Editorial Note: Originally published in March 2017, this resource is updated as needed to reflect the latest developments.
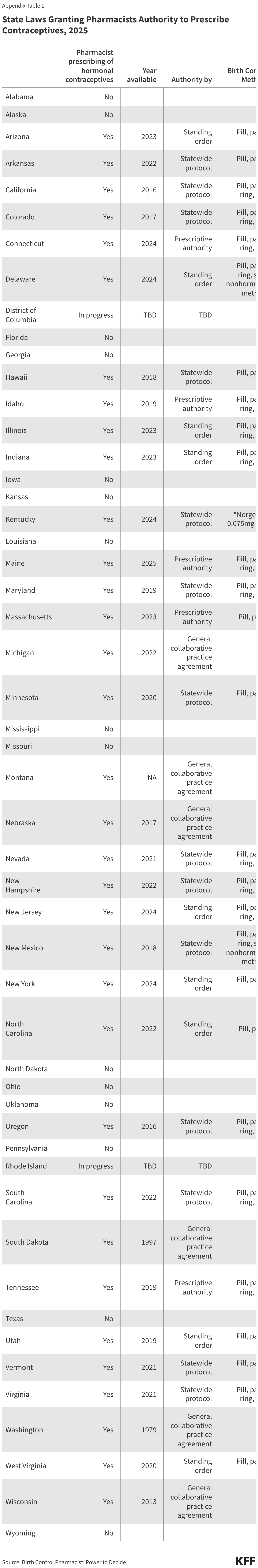
Key Facts
- The World Health Organization (WHO), founded in 1948, is a specialized agency of the United Nations with a broad mandate to act as a coordinating authority on international health issues, including helping countries mount responses to public health emergencies.
- On January 20, 2025, President Trump issued an Executive Order announcing that the U.S. would withdraw as a member of WHO and halt funding to the organization, and on January 22, 2026, the administration announced the U.S. withdrawal was complete. In 2020 during the first Trump administration, the U.S. temporarily suspended U.S. funding to WHO and initiated a process to end U.S. membership in the organization, actions that were reversed by the Biden administration in 2021.
- Prior to these actions, the U.S. government (U.S.) had been actively engaged with WHO throughout its history, providing financial and technical support as well as participating in its governance structure and had historically been one of the largest funders of WHO. U.S. contributions had ranged between $163 million and $816 million annually over the last decade.
- Since 2021, WHO has overseen negotiation processes to update the International Health Regulations (IHR) and establish a new “pandemic agreement.” In May 2024, member states approved revisions to the IHR and, in May 2025, approved a new pandemic agreement. President Trump’s January 2025 Executive Order withdrawing from WHO also stated that the U.S. would not be bound by the revised IHR or the new pandemic agreement and would no longer participate in related discussions or other WHO-based negotiations.
- WHO now faces funding shortfalls and, in the absence of U.S. contributions, had to implement staffing cuts last year. Almost 3,000 positions – 22% of the organization’s staff – were eliminated between January and June 2025, and a number of WHO’s activities were cut or curtailed. The lack of U.S. support, a shrinking institutional footprint, and other challenges raise questions about WHO’s role going forward.
What is the World Health Organization?
WHO, founded in 1948, is a specialized agency of the United Nations. As outlined in its constitution, WHO has a broad mandate to “act as the directing and coordinating authority on international health work” within the United Nations system. It has 193 member states (the U.S. is no longer a member).
The agency has played a key role in a number of past global health achievements, such as the Alma-Ata Declaration on primary health care (1978), the eradication of smallpox (formally recognized in 1980), the Framework Convention on Tobacco Control (adopted in 2003), and the 2005 and 2024 revisions of the International Health Regulations (IHR), an international agreement that outlines roles and responsibilities in preparing for and responding to international health emergencies. WHO has regularly provided member states with technical guidance and support during responses to epidemics and pandemics, such as Ebola, Zika, mpox, and COVID-19.
Mission and Priorities
WHO’s overarching mission is “attainment by all peoples of the highest possible level of health.” It supports its mission through activities such as:
- providing technical assistance to countries;
- setting international health standards and providing guidance on health issues;
- coordinating and supporting international responses to health emergencies such as disease outbreaks; and
- promoting and advocating for better global health.
The organization also serves as a convener and host for international meetings and discussions on health issues. While WHO is generally not a direct funder of health services and programs in countries, it does provide supplies and other support during emergencies and carries out programs funded by donors.
WHO’s goal for its current work period (2025-2028) is to “get the world back on track to achieve the health-related Sustainable Development Goals (SDGs) while advancing health equity and building health systems resilience.” In pursuit of this, WHO focuses on three priorities: “to promote health by addressing the root causes of disease, including climate change; to provide health by strengthening health systems based on primary health care and expanding access to health services and financial protection; and to protect health by preventing, preparing for, mitigating, detecting and responding rapidly to health emergencies.”
As part of its work to help countries be better protected against health emergencies – and propelled by the issues and challenges faced during the COVID-19 pandemic – WHO has overseen two sets of international negotiations among member states since 2021. The first was a process to update and amend the IHRs, which was completed and approved by member states in May 2024 and came into force in September 2025. While the U.S. had, through Executive Agreement, become party to prior revisions of the IHR, the Trump administration has stated that the U.S. will not be bound by the IHRs any longer. The second is a new pandemic agreement designed to institute common principles and mechanisms contributing to global prevention, preparedness, and response capacities for health emergencies such as pandemics. In May 2025, 124 member states voted to approve the agreement and initiate the process by which it can be ratified by states and then formally adopted by WHO. Eleven countries (including Poland, Israel, Italy, Russia, Slovakia and Iran) abstained from voting on the agreement, while the U.S. and other countries such as Argentina did not participate in the vote and have criticized the agreement. WHO member states continue to negotiate an annex to the agreement that details a new pathogen access and benefits sharing (PABS) system.
Organization
WHO has a global reach, with a headquarters office located in Geneva, Switzerland, six semi-autonomous regional offices that oversee activities in each region,1 and a network of country offices and representatives around the world. It is led by a Director-General (DG), currently Dr. Tedros Adhanom Ghebreyesus, who was first appointed in 2017 and was re-elected to a second five-year term in May 2022. Dr. Tedros has indicated that his priorities include continuing to strengthen WHO’s financing, staffing, and operations; building pandemic preparedness and response capacities at WHO and elsewhere; and helping countries re-orient health systems toward primary health care and universal health coverage.
World Health Assembly
The World Health Assembly (WHA), now, with the U.S. withdrawal, comprised of representatives from 193 member states, is the supreme decision-making body for WHO and is convened annually. It is responsible for selecting the Director-General, setting priorities, and approving WHO’s budget and activities. The annual WHA meeting in May also serves as a key forum for nations to debate and make decisions about health policy and WHO organizational issues. Every four years, the WHA negotiates and approves a work plan for WHO, known as the general programme of work (GPW). The current GPW (GPW 14) covers the period 2025-2028. Every two years the WHA also approves WHO’s programme budget in support of its work plan; the current programme budget covers the 2026-2027 biennium. More information about WHO’s budget provided below.
Executive Board
WHO’s Executive Board, comprised of 34 members technically qualified in the field of health, facilitates the implementation of the agency’s work plan and provides proposals and recommendations to the Director-General and the WHA. The 34 members are drawn from six regions as follows:
- 7 represent Africa,
- 6 represent the Americas,
- 5 represent the Eastern Mediterranean,
- 8 represent Europe,
- 3 represent South-East Asia, and
- 5 represent the Western Pacific.
Member states within each region designate members to serve on the Executive Board on a rotating basis.
Activities
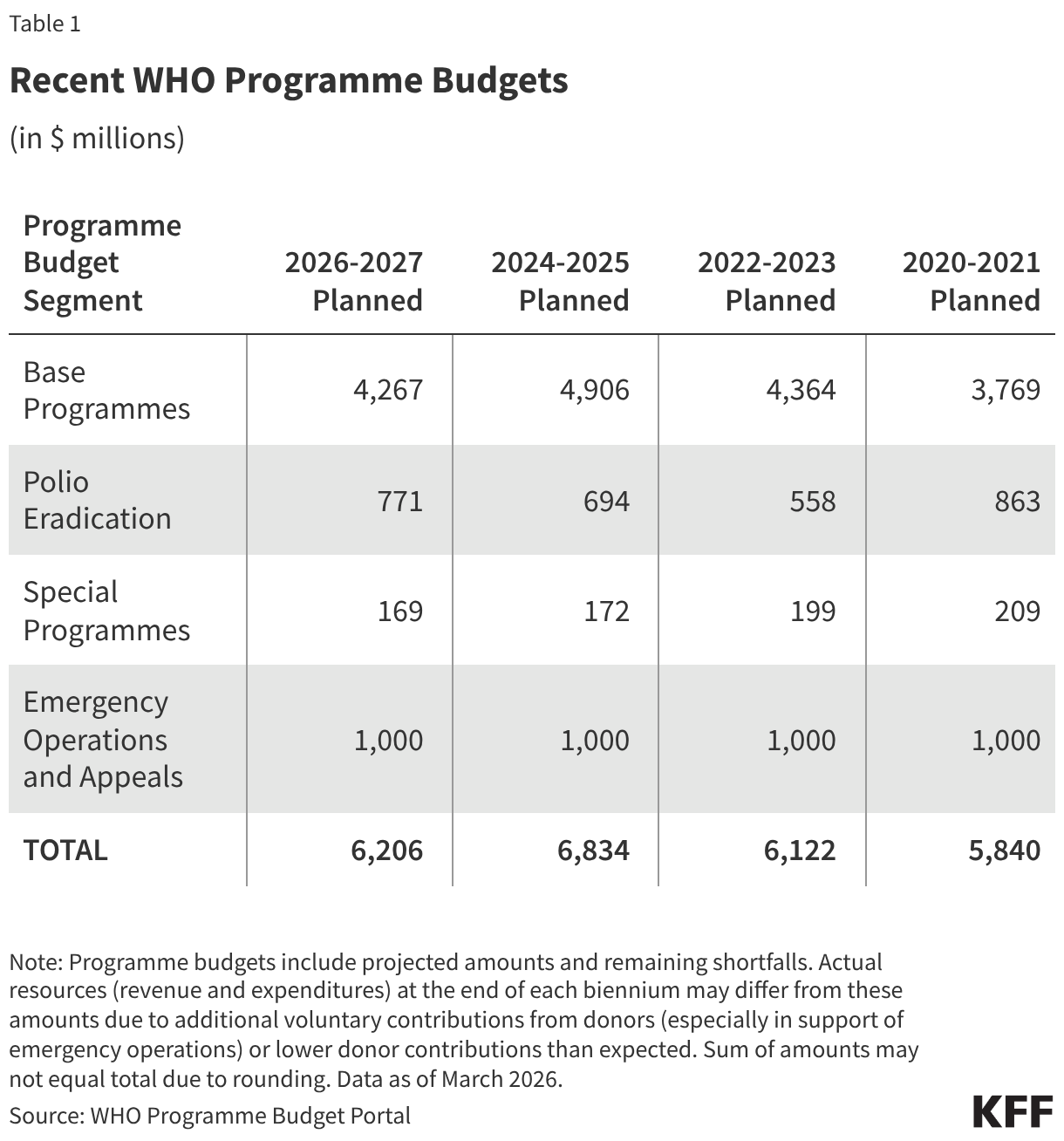
WHO supports activities across a number of key areas, organized into several “budget segments,” including “base programmes,” emergency operations, polio eradication, and “special programmes” (see Table 1). “Base programmes” refer to WHO headquarters and regional operations activities in support of the organization’s strategic objectives, such as improving access to quality essential health services, essential medicines, vaccines, diagnostics, and devices for primary health care. “Emergency operations” includes WHO efforts to help countries prepare for and respond to epidemics and other health emergencies such as COVID-19, mpox, and natural disasters. “Special programmes” include a number of WHO-led initiatives such as the Research and Training in Tropical Diseases program and Pandemic Influenza Preparedness (PIP) Framework activities.
Funding
Programme Budget
WHO has a programme budget set in advance by member states, which is meant to outline planned activities to meet its work plan over a two-year period (biennium) and describe the “resource levels required to deliver that work.” The current programme budget of $6.2 billion covers the period 2026-2027, and was approved by member states in May 2025. This amount represents a 9% decrease from the previous 2024-2025 programme budget of $6.8 billion. See Table 1.
The programme budget represents a plan for the organization’s anticipated resources, but actual resources may deviate from the initial budgeted amounts over course of the biennium due to changing or unexpected circumstances, such as additional resources provided to WHO for emergency responses or lower levels of support than expected. For example, in the 2022-2023 biennium, WHO reported a final approved budget of $10.4 billion, compared to the initial planned budget of $6.1 billion, largely reflecting additional funding received for emergency operations, including for COVID-19 and polio eradication.
Revenue
WHO has two primary sources of revenue:
- assessed contributions (set amounts expected to be paid by member-state governments, scaled by income and population) and
- voluntary contributions (other funds provided by member states, plus contributions from private organizations and individuals).
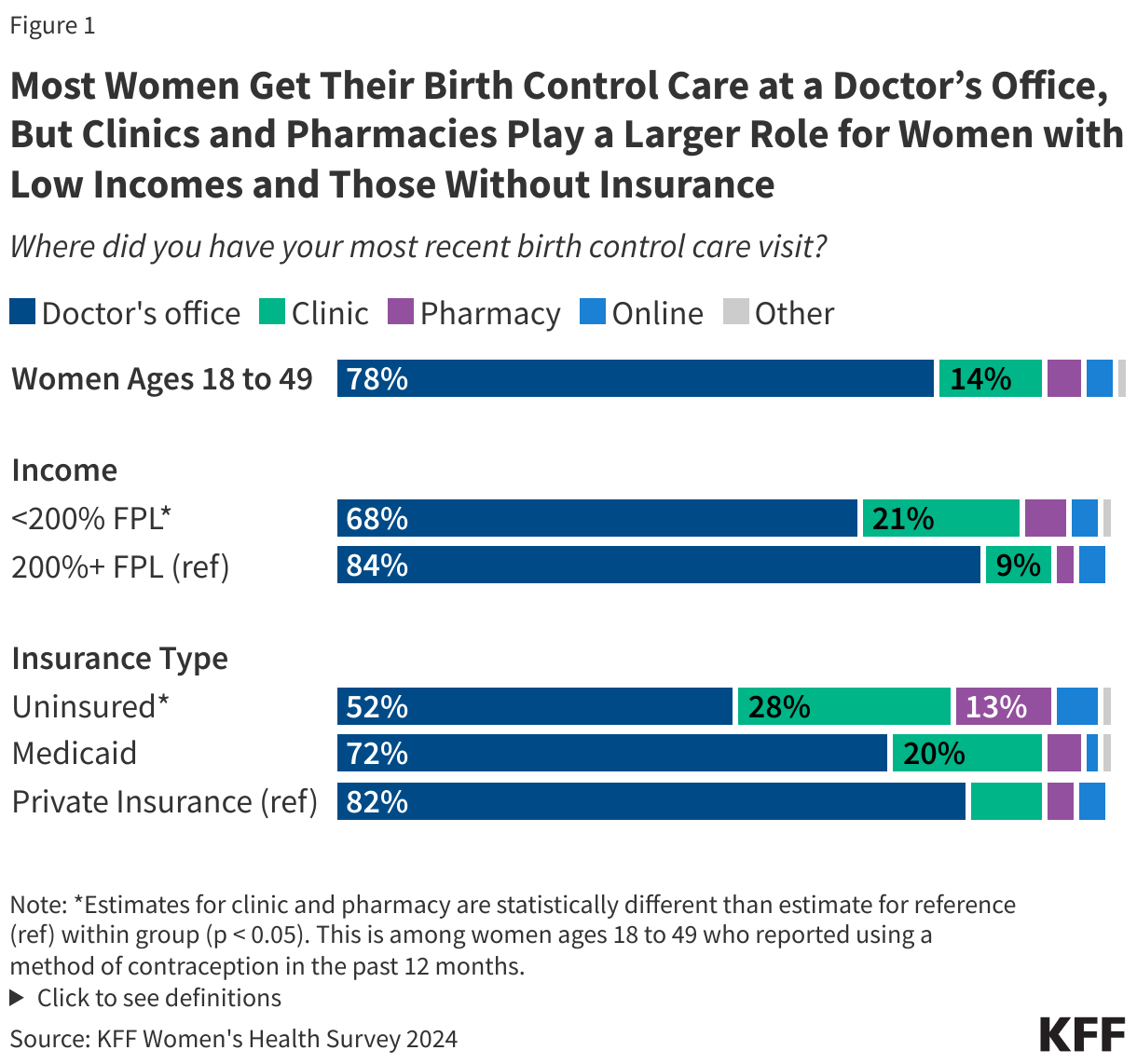
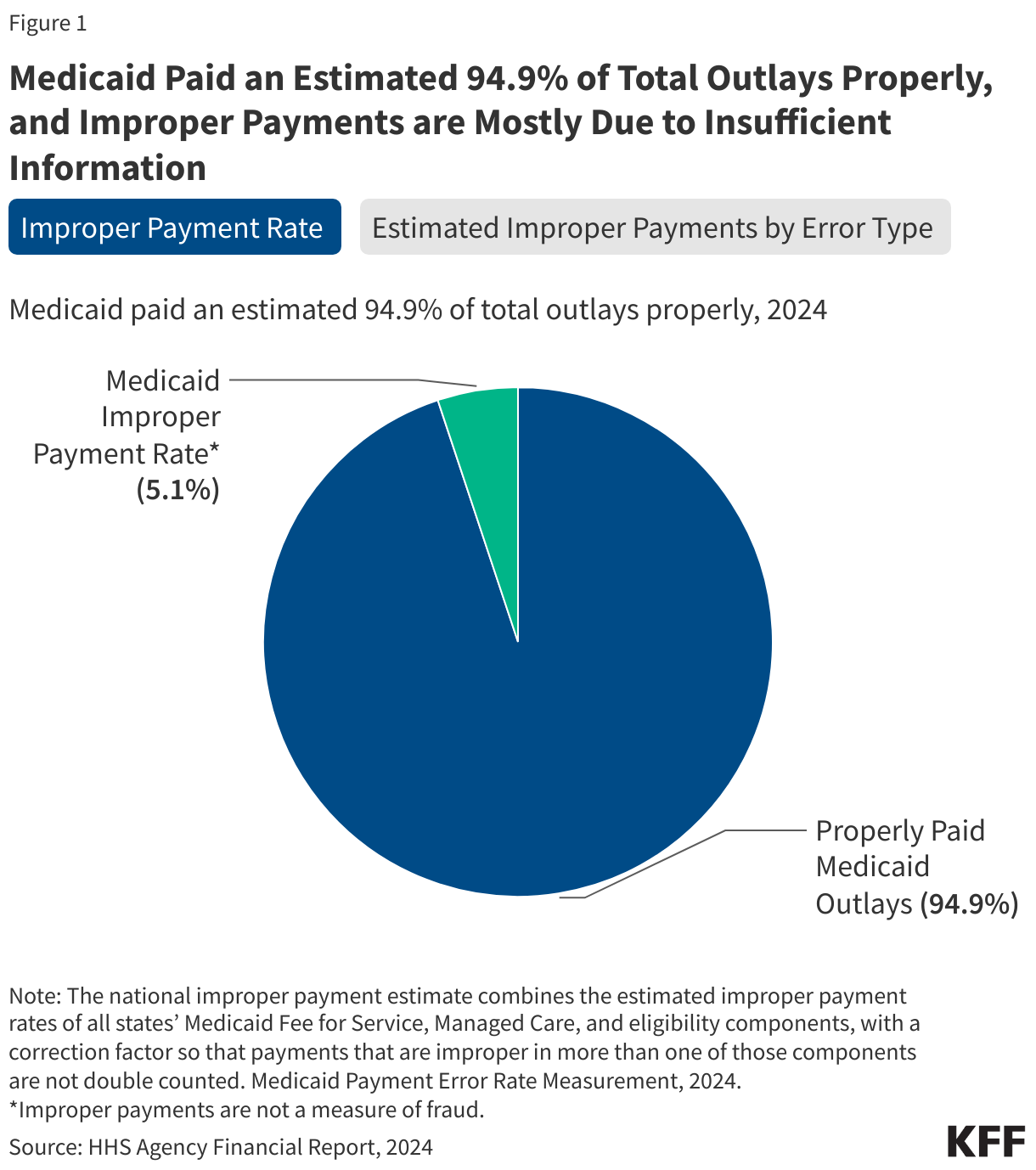
Most assessed contributions are considered “core” funding, meaning they are flexible funds that are often used to cover general expenses and program activities. Voluntary contributions, on the other hand, are often “specified” funds, meaning they are earmarked by donors for certain activities. While decades ago the majority of WHO’s revenue came from assessed contributions, in recent years voluntary contributions have comprised the larger share of WHO’s budget. For example, in the 2024-2025 budget period, voluntary contributions accounted for $5.9 billion or 83% of total revenue.2 See Figure 1.
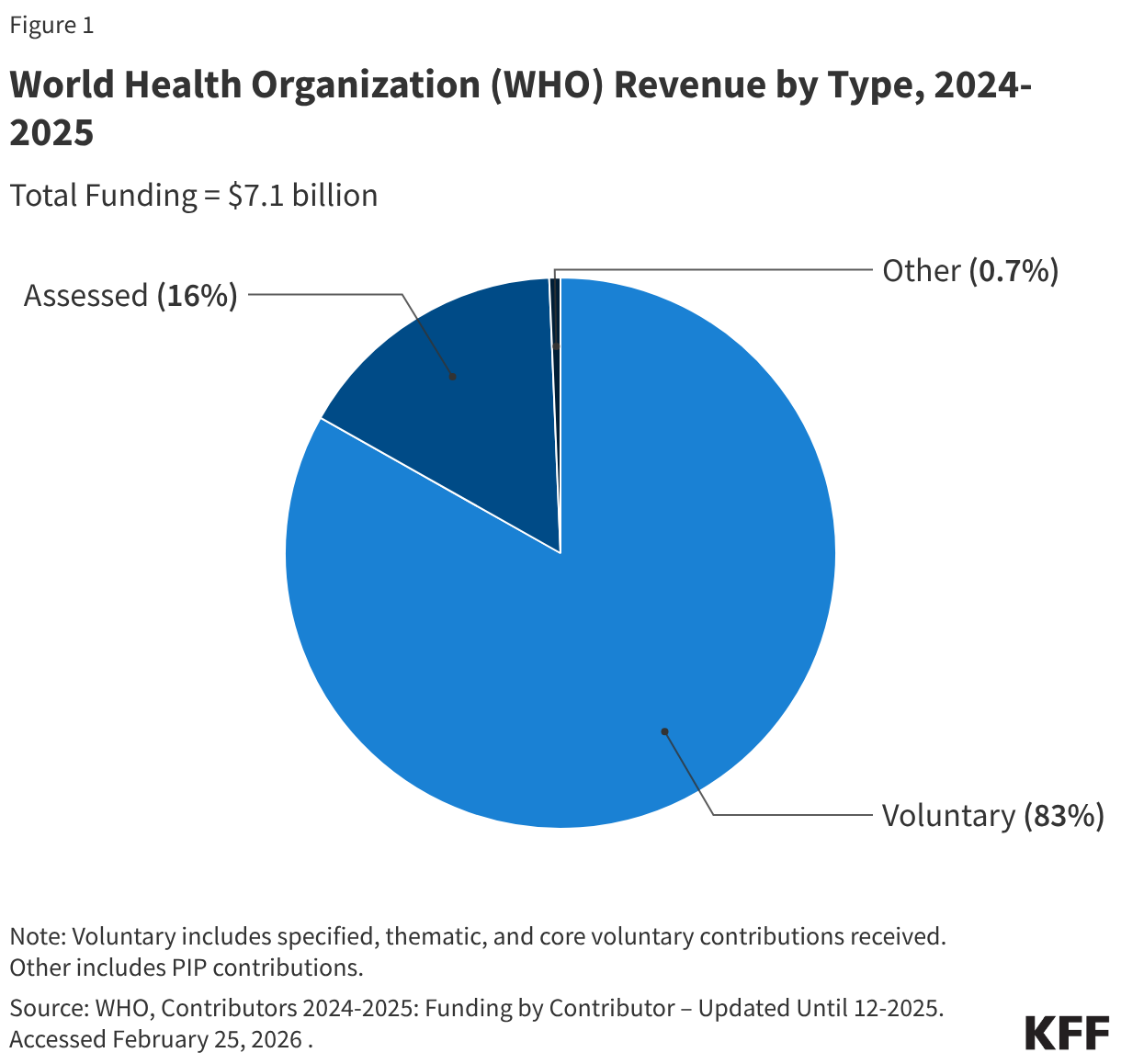
Reliance on voluntary, relatively inflexible funding has, in WHO’s view, hampered its operations and effectiveness. In 2022, member states, including the U.S. at that time, agreed in principle to move toward more predictable, flexible funding for WHO and to reduce the role of specified voluntary contributions. Since then, member states have agreed to increases in assessed contributions, with a 20% increase already implemented for the 2024-2025 biennium and another 20% increase approved for the current 2026-2027 biennium, with the goal of having 50% of WHO’s programme budget financed through assessed contributions by 2030.
In 2024, member states also approved the launch of WHO’s first-ever “investment round,” which mobilized additional funding for WHO over the subsequent four years. In its investment case for 2025-2028, WHO estimated it needs $11 billion to implement its general programme of work (GPW) over this period, but member state assessments (core contributions) are likely to amount to $4 billion, leaving a $7 billion gap to fill with voluntary contributions and other donations. Thus far, the investment round has generated $3.8 billion in additional donor pledges through 2028, or 53% of its original goal of $7 billion. The Biden administration did not announce any additional U.S. pledges to WHO through the investment round, and now the Trump administration has ceased U.S. funding for the organization.
Challenges
WHO faces a number of institutional challenges, including:
- a scope of responsibility that has expanded over time with little growth in core, non-emergency funding;
- an inflexible budget dominated in recent years by less predictable voluntary contributions often earmarked for specific activities;
- a cumbersome, decentralized, and bureaucratic governance structure;
- a dual mandate of being both a technical agency with health expertise and a political body where states debate and negotiate on sometimes divisive health issues; and
- ongoing budget and staffing challenges due to the loss of U.S. contributions and changes in the global financial landscape.
These and other challenges were particularly evident during and after perceived failures of the agency in the response to the Ebola epidemic in West Africa (2014-2015), and in the criticisms directed at WHO as it tried to help coordinate a global response to the COVID-19 pandemic. Even as many member states continue to support WHO and recognize its importance for global health, many also call for reforms to the organization that would help address its weaknesses. WHO itself supports reforms in several areas and has taken some internal reform actions.
U.S. Engagement with WHO
Prior to 2025, the U.S. government had long been engaged with WHO in multiple ways including through financial support, participation in governance and diplomacy, and joint activities (see below).
Current Status
In 2020, after the onset of the COVID-19 pandemic, the first Trump administration suspended U.S. financial support for WHO and initiated a process to withdraw the U.S. from membership in the organization.3Under the Biden administration, U.S. relations with WHO were re-established in January 2021, and U.S. funding to the organization was restored.4 However, President Trump announced on the first day of his second term, January 20, 2025, that the U.S. would be withdrawing from WHO membership and signed an Executive Order to once again suspend U.S. contributions to the organization, withdraw the U.S. from membership, and recall all U.S. personnel working with the organization. On January 22, 2025, the Trump administration submitted a formal letter of withdrawal to WHO and stated its withdrawal was completed on January 22, 2026. WHO, however, has responded by raising legal questions about whether the U.S. can complete any withdrawal without meeting its financial obligations such as assessed contributions it has not provided.
Since it began the process of withdrawing from WHO, the Trump administration has begun exploring alternative arrangements such as exploring the use of bilateral agreements to secure access to information about infectious disease outbreaks and has been considering building an alternative system for global outbreak detection and response, separate from WHO. Some U.S. states have expressed disagreement with the Trump administration’s withdrawal from WHO and are pursuing direct partnership with WHO themselves.
History
Financial Support
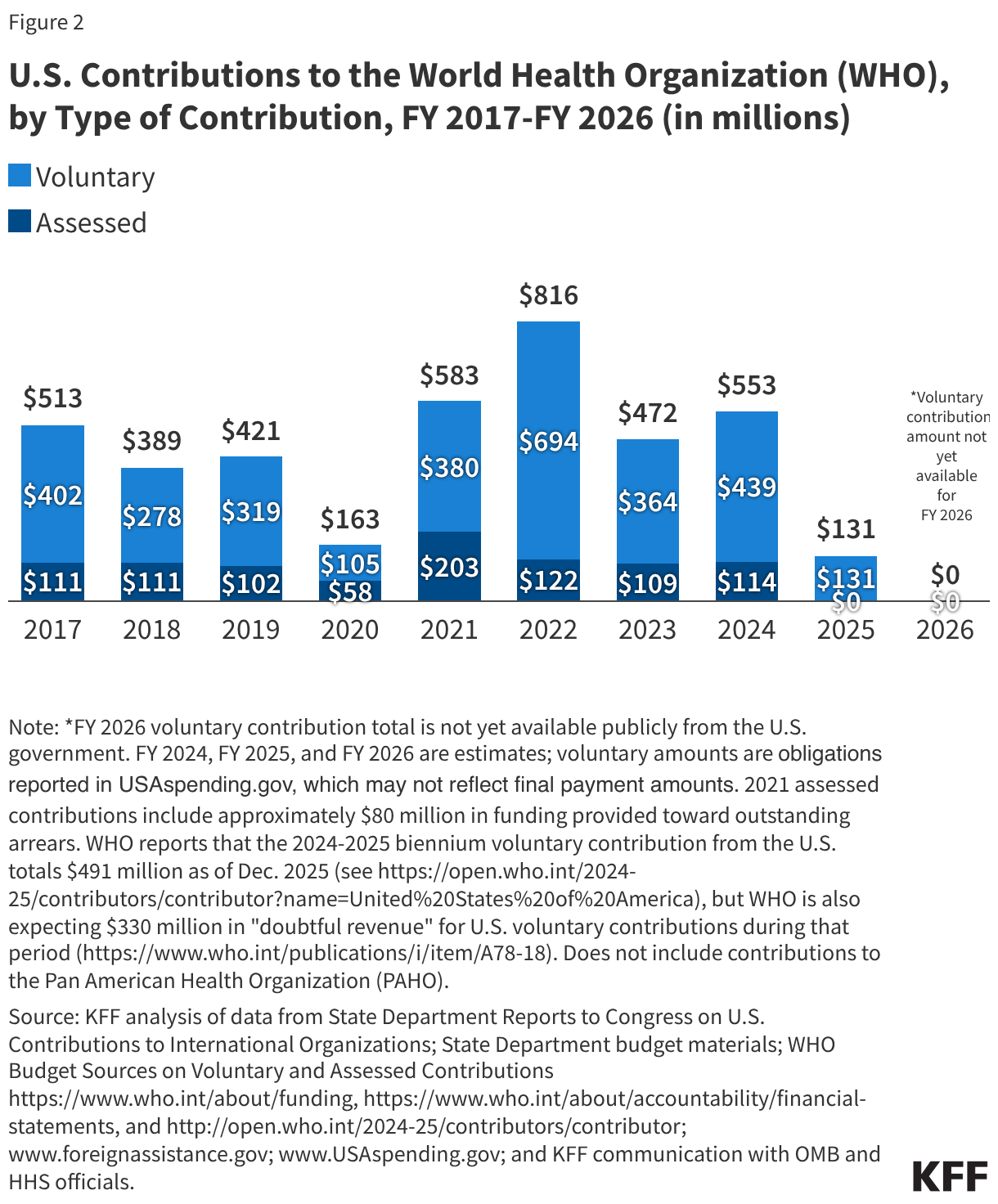
Prior to 2025, the U.S. government supported WHO through assessed and voluntary contributions (which have now been halted as directed by President Trump’s Executive Order). The U.S. had been the single largest contributor to WHO (though in the 2020-2021 period, when the first Trump administration withheld some U.S. funding during the COVID-19 pandemic, it was the third largest).
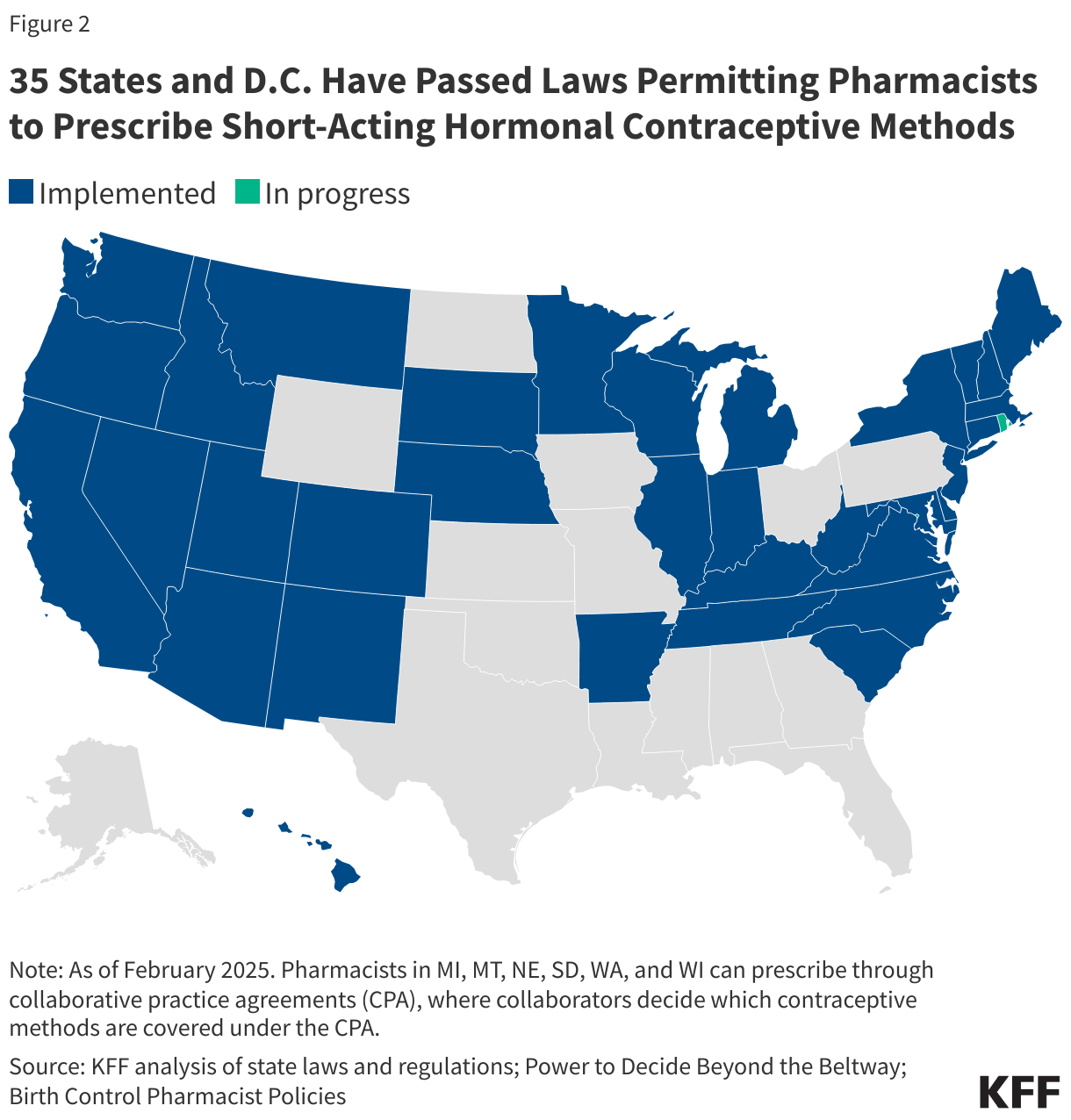
For many years, the assessed contribution for the U.S. has been set at 22% of all member state assessed contributions, the maximum allowed rate. Between FY 2017 and FY 2024, the U.S. assessed contribution was fairly stable, fluctuating between $109 million and $122 million (in FY 2019 and FY 2020 the U.S. actually paid less than its assessed amount, and in FY 2021 it paid more than that amount due to payments made toward outstanding arrears). See Figure 2.
Voluntary contributions for specific projects or activities, on the other hand, varied to reflect changing U.S. priorities and/or support during international crises. Over the past decade, U.S. voluntary contributions ranged from a low of $105 million in FY 2020 to a high of $694 million in FY 2022. Higher amounts of voluntary contributions were reflective of increased U.S. support for specific WHO activities such as emergency response. U.S. voluntary contributions also supported a range of other WHO activities such as polio eradication; maternal, newborn, and child health programs; mental health services for victims of torture and trauma; health coordination in COVID-19 response; and other infectious diseases.
For the 2022-2023 biennium period, WHO reported that U.S. assessed and voluntary contributions together represented 15.6% of WHO’s total revenue, making the U.S. the largest donor to WHO during that period.
Governance Activities
Prior to its withdrawal, the U.S. had been an active participant in WHO governance, including through the Executive Board and the World Health Assembly (WHA). This had included active engagement at the WHA, sending a large delegation each year that was typically led by a representative from the Department of Health and Human Services, with multiple other U.S. agencies and departments also participating and active participation in pandemic agreement negotiations and in the process to update and amend the IHR agreement. All official U.S. participation in WHO governance has been terminated.
Technical Support
The U.S. had, in the past, provided technical support to WHO through a variety of activities and partnerships. This included U.S. government experts and resources supporting research and reference laboratory work via WHO collaborating centres, WHO-based international partnerships such as the global outbreak and response network (GOARN), and advisory groups convened or overseen by WHO. U.S. government representatives had often been seconded to or had served as liaisons at WHO headquarters and WHO regional offices, working day-to-day with staff on technical efforts,5 though those personnel were recalled following the initiation of the U.S. withdrawal.
Partnering Activities
The U.S. had also worked in partnership with WHO before and during responses to outbreaks and other international health emergencies, including participating in international teams that WHO organized to investigate and respond to outbreaks around the world. For example, the U.S. worked with WHO and the broader multilateral response to the Ebola epidemic in West Africa that began in 2014, and U.S. scientists were part of the WHO delegation that visited China in February 2020 to assess its response to COVID-19. However, U.S. withdrawal from WHO has brought such coordination to an end.
Endnotes