cover, without cost-sharing, OTC contraception without a prescription. Nationally, 65% of workers with employer-sponsored insurance are enrolled in a self-funded plan; therefore, the majority of people with employer-sponsored insurance who live in states that require coverage of OTC contraception without a prescription are not guaranteed this coverage. While the language of New York’s law is specific to emergency contraception, the other state laws apply to a broad range of contraception. The language of these laws, with the exception of New York’s, is broad enough to include a daily oral contraceptive pill such as Opill without a change in policy. (See Over-the-Counter Oral Contraceptive Pills for the details of these state laws and others that require coverage without a prescription.)
Illinois and Oregon require private health plans to cover OTC contraception; however, while these laws do not state that a prescription is required in order for it to be covered by insurance, the laws also do not explicitly stipulate that plans must cover them without a prescription. While federal law applies to all private plans, state law applies to only individual plans and fully-insured employer plans.
Pharmacist Prescribing
In states where coverage of OTC contraception without a prescription is not possible, not required, or where billing requirements are unclear, pharmacist prescribing is a relatively novel approach that can help expand access and bridge that gap. States, rather than the federal government, establish standards that determine the scope of practice and services that may be provided by different types of licensed health practitioners practicing in that state. Twenty-seven states and D.C., including all seven in this study, have passed laws permitting pharmacists to prescribe certain methods of contraception, including OTC contraception, or to dispense it pursuant to a standing order or collaborative practice agreement (discussed in more detail later in the report). In these states, consumers can obtain a prescription for contraception, including OTC methods, at a participating pharmacy without the need for a visit to a clinic or doctor’s office. Expanded scope of practice can help facilitate private insurance and Medicaid coverage of these products and research demonstrates that it can increase access to contraception and help prevent unintended pregnancies.
However, many states do not have laws that permit pharmacist prescribing, and in the states that do, there are many limitations with pharmacy prescribing, including low pharmacy and pharmacist take-up, pharmacist training requirements, and patient counseling and evaluation requirements, which are typically provided without payment from health plans. Although the FDA did not place an age restriction on access to Opill, some states place age restrictions on pharmacists prescribing contraception to people under the age of 18. Additionally, a consumer who wants pharmacist-prescribed contraception is not able to do so outside of pharmacy hours or when the pharmacist is unavailable to provide this service. Because pharmacist prescribing is still a clinical model and patients in most states must disclose personal health data during the consultation, this approach may present barriers for people who do not wish to interact with the health care system.
Lessons Learned from Key Informant Interviews
To identify experiences and approaches for plans to cover OTC contraceptives, key players in states that require or allow for private insurance or Medicaid coverage of OTC contraception without a prescription, including some that allow pharmacists to prescribe contraception, were interviewed. After identifying states that implemented at least one of these insurance coverage or pharmacist prescribing policies, while also factoring in interviewing logistics, seven states were included in the project: Illinois, New Jersey, New Mexico, New York, Oregon, Utah, and Washington.
From January 2023 through August 2023, KFF staff completed 35 interviews with nearly 80 experts from state Medicaid programs, the Centers for Medicare and Medicaid Services (CMS), state insurance departments, private health insurance plans, Medicaid managed care organizations (MCOs), chain pharmacies, independent pharmacies, a pharmacy benefit manager (PBM), and trade associations, as well as pharmacists and contraceptive access advocates.
The information collected from the interviews highlights state policies and strategies that have been used to operationalize Medicaid and private insurance coverage of OTC contraception such as Opill, as well as identifies the approaches and challenges to help inform state and federal efforts to address coverage for Opill. To preserve interviewees’ confidentiality, individual names, affiliations, and states are not provided. (See the Methods section for more details.)
State-Funded Coverage of OTC Contraceptives Without a Prescription for Medicaid Enrollees
The specifics of coverage of family planning services and supplies vary across Medicaid programs. Four of the seven states that participated in the study confirmed they pay for non-prescribed OTC contraception for Medicaid enrollees (IL, NJ, NY, and WA). (As mentioned above, New Jersey’s coverage applies only to FFS enrollees.) Because federal Medicaid law requires a prescription to trigger coverage of drugs and products, these coverage policies are entirely state-funded. New Jersey and New York only cover non-prescribed OTC emergency contraceptive pills while Illinois and Washington cover non-prescribed OTC emergency contraceptive pills and other OTC contraceptives such as condoms.
In states that cover non-prescribed OTC contraception for Medicaid enrollees using state-only funds, there are generally two mechanisms pharmacists use to bill for them and get reimbursed from the state.
- Universal National Provider Identifier (NPI) number: NPIs are unique, 10-digit numbers issued by CMS that identify individual providers or health care entities, such as pharmacies, which are used for billing purposes. Across the Medicaid programs that cover non-prescribed OTC contraception using state funds, this is the most common mechanism pharmacists use to submit a claim without enrollee cost-sharing. According to interviewees, this process involves the pharmacist entering a dummy, blank, or state-specific universal NPI in the prescriber field. It is used by three state Medicaid programs and some MCOs in the study.
- Pharmacy NPI: In this process, the pharmacist enters the pharmacy’s NPI in the prescriber field. It is used by one state Medicaid program and several Medicaid MCOs, and it was formerly used by one state Medicaid program that used to cover non-prescribed OTC emergency contraception but no longer does.
While each of the four state Medicaid programs published billing guidance and/or resources on the coverage benefit, pharmacist and consumer awareness of the benefit is generally low. Pharmacist training and billing guidance vary across states. Some state Medicaid programs are unaware of any issues with their billing mechanism for non-prescribed OTC emergency contraception, but others have cited that some pharmacists are unaware or unsure of the billing process for contraception despite billing guidance provided through pharmacy handbooks, newsletters, and email or fax blasts. As one pharmacist pointed out, part of the confusion stems from the fact that they are dealing with many plans and there is no standardized method of communicating this information or a single source for a pharmacist to find this information.
“The consumer will attempt to obtain [OTC] contraception at a pharmacy, and the pharmacist turns them away because they’re unable. They either think that they cannot dispense it without a prescription, or they feel Medicaid requires a prescription [for coverage]. Some of them just don’t know how to do it.”
–State Medicaid official
Billing mechanisms can also differ between traditional FFS Medicaid and MCOs. For example, in one state Medicaid program, pharmacists billing for non-prescribed OTC contraception are instructed to use a dummy NPI number in the prescriber field for their FFS program, the pharmacy’s NPI for one MCO, and dummy NPIs for other MCOs. These different billing mechanisms can lead to pharmacist confusion and additional administrative burdens.
Most state Medicaid programs studied indicated they have not recently reviewed claims data for non-prescribed OTC contraception or do not know how often claims are submitted for it. State Medicaid programs do not directly advertise the benefits to beneficiaries outside of the member handbook. Several pharmacists corroborated that awareness of this benefit among pharmacists and consumers is low.
One of the three states that does not currently cover OTC contraceptives for Medicaid enrollees discontinued funding for non-prescribed contraception in 2016. The program was reportedly discontinued due to lack of utilization, and one pharmacy owner mentioned the process was tedious for both pharmacists and patients. The program was administered through the state health department and was paid for using state funds and involved pharmacists billing Medicaid using the pharmacy’s NPI. State Medicaid officials have not discussed restarting the program.
In general, Medicaid fee-for-service enrollees cannot file a claim if they did not obtain point-of-sale coverage for non-prescribed OTC emergency contraception. However, an FQHC-based Medicaid MCO in one state that covers non-prescribed OTC contraception reported that enrollees can purchase the OTC contraceptive out-of-pocket and then submit a receipt to the MCO for reimbursement.
Private Insurance Coverage of OTC Contraceptives Without a Prescription
The project included four of the states that currently require state-regulated private health insurance plans to cover at least some methods of OTC contraception without a prescription (NJ, NM, NY, and WA). Oregon and Illinois, also included in the study, have laws requiring coverage of OTC contraception, but coverage without a prescription is not explicit and interpretation of the laws varies. Nonetheless, some health plans in these states do cover OTC contraception without a prescription.
In states where private insurers are required to cover OTC contraception without a prescription, there are generally two pathways for consumers to receive coverage.
- Obtaining the OTC contraceptive product at the pharmacy counter: The private plans interviewed said that consumers can bring their OTC products to the pharmacy counter and the pharmacist would then submit a claim to the plan using their own national provider identifier (NPI) number, pharmacy NPI number, or dummy NPI depending on the plan and state protocol. Enrollees usually must purchase it from an in-network pharmacy in order for it to be covered. This approach is similar to how pharmacists billed for at-home COVID-19 tests in many situations.
- Purchasing the OTC contraceptive product at the cashier outside of the pharmacy and getting reimbursed: One private plan mentioned that consumers can also purchase the product outside of a pharmacy setting and then submit a claim with the receipt to their insurance company for reimbursement, similar to the reimbursement mechanism that was used for some at-home COVID-19 tests. While this option generally expands access to coverage in certain situations, such as when a pharmacy inside a store is closed, it could pose financial and logistical barriers by requiring enrollees to pay the cost upfront and navigate the plan’s reimbursement process, which may require the enrollees to include a prescription number.
Some health plans voluntarily provide coverage for OTC contraceptives without a prescription in states that do not mandate coverage. The state insurance department for one state without an OTC contraceptive coverage requirement noted there are a few private health plans that voluntarily cover non-prescribed OTC contraception. Corroborating this notion, one private health insurance carrier that operates in a state where coverage of non-prescribed OTC contraceptives is required also voluntarily extends the same coverage in the three other states where the carrier operates, which do not have this requirement, as long as the drug or product is included on the drug formulary. A formulary is the list of drugs covered by a specific health insurance plan. If a drug is not listed on a plan’s formulary, it is likely not covered by the plan and the enrollee would need to pay out-of-pocket for it.
Despite state coverage requirements, the health plans in the study indicated that claims for non-prescribed OTC contraception are rare, suggesting a lack of awareness of this covered benefit among pharmacists and health plan enrollees. Indeed, two independent pharmacy owners in a state with this coverage requirement indicated that they were unaware of or unfamiliar with this private insurance policy. Representatives from another state’s insurance department stated that it is the health plans’ responsibility to inform their members of any new benefits to which they are entitled. Some state insurance departments indicated that health plans are required to notify their enrollees that this coverage is available or include the information in their membership materials, though the details of how and where that information is communicated is largely up to the plan.
Some contraceptive access advocates cited concerns that raising consumer awareness and expectations about this covered benefit without better operational structures could generate confusion among enrollees with employer-sponsored insurance. Coverage requirements for OTC contraception only apply to state-regulated private health plans (individual and fully-insured employer-sponsored plans). However, the majority of covered workers are enrolled in self-funded plans, to which state coverage requirements do not apply. Two national advocates, who also work with state-level advocates, noted that broadly promoting coverage awareness can misguide and mislead many consumers who are in self-funded plans into thinking they are guaranteed this coverage because self-funded and fully-insured plans typically look the same to the enrollee so they may not know they are in a self-funded plan. Some states, such as Colorado, Maryland, and New Jersey, require insurance cards to indicate whether the plan is regulated by the state, but most states do not.
Even with state laws requiring coverage, insurance commissioners have not issued guidance on how private health plans should process claims for non-prescribed OTC contraception. Representatives from one state’s department of insurance confirmed that billing mechanisms for OTC contraceptives vary across health plans, with some plans having their pharmacy networks set up to detect the products as zero cost-sharing and others requiring enrollees to submit claims to the insurer for reimbursement. The various ways of billing private insurance can consequently lead to pharmacist confusion.
“Working in the community setting, we are incredibly busy. Having to look up a separate way to do the same thing takes a lot of time.”
– Pharmacist from a regional retail pharmacy chain
State compliance and enforcement efforts vary. One state department of insurance addresses compliance on a case-by-case basis, primarily relying on complaints from consumers or providers to trigger a compliance review. This state insurance department noted that while there was some confusion among insurers about the mechanics of the coverage law when it took effect, they have received very few complaints. Another state insurance department takes a different approach to ensuring compliance with state coverage laws by conducting systematic reviews periodically, in addition to addressing individual complaints they receive from consumers and providers, which they note are rare. However, a pharmacist and a representative from a pharmacy chain noted that even with state mandates, getting payers to operationalize coverage is difficult.
“Even though we’ll have a bill that says it’s supposed to be covered, the plans don’t necessarily start covering it. We’ve seen that with the extended supply [of hormonal contraception].”
– Clinical pharmacist and national pharmacist educator
Pharmacy Benefit Managers
Pharmacy benefit managers (PBMs) are third-party administrators contracted by health plans that perform a variety of financial and clinical services for both private insurance plans and Medicaid programs, including negotiating discounts on medications for health plans and employers, establishing prescription drug formularies, and adjudicating pharmacy claims. PBMs play a role in the provision of OTC contraception by determining what tier of a drug formulary it is placed on, which impacts enrollee cost-sharing amounts; determining pharmacy reimbursement amounts, which impacts the willingness of a pharmacy to sell a particular drug or product; processing enrollee claims; and conducting pharmacy oversight of claims to ensure proper billing techniques.
Billing for OTC contraceptives varies across PBMs. PBMs help establish standard formulary processes while ensuring the insurers and pharmacies they contract with abide by state and federal policies. One national PBM interviewee noted that coverage for OTC drugs typically requires a prescription in order for the claim to adjudicate through their pharmacy system. In states with laws that require private insurers to cover non-prescribed OTC contraceptives, a health plan interviewee explained that their members need to mail in receipts to be reimbursed for the out-of-pocket purchase. However, as other interviewees shared, there have been few claims for these non-prescribed OTC contraceptives.
Both payers/PBMs and pharmacies experience challenges with claims submission due to historical systems and processes that were not built for widespread coverage of non-prescription products. For example, one health plan explained that most chain pharmacy systems require an NPI to submit a claim and that most payer claim systems also require an NPI to adjudicate the claim. While the payer could in theory opt not to require an NPI, the pharmacy system would still require it. As a result of these collective limitations, workarounds at the pharmacy and health plan level may be necessary when billing for non-prescribed OTC contraception, creating additional burden on community pharmacy staff.
Some pharmacists are concerned that billing health plans for non-prescribed OTC contraceptives may trigger an audit from PBMs. Some pharmacists said they worry about being audited by PBMs, as these billing mechanisms appear to contradict the training they have received about billing thoroughly and carefully to avoid auditing. One pharmacy chain representative shared this concern as well since failed audits can result in penalties and loss of compensation for pharmacies. However, representatives from a private health insurance plan and a Medicaid managed care organization stated that their PBM excluded OTC categories from being auditable.
Some pharmacists also reported being concerned about potential legal liability for billing without a prescription. When a pharmacist uses their NPI, the pharmacy’s NPI, or a dummy NPI to bill for an OTC drug, it can give the impression that a prescription has been written for it. Some pharmacists in these states raised concerns about potential legal liability in the event that a patient claims a pharmacist made a mistake because no prescription was actually written.
Clear communication between PBMs, health plans, and pharmacies can help facilitate coverage for OTC contraceptives. The health plan above noted that their PBM notified network pharmacies that coverage for OTC contraceptives is a new capability that pharmacies in the state must comply with. The PBM also sent out billing guidance on how to bill for OTC contraceptives. Clear communication can help reduce confusion about different billing mechanisms and alleviate concerns about being audited.
The Role of Pharmacist Prescribing
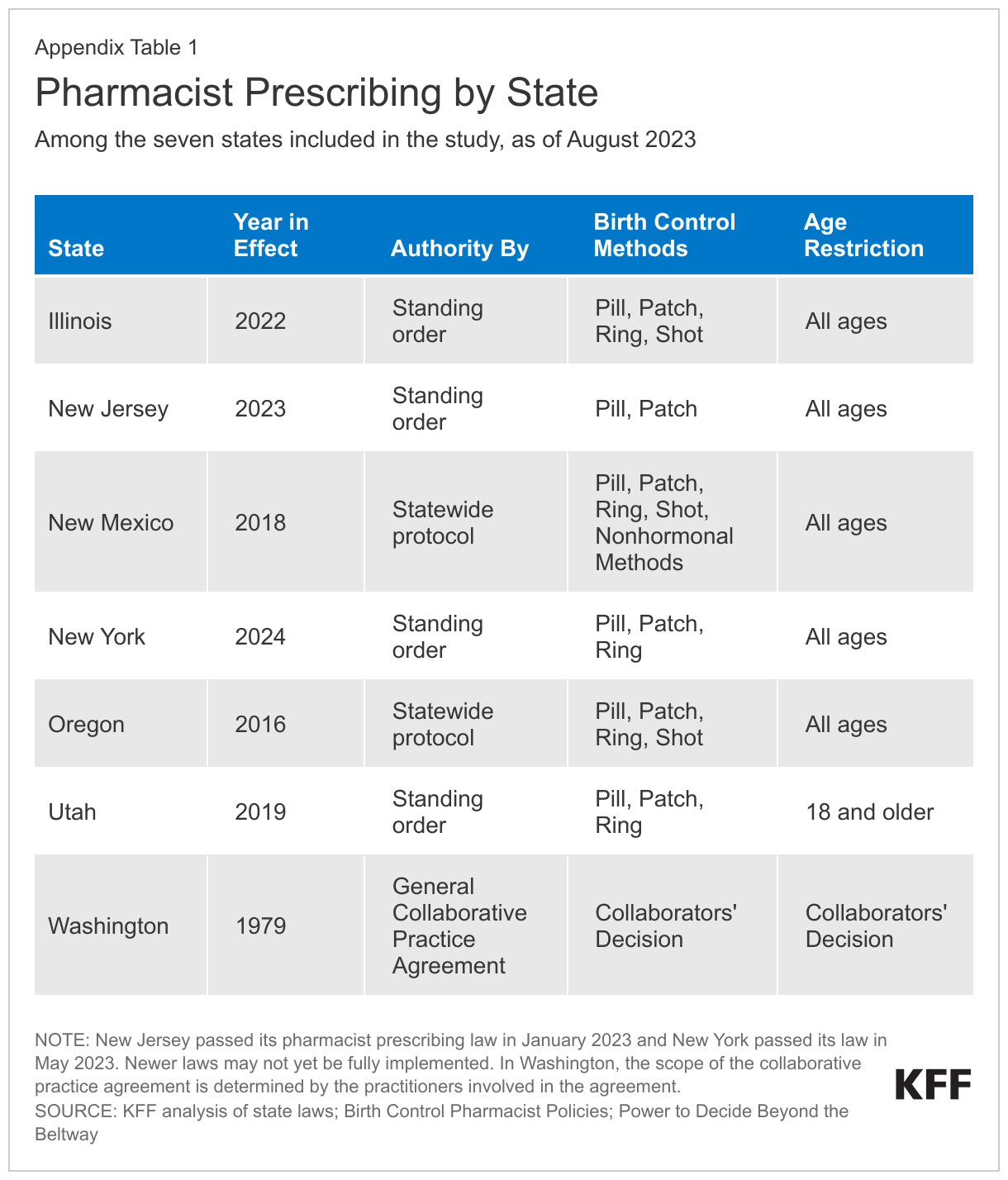
Four (NM, OR, UT, and WA) of the seven states in the study currently permit pharmacists to prescribe contraception; two states (IL and NJ) recently passed pharmacist prescribing laws and are still in the process of implementing them; and one state (NY) passed a pharmacist prescribing law in May 2023 that will take effect at the end of 2024. (See Appendix Table 1 for details on each state’s law.)
Pharmacist prescribing plays a key role in coverage of contraception without cost-sharing in some states. Many interviewees stated that pharmacist prescriptive authority helps expand access to reproductive health care. Prescribing authority varies by state and is typically issued through a standing order, collaborative practice agreement, statewide protocol, or full prescriptive authority (Table 1). Where this is an option, pharmacist prescribing helps reduce barriers to accessing contraception by removing the need to visit a clinician to obtain a prescription, and for people without insurance, it can be less expensive than getting a prescription from a clinician.
Uptake of prescriptive authority has been low among pharmacists in some states, in part because of the training requirements. In many states, including one in the study, pharmacists graduating from the state university’s pharmacy school earn prescriptive authority upon graduation. However, prescriptive authority for hormonal contraception usually involves continuing education which takes time and resources outside the standard responsibilities of a pharmacist. An executive pharmacy director for a Medicaid managed care organization stated that even though their state allows pharmacists to prescribe hormonal contraceptives via a standing order, it is uncommon for pharmacists to use this option.
While pharmacist prescribing can promote access to contraception, shortcomings of this approach may persist for several reasons. The most commonly cited reasons include:
- Lack of or limited compensation for consultation is a barrier to participation in pharmacist prescribing. Most pharmacists and independent community pharmacy leadership mentioned that pharmacists in their state are not incentivized to prescribe or dispense hormonal contraception due to the lack of payment from health plans for the service. Several pharmacy interviewees cited challenges getting private insurance to compensate pharmacists for this service. This challenge is in part due to the fact that insurance companies typically do not recognize pharmacists as providers and adhere to credentialing and enrollment processes that are typically reserved for traditional health care providers vs. pharmacists. While pharmacies are reimbursed for the cost of the drug, pharmacist services for patient counseling and evaluation, prescribing, and other administrative services associated with providing this service are often not compensated by payers or are paid at a low rate. Representatives from a pharmacy chain said that when pharmacy services are paid, whether the payment goes to the pharmacy or to the pharmacist depends on the arrangement pharmacists have with their employer. One pharmacist noted that payment typically goes to the pharmacy rather than to the pharmacist. Lack of or low compensation can sometimes lead pharmacies to charge patients consultation fees in lieu of payer compensation, which is at odds with the goal of the ACA and Medicaid to eliminate financial barriers to contraceptive services for patients. One pharmacy interviewee reported that they do not prescribe many OTC products for this reason:
“Economically, it doesn’t work out because [patients are] paying $80 [in consultation fees] to get a product that they would pay $50 for [without insurance coverage].”
–Independent pharmacy owner
- Lack of prescriptive authority where it is permitted: As mentioned above, it may be difficult to locate pharmacies with at least one prescribing pharmacist, even in states with many prescribing pharmacists. One interviewee said their data showed pharmacist prescribing is more prevalent in urban areas than rural areas. Since prescriptive ability is optional to most pharmacists, consumers may encounter inconsistent availability of pharmacists willing or able to prescribe hormonal contraception. This gap is particularly prevalent in rural areas that already face declining access to pharmacies. However, other studies suggest that pharmacists in rural communities are just as likely to prescribe contraceptives as those in urban communities.
- Lack of time: The role and scope of pharmacists in health care has rapidly expanded over the years, with a growing number of pharmacists dispensing medication, administering vaccines, and counseling patients, among other services. Several interviewees expressed that prescribing contraceptives and adjudicating coverage for OTC contraceptives adds another layer of responsibility for pharmacists who are already overstretched. The process often involves a pharmacist consultation, drug utilization review, blood pressure check, and patient education, all of which take time to complete and can interrupt workflow. For these reasons, even if there are pharmacists who can prescribe at a pharmacy, patients may have to wait to obtain a prescription for contraception. Additionally, pharmacists are generally required to enroll as a Medicaid provider in order for Medicaid to compensate for their services, which can be a burdensome process depending on the state.
When asked what suggestions they would have for other states considering implementing pharmacist prescribing, some pharmacy interviewees noted the importance of having input from pharmacies and pharmacists when developing and implementing legislation and regulations.
Considerations for Covering OTC Oral Contraceptive Pills
Discussions about coverage of an OTC daily oral contraceptive pill without a prescription have barely begun at the state level. The FDA approved the daily oral contraceptive pill, Opill, for OTC use at the end of the interview fielding period. Interviewees were asked what steps, if any, they have taken in anticipation of or in reaction to FDA approval of an OTC daily oral contraceptive pill, including agency, health plan, or pharmacy discussions, or any concrete plans. For the most part, these discussions have not yet begun or are in their very early phases. Among those that have begun planning, several interviewees expressed confidence that their current system for billing for other OTC contraception such as Plan B without a prescription is a feasible way to bill for an OTC daily oral contraceptive and that they do not believe any additional steps would need to be taken to change billing and claims systems. Private health insurance plans and a state insurance department interviewed indicated that while there have been some discussions about coverage for an OTC oral contraceptive without a prescription, they have not yet taken any concrete actions. Interviewees, including a state department of insurance, mentioned the importance of involving health care providers with clinical expertise, especially pharmacists, in efforts to expand coverage. Since their interview, one state reported it has begun a provider outreach campaign to understand barriers at the pharmacy level, particularly those in smaller towns, rural areas, and tribal communities, and is developing a communications campaign to help ensure any barriers to OTC uptake are addressed by the time Opill comes to market.
The importance of clear federal guidance about any plan coverage requirements for OTC contraception was cited by many interviewees when asked for suggestions on how to best implement coverage approaches without a prescription. One interviewee from a state insurance department noted that their state relies heavily on federal action for implementing changes for new drug coverage. Representatives for a national PBM echoed that federal guidance would be helpful, and another representative for a national health plan trade group emphasized that communication related to coverage policies from the federal government needs to be clear and cannot be confusing for patients, providers, and plans. One pharmacy interviewee commented that direction from the federal government on coverage without a prescription is mutually beneficial all around. Contraceptive access advocates noted that lack of consistency in the language between what the ACA regulations require plans to cover and the tri-agency FAQs has led to differing interpretations of policy and lack of clarity on what can be enforced.
One pharmacy chain indicated that it is currently working with the U.S. Department of Health and Human Services (HHS) to try to create a payment pathway at the federal level for pharmacist prescribing, a fallback approach to coverage where coverage without a prescription is not an option.
“Anything that’s dictated nationally versus state-by-state is always a win for the patient, and quite frankly, it’s always a win for the pharmacy.”
–Representative from a pharmacy chain
CMS has begun preparations to accommodate coverage of OTC contraception in Medicaid, but some form of a prescription is still required to obtain federal matching funds. CMS, the federal agency that administers Medicaid in partnership with state governments/Medicaid agencies, has issued guidance on how states can leverage expanded scope of pharmacist practice to cover OTC medications such as contraception. They suggest using broad and general language in their state plan regarding Medicaid coverage of OTC drugs. States submit state plan amendments (SPAs) to CMS when they plan to make changes in how they administer Medicaid program activities and are seeking federal approval to draw down federal funds or federal matching dollars for those services.
In response to the FDA approval of OTC Narcan nasal spray in March 2023, and in anticipation of the FDA approving a daily OTC oral contraceptive, CMS has encouraged state Medicaid programs to use broad implementation language in their state plan and list which therapeutic categories are covered on their state website or other public-facing documents to prevent the need for states to submit a new SPA each time their state wishes to cover an OTC drug. For example, CMS approved SPAs from Delaware, Montana, Florida, and Illinois indicating their intent to broadly cover OTC drugs. The billing mechanisms for covering these OTC products vary by state and still require the NPI field to be filled out. CMS leaders believe that this method can be leveraged by any state because even prior to Narcan’s OTC approval, all states had a protocol in place to allow pharmacists to dispense at least some drugs, for example, through a state health official’s standing order. Some state Medicaid programs also used this approach to cover at-home COVID-19 tests.
This approach is different than state Medicaid programs using state-only funds to cover OTC contraception without a clinician’s prescription. States only receive federal matching funds for OTC contraception if it is prescribed by an authorized prescriber.
Nearly all pharmacy interviewees recommended that states considering coverage for OTC contraception should include a billing structure that adequately compensates pharmacists for their services since pharmacies are not typically paid for submitting claims for non-prescribed products. A national PBM indicated that it is currently considering potential billing mechanisms for Opill and has begun partnering with its clients and pharmacies to effectuate this coverage. This PBM and a national PBM trade association also stressed that modernizing the pharmacy claims process and having a standardized billing process in place would facilitate the transition to covering OTC contraception such as Opill without a prescription.
Submitting Pharmacy Claims to Health Plans
There are several steps involved in a pharmacy submitting a claim to a health plan or PBM (see Appendix Table 2). While the processing of a pharmacy claim, from submission by the pharmacy to the return claim arriving back at the pharmacy, occurs, on average, within 3 to 4 seconds, it can take several minutes (or longer if there are problems) for the pharmacist or technician to enter all the billing information. Despite the time and resources expended to submit a claim, pharmacies are not typically paid for providing this service for non-prescribed over-the-counter medications.
Quantity limits are another consideration in coverage for OTC contraception without a prescription. Opill will be sold in packages of one-, two-, three-, and six-month supplies. Having an extended supply of oral contraception is associated with better adherence and lower rates of unintended pregnancy. Several states require plans to cover an extended supply of contraception, such as six or 12 months at a time. However, several interviewees, including some health plans and a national PBM, suggested that prohibitions on quantity limits could contribute to increased waste and costs.
“One of the big learnings we heard from our plans is that there needs to be some guardrails. If there are no quantity limits or other constraints, people could buy bulk male condoms, for example, and then resell them. It actually has happened.”
–Representative from a national health insurance trade association
While interviewees did not report these types of incidents with OTC emergency contraceptive pills, which are a closer parallel to Opill than condoms, this is an issue that at least some interviewees are thinking about in preparation for insurance coverage of Opill. On the other hand, some advocates are concerned that quantity limits can create access barriers for consumers in the event that their pack of pills gets damaged, lost, or left behind.
Stocking decisions and signage can increase opportunities for coverage. Although a few states, such as Washington and Massachusetts, require pharmacies to stock contraceptive medications such as Plan B, pharmacies in most other states will decide whether or not to carry Opill. Stocking also has implications for insurance coverage because most plans provide more coverage for drugs purchased at an in-network pharmacy than at an out-of-network pharmacy, and in some cases such as with health maintenance organizations (HMOs), drugs and services are not covered by out-of-network providers at all.
Pharmacies’ decisions on where to stock OTC contraceptives will also play a role in access, both for those with and without insurance. Some pharmacies decide to put OTC contraceptives behind the pharmacy counter or in a locked cabinet on the shelf due to shoplifting concerns. Some interviewees noted that this type of placement in a pharmacy may make it more difficult to access the contraceptives and that some consumers may have confidentiality concerns, particularly for young people and those living in smaller towns and rural areas. On the other hand, one pharmacist interviewee said that having consumers come to the pharmacy counter to obtain OTC contraception allows the pharmacist to help them understand if the product is the best option for them, understand how to use it, and assess if it is covered by their insurance.
“There is a perception that these medications, Plan B and Opill, are more likely at risk for shoplifting. So even if they do have them out at the OTC area, they could be placed behind the locked cabinet. Think about the barrier, right? We have to go and actually ask somebody to be able to have access to it.”
–Representative from a national pharmacy benefits manager
Posting signage about the availability of OTC contraception and whether it may be covered by insurance could help increase consumer awareness and uptake. An interviewee suggested that some pharmacies may be voluntarily posting signage on the shelf where emergency contraception is stocked to inform customers that if they take the product to the pharmacy counter, the pharmacist may be able to bill insurance for it, rather than the customer having to pay for it entirely out-of-pocket. A pharmacist interviewee noted that posting this type of signage may be more difficult for chain pharmacies because they typically require upper management approval for any content that is posted in their pharmacies.
Pharmacist religious refusals can also impact access to contraception in general. Multiple interviewees in the study cited religious beliefs and refusals as common barriers to accessing hormonal contraceptives through the pharmacy in at least some parts of the state, typically in rural areas and small towns. While some health plans and pharmacies try to find a second prescribing pharmacist at the pharmacy, these refusals and objections have reportedly led to delays in patient access. Even in states where a prescription is not needed for insurance coverage of OTC contraception, a pharmacist may still refuse to process the claim.
Outreach and education to pharmacies, pharmacists, and patients will be critical in fully realizing the potential of an OTC daily oral contraceptive, as discussed by several state Medicaid programs, private health plans, pharmacists, and advocates. The insights gleaned from these interviews suggest that there is a role for a variety of stakeholders including consumer advocates, health plans, state Medicaid programs, PBMs, pharmacies, and pharmacists when it comes to informing consumers of this new product and insurance coverage for it.
“If you keep [Opill] locked up, which I'm imagining will be in some of these retailers, and nobody speaks about it, I don't know how successful it will be. I hope that they're going to have a good campaign to help educate everybody about it.”
–Community pharmacist
Contraceptive access advocates expressed that both the availability of OTC contraceptives and having plan coverage of these products can especially benefit communities that have historically faced a myriad of barriers to accessing and affording contraception. They encourage policymakers at the state and federal level to evaluate all possible options for covering OTC contraceptives, with special attention to addressing the concerns facing communities without a pharmacy or people that experience other barriers to access.
“Affordability is an equity issue. We want to make sure [Opill] is not just going to people who have privilege or who could have gotten it through other mechanisms and that it’s really reaching the communities with systemic barriers in place due to inequities in our healthcare system and in our society.”
–Representative from national contraceptive access advocacy group
Discussion
State Policy Options
States have a variety of policy options if they seek to increase access to non-prescribed OTC contraception for individuals with private insurance. More states can continue passing laws requiring state-regulated health insurance plans to cover, without cost-sharing, OTC contraception without a prescription. Using broad language when developing laws and regulations, as some states have already done, can promote coverage of more drugs and products, such as Opill, emergency contraception, and non-hormonal methods such as condoms. Additionally, state laws requiring health plans to cover an extended supply of contraceptives, including those obtained over the counter without a prescription, could reduce burdens associated with having to order or go to the pharmacy every month. While these policies can increase opportunities for OTC contraceptive coverage for those who reside in those states, these policies only apply to those enrolled in state-regulated plans. States do not have the authority to require coverage for specific benefits in self-funded plans, which is the type of plan the majority of individuals with employer-sponsored coverage are enrolled in.
State Medicaid programs that do not currently have a SPA allowing broad coverage of OTC drugs will need to submit a SPA to CMS to cover OTC contraceptives (with a prescription). States that wish to cover Opill without a prescription for Medicaid enrollees may opt to use state-only funds. Most states that currently cover OTC contraception without a prescription for Medicaid enrollees only cover OTC emergency contraception and condoms, so policy changes would be required to permit coverage of a daily oral contraceptive pill such as Opill.
Expanding pharmacists’ scope of practice to permit them to prescribe contraception or enter into a standing order or other statewide protocol to dispense OTC contraception is another approach some states have taken. This approach still involves a prescription, but it can satisfy the prescription requirement to obtain coverage for the product and reduce some of the barriers often associated with having to obtain a prescription from a clinician.
Federal Policy Options
While state actions to increase access to non-prescribed OTC contraception without cost-sharing are meaningful for people who are eligible for that coverage, their reach is limited. The vast majority of states have not taken action to expand insurance coverage of OTC contraception without the need for a prescription. While some additional states may act on their own to develop systems and protocols to facilitate coverage of OTC methods without the need for a prescription, other states do not have the political climate to pass contraceptive coverage requirements in general, including OTC. Additionally, these coverage requirements will only apply to state-regulated health plans. Absent federal guidance or legislation, coverage requirements will continue to vary by state and by plan type.
At the federal level, in 2022, legislation was introduced in the U.S. House and Senate called the Affordability is Access Act that would require private health insurance plans to cover, without cost-sharing, FDA-approved OTC contraception purchased without a prescription. The legislation was re-introduced in 2023, shortly after the FDA advisory committees voted to recommend approval of Opill; however, its passage is unlikely in the current political climate.
Although the ACA requires most private health plans to cover, without cost-sharing, the full range of FDA-approved contraceptive methods, which includes OTC contraception, there is currently no specific requirement or policy that they be covered without a prescription. A prescription requirement is currently mentioned in federal FAQs clarifying ACA coverage requirements, with the most recent one issued by the Biden administration in July 2022. The FAQ references coverage of emergency contraception and states that plans must cover OTC contraceptives when the product is prescribed. It also states that plans are “encouraged to cover OTC emergency contraceptives with no cost-sharing when they are purchased without a prescription.” In June 2023, President Biden issued an executive order directing the U.S. Departments of Labor, Health and Human Services, and Treasury to consider new actions to improve access to affordable OTC contraception, including no-cost coverage. What those actions may look like and when they will occur is not yet known.
New federal guidance clarifying that coverage be provided with or without a prescription and without cost-sharing could have the effect of increasing access to non-prescribed OTC contraception such as Opill to the majority of people with private health insurance. Federal guidance under the preventive services contraceptive coverage requirements would apply to those in self-funded employer plans, as well as individual and other state-regulated plans and standardize OTC coverage for contraceptives across the country.
Medicaid programs, particularly for non-expansion populations, have considerable leeway in determining what benefits, including contraceptive methods, are covered. Although all state Medicaid programs cover outpatient prescription drugs, they are not required to do so. With limited exceptions (e.g., prenatal vitamins and certain tobacco cessation products), they are also not required to cover OTC drugs, but the majority of states do. CMS has indicated it could facilitate coverage of OTC contraception by approving SPAs permitting states to cover OTC drugs and products broadly. However, a prescription for OTC drugs is needed in order for the state to draw down federal matching funds. Congress would need to amend the federal Medicaid statute to require coverage of OTC products without a prescription. Absent that, states could adopt pharmacist prescribing to fulfill the prescription requirement.
Conclusion
As new products come to market through OTC availability, the issues of cost and coverage are raised by payors and consumers alike. In the case of COVID tests there was a rapid adoption of no cost coverage, but there was a national pandemic emergency and a cross cutting will from stakeholders across the spectrum to do what was possible to expand access to the tests. Currently, for insurers to pay for an OTC product, some kind of prescription is typically needed for the plan to process a claim. There is no national requirement that OTC contraceptive drugs and products be covered without a prescription, although some states and plans have moved forward to implement coverage absent a federal requirement.
There has been some level of bipartisan support for the availability of an OTC contraceptive pill, but there is less agreement about whether these methods should fall under the ACA’s no cost coverage requirement and be covered free of cost to policyholders. A policy that requires plans to cover OTC drugs without the requirement for a prescription will likely also have implications for the coverage and payment for other drugs that have OTC status beyond contraceptives and will shape the actions of plans, public programs, pharmacies, and policyholders alike.
While some of the interviewees for this project expressed that their state coverage laws for non-prescribed OTC contraception have increased access to some degree, most cited extensive challenges and confusion over the implementation of these laws. Issues cited include the mechanics of billing for a medication that is not accompanied by a prescription, lack of awareness about these laws (for health plans, pharmacists, and enrollees); and reliance on pharmacist prescribing as a fallback approach in states that allow it. From the perspective of many interviewees, these challenges have limited the impact of these coverage laws to meaningfully increase access to non-prescribed OTC contraception. Additionally, state benefit requirements only apply to fully-insured plans and do not apply to self-funded employer plans, which are only regulated by federal law.
Some interviewees expressed that uniform coverage of OTC oral contraceptives under private plans and Medicaid can only be achieved with federal action to clarify that no prescription is needed for coverage without cost-sharing. They commented on the importance for states with requirements for coverage to communicate about the availability of these benefits with health plans, pharmacies, and consumers to increase awareness and therefore access. Some interviewees noted that eliminating the prescription requirement for OTC oral contraceptives could also expand their availability, particularly to low-income individuals who may not have the resources to purchase the products directly.
The extent to which OTC contraceptive pills can broaden the availability of effective contraceptives to those who seek them will depend on many factors including state and federal policies, pharmacy engagement, public awareness and education, affordability, as well as the implementation of systems of coverage. Absent federal guidance or regulations, the availability of private insurance and Medicaid coverage of OTC contraception without a prescription will continue to depend on the state in which one lives and, if covered by employer-sponsored insurance, the plan’s funding structure.
Acknowledgements
Acknowledgements
KFF thanks all of the interviewees for sharing their time, expertise, insights, and feedback throughout the course of this project. All interviewees who agreed to be identified are listed below. The authors would also like to thank Donald F. Downing (University of Washington School of Pharmacy) for his contributions to this project.
AHIP (formerly America’s Health Insurance Plans)Ashley Seyfarth, PharmD, Owner, Kare DrugCat Livingston, MD, MPH, Medical Director, Health Share of OregonChristine Gilroy, MD, Chief Medical Officer, Express ScriptsIllinois Department of Healthcare and Family ServicesJenny Arnold, PharmD, CEO, Washington State Pharmacy AssociationNew Mexico MedicaidNew York State MedicaidRobyn Elliott, Managing Partner, Public Policy PartnersSally Rafie, PharmD, Founder, Birth Control PharmacistTara Pfund, PharmD, Product Manager, AssureCareVictoria Nichols, Free the Pill Project Director, Ibis Reproductive HealthWashington State Health Care Authority
Methods
KFF staff identified states and a wide range of stakeholders that are involved in aspects of the decision-making, implementation, and oversight of coverage of OTC contraceptive methods. States were selected based on whether they had implemented insurance coverage of OTC contraception without a prescription and/or expanded the scope of pharmacist practice.
To inform interviews with Medicaid officials, KFF contracted with Health Management Associates (HMA) to assist in identifying key officials from state Medicaid programs and facilitating contact with them. HMA also provided feedback on interview guides and this analysis written by KFF, and provided clarification and guidance regarding pharmacy industry policies, processes, and terminology.
Potential interviewees were contacted by KFF or HMA via email. Follow-up emails were sent to those who did not respond. KFF consulted with several experts in the fields of pharmacy and policy to develop interview guides, which were tailored to the type of industry/agency and provided to interviewees prior to the interviews. Interviews were conducted virtually (except one that was completed in writing) by the KFF research team and lasted 30 to 60 minutes. Some interviews had multiple representatives from one agency, company, or organization, while others had a single interviewee. Interviewees representing smaller private industries and organizations were offered a $200 gift card incentive to participate.
Interviewees were asked to review the draft report and provide their feedback prior to publication, which most did.
To ensure interviewee confidentiality, interviewees were instructed that they would not be personally recognized unless given express consent. For this reason, neither interviewee names nor affiliations are included in this report.
Appendix
| Appendix Table 2: Steps to Submitting a Pharmacy Claim |
| 1. Pharmacy enters information required to bill insurance into its computer system, including the patient’s insurance, the drug or product, the prescriber’s national provider identification number (NPI), and other relevant information. This can be performed by a pharmacist, pharmacy technician, or clerk. 2. Pharmacy submits the claim using the National Council for Prescription Drug Programs (NCPDP)’s standard. NCPDP sets the HIPAA standard for pharmacy claims. 3. The claim reaches what is referred to as a “switch,” which acts as a gate keeper to ensure the claim reaches the correct pharmacy benefit manager (PBM) or other payer. 4. The claim contains values associated with the patient that identifies the PBM/payer the claim should be sent to.
5. The PBM/payer receives the claim for processing. 6. After processing, the PBM/payer returns the claim back to the originating pharmacy through the “switch” with either an approval or a denial. There are other processes that might require the PBM/payer to return a claim containing more information before final processing. 7. An approved claim will provide the pharmacy with the amount being paid and the amount of copayment to collect from the patient (if any). 8. Actual payment to the pharmacy (money transfer) occurs at a later date based on the parties’ contractual agreement. |
| SOURCE: Kevin Gorospe, PharmD, Principal, Gorospe Solutions LLC |