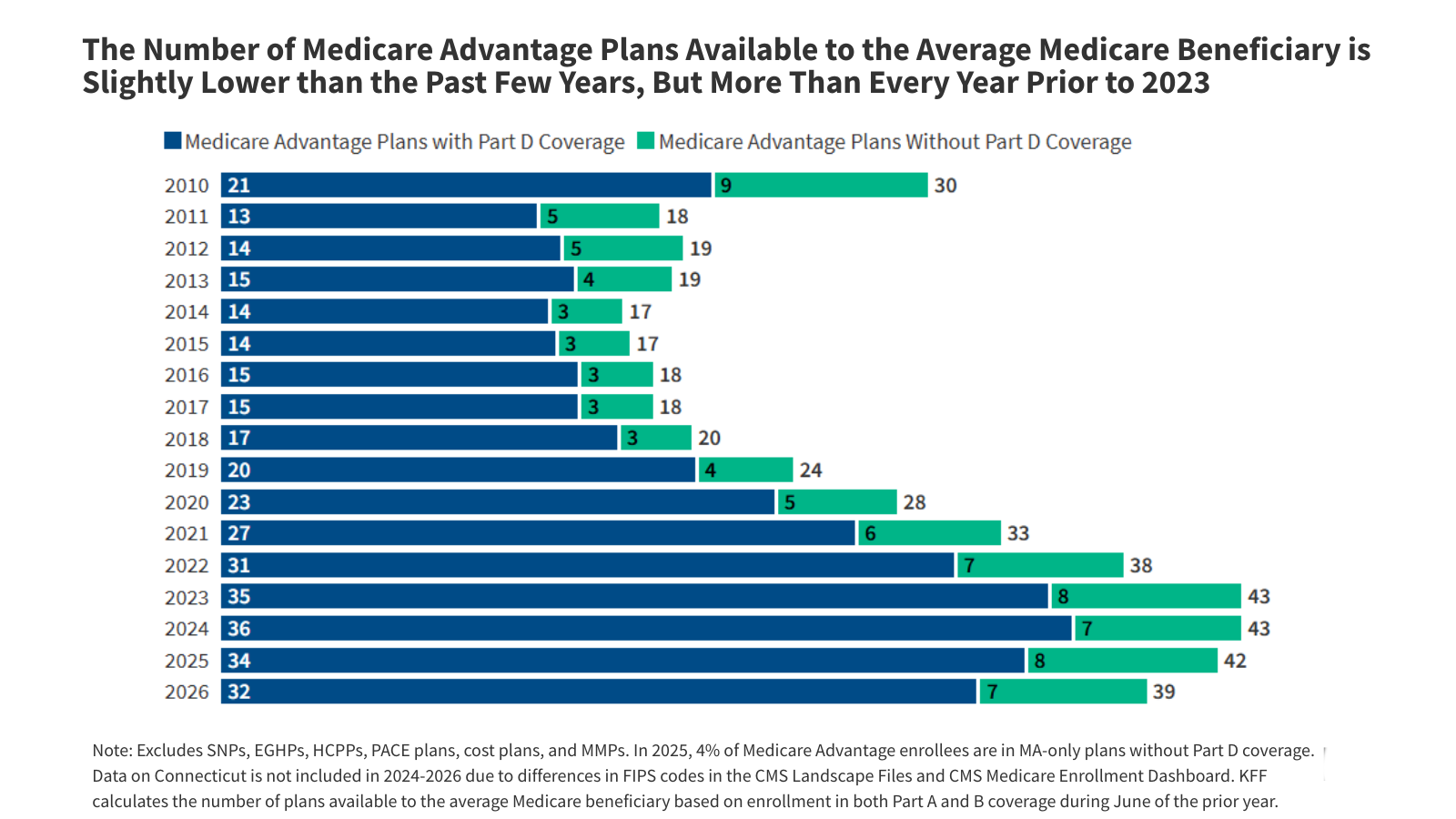
What to Know About Medicare Coverage of Telehealth
Congress has repeatedly extended pandemic-era flexibilities around Medicare coverage of telehealth, but most such flexibilities remain temporary. This brief answers key questions about the current scope of Medicare telehealth coverage, including both temporary and permanent changes adopted through legislation and regulation, and future policy considerations.