Oral Contraceptive Pills: Access and Availability
Editorial Note: Originally published on March 20, 2024, this brief was updated on March 10, 2026 to incorporate new data and the latest policy developments on contraceptive coverage.
Introduction
For over 60 years, American women have relied on oral contraceptive pills to prevent pregnancy. Oral contraceptives are the most widely used form of reversible contraception and are also commonly used to manage other health conditions. In the U.S., daily oral contraceptive pills have traditionally only been available with a prescription. In July 2023, the U.S. Food and Drug Administration approved the first over-the-counter daily oral contraceptive pill, eliminating the requirement for a prescription from a clinician. This progestin-only pill is now available for purchase in stores and online at most major retailers. This brief provides an overview of oral contraception, discusses private insurance and Medicaid coverage, and reviews strategies to promote and expand women’s access to oral contraceptives.
Background
In 1960, the Food and Drug Administration (FDA) approved the sale of Enovid for use as the first oral contraceptive. Controversial from its earliest days, in 1965, the Supreme Court ruling in Griswold v Connecticut upheld married women’s rights to contraception, followed in 1972 by the Supreme Court’s decision in Eisenstadt v Baird which extended the right to single, unmarried individuals.
Oral contraceptive pills (OCP) consist of the hormones progestin and estrogen, or only progestin, and must be taken orally once per day in order to prevent pregnancy. Currently, there are three different types available on the market: the combination pill, the progestin-only pill, and the continuous use pill. The three formulations vary in their chemical hormonal composition as well as regimen for use (Table 1). Different brands further add to the diversity of OCP available by altering the type and/or dose of hormones. Emergency contraceptive pills are also a type of OCP, consisting of the progestin levonorgestrel, but are not intended for daily use. Rather, they are used to prevent pregnancy after unprotected sex.
Both the combined and progestin-only pills are highly effective with perfect use, with a failure rate (rate at which women become pregnant while using the contraceptive) of less than 1%. However, the failure rate with “typical use” is 9%, which accounts for inconsistent or incorrect use.
Use
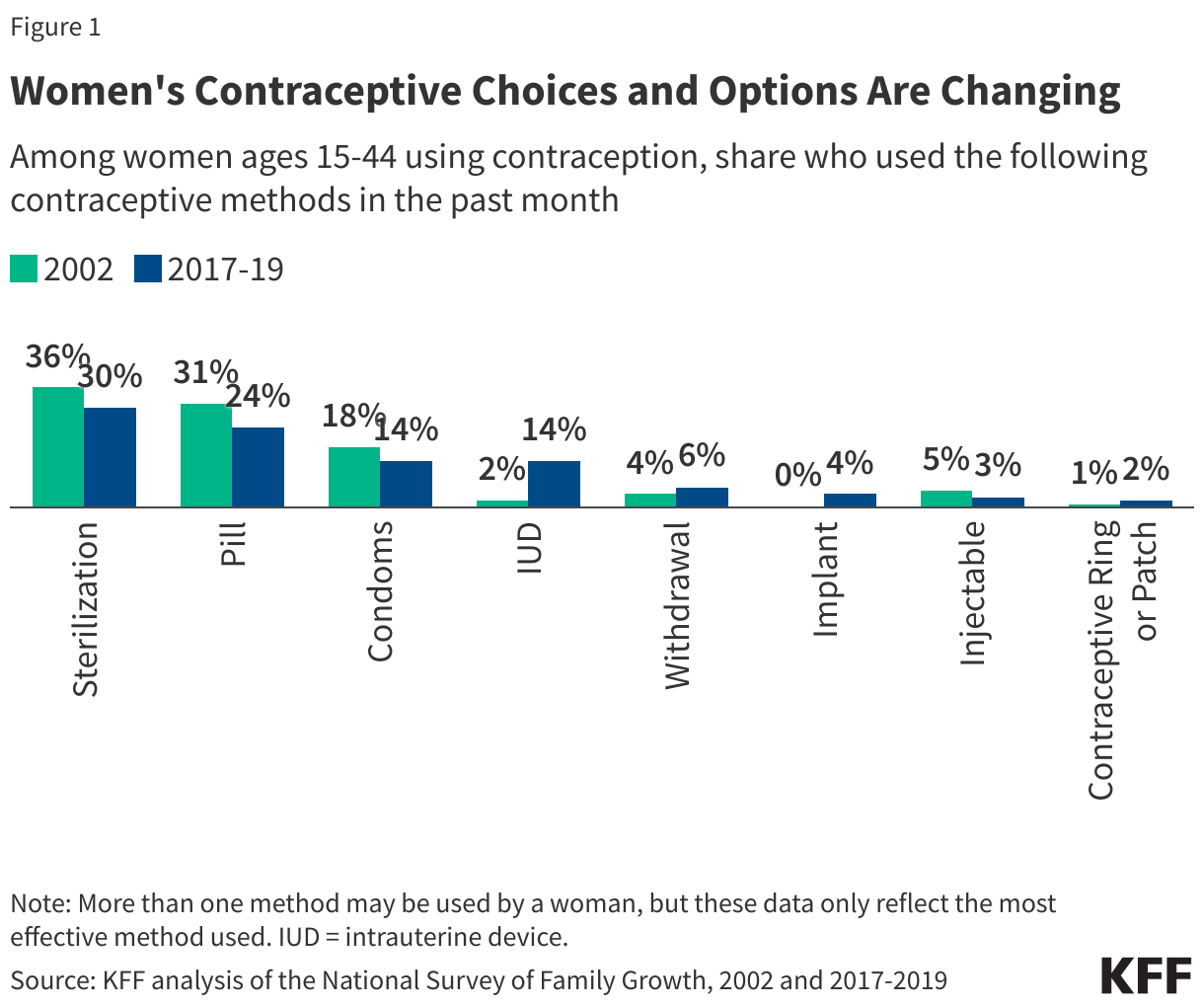
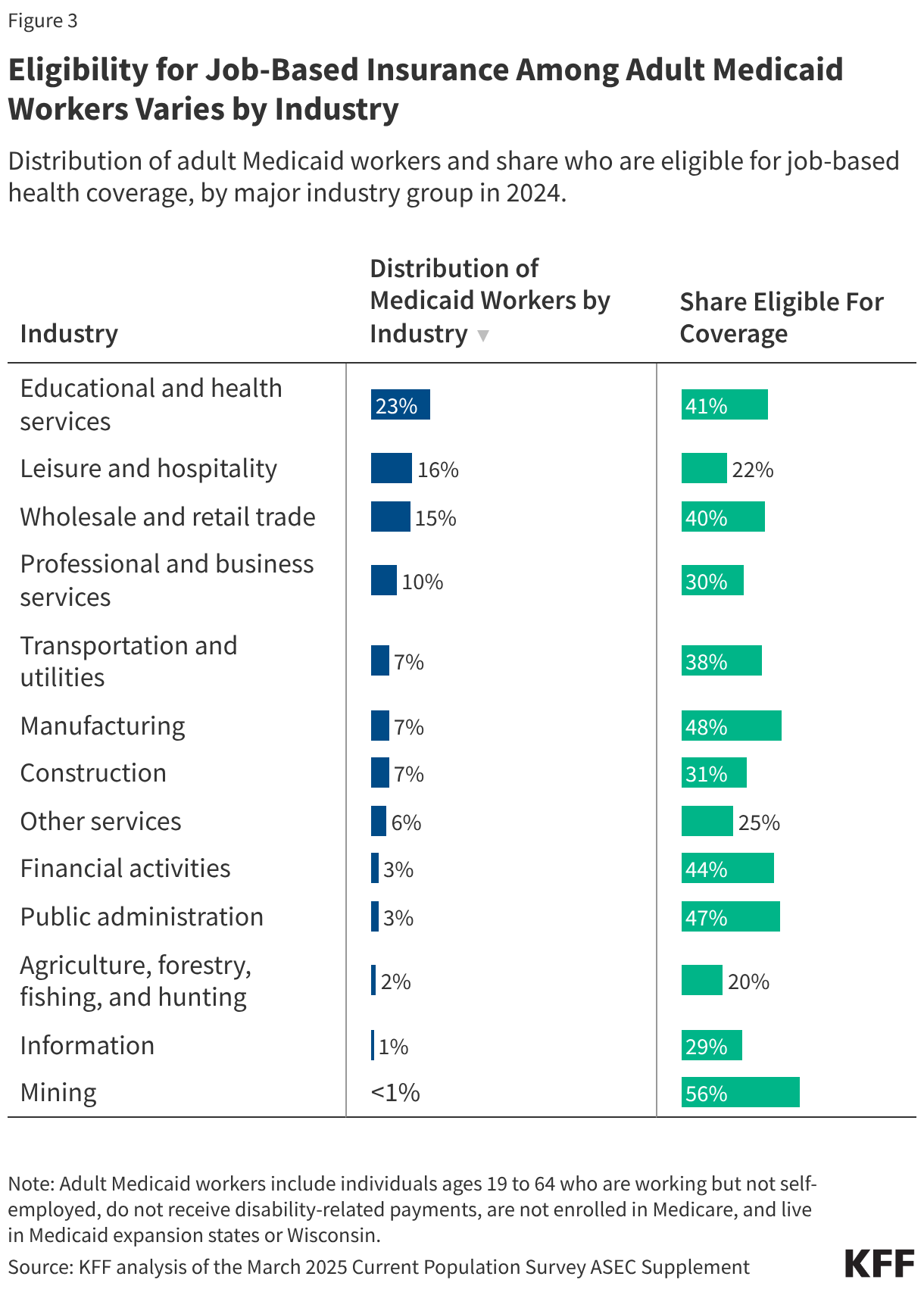
The pill was the first FDA-approved contraceptive to be used in the U.S. and is still the most commonly used form of reversible contraception. According to KFF analysis of the 2022-2023 National Survey of Family Growth, the most recent years for which data are available, about one-quarter (23%) of women ages 15-44 who currently use contraception reported using the pill as their method of choice, a decline from 31% in 2002 (Figure 1). At the same time, there has been an increase in the use of long-acting reversible contraceptives (LARCs), such as intrauterine devices (IUDs) and implants, which have been promoted by several medical groups in recent years.
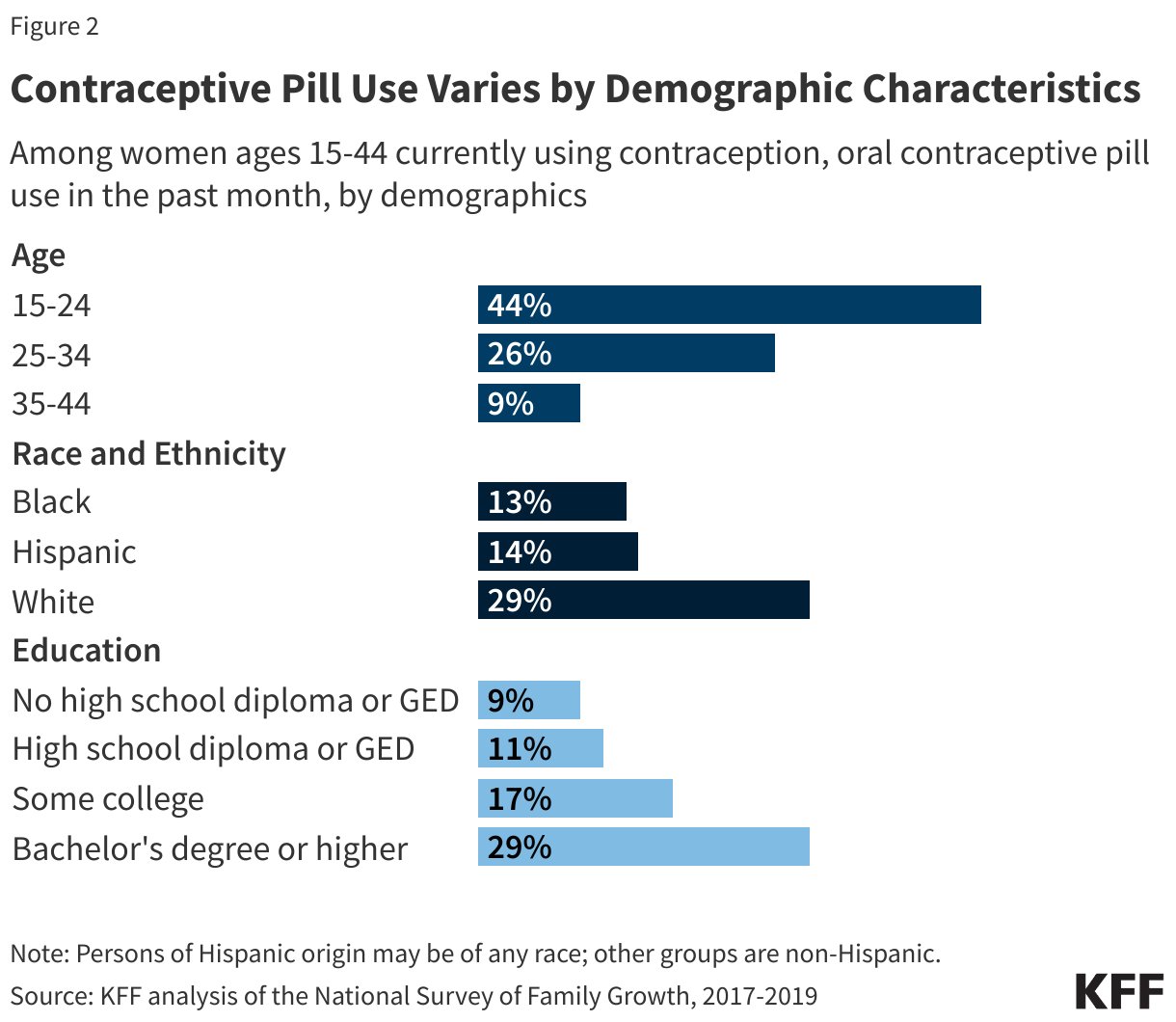
Among women ages 15-44 who use any form of contraception, OCP use is higher among younger women and decreases with age. A larger share of White women (27%) use OCP than Hispanic (19%) or Black (14%) women. OCP use increases with higher educational attainment (Figure 2).
OCPs are primarily used for pregnancy prevention, but they can also be used to address other health conditions, particularly menstrual-related disorders such as menstrual pain, irregular menstruation, fibroids, endometriosis-related pain, and menstrual-related migraines. Use of combined pills for acne has been formally approved by the FDA for specific brands. While most (82%) women who use OCP take them primarily to prevent pregnancy, 18% use them solely for non-contraceptive reasons such as to manage a medical condition (unpublished analysis from the 2022 KFF Women’s Health Survey).
Oral contraceptives are safe for most women. Possible side effects include headache, nausea, breast tenderness, and breakthrough bleeding. The combined hormonal pills may be associated with a small increased risk of deep vein thrombosis, heart attack and stroke for some women.
Insurance Coverage and Financing of Oral Contraceptives
OCPs have not always been covered by insurance plans in the same way as other prescriptions drugs. This became the focus of legislative action in the early 1990’s, first at the state and then the federal level.State legislatures began passing “contraceptive equity” laws which typically required that plans offering prescription drug coverage also cover contraceptives on the same terms as other prescriptions. Some state laws went further to require that plans cover all FDA-approved contraceptives. However, these state laws only applied to plans that were regulated by the state and did not include self-funded employer-sponsored plans, which are federally regulated through ERISA and cover most workers with employer-sponsored insurance. Minimum coverage standards for employer-sponsored plans were established in 2000, when a federal ruling from the Employment Equal Opportunity Commission found it unlawful under the Civil Rights Act for plans to deny coverage for contraceptives if they covered other preventive prescription drugs and services. By 2010, 28 states required insurers that cover prescription drugs to provide coverage for the full range of FDA-approved contraceptives.
Private Insurance and the ACA
In 2010, the Affordable Care Act (ACA) took state laws further by requiring most private plans (including self-funded, small and large group, and individual plans) to cover a wide range of recommended preventive services, without patient cost-sharing. In 2011, the Health Resources and Services Administration (HRSA), following recommendations issued by the Institute of Medicine, added that all FDA-approved, prescribed contraceptive methods and patient counseling for women with reproductive capacity be covered, without cost sharing, as a preventive service. Plans that were in effect on or before March 23, 2010, known as “grandfathered plans,” are not required to cover preventive services, or they may require cost sharing. Additionally, plans offered by an employer with a religious objection to contraception may exclude this coverage from their plan.
Under the ACA, most private health insurance plans must cover at least one form of each of the 18 FDA-approved contraceptive methods for women without cost sharing. This means that plans must cover at least one of each of the three different types of oral contraceptives – the combined pill, the progestin-only pill and the continuous use pill – though it is up to an insurer’s discretion using reasonable medical management practices whether to cover a brand name or generic contraceptive if both are available. Insurers are required to cover other contraceptives if medically necessary and must provide a process for policyholders to request coverage of a contraceptive that is not already covered without cost sharing by the plan. While some contraceptive methods are available over the counter without a prescription, plans typically require a prescription to trigger coverage.
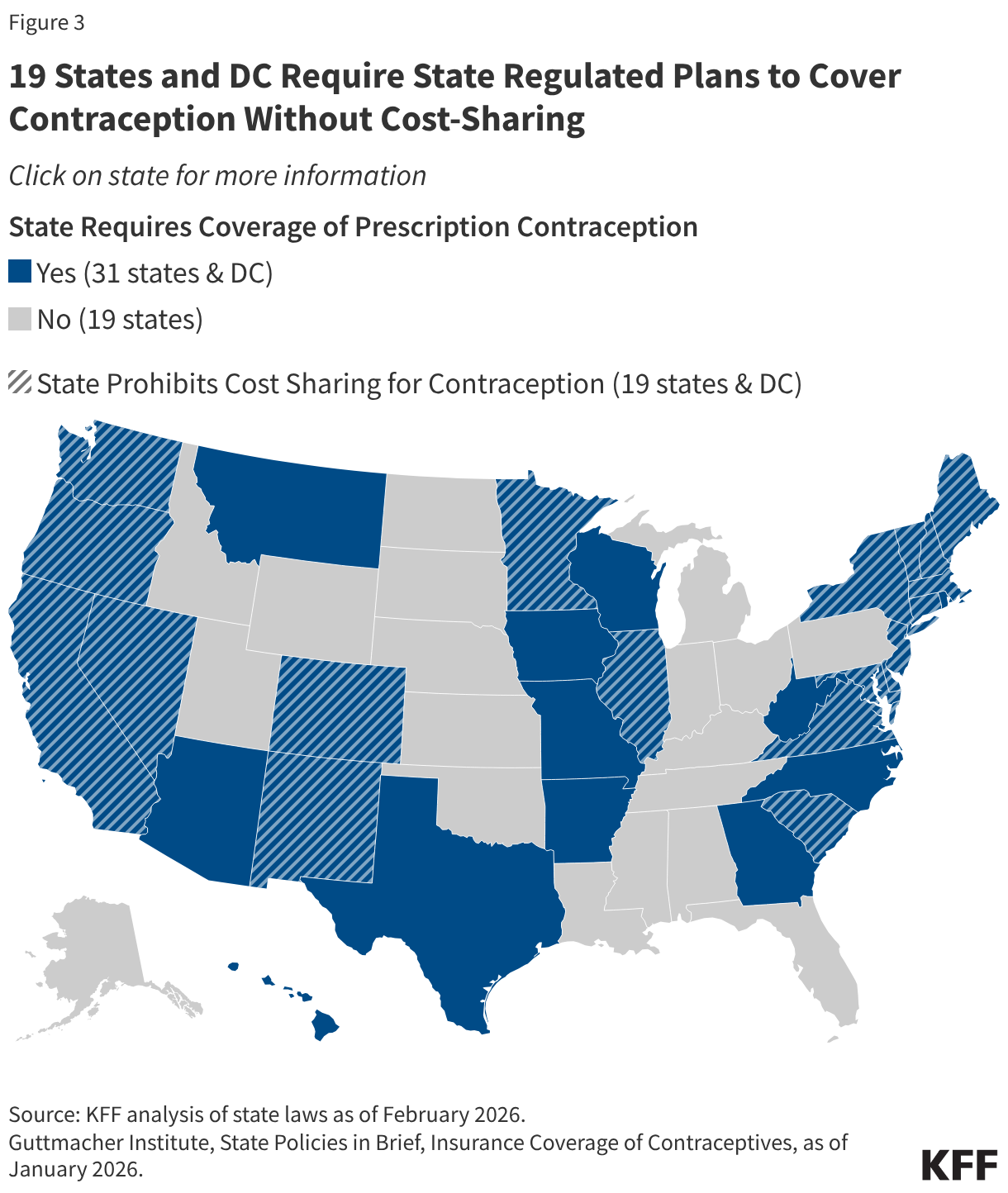
Additionally, 31 states and D.C. require state regulated plans to cover prescription contraception, and 19 of those states and D.C. passed laws that build on the federal requirement prohibiting cost-sharing for all FDA-approved contraceptive methods for women (Figure 3). Some of these states have gone beyond the ACA requirements, mandating coverage of vasectomies and/or over-the-counter contraceptives.
Today, fewer women are paying out of pocket for contraceptives as a result of the ACA’s contraceptive coverage requirement. According to a 2019 KFF analysis of the IBM MarketScan Commercial Claims and Encounters Database, among women with health insurance from a large employer who use OCP, the share experiencing out-of-pocket spending on OCP declined from 96% in 2010 to 11% in 2017.
Controversial since its inception, the provision has sparked litigation and new regulations in response to lawsuits that have reached the Supreme Court. Although the Obama administration allowed certain religious employers with an objection to contraception to request an exemption from the requirement, in 2020, the Supreme Court upheld two Trump administration rules that expanded eligibility to almost all employers that have a religious or moral objection. Female employees, dependents, and students of these exempt employers are not entitled to coverage for the full range of FDA-approved contraceptives.
Public Programs
Federal law has long required state Medicaid programs to cover family planning services and supplies without cost sharing and provides states with an enhanced federal match for providing these services. States that expanded Medicaid under the ACA must follow the ACA requirements for private plans and are required to cover at least one form of all 18 FDA-approved contraceptive methods for women. There is no similar requirement for traditional full-scope Medicaid or through a Medicaid family planning expansion program, and there is variation between states on the specific services that are covered.
Since the passage of the ACA, some states have also strengthened their contraceptive coverage requirements for Medicaid (Figure 3). For example, California passed the Contraceptive Coverage Equity Act of 2014 which extends the ACA’s coverage policy beyond private plan beneficiaries to all Medicaid managed care enrollees, regardless of whether they qualify as a result of the ACA expansion or through traditional pathways. California expanded this coverage in 2022 to cover OTC contraceptive drugs and products without a clinician’s prescription and extends this coverage to fee-for-service Medicaid beneficiaries. Thirteen (13) other states and D.C. have since enacted similar contraceptive equity laws that apply to both private insurance plans and Medicaid.
Medicare, the federal program for seniors 65 and older as well as younger adults with permanent disabilities, does not require coverage for oral contraceptives. According to KFF analysis of the 2021 Medicare Current Beneficiary Survey, over 1 million women under age 50 were enrolled in Medicare. Medicare beneficiaries that have enrolled in private Medicare Advantage plans or who have opted into the Medicare Part D prescription drug benefit may have coverage for oral contraceptives, but the scope of coverage varies between plans. Nearly 8 in 10 women of reproductive age in Medicare are dually eligible for Medicaid coverage. Coverage for oral contraceptives is also required in the Indian Health Service, the federal program that provides care on or near Indian reservations as well as in the Tricare program for active military personnel and their dependents.
Expanding Access to Contraception
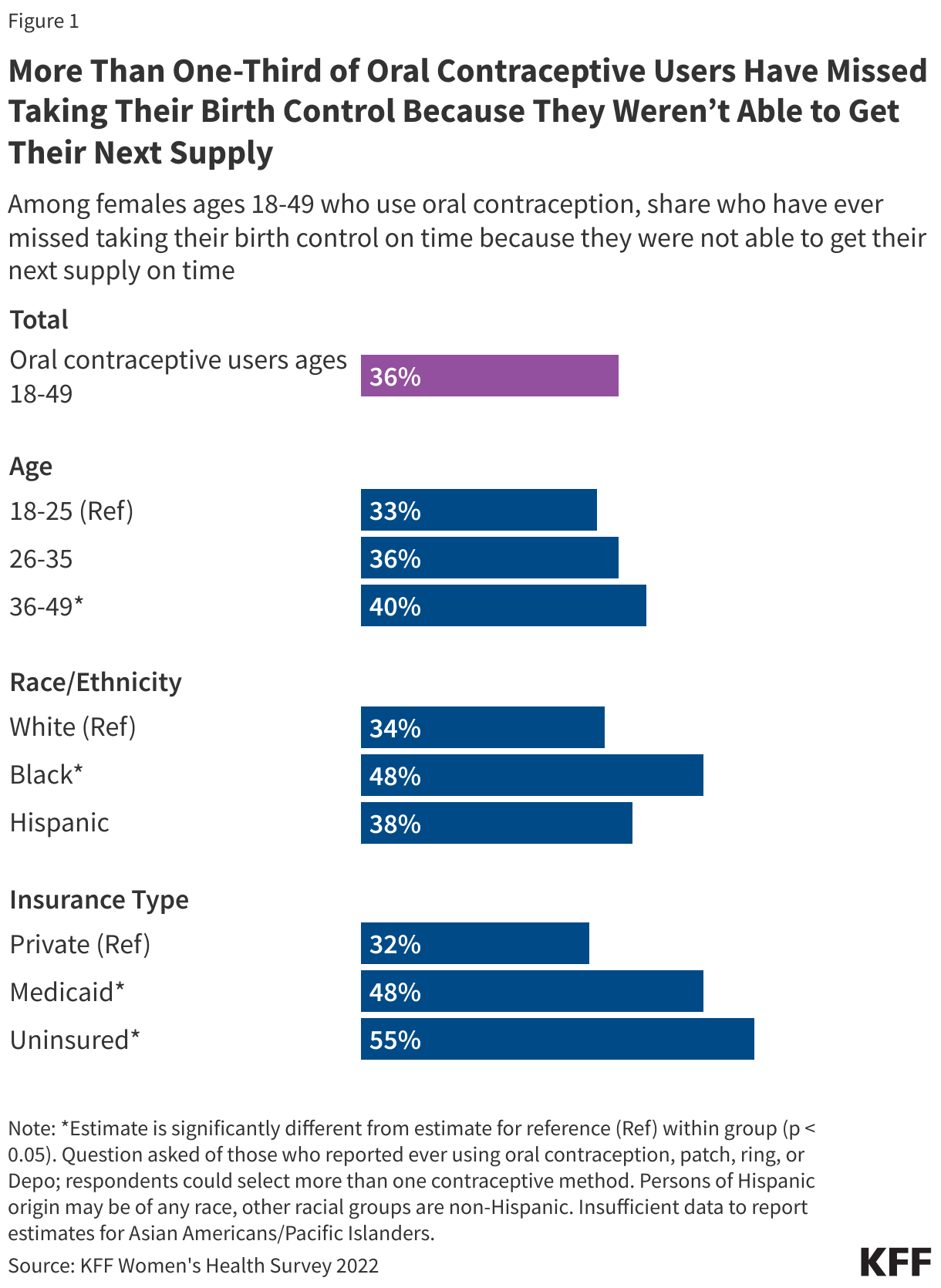
The 2022 KFF Women’s Health Survey found that one-third (33%) of female hormonal contraceptive users have missed taking their birth control because they were not able to get their next supply on time. Furthermore, it is estimated that more than 19 million women of reproductive age in need of publicly-funded contraception live in an area considered to be a contraceptive desert, meaning there is limited access to a publicly-funded provider who offers contraception. Research also points to the impacts of state and federal policies on the shrinking number of family planning providers that offer the full scope of contraceptive methods in some communities.
In recent years, there has been public discussion and state and federal policy action to reduce contraceptive access barriers by expanding the availability of daily oral contraceptive pills through different mechanisms. Approaches that have been adopted include making OCP available over the counter without a prescription; expanding the ability of pharmacists to dispense or prescribe OCP; extending the supply of contraception that is dispensed at one time; and using mail-based online services or smartphone applications.
Over the Counter (OTC) Access
In July 2023, the U.S. Food and Drug Administration (FDA) approved the progestin-only Opill for OTC use, making it the first OTC daily oral contraceptive pill. Opill is available for over-the-counter purchase without age restriction in stores and online. The suggested retail price of Opill is $19.99 for one month’s supply or $49.99 for three month’s supply. Although it is farther behind in the process, another pharmaceutical company, Cadence, is working toward FDA approval of an OTC version of its combined (progestin and estrogen) oral contraceptive pill, Zena.
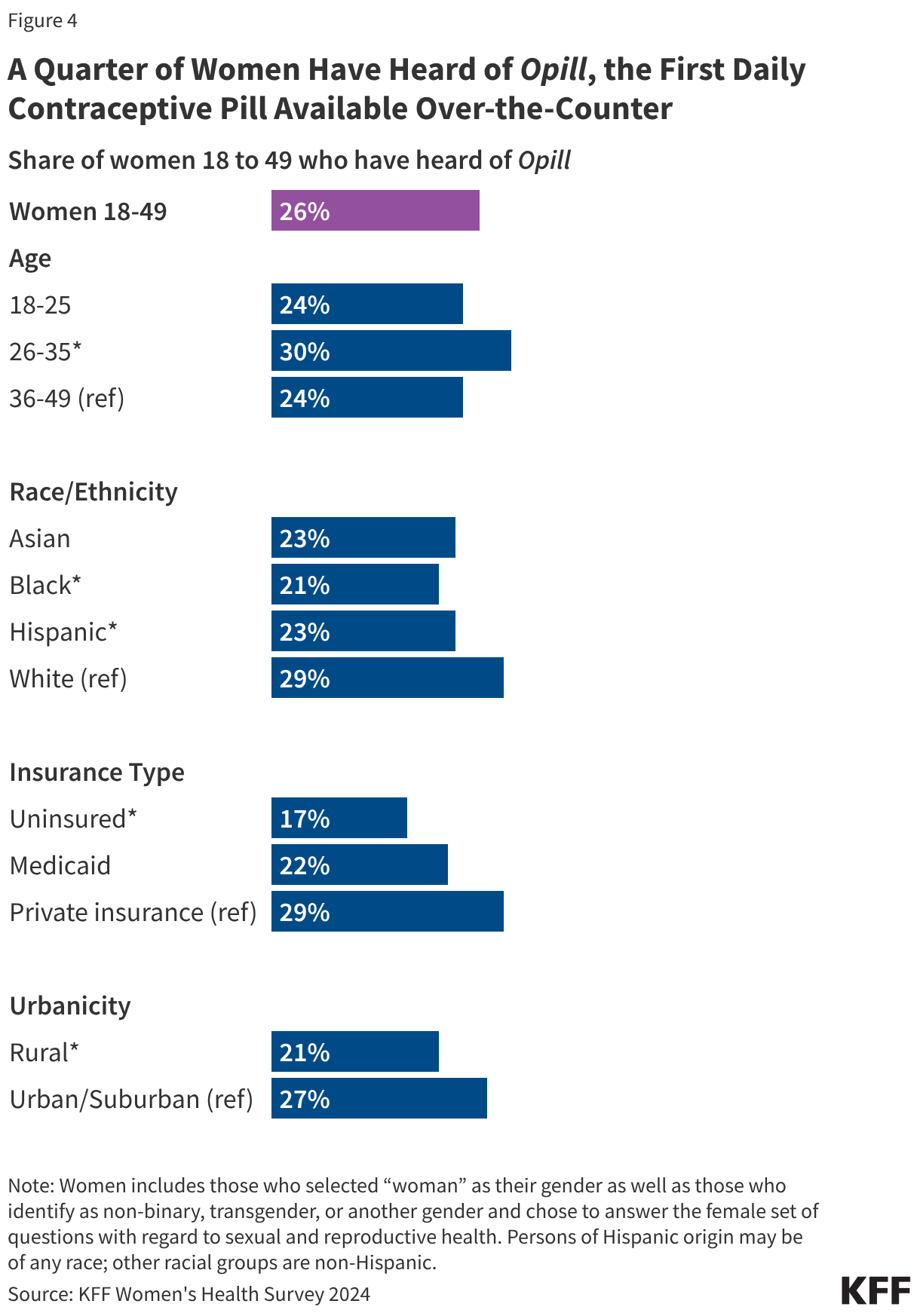
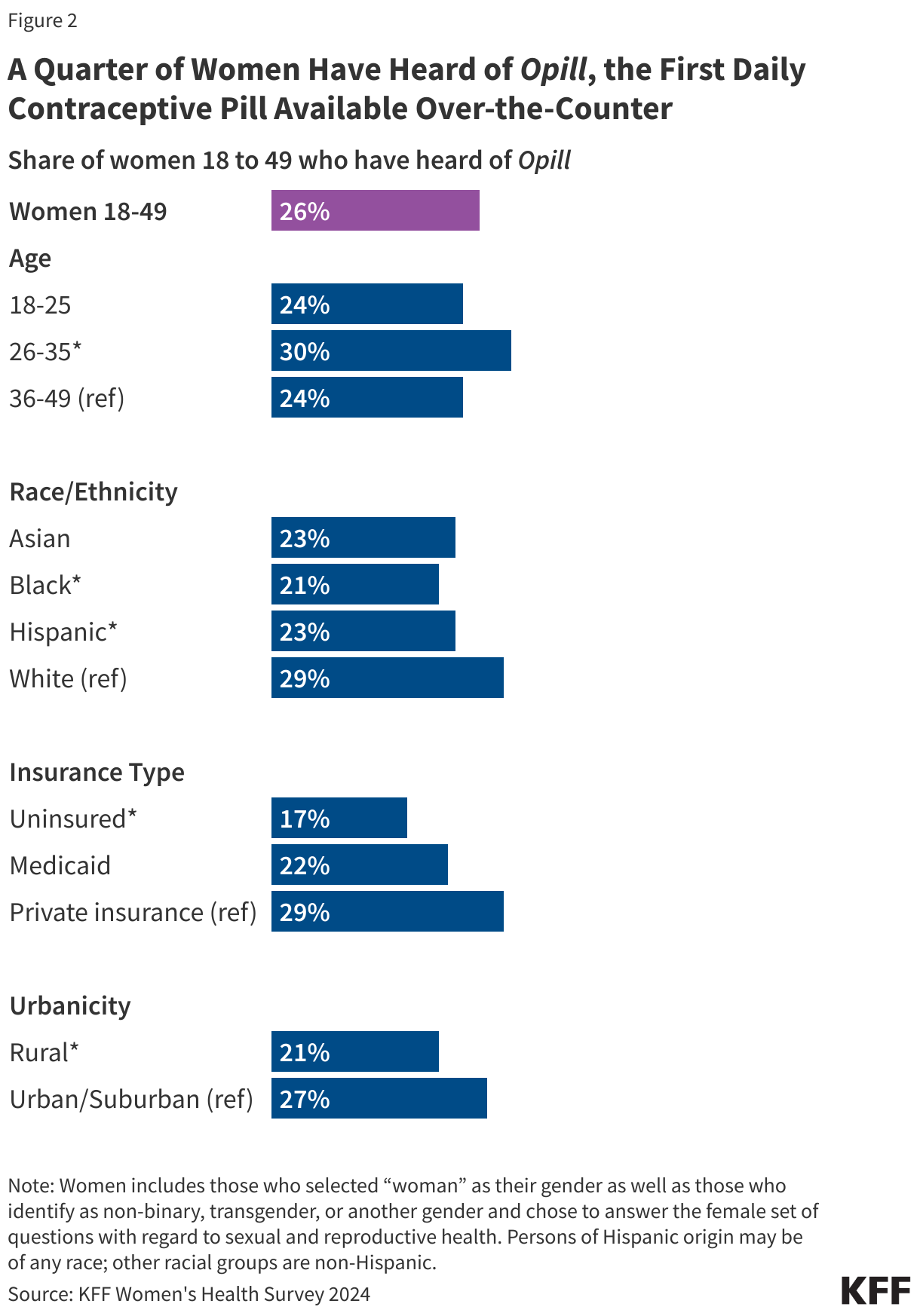
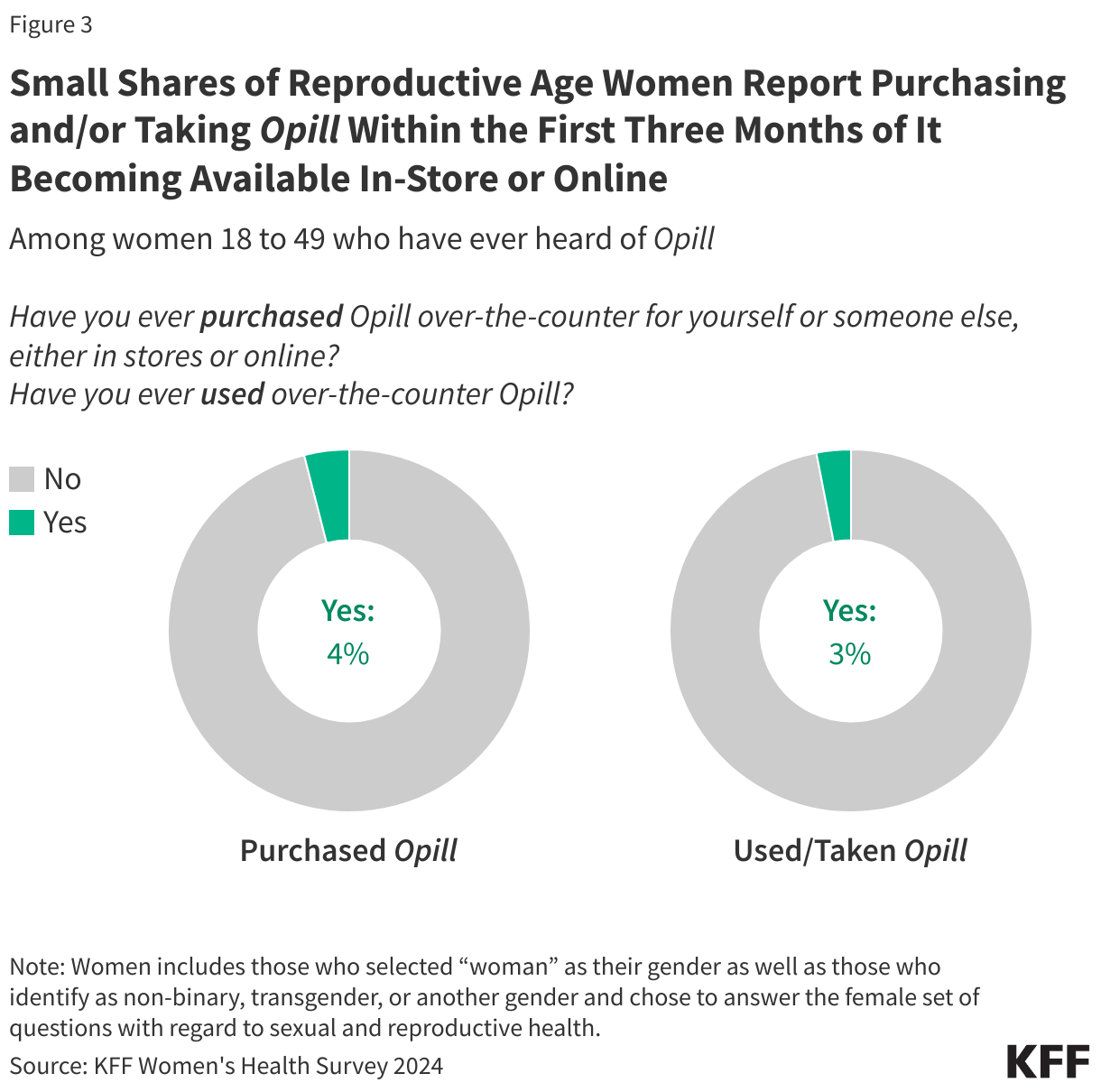
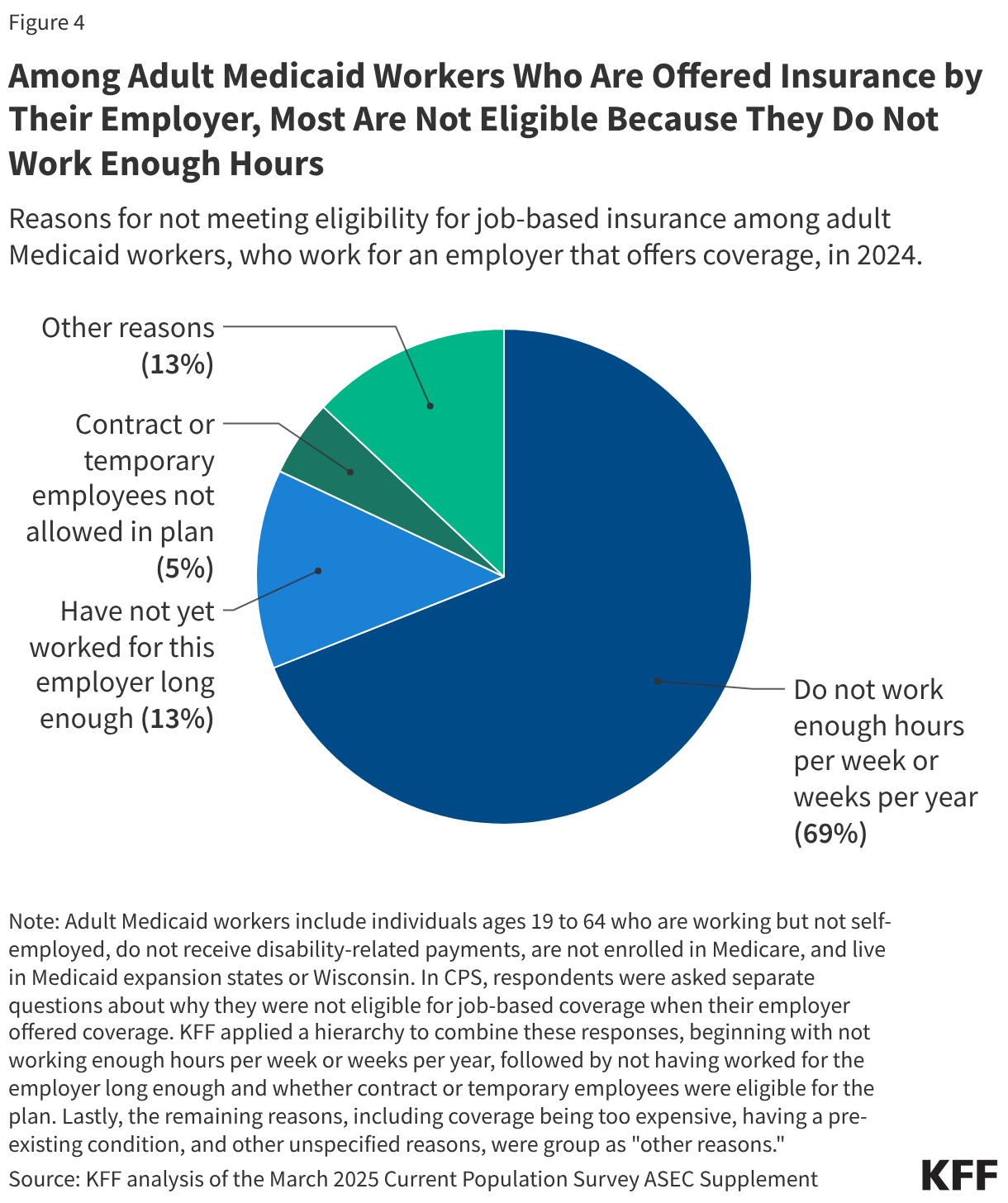
Research suggests that OTC access would increase the use of contraception and facilitate continuity of use. It could also allow women to save time spent on travel, at doctor’s office, and off work. Other research suggest that OTC oral contraceptives can especially benefit populations who have historically faced barriers to accessing contraceptive care, such as young adults and adolescents, those who are uninsured, and those living in contraceptive deserts or areas with limited access to health centers offering the full range of contraceptive methods. However, just a quarter (26%) of women 18 to 49 saying they have heard of the new daily oral contraceptive pill, and smaller shares of women who are uninsured (17%) and who live in rural areas (21%) have heard of Opill compared to those with private insurance (29%) and those living in urban or suburban areas (27%) (Figure 4).
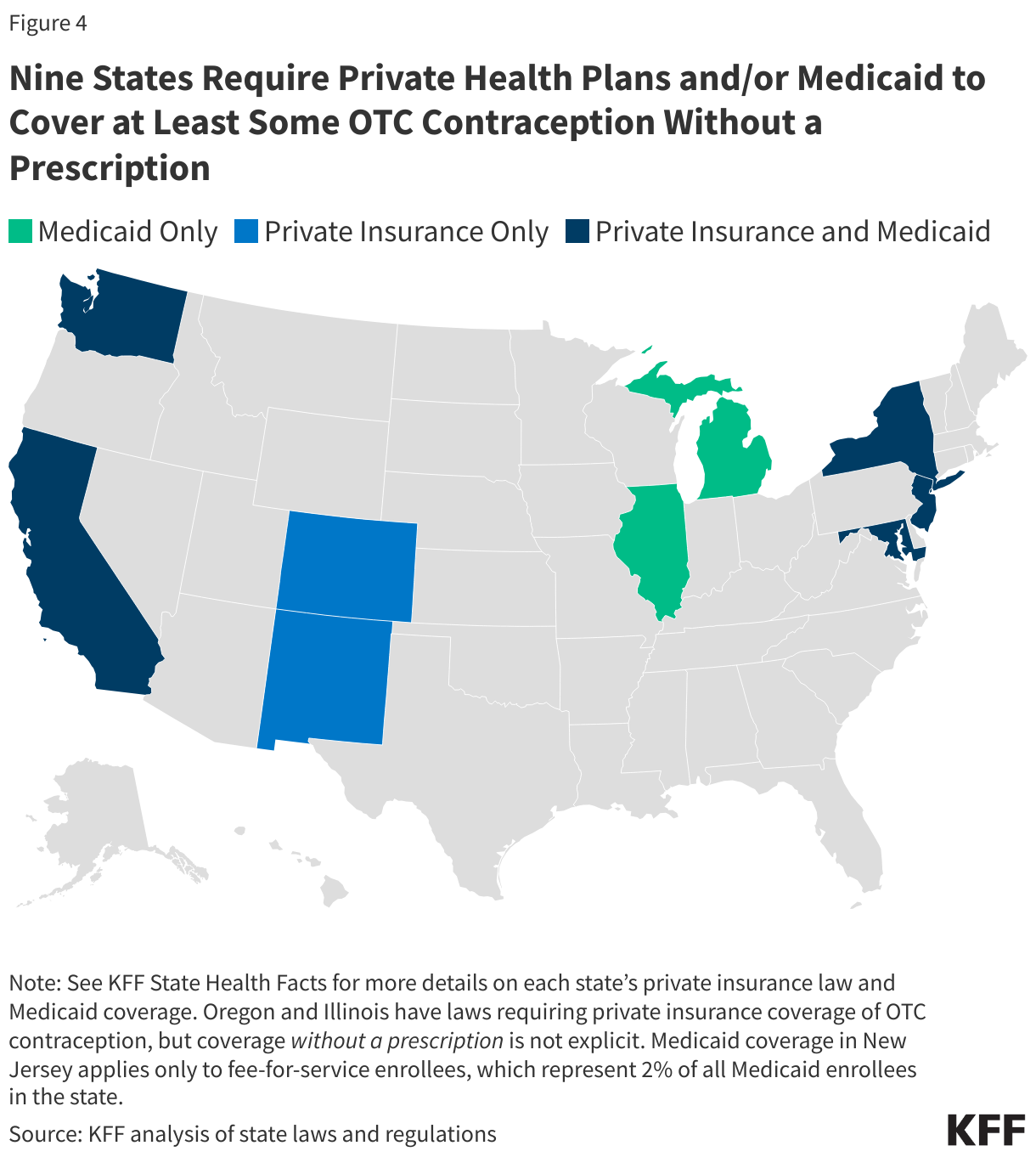
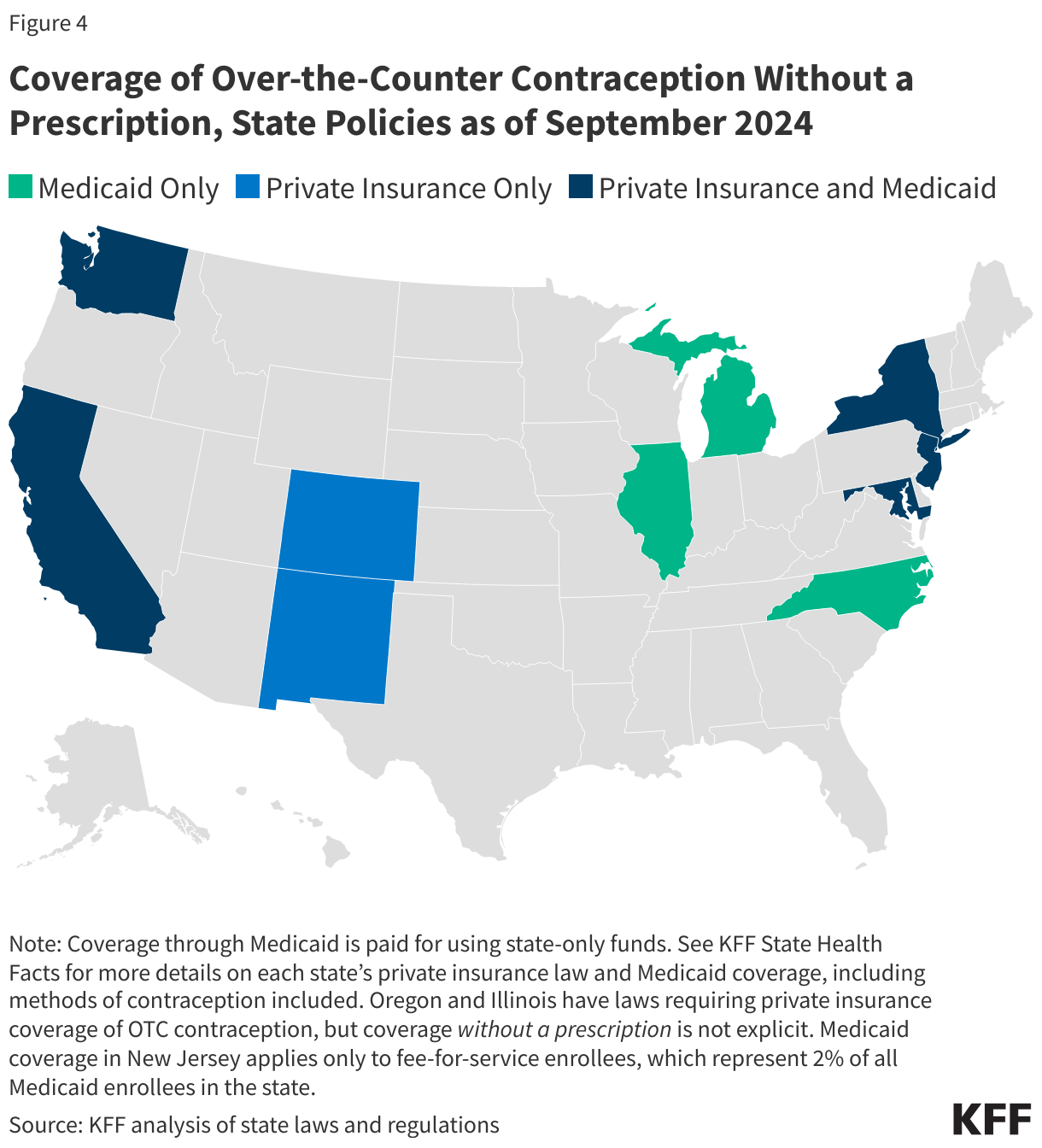
The ACA currently requires no-cost coverage for contraceptives in most private plans and for Medicaid expansion populations but plans typically require a prescription in order to trigger coverage, even for contraceptive methods that are available OTC without a prescription. Requiring plans to cover non-prescribed contraceptives would require legislation at the federal or state level, or administrative changes to the ACA’s preventive services policy. Nine states (CA, CO, DE, MD, ME, NJ, NM, NY, and WA) have laws or regulations requiring state-regulated private health insurance plans (individual, small group, and large group markets) to cover, without cost sharing, OTC contraception without a prescription. While New York’s law applies to emergency contraception only, the other state laws apply to non-prescribed contraceptive drugs broadly(Figure 5).
Eight states (CA, IL, MD, MI, NC, NJ, NY, and WA) use state-only funds to cover at least some OTC contraception without a prescription for Medicaid enrollees. However, these states, with the exception of California, cover non-prescribed emergency contraception and/or condoms only, so a change in law or policy would be needed to cover a daily oral contraceptive pill without a prescription. States wishing to cover OTC contraception without a prescription for enrollees must use state-only funds as federal funds are only available for prescribed drugs. (See KFF State Health Facts for more details on each state’s private insurance law and Medicaid coverage, including contraceptive methods covered.)
For more information on how states have implemented insurance of OTC contraceptives, see KFF’s report Insurance Coverage of OTC Oral Contraceptives: Lessons from the Field and issue brief Over-the-Counter Oral Contraceptive Pills.
Pharmacist Prescribing
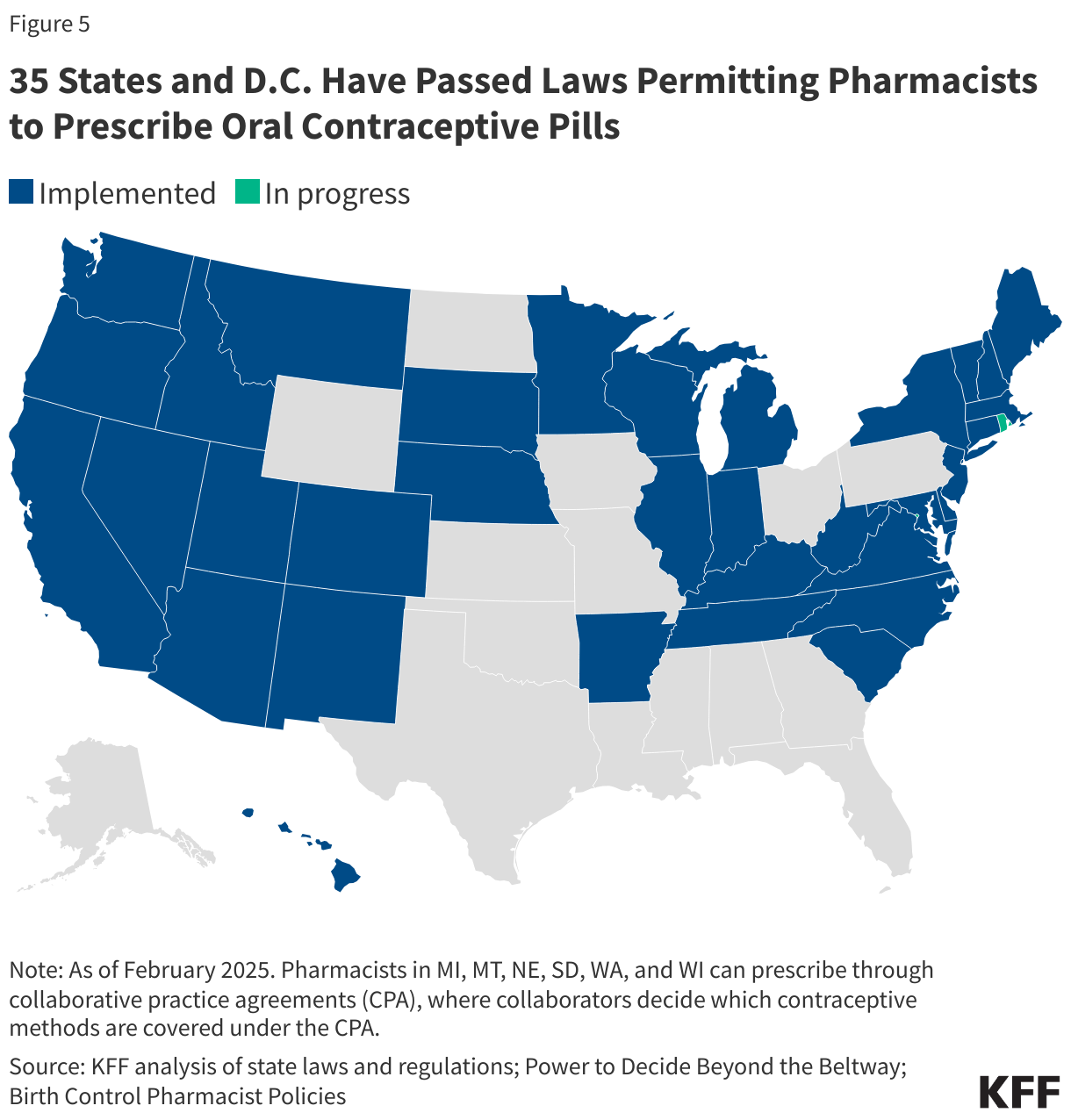
Another avenue that is gaining support in some states allows pharmacists to prescribe or dispense OCP without requiring an in-person medical visit to a physician. As of February 2026, 36 states and D.C. passed legislation to allow pharmacists to prescribe certain self-administered contraceptives to women (Figure 6). All of these states allow pharmacists to prescribe at least oral contraceptives, but states vary in other details, such as the type of prescriptive authority (e.g., collaborative practice agreements, statewide protocols, and standing orders), minimum age requirements, the type of contraceptive that pharmacists can prescribe, the length of the supply, and whether the patient needs a prior prescription from a physician.
Although expanded scope of pharmacist practice can remove some barriers to obtaining contraceptives, challenges still remain for women seeking a prescription for contraception from a pharmacist. For example, pharmacies typically charge consultation fees, which some reports suggest can be as high as $50 in certain areas. Although insurers are generally required to cover contraceptives without cost sharing, they are not obligated to cover this fee. Also, pharmacies can choose not to participate or may not have any pharmacists trained to provide this service.
From the pharmacy perspective, pharmacists must elect to complete additional education requirements, which vary by state, and often include several hours of continuing education from an accredited training program. Additionally, states may not have a reimbursement mechanism in place to pay pharmacists for providing this service. For example, while Oregon and Hawaii require plans to reimburse the dispensing entities, California’s law does not require reimbursement for payers other than Medicaid. Lack of or low reimbursement for pharmacist prescribing can result in fewer pharmacies choosing to provide this service.
12-Month Supply
Another approach to facilitate access to oral contraceptives involves increasing the dispensing period of contraceptives to 12 months per prescription. Currently, dispensing patterns vary by insurer, with many plans limiting supply of pills to 1-3 packs at a time. The 2022 KFF Women’s Health Survey found that among females who reported using birth control pills in the past year, 32% receive 1-2 packs at a time, 63% receive 3-5 packs, and just 3% receive a 12-month supply. Providing women with an extended supply of pill packs may lead to more consistent contraceptive use. Women who receive a one-year supply have been found to be 30% less likely to have an unintended pregnancy compared to women receiving a 1–3-month supply.
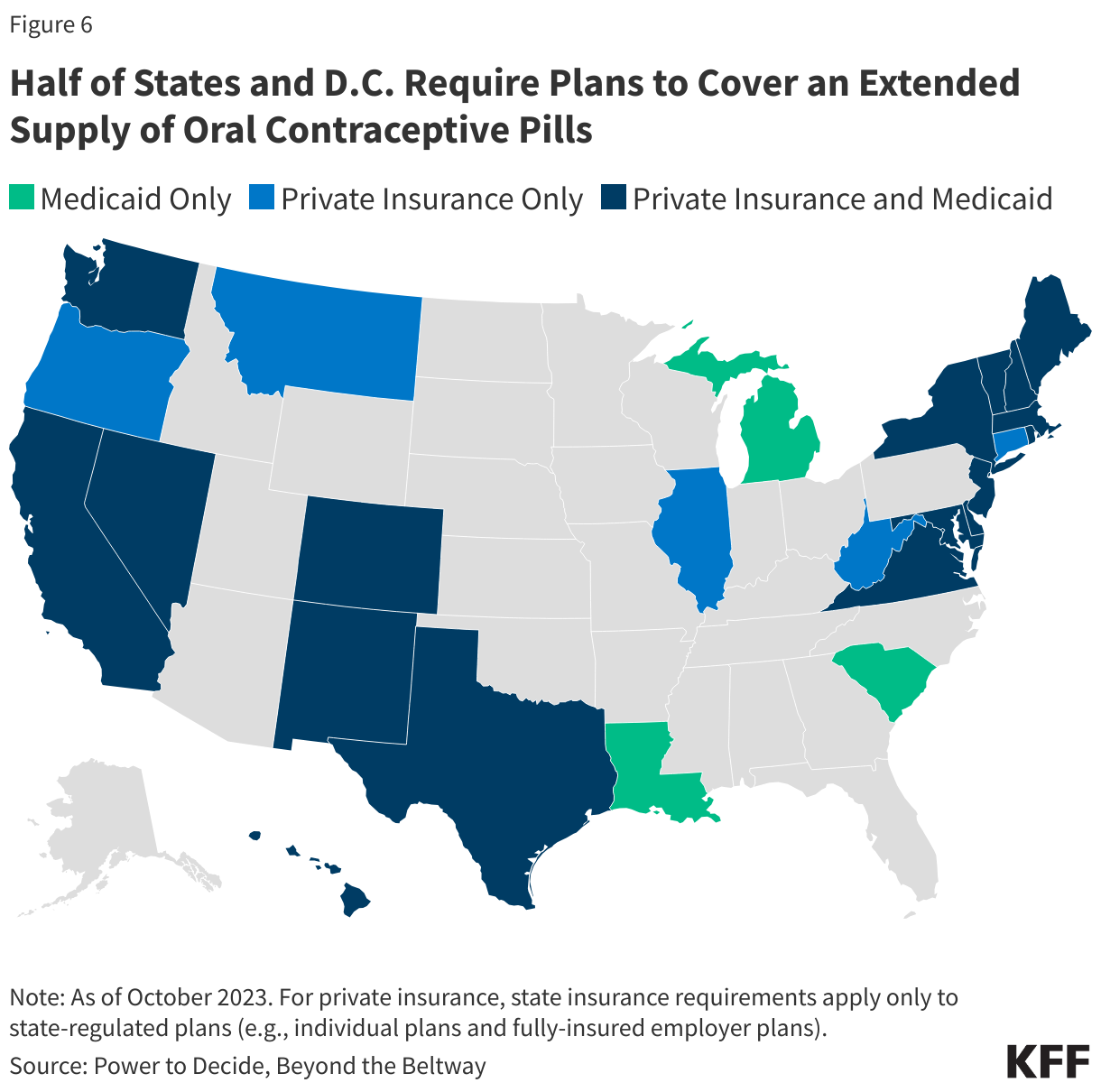
In 2015, Oregon became the first state to pass a law requiring state-regulated plans to cover a three-month supply of contraceptives when first prescribed, followed by a 12-month supply of contraceptives. Laws requiring coverage for 12 months of oral contraceptives have since been enacted in 29 additional states and DC (Figure 6). Idaho, Louisiana, and New Mexico require coverage for a 6-month supply. While most of these states have also enacted policies that require no-cost contraceptive coverage similar to the ACA’s contraceptive coverage provision, eight states (HI, ID, LA, MI, MT, OK, TX, and WV) with extended supply laws have not done so. This means that although insurers must cover a 12-month supply in these states, state law does not prohibit cost sharing; however, most plans must abide by the federal requirement and not charge any cost sharing for prescribed, FDA-approved contraceptive methods.
Telecontraception
In recent years, a growing number of companies providing contraception through online platforms (“telecontraception”) have entered the market and are providing a new option for people to obtain contraceptive supplies without the need for an in-person visit. A growing number of online services and smartphone applications offer options for patients to speak with providers by video or chat, get prescriptions, and order birth control pills through mail delivery. These services work by collaborating with physicians, pharmacies, and sometimes health insurers to prescribe and ship OCP to the patient’s home or a local pharmacy.
Costs for these services vary between companies. Most charge a fee for the service, which is typically not covered by insurance and can range from a $15 fee per consultation/prescription to a $99 yearly membership that covers the medical evaluation and customer support for the duration of the prescription.
A KFF study on telecontraception companies found considerable variation in method availability and acceptance of insurance. Many telecontraception companies accept private insurance and/or Medicaid, to pay for the cost of the pills, while others do not. The price of contraception offered by these platforms vary by method and by brand; generic pills typically range in price from $5 to $25 per pack without insurance.
Most companies ship OCPs free of charge to the patient’s home, while some require pick up from a local pharmacy. Prescriptions are often valid for 12 months and patients are sent either a one- or three-month supply of pills. Video/audio consultations are required by certain services before receiving the prescription. Services that do not require a consultation do require patients to complete a health assessment or questionnaire to determine eligibility and the appropriate pill. People in every U.S. state have access to at least one of these services, but the minimum age to use the service varies by company and state law, although many require the person to be at least 18 years old.
Oral contraceptives are the most commonly used form of reversible contraception in the U.S. Most women with private insurance or Medicaid can receive no-cost coverage for OCPs. The FDA recently approved Opill, the first ever daily OCP available over the counter, though insurance coverage of the product will largely depend on state efforts in the absence of federal guidance.Several states have enacted policies to broaden OCP access, particularly through pharmacist prescribing and insurance coverage for extended supplies and non-prescribed OTC contraceptives. The use of telemedicine to expand OCP access continues to evolve, with many women now able to obtain OCP using smartphone and web-based services.