States Focus on Quality and Outcomes Amid Waiver Changes: Results from a 50-State Medicaid Budget Survey for State Fiscal Years 2018 and 2019
Executive Summary
Medicaid covers one in five Americans, accounts for one in six dollars spent on health care in the United States, and makes up more than half of all spending on long-term services and supports. Medicaid is a state budget driver as well as the largest source of federal revenue to states. The program is constantly evolving in response to federal policy changes, the economy, and state budget and policy priorities. As states began state fiscal year (FY) 2019, the economy in most states was stable or improving and 36 states faced upcoming gubernatorial elections. With fewer budget pressures, a number of states reported expansions or enhancements to provider rates and benefits (including expansions for community-based long-term services and supports (LTSS) and behavioral health services). In addition, ballot initiatives in three states could result in adoption of the ACA Medicaid expansion. States also continue to focus on improvements in outcomes and value through delivery system changes and managed care requirements. In response to policy directions promoted by the Trump administration, an increasing number of states are pursuing demonstration waivers that include work requirements and the elimination or restriction of retroactive eligibility — policies that could result in enrollment declines (ES 1).

This report provides an in-depth examination of the changes taking place in Medicaid programs across the country. The findings are drawn from the 18th annual budget survey of Medicaid officials in all 50 states and the District of Columbia conducted by the Kaiser Family Foundation (KFF) and Health Management Associates (HMA), in collaboration with the National Association of Medicaid Directors (NAMD). This report highlights certain policies in place in state Medicaid programs in FY 2018 and policy changes implemented or planned for FY 2019. The District of Columbia is counted as a state for the purposes of this report. Given differences in the financing structure of their programs, the U.S. territories were not included in this analysis but a separate survey was fielded and results will be released in another report. Key findings include the following:
A growing number of states are pursuing federal waivers to add work requirements in Medicaid, impose new or increased premiums and lock-out periods, and waive retroactive eligibility
A growing number of states are implementing or planning Section 1115 waivers with policies that have or could result in enrollment declines, while three states (Idaho, Nebraska, and Utah) could adopt the ACA Medicaid expansion through ballot initiatives. Policies that have or are likely to result in enrollment declines are counted as restrictions in this report. Eligibility restrictions implemented in FY 2018 (by six states) or planned for implementation in FY 2019 (in 11 states) generally target broad Medicaid populations, including parents/caretakers and expansion adults. These changes are primarily occurring through Section 1115 waiver demonstration authority because they are not allowed under current law. Restrictions for FY 2018 or FY 2019 include eight states implementing or planning to implement work or community engagement requirements as a condition of Medicaid eligibility, eight states eliminating or restricting retroactive eligibility, and three states implementing or proposing lock-out periods for non-payment of premiums, failure to complete redetermination, and/or failure to timely report changes affecting eligibility. In contrast, with the exception of planned implementation of the ACA Medicaid expansion in Maine and Virginia in FY 2019, most Medicaid eligibility expansions for FY 2018 or FY 2019 are narrow and targeted to a limited number of beneficiaries. Most states are working with corrections agencies and with local jails to facilitate Medicaid enrollment prior to release and the majority of states do not terminate Medicaid coverage for enrollees who become incarcerated.
What to Watch: Three states have proposals to adopt the Medicaid expansion on the November 2018 ballot (Idaho, Nebraska, and Utah) and many states have Section 1115 waivers pending or under development that could be implemented after FY 2019 and would impact eligibility, if approved by CMS.
Risk-based managed care continues to be the predominant delivery system for Medicaid services, and states are focused on implementing alternative payment models and improving quality within MCOs. Among the 39 states with comprehensive risk-based managed care organizations (MCOs), 33 states reported that 75% or more of their Medicaid beneficiaries were enrolled in MCOs as of July 1, 2018. Although many states still carve-out behavioral health services from MCO contracts, movement to carve-in these services continues. Nearly all states have managed care quality initiatives in place such as pay for performance or capitation withholds and an increasing number of states (23 in FY 2018) set a target percentage of MCO provider payments that must be in alternative payment models (APMs).
What to Watch: Alaska and Arkansas reported plans to implement an MCO program for the first time in FY 2019. States continue to focus on improving value, quality, and outcomes through managed care arrangements. CMS has announced plans to release revised Medicaid managed care regulations for public comment later this fall.
States are working to address social determinants of health both within and outside of MCO contract requirements. Medicaid programs have been expanding their use of other service delivery and payment reform models to achieve better outcomes and lower costs. Forty-three states had one or more delivery system or payment reform initiatives in place in FY 2018 (e.g., patient-centered medical homes (PCMHs), ACA Health Homes, accountable care organizations (ACOs), episode of care payments, or delivery system reform incentive programs (DSRIPs)).
What to Watch: About one-third of the states reported a wide variety of initiatives implemented in FY 2018 or planned for FY 2019 that address the social determinants of health (SDOH) outside of managed care and more than one-third reported collecting or plans to collect SDOH data from various sources including screenings and assessments, data collected for other state programs, claims data, beneficiary surveys, or as part of a care management or home visiting program.
Nearly all states in FY 2018 (46 states) and FY 2019 (48 states) are employing one or more strategies to expand the number of people served in home and community-based settings. Almost all states continue to report using home and community-based services (HCBS) waivers and/or state plan options (i.e., 1915(c), 1115, 1915(i), and 1915(k)) to serve more individuals in the community. As of July 1, 2018, 24 states covered LTSS through one or more capitated managed care arrangements (“MLTSS”). Pennsylvania introduced MLTSS in FY 2018, with a plan to phase-in statewide over time. Virginia ended its Financial Alignment Demonstration (FAD) but adopted statewide MLTSS for a broader population, including dual eligible individuals. To address challenges in finding and retaining LTSS direct care workers, a number of states reported wage increases for these workers in FY 2018 and/or FY 2019.
What to Watch: Housing-related supports remain an important part of state LTSS benefits, even as Money Follows the Person (MFP) grant funds expire. While 30 states reported that they expect to continue to offer housing-related supports after MFP funds are exhausted, about half of states reported plans to discontinue at least some housing-related services or administrative functions when MFP ends.
In FY 2018 and FY 2019, with favorable economic conditions in most states, more states made or are planning provider rate increases compared to restrictions, and there is little new activity around provider taxes. More rate increases relative to decreases holds true across major provider types, with the exception of inpatient hospitals (where inpatient hospital rate “restrictions” are primarily rate freezes that are counted as restrictions in this report). About half of MCO states (21 of 39) require MCO payments to some or all types of providers to follow percent or level changes in fee-for-service (FFS) rates. Twenty-seven states require minimum MCO payments (rate floors) for some provider types, and five states reported minimum MCO payment requirements for all types of Medicaid providers. All states except Alaska rely on provider taxes and fees to fund a portion of the non-federal share of the costs of Medicaid. Two states indicated plans for new provider taxes in FY 2019, including Virginia that plans a new hospital provider tax to finance state costs of the newly adopted Medicaid expansion.
What to Watch: As enrollees are predominantly in MCOs, the significance of changes in FFS payment rates is difficult to assess without a better understanding of how changes in FFS rates affect changes in MCO rates paid to providers. Twenty-nine states have at least one provider tax that is at or above 5.5% of net patient revenues (close to the maximum safe harbor threshold of 6%). Therefore, federal action to lower that threshold as proposed in the past would have financial implications for many states.
Positive economic conditions and state priorities resulted in a number of states increasing benefits, particularly for mental health and substance use disorder (SUD) treatment. A total of 19 states expanded or enhanced covered benefits in FY 2018 and 24 states plan to add or enhance benefits in FY 2019. The most common benefit enhancements reported were for mental health/SUD services (including waiver of the IMD exclusion). A handful of states also reported expansions related to dental services, telemonitoring/telehealth, physical or occupational therapy services, and screening or home visiting services for pregnant women and/or children.
What to Watch: Medicaid continues to play an important role in addressing the opioid epidemic and more broadly in connecting Medicaid beneficiaries to behavioral health services. Going forward, it will be important to follow trends and innovations in how states use Medicaid to increase access to behavioral health services and contain the growth of the opioid and broader SUD crisis.
States continue to focus on cost containment efforts to address rising prescription drug costs and on pharmacy benefit management strategies to address the opioid crisis. Prescription drug costs continue to exert pressure on Medicaid spending, and most states identified specialty and high-cost drugs (individually or in general) as the most significant driver of these costs. Many states reported implementing or making changes to a wide variety of prescription drug cost containment initiatives in FY 2018 or FY 2019, especially initiatives to generate greater rebate revenue and implement new utilization controls (e.g., prior authorization requirements, step therapy, other clinical criteria, or dose optimization). Almost all MCO states generally carve the pharmacy benefit into managed care, and many MCO states are moving to align MCO pharmacy policies with FFS policies. In FY 2018, all states report implementing one or more FFS pharmacy management strategies targeted at opioid harm reduction, including quantity limits, clinical criteria claim system edits, step therapy, other prior authorization requirements, and requirements for Medicaid prescribers to check their states’ Prescription Drug Monitoring Program before prescribing opioids to a Medicaid patient. States continue to increase access to Medication Assisted Treatment (MAT) for opioid use disorder, and 38 states reported coverage of methadone in FY 2018.
What to Watch: Several states noted the emerging cost challenge posed by gene therapies and immunotherapies like “CAR-T” (Chimeric Antigen Receptor-T cell) therapies,1 designated by CMS as covered outpatient drugs. While states are expanding coverage of MAT, many reported access challenges, especially in rural areas.
Challenges and Priorities FY 2019 and Beyond: Continuing to tackle the opioid epidemic is a key priority for states in FY 2019 and beyond. New federal legislation expected to be signed into law as this report was being finalized, the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities (SUPPORT) Act, contains a number of provisions related to Medicaid’s role in helping states provide coverage and services to people who need SUD treatment,2 particularly those needing opioid use disorder (OUD) treatment. These provisions include the ability to use federal Medicaid funds for services in “institutions for mental disease” (IMDs) for nonelderly adults for up to 30 days from October 1, 2019 to September 30, 2023; required coverage of all FDA-approved drugs for medication-assisted treatment (MAT) as well as counseling and behavioral therapy services from October 2020 through September 2025; required suspension of Medicaid eligibility for individuals under age 21 or former foster care youth up to age 26 while incarcerated, and restoration of coverage upon release; creation of new demonstrations to help states increase Medicaid SUD provider capacity; and tighter prescription drug oversight.
In addition, states reported a wide variety of other priorities for FY 2019 and beyond, including implementing managed care, payment, and delivery system reform initiatives; undertaking major information technology system procurements and upgrades; pursuing new Section 1115 demonstration waivers, and managing their Medicaid budgets. The trajectory of the economy, the direction of federal policies around Medicaid Section 1115 waivers, and the outcomes of state and federal elections in November 2018 will be factors that shape Medicaid in FY 2019 and beyond.
Report: Introduction
Medicaid now provides health insurance coverage to one in five Americans and accounts for nearly one-sixth of all U.S. health care expenditures.3 The Medicaid program constantly evolves due to changes in federal and state policies, the economy, and other state budget and policy priorities. As of July 26, 2018, all states had enacted a new or revised budget for FY 2019 with only three states enacting a budget after the fiscal year began.4 ,5 This represented a significant improvement from the prior year when a number of states called special sessions in 2017 to complete their FY 2018 budgets and 11 states started FY 2018 without a fully enacted budget.6 A stable economy and improvements in state revenue growth resulted in surpluses in many states at the beginning of FY 2019 when this survey was conducted.7
Report findings are drawn from the 18th annual budget survey of Medicaid officials in all 50 states and the District of Columbia conducted by the Kaiser Family Foundation (KFF) and Health Management Associates (HMA), in collaboration with the National Association of Medicaid Directors (NAMD). (Previous reports are archived here.8 ) This year’s KFF/HMA Medicaid budget survey was conducted from June through September 2018 via a survey sent to each state Medicaid director in June 2018 and then a follow-up telephone interview. An acronym glossary and the survey instrument are included as appendices to this report.
The District of Columbia is counted as a state for the purposes of this report; the counts of state policies or policy actions that are interspersed throughout this report include survey responses from the 51 “states” (including DC). All 50 states and DC completed surveys and participated in telephone interview discussions between July and September 2018.9 Given differences in the financing structure of their programs, the U.S. territories were not included in this analysis but a separate survey was fielded and results will be released in another report.
This report examines Medicaid policies in place or implemented in FY 2018, policy changes implemented at the beginning of FY 2019, and policy changes for which a definite decision has been made to implement in FY 2019 (which began for most states on July 1, 201810 ). Policies adopted for the upcoming year are occasionally delayed or not implemented for reasons related to legal, fiscal, administrative, systems, or political considerations, or due to delays in approval from CMS. Key findings of this survey, along with state-by-state tables, are included in the following sections of this report:
Report: Eligibility And Premiums
Key Section Findings
Since 2014, most major Medicaid eligibility policy changes have been related to adoption of the ACA Medicaid expansion. Thirty-two states have implemented the expansion to date and two additional states adopted the expansion in FY 2018 and plan to implement the policy in FY 2019 (Maine and Virginia). Other Medicaid eligibility expansions for FY 2018 or FY 2019 were narrow and targeted to a limited number of beneficiaries. In contrast, eligibility restrictions implemented in FY 2018 (by six states) or planned for implementation in FY 2019 (in 11 states) generally target broader Medicaid populations including expansion adults and parents/caretakers. Policies that have or are likely to result in enrollment declines are counted as restrictions in this report. The vast majority of states implementing or planning eligibility policies that are counted as restrictions in FY 2018 or FY 2019 are doing so through Section 1115 waiver authority, whereas most states implementing or planning eligibility expansions are doing so through state plan amendment (SPA) authority.
What to watch:
- Three states have proposals to adopt the Medicaid expansion on the November 2018 ballot (Idaho, Nebraska, and Utah).
- In FY 2019, eleven states are planning to implement Medicaid Section 1115 waivers with policies that would result in eligibility restrictions including the addition of work requirements and the elimination or limitation of retroactive eligibility. Some states indicated that significant administrative resources will be needed to implement these initiatives, including one-time costs such as systems modifications and ongoing annual costs such as increased staffing.
- Most states are working with corrections agencies and with local jails to facilitate Medicaid enrollment prior to release and the majority of states do not terminate Medicaid coverage for enrollees who become incarcerated. The SUPPORT Act would be prohibit states from terminating Medicaid eligibility for an individual under age 21 or former foster care youth up to age 26 while incarcerated beginning in October 2019.
- Seven states reported planned changes related to new or increased premiums in FY 2019, six of which are through Section 1115 waivers.
Tables 1, 2, and 3 at the end of this section include additional details on eligibility, premium, and corrections-related policy changes in FY 2018 and FY 2019.
Changes to Eligibility Standards
Eligibility Restrictions
A growing number of states are pursuing Section 1115 waivers that include policies that would result in eligibility restrictions in FY 2018 and FY 2019 (Exhibit 1). Some of these policies are in response to January 2018 CMS guidance11 indicating the agency’s support for Section 1115 waiver proposals that would require certain Medicaid enrollees to meet a work requirement in order to maintain coverage. Policies that have or are likely to result in enrollment declines are counted as restrictions in this report.
| Exhibit 1: Eligibility Restrictions by Policy Authority | ||||
| FY 2018 | FY 2019 | |||
| SPA | 2 States | CT, NM | 0 States | |
| Section 1115 Waiver | 4 States | AR, IA, IN, UT | 11 States | AL*, FL*, IN, KY*^, MA, ME*, MI*, NH, NM*, OH*, SD* |
| *Indicates the Section 1115 Waiver has not yet been approved by CMS.^CMS’ approval of Kentucky’s waiver authorizing FY 2019 restrictions was set aside by a court order in June 2018 that also remanded the waiver to CMS for reconsideration regarding how the waiver would meet the medical assistance objectives of the Medicaid statute. | ||||
Six states reported implementing restrictions in FY 2018 and 11 states reported restrictions already implemented or planned for implementation in FY 2019 (Exhibit 1 and Table 1). This year’s survey reports changes that states plan to implement in FY 2019, even if the changes are in Section 1115 waiver proposals that are still pending approval12 at CMS. Waiver provisions that states plan to implement in FY 2020 or after are described later in the “Challenges and Priorities” section of this report.
Eight states reported implementing or plans to implement work requirements under Section 1115 waiver authority in FY 2018 or FY 2019 (Table 2).13 These policies generally require beneficiaries to verify their participation in approved activities, such as employment, job search, or job training programs, for a certain number of hours per week or month to receive health coverage or qualify for an exemption. Data show, however, that most Medicaid enrollees are already working or would qualify for exemptions from these requirements, yet these individuals would still need to navigate a reporting or exemption process to retain their Medicaid coverage. In this report, work requirement policies are counted based on the initial date of implementation rather than the date on which the first coverage terminations will occur.
- Three states have approved waivers to implement a work requirement, including one state (Arkansas) that implemented the policy in June of FY 2018 and two states (Indiana and New Hampshire) that plan to implement in FY 2019.
- Five additional states reported pending work requirement proposals that they plan to implement in FY 2019. Four of these states (Alabama, Maine, Ohio, and South Dakota) have proposals pending approval by CMS and one state (Kentucky) received approval for a work requirement waiver that was set aside by court order. That waiver is now back with CMS for reconsideration (see the Kentucky HEALTH waiver box below). Kentucky’s waiver is referred to as “pending” throughout the rest of this report.
Many of these states reported new administrative requirements or costs associated with implementation of work requirement policies, including those related to vendor contracts for call centers, systems modifications, increased state and local office staff, outreach to beneficiaries and providers, and increased MCO personnel costs.
Kentucky HEALTH Waiver
In January 2018, Kentucky received initial CMS approval for the Kentucky HEALTH Section 1115 waiver. The waiver included several provisions approved for the first time in Medicaid — a work requirement, monthly premiums up to 4% of income, and coverage lock-outs for failure to timely renew eligibility or timely report changes affecting eligibility — as well as heightened cost-sharing for non-emergency ER use, elimination of retroactive eligibility, and elimination of non-emergency medical transportation. A court order issued in Stewart v. Azar on June 29, 2018, however, set aside the CMS approval and remanded the waiver to CMS for reconsideration regarding how the Kentucky waiver would meet the medical assistance objectives of the Medicaid statute. At the time of the survey, CMS had completed an additional federal public comment period and the state was preparing for FY 2019 implementation, but the waiver remained under consideration at CMS.
Eight states indicated that they have eliminated or propose to eliminate or restrict retroactive eligibility for one or more groups in FY 2018 and FY 2019 (Table 2).14 In FY 2018, Arkansas limited retroactive eligibility from 90 to 30 days, Iowa eliminated retroactive eligibility for all groups other than children under age one and pregnant women (but restored retroactive eligibility for nursing facility residents in July 2018), and Utah eliminated retroactive eligibility for Primary Care Network (PCN) waiver adults. In FY 2019, five states (Florida, Kentucky, Maine, New Hampshire, and New Mexico) plan to eliminate retroactive eligibility or limit it to a single month for most enrollees.
Other examples of reported eligibility restrictions in FY 2018 or FY 2019 include:
- In FY 2018, as part of their HIP 2.0 waiver renewal (a waiver that already included other eligibility restrictions), Indiana15 imposed a three-month lock-out from coverage on expansion adults who fail to comply with redetermination. Enrollees who do not verify eligibility at renewal are disenrolled but can re-enroll without a new application if they provide necessary documentation within 90 days. After 90 days, individuals are subject to a three-month lock-out before they can re-enroll.16
- In FY 2019, New Mexico plans to implement proposals included in a pending waiver application to disenroll and lock-out expansion adults who do not pay required Medicaid premiums and a “reasonable promptness” waiver that would delay coverage until the first day of the first month following receipt of required premiums.
- In FY 2019, Maine17 plans to implement (if their pending waiver is approved) several restrictions on their traditional Medicaid populations in addition to their planned waiver of retroactive eligibility and the work requirement. These include applying a $5,000 asset test to all coverage groups that currently do not have an asset test and eliminating hospital presumptive eligibility for all coverage groups. The state’s pending waiver application proposes to implement these initiatives within six months of demonstration approval.
Eligibility expansions
Aside from planned implementation of the ACA Medicaid expansion in two states in FY 2019, most other eligibility expansions for FY 2018 and FY 2019 are narrow in scope. Overall, ten states implemented Medicaid eligibility expansions in FY 2018, and seven states plan to implement expansions in FY 2019.
Two states (Maine and Virginia) plan to implement the ACA Medicaid expansion in FY 2019. These expansions will add to the 32 states that had already implemented the ACA Medicaid expansion as of July 2018 (Figure 1). In Virginia, the expansion was adopted as part of the FY 2019-2020 Medicaid budget, with implementation planned for January 1, 2019. Maine voters adopted the Medicaid expansion through a ballot initiative in November 2017 that required submission of a state plan amendment (SPA) within 90 days and implementation of expansion within 180 days of the measure’s effective date.18 After failing to meet the SPA submission deadline (April 3, 2018), Maine’s Governor complied with a Maine Supreme Judicial Court order to submit an expansion SPA on September 4, 2018 but also sent a letter to the federal government asking CMS to reject the SPA. The expansion had not yet been implemented at the time of the survey, and implementation will fall to the new governor after the November 2018 election.
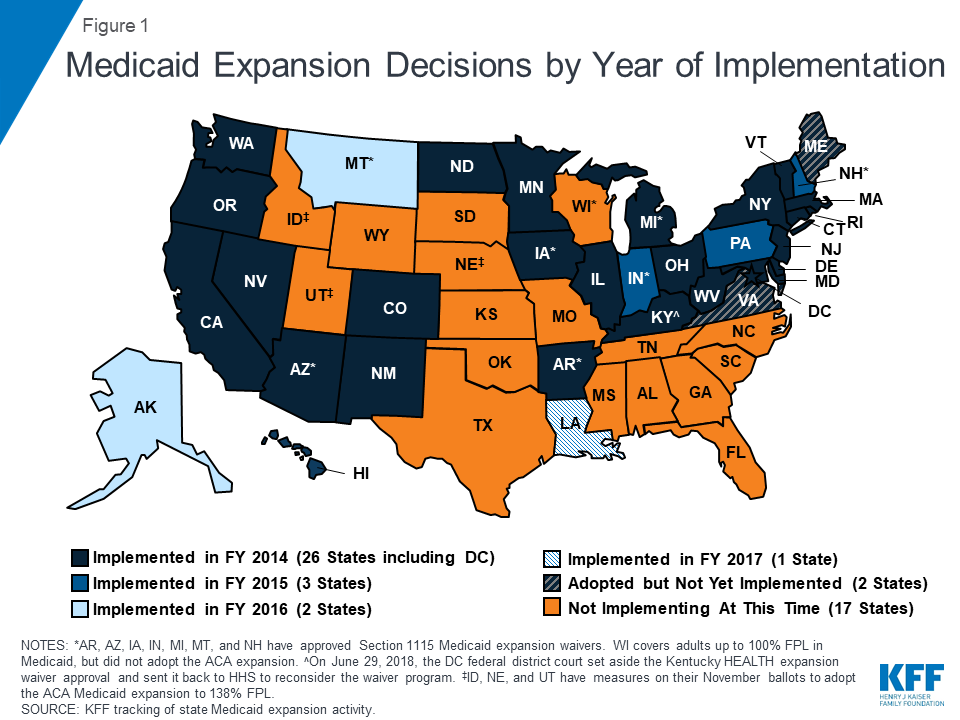
Three additional states (Idaho, Nebraska, and Utah) will have referendum initiatives to adopt the Medicaid expansion on the November 2018 ballot. Montana also has an expansion-related initiative for voters on the November ballot that would eliminate the state’s June 2019 expansion program sunset date and raise tobacco taxes to fund the state’s share of expansion costs.
| Exhibit 2: Eligibility Expansions by Policy Authority | ||||
| FY 2018 | FY 2019* | |||
| SPA | 8 States | AR, CO, ID, MO, PA, SC, UT, VT | 6 States | CT, MD, ME, MO, NJ, VA |
| Section 1115 Waiver | 3 States | DE, UT, VA | 0 States | |
| *Iowa’s FY 2019 eligibility expansion that reinstated 3-month retroactive eligibility for Medicaid-eligible nursing facility residents did not require new Section 1115 or SPA authority. | ||||
Ten states implemented more narrow eligibility expansions in FY 2018 and five states plan to implement more limited expansions in FY 2019. In contrast to eligibility restrictions, which states are primarily implementing under Section 1115 authority, most states implementing or planning eligibility expansions in FY 2018 or FY 2019 are doing so through optional authority using a state plan amendment (SPA) (Exhibit 2). Some examples of these other expansions include the following:
- In FY 2018 under Section 1115 waiver authority, Utah implemented coverage for individuals with income below 5% of the federal poverty level (FPL) who are chronically homeless, justice-involved, or in need of substance use and/or mental health treatment. The state also has approval to implement 12 month continuous eligibility for this population.
- Missouri increased asset limits in both FY 2018 and FY 2019 for aged and disabled beneficiaries and Vermont increased asset limits in FY 2018 for their Working People with Disabilities Program.
- In FY 2018, Arkansas and South Carolina eliminated the five-year waiting period on Medicaid eligibility for lawfully-residing immigrant children and pregnant women.19
Premiums
The Medicaid statute generally does not allow states to charge premiums to most Medicaid beneficiaries. Historically, premiums were limited to special higher income categories of beneficiaries such as expanded Medicaid for the working disabled. However, some states have obtained waiver authority to charge higher premiums and/or copayments than otherwise allowed.
Only Indiana reported activity related to Medicaid premiums in FY 2018 (Table 2). Effective January 1, 2018, the state changed its monthly premiums (which are paid into a health account) for expansion adults from 2% of income to tiered amounts based on income ranges (expressed as a percentage of the FPL). The result is reduced fluctuation of premiums. This report considers the change neutral from the beneficiary’s perspective.
Seven states report proposed implementation of new premium programs or changes to existing premiums for FY 2019 (Table 2). Three of these states (Idaho, Indiana, and Iowa) have already received approval for these changes, while they are still pending as part of Section 1115 waiver requests in four states (Kentucky, Maine, Michigan, and New Mexico). Notable FY 2019 premium policy changes include an approved waiver provision in Indiana to implement a premium surcharge for tobacco users and a pending waiver request in Michigan to require premiums of up to 5% of income (a higher amount than CMS has ever approved for any state) for expansion adults after 48 cumulative months of expansion eligibility.
Coverage Initiatives for the Criminal Justice Population
In recent years, many states have implemented new policies to connect individuals involved with the criminal justice system to Medicaid, as the Medicaid expansion extended new coverage to these individuals in many states (especially childless adults who were not previously eligible in most states). Connecting these individuals to health coverage20 can facilitate their integration back into the community upon release. While individuals may be enrolled in Medicaid while they are incarcerated, Medicaid cannot cover the cost of their care during incarceration, except for inpatient services. Nearly all states have policies in place to cover inpatient care under Medicaid for eligible, incarcerated individuals (Exhibit 3 and Table 3). Most states are also working with corrections agencies and with local jails to facilitate Medicaid enrollment prior to release. The majority of states do not terminate Medicaid coverage for enrollees who become incarcerated: states either maintain the eligibility of the incarcerated individual with benefits limited to inpatient hospital care, or they suspend eligibility. When coverage is suspended, it can be reinstated more easily and quickly upon release from incarceration or when an inpatient hospital stay occurs.
As this report was being finalized, the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities (SUPPORT) Act was expected to be signed into law. Beginning in October 2019, the Act would prohibit states from terminating Medicaid eligibility for an individual under age 21 or former foster care youth up to age 26 while incarcerated. States would also be required to redetermine eligibility prior to release without requiring a new application and restore coverage upon release.
While both Medicaid expansion and non-expansion states have adopted strategies to connect justice-involved individuals to Medicaid coverage, these initiatives affect many more people in expansion states compared to non-expansion states where Medicaid eligibility for adults remains restrictive.
| Exhibit 3: Coverage Initiatives for the Criminal Justice Population in FY 2018 and/or FY 2019 (# of States) | |||
| Select Medicaid Coverage Policies for the Criminal Justice Population | Jails | Prisons* | Parolees |
| Medicaid coverage for inpatient care provided to incarcerated individuals | 42 | 48 | N/A |
| Medicaid outreach/assistance strategies to facilitate enrollment prior to release from incarceration or for parolees | 34 | 39 | 26 |
| Eligibility maintained or suspended (rather than terminated) for Medicaid enrollees who become incarcerated^ | 36 | 38 | N/A |
| ^States that continue Medicaid eligibility for incarcerated individuals but limit covered benefits to inpatient hospitalization are also included in the count of states that suspend eligibility. *The District of Columbia has jails but not a prison system. However, DC is counted under Medicaid outreach/assistance strategies because some individuals who serve prison terms outside of DC may be placed in residential re-entry centers upon returning to DC and may apply for Medicaid to access coverage for 24-hour inpatient care and to facilitate enrollment prior to release. | |||
Table 1: Changes to Eligibility Standards in all 50 States and DC, FY 2018 and FY 2019
| Eligibility Standard Changes | ||||||
| States | FY 2018 | FY 2019 | ||||
| (+) | (-) | (#) | (+) | (-) | (#) | |
| Alabama | X | |||||
| Alaska | ||||||
| Arizona | ||||||
| Arkansas | X | X | X | |||
| California | ||||||
| Colorado | X | |||||
| Connecticut | X | X | ||||
| Delaware | X | |||||
| DC | X | |||||
| Florida | X | |||||
| Georgia | ||||||
| Hawaii | ||||||
| Idaho | X | |||||
| Illinois | ||||||
| Indiana | X | X | X | |||
| Iowa | X | X | ||||
| Kansas | ||||||
| Kentucky | X | |||||
| Louisiana | ||||||
| Maine | X | X | ||||
| Maryland | X | |||||
| Massachusetts | X | |||||
| Michigan | X | |||||
| Minnesota | ||||||
| Mississippi | ||||||
| Missouri | X | X | ||||
| Montana | ||||||
| Nebraska | ||||||
| Nevada | ||||||
| New Hampshire | X | |||||
| New Jersey | X | |||||
| New Mexico | X | X | ||||
| New York | X | |||||
| North Carolina | ||||||
| North Dakota | ||||||
| Ohio | X | |||||
| Oklahoma | ||||||
| Oregon | ||||||
| Pennsylvania | X | |||||
| Rhode Island | ||||||
| South Carolina | X | |||||
| South Dakota | X | |||||
| Tennessee | ||||||
| Texas | ||||||
| Utah | X | X | ||||
| Vermont | X | |||||
| Virginia | X | X | ||||
| Washington | ||||||
| West Virginia | ||||||
| Wisconsin | ||||||
| Wyoming | ||||||
| Totals | 10 | 6 | 3 | 7 | 11 | 1 |
NOTES: From the beneficiary’s perspective, eligibility expansions or policies likely to increase Medicaid enrollment are denoted with (+), eligibility restrictions or policies likely to decrease enrollment are denoted with (-), and neutral changes are denoted with (#). This table captures eligibility changes that states have implemented or plan to implement in FY 2018 or FY 2019, including changes that are part of pending Section 1115 waivers. For pending waivers, only provisions planned for implementation before the end of FY 2019 (according to waiver application documents and/or interviews with state Medicaid staff) are counted in this table. Waiver provisions in pending waivers that states plan to implement in FY 2020 or after are not counted here. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||
Table 2: States Reporting Eligibility21 and/or Premium22 Changes in FY 2018 and FY 201923
| State | Fiscal Year | Eligibility Changes |
| Alabama | 2019 | Parents & Caretaker Relatives (-): Pending Section 1115 Waiver: Add a work/community engagement requirement for non-disabled, non-pregnant individuals under age 60 (75,000 individuals). |
| Arkansas | 2018 | Expansion Adults (-) Approved Sec. 1115 Waiver: Limit retroactive coverage from 90 to 30 days. Expansion Adults (-) Approved Sec. 1115 Waiver: Work requirement for expansion adults (40,000 individuals) (first case closures occurred 9/1/2018). Expansion Adults (#) Approved Sec. 1115 Waiver: End premium assistance program for employer sponsored insurance and transition individuals to QHP coverage (40 individuals). Children and Pregnant Women (+): Implement the CHIPRA option to eliminate the 5-year waiting period on Medicaid eligibility for lawfully-residing immigrant children and pregnant women. |
| Colorado | 2018 | Aged & Disabled (+): Medicaid buy-in option for individuals in support living services, spinal cord injury, & brain injury waivers (40 individuals). |
| Connecticut | 2018 | Parents & Caretaker Relatives (-): Reduce the income threshold for Husky A from 155% FPL to 138% FPL (12,000 individuals). |
| 2019 | Parents & Caretaker Relatives (+): Increase the income threshold for Husky A from 138% FPL to 155% FPL (12,000 individuals). | |
| Delaware | 2018 | Former Foster Youth (+) Approved Sec. 1115 Waiver: Add coverage for individuals who were in foster care and on Medicaid in another state at the time they aged out of the foster care system (under age 26 and under 133% FPL) (3 individuals). |
| District of Columbia | 2019 | Medically Needy (#): Clarification of Medically Needy eligibility criteria (with regard to countable medical expenditures) (7,000 individuals). |
| Florida | 2019 | Non-Pregnant Adults (-): Pending Sec. 1115 Waiver:Discontinue retroactive eligibility beyond the current application month. |
| Idaho | 2018 | Children (+): Cover children with serious emotional disturbance (SED) in families with income between 185% and 300% FPL (SED YES program) (2,000 to 10,000 children). |
| 2019 | Premiums (New): Children enrolled in the 1915(i) SED YES program will be subject to a premium in early 2019. | |
| Indiana | 2018 | Expansion Adults (-) Approved Sec. 1115 Waiver: Three-month lock-out of coverage following a 90-day period of disenrollment for failure to comply with redetermination. Expansion Adults (#) Approved Sec. 1115 Waiver: End HIP Link premium assistance program for employer-sponsored insurance (enrollees will be moved to other HIP 2.0 coverage). Parents & Caretaker Relatives (#) Approved Sec. 1115 Waiver: Transitional Medical Assistance (TMA) is now for families in which a qualified HIP low-income parent/caretaker would otherwise fail financial eligibility due to new or increased earned income from a job or from self-employment exceeding the 133% FPL MAGI income limit. Premiums (Neutral) for Expansion and Other Adults Approved Sec. 1115 Waiver: Monthly POWER Account contributions are now tiered based on income. The tiered amounts, effective January 1, 2018, are $1.00, $5.00, $10.00, $15.00, and $20.00. |
| 2019 | Expansion and Other Adults (-) Approved Sec. 1115 Waiver: Work/community engagement requirement, phased implementation beginning 1/1/2019. Premiums (New) for Expansion Population Approved Sec. 1115 Waiver: Add a 50% premium surcharge for tobacco users beginning in the second year of enrollment. | |
| Iowa | 2018 | All Groups but Pregnant Women and Children Under Age 1 (-) Approved Sec. 1115 Waiver: Eliminate retroactive eligibility. |
| 2019 | Nursing Facility Residents (+): Reinstate 3-month retroactive eligibility for Medicaid-eligible nursing facility residents, effective July 1, 2018. Premiums (New) Approved Section 1115 Waiver: Adults must complete healthy behaviors (preventive dental visit and health risk assessment) or pay a $3 monthly premium for the Dental Wellness Plan. | |
| Kentucky | 2019 | Expansion Adults and Parents/Caretakers (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering:24 Work/community engagement requirement. Expansion Adults and Parents/Caretakers (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Eliminate retroactive eligibility. Expansion Adults and Parents/Caretakers (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Reasonable promptness waiver to delay coverage until the first day of the first month following receipt of the required premium. Expansion Adults and Parents/Caretakers (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Disenrollment and lock-out of coverage if renewal is not completed timely. Expansion Adults and Parents/Caretakers (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Disenrollment and lock-out of coverage if changes affecting eligibility are not reported timely. Expansion Adults (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Disenrollment and lock-out of coverage for non-payment of premiums. Premiums (New) for Expansion Adults and Parents/Caretakers Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Up to 4% of income but at least $1. |
| Maine | 2019 | Adults (-) Pending Sec. 1115 Waiver: Add a work requirement for many groups of traditional adults. Those who do not comply with work requirement would be limited to no more than 3 months coverage in a 36-month period. All Groups (-) Pending Sec. 1115 Waiver: Eliminate retroactive eligibility. Adults (-) Pending Sec. 1115 Waiver: Apply a $5,000 asset test to all coverage groups that do not currently have an asset test (under current law there is no asset test for coverage groups based solely on low income (vs. old age/disability)). All Groups (-) Pending Sec. 1115 Waiver: Eliminate hospital presumptive eligibility. Expansion Adults (+): Implement ACA Medicaid expansion (approved by referendum in November 2017) via a SPA.25 Premiums (New) for Adults Ages 19 to 64 Pending Sec 1115 Waiver: Premiums of between $10 and $40 per month, not to exceed 2% of income. |
| Maryland | 2019 | Adults (+): Increased income threshold for limited family planning benefit from 200% FPL to 250% FPL, remove age limit, and expanded coverage to include men. Switched from 1115 to SPA authority (9,000 individuals). |
| Massachusetts | 2019 | Adults (-) Approved Sec. 1115 Waiver: Eliminate 90-day period of provisional eligibility for adults under age 65 without verified income. The following groups will continue to be eligible for 90-day provisional eligibility pending verification of income: (1) pregnant women with self-attested MAGI income less than or equal to 200% FPL; (2) individual with HIV-positive status with self-attested MAGI income less than or equal to 200% FPL; or (3) individual in active treatment for breast or cervical cancer with self-attested MAGI income less than or equal to 250% FPL. (135,000).26 |
| Michigan | 2019 | Expansion Adults 100-138% FPL (-) Pending Sec. 1115 Waiver: Disenroll individuals for non-payment of required premiums after reaching 48 months of cumulative Healthy Michigan Plan eligibility. Expansion Adults 100-138% FPL (-) Pending Sec. 1115 Waiver: Disenroll individuals for failure to complete an annual healthy behavior requirement after reaching 48 months of cumulative Healthy Michigan Plan eligibility. Premiums (Increased) Pending Sec. 1115 Waiver: Require premium of 5% of income for expansion population with incomes between 100% and 133% FPL. |
| Missouri | 2018 | Aged & Disabled (+): Asset limit increase (phased increase from FY 2018 through FY 2022) (2,865 individuals). |
| 2019 | Aged & Disabled (+): Asset limit increase (phased increase from FY 2018 through FY 2022) (992 individuals) | |
| New Hampshire | 2019 | Expansion Adults (-): Approved Sec. 1115 Waiver: Work/community engagement requirement for expansion population, implementation beginning 1/1/2019. Expansion Adults (-): Approved Sec. 1115 Waiver: Eliminate retroactive eligibility for expansion population. |
| New Jersey | 2019 | Family Planning Enrollees (+): Expansion of family planning to cover men and women ages 19 to 65 between 138% and 205% FPL (over 30,000 individuals). |
| New Mexico | 2018 | Aged & Disabled (-): Home equity exclusion changed from the federal maximum of $840,000 to the federal minimum of $560,000 (fewer than 5 individuals). |
| 2019 | Family Planning (-) Pending Sec. 1115 Waiver: Limit family planning to age 51 and under (or under 65 with Medicare) (15,200 individuals). Most Managed Care Members (-) Pending 1115 waiver: Limit retroactive Medicaid to one month for most managed care members (8,000 individuals). Expansion Adults (-): Pending Sec. 1115 Waiver: Reasonable promptness waiver to delay coverage until the first day of the first month following receipt of the required premium (pending CMS approval of proposed premiums). Expansion Adults (-): Pending Sec. 1115 Waiver: Disenrollment and lock-out of coverage for non-payment of premiums. Transitional Medical Assistance Parents/Caretakers (-): Pending Sec. 1115 Waiver: Eliminate TMA coverage pathway for parents/caretakers. Premiums for Expansion Adults above 100% FPL (New) Pending Sec. 1115 Waiver: New monthly premiums, which could be lowered under provisions of the healthy behavior incentive program. | |
| New York | 2018 | Former Foster Youth (#): Eliminate coverage category for individuals under age 26 who were in foster care and on Medicaid in another state at the time they aged out of the foster care system (0 individuals in this category). |
| Ohio | 2019 | Expansion Adults (-): Pending Sec. 1115 Waiver: Work/community engagement requirement for Group VIII (expansion) MAGI adults (701,707 individuals). |
| Pennsylvania | 2018 | Children Under Age 4 (+): Continuous eligibility (7,746 children). |
| South Carolina | 2018 | Children & Pregnant Women (+): Implemented the CHIPRA option to eliminate the 5-year waiting period on Medicaid eligibility for lawfully-residing immigrant children and pregnant women. |
| South Dakota | 2019 | Adults in Minnehaha and Pennington Counties (-): Pending Sec. 1115 Waiver: Work/community engagement requirement, with offer of 12 months TMA and an additional 12 months premium assistance to individuals who continue to meet the work requirement but whose income increases above the Medicaid eligibility limit as a result of meeting the requirement (1,300 individuals). |
| Utah | 2018 | Parents & Caretakers (+): Increased the Basic Maintenance Standard to 55% FPL (3,000 individuals). Adults (+) Approved Sec. 1115 Waiver: New eligibility group for chronically homeless, justice-involved individuals and those in need of substance abuse and/or mental health treatment, with income below 5% FPL (2,800 individuals). Adults (-) Approved Sec. 1115 Waiver: Eliminate retroactive eligibility for Primary Care Network (PCN) adults. Adults (+) Approved Sec. 1115 Waiver: Twelve months continuous eligibility for targeted childless adult population. |
| Vermont | 2018 | Aged & Disabled (+): Increased asset level for Working People with Disabilities program (from $5,000 to $10,000 for an individual and from $6,000 to $15,000 for a couple) (70 individuals). |
| Virginia | 2018 | Disabled (+) Approved Sec. 1115 Waiver: Increased eligibility from 80% to 100% FPL for waiver services for people with serious mental illness (GAP waiver program, full restoration to pre-2016 level) (2,000 adults with SMI). |
| 2019 | Expansion Adults (+): ACA expansion of eligibility to non-caretaker, low-income adults between 0% and 138% of FPL (400,000 individuals). |
Table 3: Corrections-Related Enrollment Policies in all 50 States and DC, in Place in FY 2018 And Actions Taken in FY 2019
| States | Medicaid Coverage For Inpatient Care Provided to Incarcerated Individuals | Medicaid Outreach/Assistance Strategies to Facilitate Enrollment Prior to Release^ | Medicaid Eligibility Suspended Rather Than Terminated For Enrollees Who Become Incarcerated^ | |||||||||
| Jails | Prisons | Jails | Prisons | Jails | Prisons | |||||||
| In place FY 2018 | New or Expanded FY2019 | In place FY 2018 | New or Expanded FY2019 | In place FY 2018 | New or Expanded FY2019 | In place FY 2018 | New or Expanded FY2019 | In place FY 2018 | New or Expanded FY2019 | In place FY 2018 | New or Expanded FY2019 | |
| Alabama | X | X | X | X | X | X | ||||||
| Alaska | X | X | X | X | X | X | ||||||
| Arizona | X | X | X | X | X | X | ||||||
| Arkansas | X | X | X | X | X | X | ||||||
| California | X | X | X | X | X | X | ||||||
| Colorado | X | X | X | X | X | X | ||||||
| Connecticut | X | X | X | X | X | X | ||||||
| Delaware | X | X | X | X | X* | X* | ||||||
| DC | X | N/A | N/A | X | X | X | N/A | N/A | ||||
| Florida | X | X | ||||||||||
| Georgia | X | |||||||||||
| Hawaii | X | X | X | X | ||||||||
| Idaho | X | X | ||||||||||
| Illinois | X | X | X | X | ||||||||
| Indiana | X | X | X | X | X | X | ||||||
| Iowa | X | X | X | X | X | |||||||
| Kansas | X | X* | X | |||||||||
| Kentucky | X | X | X | X | X | X | X | X | X | X | ||
| Louisiana | X | X | X* | X | X | X | ||||||
| Maine | X | X | X | X | ||||||||
| Maryland | X | X | X | X | X* | X | X | |||||
| Massachusetts | X | X | X | X | X | X | ||||||
| Michigan | X | X | X | X | X | X | ||||||
| Minnesota | X | X | ||||||||||
| Mississippi | X | X | X | |||||||||
| Missouri | X | X | X | X | ||||||||
| Montana | X | X | X | X | X | X | ||||||
| Nebraska | X | X | X | X | ||||||||
| Nevada | X | X | X | X | X | |||||||
| New Hampshire | X | X | X | X | X | X | ||||||
| New Jersey | X | X | X | X | X | X | ||||||
| New Mexico | X | X | X | X | X | X | ||||||
| New York | X | X | X | X | X | X | ||||||
| North Carolina | X | X | ||||||||||
| North Dakota | X | X | X | |||||||||
| Ohio | X | X | X | X | X | |||||||
| Oklahoma | X | |||||||||||
| Oregon | X | X | X | X | X | X | ||||||
| Pennsylvania | X | X | X | X | X | X | ||||||
| Rhode Island | X | X | X | X | X | X | ||||||
| South Carolina | X | X | X | X | X | X | ||||||
| South Dakota | X | X | X | X | ||||||||
| Tennessee | X | X | X | X | ||||||||
| Texas | X | X | X | X | X | |||||||
| Utah | X | X | X | X | X | X | ||||||
| Vermont | X | X | X | X | ||||||||
| Virginia | X | X | X | X | X | X | ||||||
| Washington | X | X | X | X | X | X | ||||||
| West Virginia | X | X | X | X | X | X | ||||||
| Wisconsin | X | X | X | X | ||||||||
| Wyoming | ||||||||||||
| Totals | 42 | 1 | 48 | 1 | 32 | 5 | 38 | 4 | 35 | 2 | 37 | 1 |
NOTES: ^States with “Medicaid outreach assistance strategies to facilitate enrollment prior to release” include those implementing a variety of strategies. In many cases, staff of the prison or jail provide most of the assistance in collaboration with the Medicaid agency. ^States that continue Medicaid eligibility for incarcerated individuals but limit covered benefits to inpatient hospitalization are also included in the count of states that suspend eligibility. “*” indicates that a policy was newly adopted in FY 2019, meaning that the state did not have any policy in that category/column in place in FY 2018. N/A: The District of Columbia has jails but no prisons (however, individuals returning to DC from federal prisons may be placed in residential re-entry centers and have the opportunity to apply for Medicaid from there in order to attain coverage for 24 hour inpatient care and to facilitate enrollment prior to release). SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||||||
Report: Managed Care Initiatives
Key Section Findings
Managed care is the predominant delivery system for Medicaid in most states. As of July 1, 2018, among the 39 states with comprehensive risk-based managed care organizations (MCOs), 33 states reported that 75% or more of their Medicaid beneficiaries were enrolled in MCOs, a notable increase from the 29 states reporting 75% or more as of July 1, 2017. Although many states still carve-out behavioral health services from MCO contracts, movement to carve-in these services continues – two states in FY 2018 and 11 states in FY 2019 reported BH service carve-ins or implementation of an integrated MCO arrangement. Nearly all states have managed care quality initiatives in place such as pay for performance or capitation withholds.
What to watch:
- The current administration is expected to release revised Medicaid managed care regulations for public comment.
- Alaska and Arkansas reported plans to implement an MCO program for the first time in FY 2019. Alaska is contracting with one MCO to serve one geographic area, and Arkansas will begin making actuarially sound “global payments” to “Provider-led Arkansas Shared Savings Entities” (PASSEs) that serve Medicaid beneficiaries who have complex behavioral health and intellectual and developmental disabilities service needs.
- The 2016 Medicaid MCO rule allows states to use “in lieu of” authority to cover services for adults who receive inpatient psychiatric or SUD treatment services in an IMD for no more than 15 days in a month. In this survey, 28 of the 39 MCO states reported that they are using this authority for both FYs 2018 and 2019, and three states reported plans to begin using this authority in FY 2019. The SUPPORT Act would create a new state option from October 1, 2019 to September 30, 2023 to cover IMD services for up to 30 days in a year for individuals with an SUD and also would codify the provision allowing MCOs to offer “in lieu of” IMD coverage for up to 15 days in a month.
- An increasing number of states (23 in FY 2018) set a target percentage of MCO provider payments that must be in alternative payment models (APMs), up from 13 states in FY 2017 and 5 states in FY 2016. States are also increasingly requiring MCOs to screen beneficiaries for social needs (16 states in FY 2018 and 3 additional states in FY 2019); and to provide care coordination pre-release to incarcerated individuals (6 states in FY 2018 and 2 additional states in FY 2019).
Tables 4 through 7 include more detail on the populations covered under managed care (Tables 4 and 5), behavioral health services covered under MCOs (Table 6), and managed care quality initiatives (Table 7).
NEW: 50-state Medicaid survey finds that states are increasingly relying on managed care. In 33 states of 39 states with managed care organizations (MCOs), at least 75% of all Medicaid beneficiaries are in managed care.
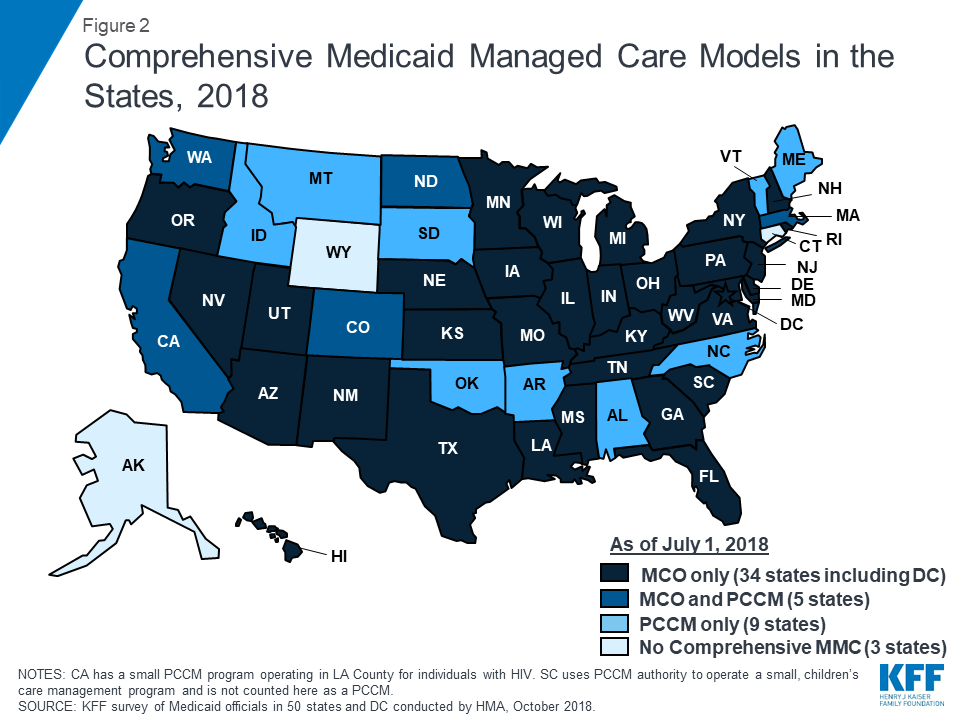
Managed care remains the predominant delivery system for Medicaid in most states. As of July 2018, all states except three – Alaska, Connecticut,27 and Wyoming – had some form of managed care in place, unchanged from July 2017. Compared to the prior year, the number of states contracting with comprehensive risk-based managed care organizations (MCOs) (39 states) also remained unchanged while two fewer states reported operating a Primary Care Case Management (PCCM) program (14 states). PCCM is a managed fee-for-service (FFS) based system in which beneficiaries are enrolled with a primary care provider who is paid a small monthly fee to provide case management services in addition to primary care.
Of the 48 states that operate some form of managed care, five operate both MCOs and a PCCM program while 34 states operate MCOs only and nine states operate PCCM programs only28 (Figure 2 and Table 4). In total, 28 states contracted with one or more PHPs to provide Medicaid benefits including, behavioral health care, dental care, vision care, non-emergency medical transportation (NEMT), or long-term services and supports (LTSS).
The current administration is expected to release revised Medicaid managed care regulations for public comment. Under the previous administration in April 2016, CMS issued a final rule on Medicaid managed care providing a framework of plan standards and requirements designed to improve the quality, performance, and accountability of these programs.29 ,30 The new rule represented a major revision and modernization of federal regulations in this area. The Trump Administration, however, is expected to release revised Medicaid managed care regulations before the end of CY 2018.31
In advance of releasing revised Medicaid managed care regulations, CMS released an Informational Bulletin32 in June 2017 indicating they would use “enforcement discretion” to work with states on achieving compliance with the new managed care regulations, except for specific areas that “have significant federal fiscal implications.” In this year’s survey, MCO states were asked whether they have asked CMS for flexibility in meeting managed care regulation deadlines. States answering “yes” most frequently reported requesting relief on deadlines related to member services (e.g., grievance and appeals procedures or member handbooks and enrollment requirements) and provider/network requirements (e.g., screening and enrollment of network providers or provider directory requirements).
Populations Covered by Risk-Based Managed Care
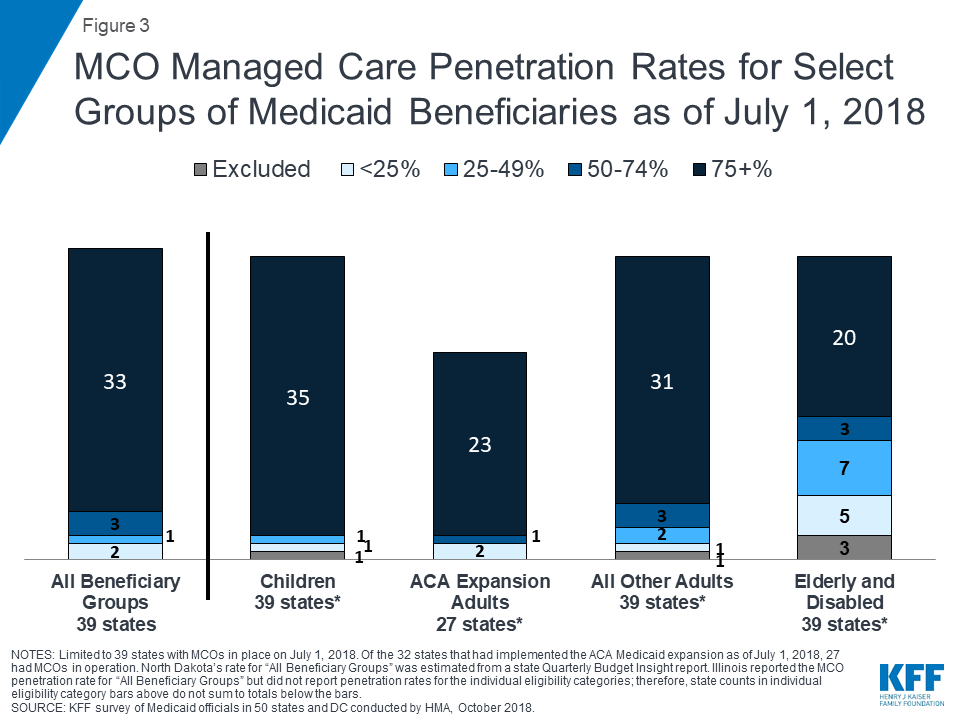
The share of Medicaid beneficiaries enrolled in MCOs has steadily increased as states have expanded their managed care programs to new regions and new populations and made MCO enrollment mandatory for additional eligibility groups. This year’s survey showed continued notable growth. Among the 39 states with MCOs, 33 states33 reported that 75% or more of their Medicaid beneficiaries were enrolled in MCOs as of July 1, 2018 (up from 29 states in last year’s survey), including nine of the ten states with the largest total Medicaid enrollment. These nine states (California, New York, Texas, Florida, Pennsylvania, Illinois, Ohio, Michigan, and Georgia) account for over half of all Medicaid beneficiaries across the country (Figure 3 and Table 4).34
Children and adults, particularly those enrolled through the ACA Medicaid expansion, are much more likely to be enrolled in an MCO than elderly Medicaid beneficiaries or persons with disabilities. Thirty-five35 of the 39 MCO states reported covering 75% or more of all children through MCOs. Of the 32 states that had implemented the ACA Medicaid expansion as of July 1, 2018, 27 were using MCOs to cover newly eligible adults.36 The large majority of these states (23 states) covered more than 75% of beneficiaries in this group through capitated managed care. New Hampshire reported that most of its ACA expansion adults are enrolled in Qualified Health Plans (with premium assistance, under Section 1115 authority) and only 20% are enrolled in MCOs.37 The state reported, however, that it will end its premium assistance waiver (as of December 31, 2018) and will transition QHP-enrolled members to MCOs. Thirty-one of the 39 MCO states reported covering 75% or more of low-income adults in pre-ACA expansion groups (e.g., parents, pregnant women) through MCOs. In contrast, the elderly and people with disabilities were the group least likely to be covered through managed care contracts, with only 20 of the 39 MCO states reporting coverage of 75% or more such enrollees through MCOs (Figure 3).
In states with both MCOs and PCCM programs, MCOs cover a larger share of beneficiaries than PCCM programs in a majority of these states. However, Colorado is an exception: as of July 1, 2018, a majority of Colorado’s enrollees were in the PCCM program, which is the foundation of the state’s “Accountable Care Collaboratives.”
Alaska and Arkansas reported plans to implement an MCO program for the first time in FY 2019. Alaska is contracting with one MCO to serve one geographic area that includes Anchorage and the Mat-Su Valley, and Arkansas will begin making actuarially sound “global payments” (beginning January 1, 2019) to “Provider-led Arkansas Shared Savings Entities” (PASSEs) that will serve Medicaid beneficiaries who have complex behavioral health and intellectual and developmental disabilities service needs. Only one state reported policies that reduced the state’s reliance on the MCO model of managed care: Massachusetts reported that the implementation of its Accountable Care Organization (ACO) program beginning in FY 2018 has resulted in decreased MCO enrollment as MCO enrollees may choose to enroll in ACOs instead.
Populations with Special Needs
For geographic areas where MCOs operate, this year’s survey asked MCO states whether, as of July 1, 2018, certain subpopulations with special needs were enrolled in MCOs for their acute care services on a mandatory or voluntary basis or were always excluded. This year’s survey further grouped subpopulations by dual eligible and by LTSS status (Exhibit 4 and Table 5).
Pregnant women were the group most likely to be enrolled on a mandatory basis while persons with I/DD were among the least likely to be enrolled on mandatory basis. As a group, foster children were most likely to be enrolled on a voluntary basis, although they were enrolled on a mandatory basis in a larger number of states. Among states indicating that the enrollment approach for a given group or groups varied, the location of LTSS services provided (residential versus community-based) was a frequently cited basis of variation. States with Financial Alignment Demonstrations for dual eligibles in addition to other managed care programs also often cited varying enrollment criteria for dual eligibles.
| Exhibit 4: MCO Enrollment of Populations with Special Needs, July 1, 2018(# of States) | ||||||||
| Non-Dual/Non-LTSS: | Non-Dual/Receives LTSS: | Dual Eligibles | ||||||
| Pregnant women | Foster children | CSHCNs | Persons with SMI/SED | Persons with I/DD | Persons w/ physical disabilities | Seniors | ||
| Always mandatory | 36 | 22 | 25 | 23 | 10 | 17 | 15 | 11 |
| Always voluntary | 2 | 8 | 6 | 3 | 6 | 4 | 4 | 4 |
| Varies | 0 | 6 | 6 | 11 | 13 | 10 | 9 | 14 |
| Always excluded | 1 | 3 | 2 | 2 | 10 | 8 | 11 | 10 |
| Notes: “CSHCNs” – children with special health care needs, “SMI/SED” – persons with serious mental illness or serious emotional disturbance, “I/DD” – persons with intellectual and developmental disabilities. | ||||||||
Acute Care Managed Care Population Changes
In both FY 2018 and FY 2019, only a few states reported actions to increase enrollment in acute care managed care, reflecting full or nearly full MCO saturation in most MCO states. As described above, Alaska and Arkansas reported plans to implement an MCO program for the first time in FY 2019. Of the 39 states with MCOs already in place as of July 1, 2018, five states in FY 2018 and five states in FY 2019 indicated that they made specific policy changes to increase the number of enrollees in MCOs through geographic expansion, voluntary or mandatory enrollment of new groups into MCOs, or mandatory enrollment of specific eligibility groups that were formerly enrolled on a voluntary basis (Exhibit 5). Thirty-seven states reported that acute care MCOs were operating statewide as of July 2018, including Illinois, which expanded MCOs statewide in FY 2018. The remaining two MCO states without statewide programs (Colorado and Nevada) did not report a geographic expansion planned for FY 2019.
| Exhibit 5: Medicaid Acute Care Managed Care Population Expansions, FY 2018 and FY 2019 | ||
| FY 2018 | FY 2019 | |
| Geographic Expansions | IL | — |
| New Population Groups Added | PA, TX, VA | IL, NH, OH, PA, VA |
| Voluntary to Mandatory Enrollment | WI | — |
| Implementing an MCO program for the first time | — | AK, AR |
Two notable acute care MCO expansions relate to programs that combine both acute care and LTSS:
- In January 2018, Pennsylvania began to phase-in its Community HealthChoices (CHC) program which provides both physical health and long-term services and supports through newly contracted MCOs. CHC enrollees include individuals in nursing facilities (currently carved out of managed care after 30 days), full benefit dually-eligible individuals, and individuals receiving home and community-based services.
- Virginia implemented its CCC Plus (Commonwealth Coordinated Care) program in FY 2018 making MCO enrollment mandatory for most LTSS populations.
In FY 2018 and FY 2019, states expanded MCO enrollment (either voluntary or mandatory) to other groups including state wards and foster children (Illinois), expansion adults transitioning from the state’s premium assistance program to MCO coverage (New Hampshire), workers with disabilities and persons receiving Specialized Recovery Services (Ohio), the Breast and Cervical Cancer and Adoption Assistance eligibility groups (Texas), and persons with third party liability coverage (Virginia).
Only one state made enrollment mandatory for a specific eligibility group that was formerly enrolled on a voluntary basis: Wisconsin made enrollment mandatory in FY 2018 for SSI adults with disabilities that do not have long-term care needs, are not enrolled in HCBS or MLTSS, are not tribal members, and are not dual eligibles.
Services Covered Under MCO Contracts
Behavioral Health Services Covered Under MCO Contracts
Although MCOs are at risk financially for providing a comprehensive set of acute care services, nearly all states exclude or “carve-out” certain services from their MCO contracts, frequently behavioral health services. States with acute care MCOs were asked to indicate whether specialty outpatient mental health (MH) services, inpatient mental health services, and outpatient and inpatient substance use disorder (SUD) services are always carved-in (i.e., virtually all services are covered by the MCO), always carved-out (to PHP or FFS), or the carve-in status varies by geographic or other factors. More than half of the 39 MCO states reported that specific behavioral health service types were carved into their MCO contracts, with specialty outpatient mental health services somewhat less likely to be carved in (Exhibit 6 and Table 6).
| Exhibit 6: MCO Coverage of Behavioral Health, July 1, 2018 (# of States) | ||||
| Specialty Outpatient MH* | Inpatient MH | Outpatient SUD | Inpatient SUD | |
| Always carved-in | 22 | 24 | 27 | 26 |
| Always carved-out | 10 | 7 | 7 | 6 |
| Varies | 7 | 8 | 5 | 7 |
| *“Specialty outpatient mental health” services mean services used by adults with Serious Mental Illness (SMI) and/or youth with serious emotional disturbance (SED), commonly provided by specialty providers such as community mental health centers. | ||||
In FY 2018, Mississippi and South Carolina reported actions to carve behavioral health services into their MCO contracts and Washington reported implementing integrated MCO contracts in additional geographic areas.
In FY 2019, 11 states reported actions impacting the coverage of behavioral health services under MCO contracts:
- Six states (Mississippi, New Jersey, New York, South Carolina, Virginia, and West Virginia) reported actions to carve behavioral health services into their MCO contracts.
- Arizona and Washington reported plans to implement additional integrated MCO contracts.
- Michigan reported a plan to implement pilot programs that would provide both physical and behavioral health services.
- Ohio reported a full carve-in of behavioral health services as of July 1, 2018.
- Arkansas reported plans to implement a new MCO program to serve beneficiaries who have complex behavioral health and intellectual and developmental disabilities needs.
Institutions for Mental Disease (IMD) “In Lieu OF” Rule
The 2016 Medicaid Managed Care Final Rule38 allows states (under the authority for health plans to cover services “in lieu of” those available under the Medicaid state plan), to receive federal matching funds for capitation payments on behalf of adults who receive inpatient psychiatric or substance use disorder (SUD) treatment or crisis residential services in an IMD for no more than 15 days in a month.39 States were asked whether or not they planned to use this new authority. Of the 39 states with MCOs as of July 1, 2018, 28 states answered “yes” for both FYs 2018 and 2019; three states reported plans to begin using this authority in FY 2019; and five states answered “no.”40 Also, North Carolina reported using “in lieu of” authority in its PHP contract and Arkansas reported plans to use this authority when it launches its new MCO program in January 2019. At the time this report was being finalized, however, the SUPPORT Act41 was expected to be signed into law creating a new state option from October 1, 2019 to September 30, 2023 to cover IMD services for up to 30 days in a year for individuals with an SUD. The SUPPORT Act also codified the 2016 Medicaid Managed Care Final Rule provision allowing MCOs to offer “in lieu of” IMD coverage for up to 15 days in a month.
Additional Services
Thirty-seven states with MCO contracts as of July 1, 2018, reported that MCO plans in their states may offer a range of services beyond those described in the state plan or waivers. The most common additional services reported were limited or enhanced adult dental services beyond contractually required state plan benefits, enhanced vision services for adults, and enhanced transportation services. Other value-added services reported included enhanced care coordination, wellness incentives, waiving co-payments, routine or school/sports physicals, and diabetic, weight-loss, tobacco cessation, and chiropractic services. Several states noted that MCOs offer services that address social determinants of health, including GED coaching, housing support, mother and baby supports, educational services and food access services. Others reported offering services/items to promote safety, including helmets and infant car seats.
Managed Care (Acute and LTSS) Quality, Contract Requirements, and Administration
Quality Initiatives
Over time, the expansion of comprehensive risk-based managed care in Medicaid has been accompanied by greater attention to measuring quality and plan performance and, increasingly, to measuring health outcomes. After years of comprehensive risk-based managed care experience within the Medicaid program, many states now incorporate quality into the procurement process, as well as into ongoing program monitoring.42
States procure MCO contracts using different approaches; however, most states use competitive bidding, in part because the dollar value is so large. Under these procurements, states can specify requirements and criteria that go beyond price, and may expect plans to compete on the basis of value-based payment arrangements with network providers, specific policy priorities such as improving birth outcomes, strategies to address social determinants of health, and/or other specific performance and quality criteria. In this year’s survey, states were asked if they used, or planned to use, the National Committee for Quality Assurance’s (NCQA’s) Healthcare Effectiveness Data and Information Set (HEDIS®) scores as criteria for selecting MCOs. Of the 39 states with MCOs, 14 indicated that they use or plan to use HEDIS scores as criteria for selecting MCOs.
Nearly all MCO states (35 states) reported using at least one select Medicaid managed care quality initiative in FY 2018 (Figure 4 and Table 7). States were asked to indicate whether they had specific managed care quality strategies (acute and/or MLTSS) in place in FY 2018 and to identify newly added or expanded initiatives for FY 2019. More than three quarters of MCO states reported having initiatives in place in FY 2018 that make MCO comparison data publicly available. More than half of MCO states reported capitation withhold arrangements and/or pay for performance incentives in FY 2018. Fewer states reported use of an auto-assignment algorithm that includes quality performance measures.
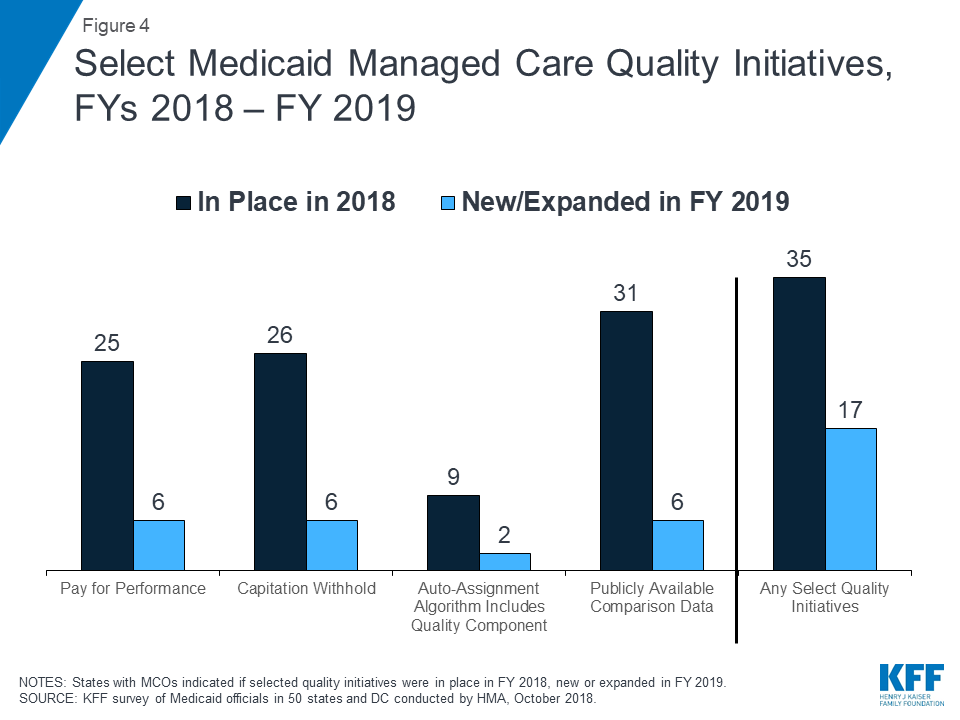
In FY 2019, 17 MCO states expect to implement new or expanded quality initiatives (Figure 4). The predominance of states reporting new or expanded activity in FY 2019 reported activity related to enhancing/expanding existing initiatives. However, two states reported new initiatives. Utah is planning to create a public facing dashboard for its acute care MCOs to make comparison data publicly available in FY 2019 and Louisiana is planning to add quality as a component to its auto-assignment algorithm for acute care contracts in calendar year 2019 (Table 7).
State-mandated Performance Improvement Project Examples
Federal regulations mandate that states must require, under contracts starting on or after July 1, 2017, each MCO or PHP to establish and implement an ongoing comprehensive quality assessment and performance improvement program for Medicaid services. Performance Improvement Projects (PIPs) may be designated by CMS, by states, or developed by health plans, but must be designed to achieve significant, sustainable improvement in health outcomes and enrollee satisfaction. In this year’s survey, states were asked to indicate whether they mandate MCO PIPs in a particular focus area. States reported a range of state-mandated PIP focus areas, including prenatal smoking and antipsychotic medication use in children (Kentucky), diabetes prevention and management and clinical depression screening and management (New Mexico), child health (Ohio), strengthening care coordination and encouraging transitions from nursing facilities to community care (Pennsylvania), and individuals with complex needs, especially those with comorbid anxiety and depression (Texas).
Contract Requirements
Alternative [Provider] Payment Models Within MCO Contracts
Value-based purchasing (VBP) strategies are important tools for states pursuing improved quality and outcomes and reduced costs of care within Medicaid and across payers. Generally speaking, VBP strategies include activities that hold a provider or MCO accountable for cost and quality of care.43 This often includes efforts to implement alternative payment models (APMs) which replace FFS/volume-driven provider payments with payment models that incentivize quality, coordination, and value (e.g., shared savings/shared risk arrangements and episode-based payments). Many states included a focus on adopting and promoting APMs as part of their federally-supported State Innovation Model (SIM) projects and as part of delivery system reform efforts approved under Section 1115 Medicaid waivers.44 A growing number of states are encouraging or requiring Medicaid MCOs to adopt APMs to advance VBP in Medicaid.
More than half of MCO states (23 states) identified a specific target in their MCO contracts for the percentage of provider payments, network providers, or plan members that MCOs must cover via alternative provider payment models in FY 2018 (Exhibit 7). (Only 13 states identified having a target percentage in place in FY 2017 and five states in FY 2016.) Four additional states plan to add a target percentage in FY 2019.
| Exhibit 7: States that Require MCOs to Meet a Target % for Provider APMs | ||
| # of States | States | |
| In Place FY 2018 | 23 | AZ, CA, DC, DE, HI, IA, LA, MA, MN, MO, NE, NH, NM, NY, OH, PA, RI, SC, TN, TX, WA, WI, WV |
| Plan to Begin in FY 2019 | 4 | FL, OR, UT, VA |
In FY 2018, 10 states had contracts that required Medicaid MCOs to adopt specific alternative provider payment models (e.g., episode of care payments, shared savings/shared risk, etc.), while eight states had contracts that encouraged MCOs to adopt specific APMs (Exhibit 8). In FY 2019, three additional states plan to require the use of specific APMs while four additional states plan to encourage specific APMs. CMS launched a Learning and Action Network (LAN) in 2015 to encourage alignment across public and private sector payers by providing a forum for sharing best practices and developing common approaches to designing and monitoring of APMs, as well as by developing evidence on the impact of APMs.45 Several states reported use of the LAN framework in devising MCO APM requirements (see examples below).
| Exhibit 8: States that Require vs. Encourage the Use of Specific APMs | ||||
| FY 2018 | FY 2019 | |||
| Require | 10 States | IA, LA, NE, NM, OH, PA, RI, TN, WA, WV | 3 States | AZ, KS, MI |
| Encourage | 8 States | DC, DE, IL, MA, NH, NY, TX, VA | 4 States | NJ, OR, UT, WI |
State APM Strategies for Medicaid MCO Contracts
- Arizona has set value-based APM targets at 50% for acute care MCOs, 35% for LTSS MCOs, and 25% for its seriously mentally ill (SMI) integrated population. The state plans to require use of strategies in LAN-APM46 categories 3 and 4 with targets of 40% for acute care MCOs, 25% for MLTSS, and 10% for SMI plans in FY 2019.
- Louisiana requires MCOs to implement APMs that fall within the LAN category 2A, 2C, 3, or 4, with an expectation that the MCO will implement one new contract in category 3 or 4 by 2019.
- Massachusetts requires that 60% of enrollees are covered by APM arrangements in years 1 and 2 of the current MCO contract, with 70% of enrollees in APM arrangements by years 3 and 4.
- Michigan plans to require use of the state preferred patient-centered medical home (PCMH) model in FY 2019.
- New Mexico requires that MCOs implement APM arrangements that: include a quality/outcome-based bonus or withhold for a minimum of 7% of all MCO provider payments; have an upside-only shared savings arrangement or two or more bundled payments (for episodes of care) for a minimum of 10% of all MCO provider payments; and have an upside and downside risk or a full-risk global payment arrangement for at least 3% of all MCO provider payments.
Social Determinants of Health
In April 2017, the CMS Center for Medicare and Medicaid Innovation selected 32 organizations to implement and test models to support local communities in addressing the health-related social needs of Medicare and Medicaid beneficiaries, aiming to bridge the gap between clinical and community service providers. This “Accountable Health Communities” (AHC) model represents the first CMS innovation model that focuses on social determinants of health. The goal of the five-year program is to encourage innovation to deliver local solutions that improve access to community-based services.47 As part of this effort, CMS has developed an AHC Health-Related Social Needs Screening Tool. This increased attention to social determinants of health is also seen at the state level, where many states have looked to Medicaid MCOs to develop strategies to identify and address social determinants of health.
The survey found that 16 states required, while 10 states encouraged MCOs to screen enrollees for social needs and/or provide referrals to social services in FY 2018 (Exhibit 9). In FY 2019, three additional states plan to require, while six additional states plan to encourage MCOs to screen/refer enrollees for social needs in FY 2019.
| Exhibit 9: States that Require vs. Encourage MCOs to Screen for Social Needs and/or Provide Referrals to Social Services | ||||
| FY 2018 | FY 2019 | |||
| Require | 16 States | CA, DC, DE, GA, IL, LA, MA, MD, MO, NE, NM, PA, RI, TN, VA, WI | 3 States | FL, HI, WV |
| Encourage | 10 States | AZ, CO, IA, KY, MI, NJ, NV, NY, TX, WA | 6 States | AR,48 IN, MS, NH, OH, OR |
State Strategies to Address Social Determinants of Health
- Hawaii will require MCOs in FY 2019 to identify individuals who are chronically homeless and to develop strategies to address housing instability; the state will also offer a new MCO-delivered community-based benefit to this target population under its Section 1115 waiver.
- Michigan requires MCOs to incorporate social determinants of health into their process for analyzing data to support population health management.
- Missouri requires MCOs to offer local community care coordination programs that promote improved outcomes through use of a physician-directed, integrated team that also provides referrals to community and social supports. Missouri MCOs also offer members with multiple chronic conditions an opportunity to participate in primary care health homes and behavioral health homes, which provide care coordination and linkages/referrals to social supports.
- Ohio is developing a uniform health risk assessment for MCOs to use that includes questions on social determinants of health.
- Wisconsin requires MCOs that provide services to SSI-eligible individuals to include social needs in the member health assessment and to refer as needed for housing services, adult protective services, crisis resolution for long-term care, etc.
In response to a new survey question, three states (Iowa, Massachusetts, and New Jersey) reported that they tie MCO incentive payments or withholds to a social determinants-related measure. One state (Colorado) reports that Rocky Mountain Health Plan, which participates in the federal AHC pilot described above, will adopt a social determinant-related measure in FY 2019. In Massachusetts, the state’s approach to accountable care organizations includes shared savings and losses that they expect will also be impacted by an organization’s attention to social determinants. In addition, Massachusetts uses a social determinants of health model to risk adjust MCO capitation rates for Medicaid ACO/MCO contracts, and the state plans to have a continuous risk adjuster for Senior Care Options and One Care that will include social determinants of health in a similar manner.
Criminal Justice-Involved PopulationsEngaging Medicaid MCOs in efforts to improve continuity of care for individuals released from correctional facilities into the community is important to ensure that individuals with complex or chronic health conditions, including behavioral health needs, have an effective transition to treatment in the community. In FY 2018, six states required MCOs to provide care coordination services to at least some enrollees prior to release from incarceration, while five states encouraged MCOs to provide care coordination services prior to release. Four states intend to use contracts to require or encourage such care coordination in FY 2019 (Exhibit 10). New Mexico will move from encouraging to requiring plans to participate in care coordination to facilitate the transition of members from prisons, jails, and detention facilities into the community.
| Exhibit 10: States that Require vs. Encourage MCOs to Provide Care Coordination Services to Enrollees Prior to Release from Incarceration | ||||
| FY 2018 | FY 2019 | |||
| Require | 6 States | AZ, KS, LA, OH, WA, WI | 2 States | NM, VA |
| Encourage | 5 States | CO, IA, KY, NM, PA | 2 States | DE, MA |
State Care Coordination Examples for Enrollees Pre-Release from Incarceration
- Arizona requires MCOs to participate in “reach-in” care coordination, allowing for immediate service delivery and care coordination activity upon release from incarceration.
- Louisiana specifies that individuals are eligible for at least one pre-release case management visit, conducted via teleconference with the MCO, to allow a case manager to determine post-release healthcare and other needs and to facilitate a linkage to care.
- Wisconsin requires pre-release care management for individuals who require long-term services and supports to better assure smooth transitions back to the community.
Administrative Policies
Minimum Medical Loss Ratios
The Medical Loss Ratio (MLR) is the proportion of total capitation payments received by an MCO spent on clinical services and quality improvement. CMS published a final rule in 2016 that requires states to develop capitation rates for Medicaid to achieve an MLR of at least 85% in the rate year, for rating periods and contracts starting on or after July 1, 2019. States must include requirements for plans to calculate and report an MLR for contracts that take effect on or after July 1, 2017.49 This 85% minimum MLR is the same standard that applies to Medicare Advantage and private large group plans. There is no federal requirement that Medicaid plans must pay remittances to the state if they fail to meet the MLR standard, but states have discretion to require remittances.
Some states reported that their MLR requirement for acute care contracts and/or MLTSS exceeds the 85% minimum, while other states noted that they do not yet specify an MLR.50 States were asked whether they require MCOs that do not meet the minimum MLR requirement to pay remittances. Twenty states reported that they always require MCOs to pay remittances, while five states indicated they sometimes require MCOs to pay remittances (Exhibit 11).
| Exhibit 11: Medicaid MCO Minimum Medical Loss Ratio (MLR) Remittance Requirements as of July 1, 2018 | ||
| # of states | States | |
| State always requiring remittance | 20 | CO, DE, IA, IL, IN, KY, LA, MD, MN, MO, NE, NV, NJ, NY, OR, PA, SC, VA, WA, WV |
| State sometimes requiring remittance | 5 | CA, KS, MA, MS, OH |
PCCM and PHP Program Changes
Primary Care Case Management (PCCM) Program Changes
Of the 14 states with PCCM programs, two reported enacting policies to increase PCCM enrollment in FY 2018 or FY 2019. Colorado reported growth in its PCCM-based Accountable Care Collaboratives in both FY 2018 and FY 2019. Massachusetts reported that member transitions related to the implementation of its Accountable Care Organization (ACO) program in FY 2018 had the overall effect of increasing PCCM enrollment in FY 2018.
Two other states reported new PCCM programs:
- Alabama reported plans for FY 2019 to replace its current PCCM program (Patient 1st) and Maternity PHP program with a new PCCM entity program (the Alabama Coordinated Health Network) that will cover care coordination services. Alabama is also planning to implement a second PCCM entity program in FY 2019 (the Alabama Integrated Care Network) that will provide enhanced case management, education, and outreach services to most LTSS recipients in both HCBS and institutional settings.
- Arizona implemented an American Indian Medical Home in FY 2018 using PCCM authority and enrollment is expected to expand in FY 2019.
Four states (Idaho, Illinois, Nevada, and Vermont) reported actions to decrease enrollment in a PCCM program in FY 2018 or FY 2019. Idaho is transitioning dual eligibles from PCCM to its Medicaid-Medicare Coordinated Plan in FY 2019; Illinois reported ending its PCCM program when its MCO program was expanded statewide in FY 2018; Nevada ended its Health Care Guidance Program on June 30, 2018; and Vermont is decreasing its PCCM payment over the first half of FY 2019 and will end the payment effective January 1, 2019.
Limited-Benefit Prepaid Health Plans (PHP) Changes
In this year’s survey, the 28 states contracting with at least one PHP as of July 1, 2018, were asked to indicate whether certain services (listed in Exhibit 12 below) were provided under these arrangements. The most frequently cited services provided (of those included in the question) were outpatient mental health services (14 states), followed by outpatient substance use disorder (SUD) treatment services, non-emergency medical transportation (NEMT), and dental services (13 states each).
| Exhibit 12: Services Covered Under PHP Contracts, July 1, 2018 | ||
| # of States | States51 | |
| Outpatient Mental Health | 14 | CA, CO, HI, ID, LA, MA, MI, NC, OR, PA, TN*, UT, WA, WI |
| Inpatient Mental Health | 12 | CA, CO, HI, LA, MA, MI, NC, PA, TN*, UT, WA, WI |
| Outpatient SUD Treatment | 13 | CA, CO, ID, LA, MA, MI, NC, OR, PA, TN*, UT, WA, WI |
| Inpatient SUD Treatment | 10 | CA, CO, MA, MI, NC, PA, TN*, UT, WA, WI |
| Non-Emergency Medical Transportation (NEMT) | 13 | FL, IA, IN, KY, ME, MI, NJ, OK, RI, TN*, TX, UT, WI |
| Dental | 13 | AR, CA, IA, ID, LA, MI, NE, NV, RI, TN*, TX, UT, WI |
| Long-Term Services and Supports | 6 | ID, MI, NC, NY, TN*, WI |
| Vision | 2 | TN*, WI |
| * In addition to separate dental and vision PHPs, TN contracts with a non-risk PHP to provide comprehensive benefits (physical health, behavioral health and LTSS) to children who are in foster care, receive Supplemental Security Income (SSI), or receive care in certain institutional settings. | ||
Twelve states reported implementing policies to increase PHP enrollment in FY 2018 or FY 2019. Seven states (Arkansas, Florida, Iowa, Nebraska, Nevada, Utah, and Washington) reported new or expanded dental PHPs in FY 2018 or planned for FY 2019. Other states reported the following changes: increased enrollment in its Drug Medi-Cal PHPs (California); increased enrollment in its dual eligible PHP (Idaho); implementation of a NEMT PHP (Indiana); enrollee transitions related to the implementation of its ACO initiative which increased behavioral health PHP enrollment (Massachusetts); and increased enrollment in its LTSS PHPs (New York).
Four states also reported actions that decreased PHP enrollment in FY 2018 or FY 2019. Alabama reported plans to end its maternity care PHP (when its new PCCM-entity program is implemented); Kentucky reported that it is planning to eliminate coverage under its NEMT PHP for expansion adults; Washington reported that enrollment in its behavioral health PHPs is decreasing as the state converts behavioral health PHPs to fully integrated MCO contracts in additional geographic areas; and Wyoming reported ending its PHP arrangement for children with emotional disturbance.
In this year’s survey, states with PHPs were also asked to briefly describe PHP contract quality strategies in place in FY 2018 or planned for FY 2019. Nearly two-thirds of states with PHPs reported a variety of quality strategies including tracking of HEDIS and/or other measures; requiring Performance Improvement Projects; incentive payments; withholds tied to performance measures; public reporting of performance results (e.g., report cards or dashboards); imposition of penalties or liquidated damages; and use of alternative payment methods.
Table 4: Share of the Medicaid Population Covered Under Different Delivery Systems in all 50 States and DC, as of July 1, 2018
| States | Type(s) of Managed Care In Place | Share of Medicaid Population in Different Managed Care Systems | ||
| MCO | PCCM | FFS / Other | ||
| Alabama | PCCM | — | 93.5% | 6.5% |
| Alaska | FFS | — | — | 100.0% |
| Arizona | MCO | 93.0% | — | 7.0% |
| Arkansas* | PCCM | — | NR | NR |
| California | MCO and PCCM* | 83.0% | — | 17.0% |
| Colorado | MCO and PCCM* | 10.1% | 89.9% | 0.0% |
| Connecticut | FFS* | — | — | 100.0% |
| Delaware | MCO | 97.0% | — | 3.0% |
| DC | MCO | 77.0% | — | 23.0% |
| Florida | MCO | 92.0% | — | 8.0% |
| Georgia | MCO | 83.0% | — | 17.0% |
| Hawaii | MCO | 99.9% | — | 0.1% |
| Idaho* | PCCM | — | 92.0% | 8.0% |
| Illinois | MCO | 80.0% | — | 20.0% |
| Indiana | MCO | 84.0% | — | 16.0% |
| Iowa | MCO | 92.6% | — | 7.4% |
| Kansas | MCO | 95.0% | — | 5.0% |
| Kentucky | MCO | 91.0% | — | 9.0% |
| Louisiana | MCO | 91.2% | — | 8.8% |
| Maine | PCCM | — | 50.0% | 50.0% |
| Maryland | MCO | 86.0% | — | 14.0% |
| Massachusetts | MCO and PCCM | 43.0% | 25.0% | 32.0% |
| Michigan | MCO | 77.6% | — | 22.4% |
| Minnesota | MCO | 84.0% | — | 16.0% |
| Mississippi | MCO | 65.0% | — | 35.0% |
| Missouri | MCO | 76.0% | — | 24.0% |
| Montana | PCCM | — | 73.0% | 27.0% |
| Nebraska | MCO | 99.7% | — | 0.4% |
| Nevada | MCO | 79.0% | — | 21.0% |
| New Hampshire* | MCO | 73.8% | — | 3.9% |
| New Jersey | MCO | 95.0% | — | 5.0% |
| New Mexico | MCO | 90.1% | — | 9.9% |
| New York | MCO | 77.2% | — | 22.8% |
| North Carolina | PCCM | — | 90.0% | 10.0% |
| North Dakota | MCO* and PCCM | 22.0% | NR | NR |
| Ohio | MCO | 89.5% | — | 10.5% |
| Oklahoma | PCCM | — | 74.9% | 25.1% |
| Oregon | MCO* | 93.0% | — | 7.0% |
| Pennsylvania | MCO | 84.6% | — | 15.4% |
| Rhode Island | MCO | 91.0% | — | 9.0% |
| South Carolina | MCO* | 77.0% | — | 23.0% |
| South Dakota | PCCM | — | 80.0% | 20.0% |
| Tennessee | MCO | 100.0% | — | 0.0% |
| Texas | MCO | 94.0% | — | 6.0% |
| Utah | MCO | 80.2% | — | 19.9% |
| Vermont | PCCM | — | 63.0% | 37.0% |
| Virginia | MCO | 95.0% | — | 5.0% |
| Washington | MCO and PCCM | 92.0% | 2.0% | 6.0% |
| West Virginia | MCO | 80.0% | — | 20.0% |
| Wisconsin | MCO | 67.0% | — | 33.0% |
| Wyoming | FFS | — | — | 100.0% |
| ‘NOTES: NR – not reported. MCO refers to risk-based managed care; PCCM refers to Primary Care Case Management. FFS/Other refers to Medicaid beneficiaries who are not in MCOs or PCCM programs. *AR – Most expansion adults served by Qualified Health Plans through “Arkansas Works” premium assistance waiver. *CA – PCCM program operates in LA county for those with HIV. *CO – PCCM enrollees are part of the state’s Accountable Care Collaboratives (ACCs). *CT – Terminated its MCO contracts in 2012 and now operates its program on a fee-for-service basis using three ASO entities. *ID – The Medicaid-Medicare Coordinated Plan (MMCP) has been recategorized by CMS as an MCO but is not counted here as such since it is secondary to Medicare. *ND’s total MCO penetration rate estimated from ND DHS Quarterly Budget Insight data for quarter ending 6/30/2018. *NH – 22.3% of overall population and 77% of expansion adults are served by Qualified Health Plans under NH’s premium assistance program waiver *OR – MCO enrollees include those enrolled in the state’s Coordinated Care Organizations. *SC – Uses PCCM authority to provide care management services to approximately 200 medically complex children. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||
Table 5: Enrollment of Special Populations Under Medicaid Managed Care Contracts for Acute Care in all 50 States and DC, as of July 1, 2018
| States | Non-Dual, Non-LTSS Populations | Non-Dual LTSS populations | Duals | |||||
| Pregnant Women | Foster Children | CSHCNs | Persons with SMI/SED | Persons with ID/DD | Persons w/ physical disabilities | Seniors | ||
| Alabama | — | — | — | — | — | — | ||
| Alaska | — | — | — | — | — | — | — | — |
| Arizona | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| Arkansas | — | — | — | — | — | — | — | — |
| California | Mandatory | Varies | Mandatory | Mandatory | Varies | Varies | Varies | Varies |
| Colorado | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary |
| Connecticut | — | — | — | — | — | — | — | — |
| Delaware | Mandatory | Mandatory | Mandatory | Mandatory | Varies | Mandatory | Mandatory | Varies |
| DC | Mandatory | Varies | Voluntary | Varies | Excluded | Varies | Excluded | Varies |
| Florida | Mandatory | Mandatory | Mandatory | Mandatory | Voluntary | Mandatory | Mandatory | Mandatory |
| Georgia | Mandatory | Mandatory | Excluded | Excluded | Excluded | Excluded | Excluded | Excluded |
| Hawaii | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| Idaho | — | — | — | — | — | — | — | — |
| Illinois | Mandatory | Excluded | Varies | Varies | Varies | Varies | Varies | Varies |
| Indiana | Mandatory | Voluntary | Mandatory | Mandatory | Excluded | Excluded | Excluded | Excluded |
| Iowa | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| Kansas | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| Kentucky | Mandatory | Mandatory | Mandatory | Mandatory | Excluded | Excluded | Excluded | Varies |
| Louisiana | Mandatory | Mandatory | Mandatory | Varies | Varies | Varies | Varies | Excluded |
| Maine | — | — | — | — | — | — | — | — |
| Maryland | Mandatory | Mandatory | Varies | Varies | Varies | Varies | Excluded | Excluded |
| Massachusetts | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary | Voluntary |
| Michigan | Mandatory | Mandatory | Mandatory | Mandatory | Varies | Varies | Varies | Voluntary |
| Minnesota | Mandatory | Voluntary | Voluntary | Varies | Voluntary | Voluntary | Mandatory | Varies |
| Mississippi | Mandatory | Voluntary | Voluntary | Varies | Excluded | Excluded | Excluded | Excluded |
| Missouri | Mandatory | Mandatory | Varies | Varies | Excluded | Excluded | Excluded | Excluded |
| Montana | — | — | — | — | — | — | — | — |
| Nebraska | Mandatory | Mandatory | Mandatory | Varies | Mandatory | Mandatory | Mandatory | Mandatory |
| Nevada | Mandatory | Voluntary | Voluntary | Voluntary | Excluded | Excluded | Excluded | Excluded |
| New Hampshire | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| New Jersey* | Mandatory | Mandatory | Mandatory | Mandatory | Varies | Mandatory | Voluntary | Mandatory |
| New Mexico | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| New York | Mandatory | Varies | Mandatory | Mandatory | Excluded | Mandatory | Mandatory | Excluded |
| North Carolina | — | — | — | — | — | — | — | — |
| North Dakota | Excluded | Excluded | Excluded | Excluded | Excluded | Excluded | Excluded | Excluded |
| Ohio | Mandatory | Mandatory | Mandatory | Mandatory | Varies | Varies | Varies | Varies |
| Oklahoma | — | — | — | — | — | — | — | — |
| Oregon | Mandatory | Varies | Mandatory | Mandatory | Varies | Mandatory | Mandatory | Voluntary |
| Pennsylvania | Mandatory | Mandatory | Mandatory | Mandatory | Varies | Varies | Varies | Varies |
| Rhode Island | Mandatory | Mandatory | Mandatory | Mandatory | Voluntary | Voluntary | Voluntary | Varies |
| South Carolina | Mandatory | Voluntary | Varies | Varies | Varies | Varies | Excluded | Varies |
| South Dakota | — | — | — | — | — | — | — | — |
| Tennessee | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| Texas | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Varies |
| Utah | Mandatory | Varies | Mandatory | Mandatory | Varies | Varies | Varies | Varies |
| Vermont | — | — | — | — | — | — | — | — |
| Virginia | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory | Mandatory |
| Washington | Mandatory | Voluntary | Mandatory | Mandatory | Varies | Mandatory | Varies | Varies |
| West Virginia | Mandatory | Excluded | Varies | Varies | Excluded | Excluded | Excluded | Excluded |
| Wisconsin | Mandatory | Varies | Varies | Varies | Voluntary | Mandatory | Varies | Varies |
| Wyoming | — | — | — | — | — | — | — | — |
| Mandatory | 36 | 22 | 25 | 23 | 10 | 17 | 15 | 11 |
| Voluntary | 2 | 8 | 6 | 3 | 6 | 4 | 4 | 4 |
| Varies | 0 | 6 | 6 | 11 | 13 | 10 | 9 | 14 |
| Excluded | 1 | 3 | 2 | 2 | 10 | 8 | 11 | 10 |
| ‘NOTES: “–” indicates there were no MCOs operating in that state’s Medicaid program as of July 1, 2018. I/DD – intellectual and developmental disabilities, CSHCN – Children with special health care needs, SMI – Serious Mental Illness, SED – Serious Emotional Disturbance. States were asked to indicate for each group if enrollment in MCOs is “Mandatory,” “Voluntary,” “Varies,” or if the group is “Excluded” from MCOs as of July 1, 2018. *NJ: Nursing facility residents as of July 1, 2014 were grandfathered and remain excluded from MCO enrollment unless they experience a change in eligibility status or are discharged from the nursing facility. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||
Table 6: Behavioral Health Services Covered Under Acute Care MCO Contracts in all 50 States and DC, as of July 1, 2018
| States | Specialty OP Mental Health | Inpatient Mental Health | Outpatient SUD | Inpatient SUD |
| Alabama | — | — | — | — |
| Alaska | — | — | — | — |
| Arizona | Varies | Varies | Varies | Varies |
| Arkansas | — | — | — | — |
| California | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Colorado | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Connecticut | — | — | — | — |
| DC | Varies | Varies | Always Carved-in | Always Carved-in |
| Delaware | Always Carved-out | Always Carved-in | Always Carved-out | Always Carved-in |
| Florida | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Georgia | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Hawaii | Always Carved-out | Always Carved-out | Always Carved-in | Always Carved-in |
| Idaho | — | — | — | — |
| Illinois | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Indiana | Varies | Always Carved-in | Always Carved-in | Always Carved-in |
| Iowa | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Kansas | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Kentucky | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Louisiana | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Maine | — | — | — | — |
| Maryland | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Massachusetts | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Michigan | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Minnesota | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Mississippi | Always Carved-in | Always Carved-in | Varies | Varies |
| Missouri | Always Carved-out | Varies | Varies | Varies |
| Montana | — | — | — | — |
| Nebraska | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Nevada | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| New Hampshire | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| New Jersey | Varies | Varies | Varies | Varies |
| New Mexico | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| New York | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| North Carolina | — | — | — | — |
| North Dakota | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Ohio | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Oklahoma | — | — | — | — |
| Oregon | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Pennsylvania | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Rhode Island | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| South Carolina | Always Carved-in | Varies | Always Carved-in | Always Carved-in |
| South Dakota | — | — | — | — |
| Tennessee | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Texas | Varies | Always Carved-in | Always Carved-in | Always Carved-in |
| Utah | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Vermont | — | — | — | — |
| Virginia | Always Carved-out | Varies | Always Carved-in | Varies |
| Washington | Varies | Varies | Varies | Varies |
| West Virginia | Always Carved-in | Varies | Always Carved-in | Varies |
| Wisconsin | Varies | Always Carved-in | Always Carved-in | Always Carved-in |
| Wyoming | — | — | — | — |
| Always Carved-in | 22 | 24 | 27 | 26 |
| Always Carved-out | 10 | 7 | 7 | 6 |
| Varies | 7 | 8 | 5 | 7 |
| NOTES: OP – Outpatient. SUD – Substance Use Disorder. “–” indicates there were no MCOs operating in that state’s Medicaid program in July 2018. For beneficiaries enrolled in an MCO for acute care benefits, states were asked to indicate whether these benefits are always carved-in (meaning virtually all services are covered by the MCO), always carved-out (to PHP or FFS), or whether the carve-in varies (by geography or other factor). “Specialty outpatient mental health” refers to services utilized by adults with Serious Mental Illness (SMI) and/or youth with serious emotional disturbance (SED) commonly provided by specialty providers such as community mental health centers. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||
Table 7: Select Medicaid Managed Care Quality Initiatives in all 50 States and DC, In Place in FY 2018 and Actions Taken in FY 2019
| States | Pay for Performance/Performance Bonus | Capitation Withhold | Auto-Assignment Algorithm Includes Quality Performance Measures | Publicly Available Comparison Data About MCOs | Any Select Quality Initiatives | |||||
| In PlaceFY 2018 | New or ExpandedFY 2019 | In PlaceFY 2018 | New or ExpandedFY 2019 | In PlaceFY 2018 | New or ExpandedFY 2019 | In PlaceFY 2018 | New or ExpandedFY 2019 | In PlaceFY 2018 | New or ExpandedFY 2019 | |
| Alabama | ||||||||||
| Alaska | ||||||||||
| Arizona | X | X | X | |||||||
| Arkansas | ||||||||||
| California | X | X | X | X | ||||||
| Colorado | X | X | X | |||||||
| Connecticut | ||||||||||
| DC | X | X | X | X | X | X | ||||
| Delaware | X | X | X | X | ||||||
| Florida | X | X | X | X | X | X | ||||
| Georgia | X | X | ||||||||
| Hawaii | X | X | X | X | X | X | X | |||
| Idaho | ||||||||||
| Illinois | X | X | X | X | ||||||
| Indiana | X | X | X | |||||||
| Iowa | X | X | X | X | X | X | X | |||
| Kansas | X | X | X | X | X | X | ||||
| Kentucky | X | X | ||||||||
| Louisiana | X | X* | X | X | X | |||||
| Maine | ||||||||||
| Maryland | X | X | X | X | X | X | ||||
| Massachusetts | X | X | X | X | X | X | ||||
| Michigan | X | X | X | X | X | |||||
| Minnesota | X | X | X | |||||||
| Mississippi | ||||||||||
| Missouri | X | X | X | X | X | X | ||||
| Montana | ||||||||||
| Nebraska | X | X | X | |||||||
| Nevada | X | X | X* | X | X | X | ||||
| New Hampshire | X | X | ||||||||
| New Jersey | X | X | X | X | X | X | ||||
| New Mexico | X | X | X | |||||||
| New York | X | X | X | |||||||
| North Carolina | ||||||||||
| North Dakota | ||||||||||
| Ohio | X | X | X | X | X | X | X | |||
| Oklahoma | ||||||||||
| Oregon | X | X | X | |||||||
| Pennsylvania | X | X | X | X | X | |||||
| Rhode Island | X | X | X | X | X | X | ||||
| South Carolina | X | X | X | X | X | |||||
| South Dakota | ||||||||||
| Tennessee | X | X | X | X | ||||||
| Texas | X | X | X | X | ||||||
| Utah | X* | X | ||||||||
| Vermont | ||||||||||
| Virginia | X | X | X | X | X | X | ||||
| Washington | X | X | X | X | X | X | ||||
| West Virginia | ||||||||||
| Wisconsin | X | X | X | X | ||||||
| Wyoming | ||||||||||
| Totals | 25 | 6 | 26 | 6 | 9 | 2 | 31 | 6 | 35 | 17 |
| NOTES: States with MCO contracts were asked to report if select quality initiatives were included in contracts in FY 2018, or are new or expanded in FY 2019. The table above does not reflect all quality initiatives states have included as part of MCO contracts. “*” indicates that a policy was newly adopted in FY 2019, meaning that the state did not have any policy in that category/column in place in FY 2018. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||||
Report: Emerging Delivery System And Payment Reforms
Key Section Findings
Medicaid programs have been expanding their use of other service delivery and payment reform models to achieve better outcomes and lower costs. Forty-three states have one or more delivery system or payment reform initiatives in place in FY 2018 (e.g., patient-centered medical home (PCMH), ACA Health Home, accountable care organization (ACO), episode of care payment, or delivery system reform incentive program (DSRIP)).
What to watch:
- About one-third of the states reported a wide variety of initiatives implemented in FY 2018 or planned for FY 2019 that address the social determinants of health (SDOH) outside of managed care and more than one-third reported collecting or plans to collect SDOH data from various sources including screenings and assessments, data collected for other state programs, claims data, beneficiary surveys, or as part of a care management or home visiting program.
- Nearly three-quarters of the states reported broader initiatives to expand access to mental health or SUD services in FY 2018 or FY 2019, however, the most common element is to expand coverage for mental health or SUD services (especially IMD services).
Table 8 contains more detailed information on emerging delivery system and payment reform initiatives in place in FY 2018 and new or expanded initiatives in FY 2019.
Over three-quarters of all state Medicaid programs (43 states) had at least one of the specified delivery system or payment reform models in place in FY 2018, a modest increase over the 40 states reporting at least one model in place in 2017 (Figure 5 and Table 8). This year’s survey asked states whether certain delivery system and payment reform models (defined in the box below) designed to improve health outcomes and constrain cost growth were in place in FY 2018, and whether they planned to adopt or enhance these models in FY 2019. For FY 2019, 19 states (all states with at least one model already in place) reported plans to adopt or expand one or more of the models to reward quality and encourage integrated care. Key initiatives include patient-centered medical homes (PCMHs), ACA Health Homes, and Accountable Care Organizations (ACOs).
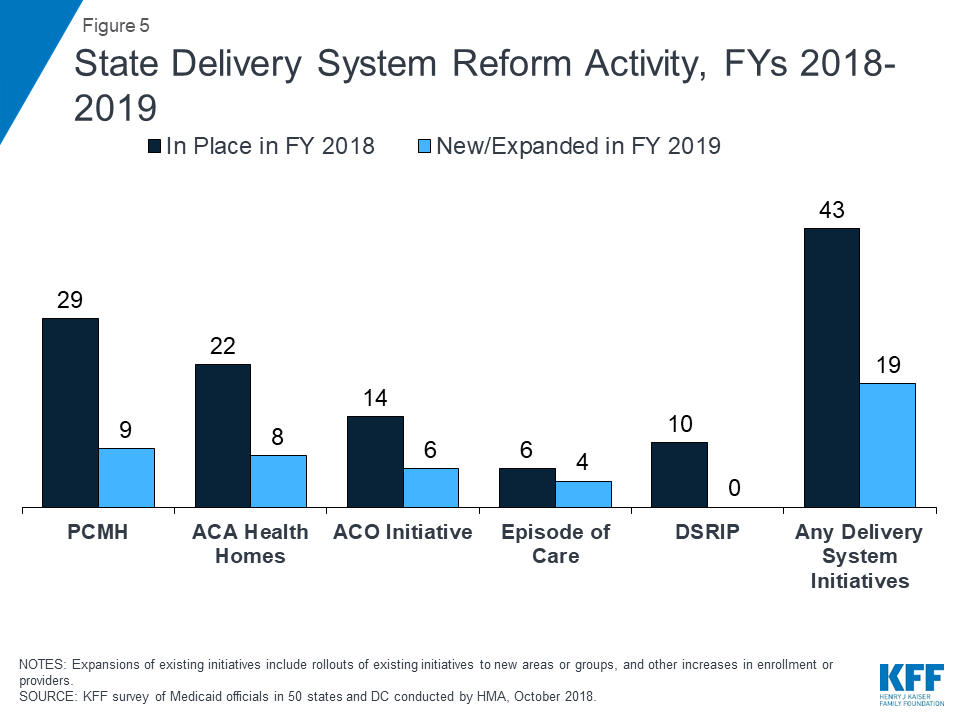
Delivery System Reform Initiatives Defined
- Patient-Centered Medical Home (PCMH). Under a PCMH model, a physician-led, multi-disciplinary care team holistically manages the patient’s ongoing care, including recommended preventive services, care for chronic conditions, and access to social services and supports. Generally, providers or provider organizations that operate as a PCMH seek recognition from organizations like the National Committee for Quality Assurance (NCQA).52 PCMHs are often paid (by state Medicaid agencies directly or through MCO contracts) a per member per month (PMPM) fee in addition to regular FFS payments for their Medicaid patients.
- ACA Health Home. The ACA Health Homes option, created under Section 2703 of the ACA, builds on the PCMH concept. By design, Health Homes must target beneficiaries who have at least two chronic conditions (or one and risk of a second, or a serious and persistent mental health condition), and provide a person-centered system of care that facilitates access to and coordination of the full array of primary and acute physical health services, behavioral health care, and social and long-term services and supports. This includes services such as comprehensive care management, referrals to community and social support services, and the use of health information technology (HIT) to link services, among others. States receive a 90% federal match rate for qualified Health Home service expenditures for the first eight quarters under each Health Home State Plan Amendment; states can (and have) created more than one Health Home program to target different populations.53
- Accountable Care Organization (ACO). While there is no uniform, commonly accepted federal definition of an ACO, an ACO generally refers to a group of health care providers or, in some cases, a regional entity that contracts with providers and/or health plans, that agrees to share responsibility for the health care delivery and outcomes for a defined population.54 An ACO that meets quality performance standards that have been set by the payer and achieves savings relative to a benchmark can share in the savings. States use different terminology in referring to their Medicaid ACO initiatives, such as Regional Care Collaborative Organizations (RCCOs) in Colorado55 and Accountable Entities in Rhode Island.
- Episode of Care Initiatives. Unlike FFS reimbursement, where providers are paid separately for each service, or capitation, where a health plan receives a PMPM payment for each enrollee intended to cover the costs for all covered services, episode-of-care payment provides a set dollar amount for the care a patient receives in connection with a defined condition or health event (e.g., heart attack or knee replacement). Episode-based payments usually involve payment for multiple services and providers, creating a financial incentive for physicians, hospitals, and other providers to work together to improve patient care and manage costs.
- Delivery System Reform Incentive Payment (DSRIP) Programs. DSRIP initiatives,56 which emerged under the Obama administration, provide states with significant federal funding to support hospitals and other providers in changing how they provide care to Medicaid beneficiaries.[endnote 375783-11] DSRIP initiatives link funding for eligible providers to process and performance metrics. Although some states may be interested in developing new DSRIP initiatives, the Trump administration has not indicated an intent to use this tool to advance delivery system reform.
PCMH and Health Home initiatives were the most common delivery system reform initiatives in place in states in FY 2018 (Table 8). PCMH initiatives operated in over half (29 states) of Medicaid programs in FY 2018. Nine states reported plans to expand or enhance existing PCMH programs in FY 2019, often citing increased provider participation. Over one-third of states (22 states) had at least one Health Home initiative in place in FY 2018. Six states reported plans to adopt and two states reported plans to expand Health Homes in FY 2019. One state (Alabama) reported that its PCMH and Health Home programs would end in FY 2019 when its new PCCM-entity program is implemented.
About a quarter of states had ACO initiatives in place and fewer states have episode of care initiatives in place in FY 2018 (Table 8). Fourteen states reported having ACOs in place for at least some of their Medicaid beneficiaries in FY 2018.57 Six states reported plans to expand an existing initiative in FY 2019. Six states reported that they had episode-of-care payment initiatives in place in FY 2018, unchanged from 2017. Three of these states also reported planned expansions of these initiatives in FY 2019. Idaho reported plans to implement a new episode-of-care payment related to births in FY 2019.
State Delivery System Reform Examples
- Montana reported plans to add an additional tier level for complex patients that require in-home visits to its PCMH program in FY 2019.
- Michigan reported that a new opioid Health Home is planned for FY 2019.
- Massachusetts reported that its ACO pilot program expanded statewide with over 870,000 of the state’s 1.2 million enrollees transferred to ACOs as of March 1, 2018. Massachusetts employs three different ACO models: an ACO/MCO partnership model; an ACO contracting directly with the state (without an MCO partner); and an exclusively MCO administered model (currently one small plan).
- Rhode Island reported that its “Accountable Entity” program, administered in partnership with its existing MCOs, continues to expand.
Ten states reported DSRIP initiatives in place in FY 2018, unchanged from FY 2017 (Table 8). DSRIP initiatives, which emerged under the Obama administration, have provided states with significant federal funding to support hospitals and other providers in changing how they provide care to Medicaid beneficiaries. No states reported expansions or enhancements to existing initiatives or reported new DSRIP initiatives planned for FY 2019. One state (Kansas) reported that its DSRIP program would end in FY 2019. These initiatives were not intended to be permanent and the Trump administration has not signaled an intent to promote these initiatives going forward.
OTHER INITIATIVES
All-payer claims database (APCD) systems are large-scale databases that systematically collect medical claims, pharmacy claims, dental claims (typically, but not always), and eligibility and provider files from both private and public payers. APCDs can be used to help identify areas to focus reform efforts and for other purposes. Seventeen states reported having an APCD in place. Two states (Florida and New York) reported that their APCD would be expanded in FY 2019 and four states (Alaska, Connecticut, Delaware, and Hawaii) reported plans for new APCDs in FY 2019.
In addition to the initiatives discussed above, states mentioned a variety of other delivery system and payment reform initiatives (not counted in the totals for Figure 5 and Table 8), including value-based purchasing initiatives, pay for performance payments, or other incentive arrangements targeted at hospitals, nursing facilities, federally qualified health centers (FQHCs), or other provider types. Examples of other initiatives reported include the following: Alaska is contracting with a health system in FY 2019 to provide case management services on a non-risk basis; Maryland reported on its recently approved Total Cost of Care All-Payer Model (an expansion of the existing all-payer model for hospital services) that will take effect in January 2019; Michigan reported on its Direct Primary Care Pilot that began August 1, 2018 (providing same or next business day primary care appointments for a fixed monthly fee); Rhode Island reported plans to implement a bundled payment for mental health and SUD crisis stabilization services limited to state certified providers; and Wyoming reported on its plans to implement a super-utilizer program targeted at high-cost, high-risk adults.
States with significant populations and/or services delivered outside of contracted MCO arrangements also reported on a wide range of non-MCO quality activities including collection of HEDIS data and other performance measures, conducting beneficiary satisfaction (CAHPS58 ) surveys, collecting LTSS measures, conducting performance improvement projects, public reporting quality data, and taking steps to expand the use of health information technology and health information exchange. One state (Connecticut) with a comprehensive quality strategy for its managed fee-for-service delivery system approach is highlighted below.
Connecticut Managed Fee-For-Service Quality Activities
Connecticut operates a self-insured, managed fee-for-service model, contracting with three Administrative Service Organizations (ASOs) that focus, respectively, on medical, behavioral health, and dental services. The state withholds a percentage of the ASO administration fees on a rolling basis in consideration of performance on a range of indicators related to member health outcomes and member and provider satisfaction. The medical ASO monitors a broad range of HEDIS and hybrid measures, administers CAHPS through a subcontractor, employs the Johns Hopkins CareAnalyzer tool and a fully integrated statewide claims data set to do predictive modeling, risk stratification, and other quality activities, and conducts mystery shopper and other analyses of access and provider capacity. The state pushes considerable claims data, some clinical data, and admission, discharge, or transfer (ADT) data through a portal to PCMH practices, which they use in support of interventions for their panels. Finally, the medical ASO produces an annual provider practice profile report for each provider that details their performance, in context of peers.
STATE initiatives to address social Determinants of Health
In addition to imposing requirements on MCOs to screen enrollees for social needs and make referrals to other services, many states have initiatives outside of their MCO programs to address one or more of their enrollees’ social determinants of health (SDOH), such as food insecurity, housing, employment, or education. In this year’s survey, states were asked to briefly describe any initiatives implemented in FY 2018 or planned for FY 2019 that address SDOH outside of managed care and/or the housing supports discussed in the “Long-Term Services and Supports” section below.
About one-third of the states described a wide variety of initiatives including case management strategies that include the identification of social factors and appropriate referrals; initiatives that employ community health workers; and participation in the CMS Accountable Health Communities grant program. Other examples of initiatives reported for non-MCO populations include the following: Colorado reported plans to implement multiple SDOH initiatives in FY 2019, mostly through its Regional Accountable Entities that are responsible for collaborating with community-based organizations and promoting member engagement with SNAP, WIC, and other local programs; DC reported that it is working with its NEMT provider to promote the increased use of transportation benefits for fee-for-service beneficiaries in order to alleviate challenges with transport to urgent care and primary care; Maine reported that employment is a focus area for its behavioral health homes; and Massachusetts is seeking federal approval to allow its ACOs to use “flex funds” (under DSRIP expenditure authority) to pay for certain health-related social services.
Colorado Opportunity Framework
The Colorado Opportunity Framework is a life stage, indicator-based framework designed to develop a health care delivery system that incorporates key SDOH. The Framework separates the human life cycle into 9 stages and tracks over 27 unique metrics from 12 sources including Medicaid claims data, the Behavioral Risk Factor Surveillance System (BRFSS), Public Health and State Education data sets, and other population surveys. Colorado will be collecting data on these metrics on an ongoing basis, as well as data on a set of public reporting measures that include a number of SDOH such as high school graduation rates and math and reading testing scores.
States were also asked if the Medicaid agency collected SDOH-related data for enrollees and, if so, how that data was used. Over one-third of states, including both MCO and non-MCO states, reported that SDOH data was collected as of FY 2018 or would be collected beginning in FY 2019. These states reported a variety of SDOH data sources including screenings or assessments at the time of or following the eligibility determination process, data collected for other state programs, data derived from claims or encounter data, beneficiary surveys, information collected as a part of a care management or home visiting program, and other data submitted by providers. The most common uses reported for this data were to inform care coordination or care management, for quality improvement initiatives, and for performance measurement. Other uses mentioned included rate setting, to fulfill federal reporting requirements, and to make referrals.
Several states also reported providing or plans to provide fee-for-service care coordination services to incarcerated persons prior to release. For example, Colorado reported that its Regional Accountable Entities were encouraged, but not required, to provide pre-release care coordination services; Connecticut connects incarcerated individuals, pre-release, with the state’s medical Administrative Services Organization (ASO) to help smooth the transition to community-based medical services; and Kentucky reported plans for a care coordination pilot program targeting incarcerated individuals with SMI.
Access Improvement Focus Areas
This year’s survey included additional questions for states with initiatives to increase access to care in rural areas and to increase access to mental health and SUD services. States were asked to briefly describe initiatives implemented in FY 2018 or planned for FY 2019.
Improving Access to Care in Rural Areas
Medicaid is a vital source of health care coverage in rural areas and small towns that tend to have lower household incomes, lower rates of workforce participation, and higher rates of disability compared to the rest of the nation.59 Nearly half of states reported a variety of new or expanded initiatives to improve access to care in rural areas implemented in FY 2018 or FY 2019. The most frequently mentioned type of initiative related to telehealth, including e-Consult, telemedicine, tele-monitoring, and Project ECHO60 programs. Strategies mentioned by at least two states included funding increases for rural providers, expanded SUD treatment services in rural areas, expanded funding for primary care residency programs, and participation in multi-payer initiatives that promote rural access to care.
Improving Access to Mental Health and SUD Services
Nearly three-quarters of states reported initiatives to expand access to mental health (MH) or SUD services in FY 2018 or FY 2019 (MH/SUD policy changes that expand access are also discussed in the “Eligibility and Premiums” and “Benefits and Copayments” sections of this report). The most commonly reported actions were seeking waiver authority to expand coverage of services provided in an Institute for Mental Disease (IMD) and adding coverage for new MH or SUD services, often under waiver authority. Several states also reported delivery system reforms expected to improve access, such as adoption of the Hub & Spoke model, MCO integration efforts and establishment of mental health and SUD Health Homes.61
Other initiatives reported included access-related incentive payments; allowing up to 5% of the cost of MCO behavioral health-related “community investments” to be counted as benefit expenditures rather than administrative costs; promoting the use of the SBIRT (Screening, Brief Intervention, and Referral to Treatment) tool in physician offices, FQHCs, and intake areas of institutional settings; participating in the CMS Innovation Accelerator Program, the Opioid Data Analytic Cohort; participating in a state SUD Treatment and Reentry Center Project for offenders; eliminating copays for pharmacy SUD treatment services; and geographically expanding an HCBS program for persons with severe disabling mental illness.
Table 8: Select Delivery System and Payment Reform Initiatives in all 50 States and DC, In Place in FY 2018 and Actions Taken in FY 2019
| States | Patient-Centered Medical Homes(PCMH) | ACA Health Homes | Accountable Care Organizations (ACO) | Episode of Care Payments | Delivery System Reform Incentive Payment Program(DSRIP) | Any Delivery System or Payment Reform Initiatives | ||||||
| In Place FY 2018 | New/ Expand FY 2019 | In Place FY 2018 | New/ Expand FY 2019 | In Place FY 2018 | New/ Expand FY 2019 | In Place FY 2018 | New/ Expand FY 2019 | In Place FY 2018 | New/ Expand FY 2019 | In place FY 2018 | New/ Expand FY 2019 | |
| Alabama | X | X | X | |||||||||
| Alaska | ||||||||||||
| Arizona | X | X | ||||||||||
| Arkansas | X | X | X | |||||||||
| California | X* | X | X | X | ||||||||
| Colorado | X | X | X | X | X | X | ||||||
| Connecticut | X | X | X | X | X | X | ||||||
| Delaware | ||||||||||||
| DC | X | X | ||||||||||
| Florida | X | X | X | X | ||||||||
| Georgia | X | X | ||||||||||
| Hawaii | ||||||||||||
| Idaho | X | X | X* | X | X | |||||||
| Illinois | X | X* | X | X | ||||||||
| Indiana | ||||||||||||
| Iowa | X | X | X | |||||||||
| Kansas | X* | X | X | X | ||||||||
| Kentucky | ||||||||||||
| Louisiana | X | X | ||||||||||
| Maine | X | X | X | |||||||||
| Maryland | X | X | ||||||||||
| Massachusetts | X | X | X | X | X | |||||||
| Michigan | X | X | X | X | X | |||||||
| Minnesota | X | X | X | X | ||||||||
| Mississippi | ||||||||||||
| Missouri | X | X | X | X | ||||||||
| Montana | X | X | X | X | ||||||||
| Nebraska | X | X* | X | X | X | |||||||
| Nevada | X | X | ||||||||||
| New Hampshire | X | X | ||||||||||
| New Jersey | X | X | X | X | ||||||||
| New Mexico | X | X | X | X | X | X | X | X | ||||
| New York | X | X | X | X | X | X | X | X | X | X | ||
| North Carolina | X | X | X | |||||||||
| North Dakota | ||||||||||||
| Ohio | X | X | X | X | X | X | X | X | ||||
| Oklahoma | X | X | X | |||||||||
| Oregon | X | X | ||||||||||
| Pennsylvania | X | X | X | X | X | X | X | |||||
| Rhode Island | X | X | X | X | X | X | ||||||
| South Carolina | X | X | ||||||||||
| South Dakota | X | X | ||||||||||
| Tennessee | X | X | X | X | X | X | ||||||
| Texas | X | X | X | |||||||||
| Utah | ||||||||||||
| Vermont | X | X | X | X | X | X | ||||||
| Virginia | X | X | ||||||||||
| Washington | X | X | X | |||||||||
| West Virginia | X | X | ||||||||||
| Wisconsin | X | X | X | |||||||||
| Wyoming | X | X | X | X | ||||||||
| Totals | 29 | 9 | 22 | 8 | 14 | 6 | 6 | 4 | 10 | 0 | 43 | 19 |
| NOTES: Expansions of existing initiatives include rollouts of existing initiatives to new areas or groups and significant increases in enrollment or providers. “*” indicates that a policy was newly adopted in FY 2019, meaning that the state did not have any policy in that category/column in place in FY 2018. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||||||
Report: Long-term Services And Supports Reforms
Key Section Findings
Nearly all states in FY 2018 (46 states) and FY 2019 (48 states) are employing one or more strategies to expand the number of people served in home and community-based settings. A majority of states continue to report using HCBS waivers and/or state plan options (i.e., 1915(c), 1115, 1915(i), and 1915(k)) to serve more individuals in the community. As of July 1, 2018, 24 states covered LTSS through one or more capitated managed care arrangements.
What to watch:
- States continue to report challenges finding and retaining LTSS direct care workers. Fifteen states raised wages for direct care workers in FY 2018 and 24 states report wage increases in FY 2019.
- Housing supports remain an important part of state LTSS benefits, even as Money Follow the Person (MFP) grant funds expire. Thirty states reported that they expect to continue to offer housing-related supports even after MFP funds are exhausted. However, about half of the states reported plans to discontinue at least some housing-related services or administrative functions when MFP ends.
- Pennsylvania introduced MLTSS in FY 2018, with a plan to phase-in statewide over time. Virginia ended its Financial Alignment Demonstration (FAD) but adopted statewide MLTSS for a broader population, including dual eligible individuals. Only one state expects to adopt MLTSS in FY 2019.
Additional information on HCBS expansions implemented in FY 2018 or planned for FY 2019 as well as state-level details on capitated MLTSS models can be found in Tables 9 and 10.
Medicaid is the nation’s primary payer for long-term services and supports (LTSS), covering a continuum of services ranging from home and community-based services (HCBS) that allow people to live independently in their own homes or in other community settings to institutional care provided in nursing facilities (NFs) and intermediate care facilities for individuals with intellectual disabilities (ICF-IDs). In federal fiscal year 2016, spending on Medicaid LTSS totaled $167 billion, and HCBS represented 57% of these expenditures. In recent years, growth in Medicaid LTSS expenditures has been largely concentrated in HCBS. In 2016, spending on HCBS grew by 10% while spending on institutional LTSS decreased 2%.62
This year’s survey shows the vast majority of states in FY 2018 (46 states) and the vast majority of states in FY 2019 (48 states) are using one or more strategies to expand the number of people served in home and community-based settings (Figure 6). States were asked about their use of the following rebalancing tools/methods: use of HCBS waivers and/or State Plan Amendments (SPAs) (including 1915(c), Section 1115, 1915(i), and 1915(k)); use of rebalancing incentives in managed care contracts; use of Programs of All-Inclusive Care for the Elderly (PACE); and efforts to downsize state institutions. A large majority of states in FY 2018 (40 states) and in FY 2019 (42 states) reported adopting new HCBS waivers/SPAs and/or serving more individuals through existing HCBS waivers/SPAs. About a third of states reported using rebalancing incentives in managed care contracts and about the same share reported implementing PACE expansions in FY 2018 and FY 2019. Fewer states report efforts to downsize state institutions. Table 9 shows state use of selected LTSS rebalancing tools in FY 2018 and FY 2019.

Few states reported actions to reduce or restrict the number of persons served in home and community-based settings in FY 2018 or in FY 2019. In FY 2018, Missouri increased the state’s institutional level of care standard, which affected the eligibility of 445 waiver participants upon reassessment and of over 1,200 waiver applicants on pre-assessment screening. In FY 2019, Michigan may reduce the number of slots available under its MI Choice 1915(c) waiver, which serves seniors and adults with physical disabilities, to reflect available funding.
Tennessee Rebalancing Incentives in MCO Contracts
Tennessee pays its MLTSS health plans a blended capitation rate for older adults and adults with physical disabilities who meet Nursing Facility (NF) level of care (LOC) and are receiving services in a NF or HCBS. First, the state develops actuarially sound rates for each service setting. The mix of individuals receiving services in each setting (NF vs. HCBS) is determined and a target is established for how the percentages are expected to change during the rating period. The two capitation rates are blended according to those percentages, resulting in a single capitation payment for all persons who meet NF LOC. This is done separately for the dual eligible and the non-dual populations in each region. Because reimbursement is the same for NFs or HCBS, there is an incentive to serve people in the community whenever possible (both delaying or preventing NF placement as well as transitioning from NF placement to the community when appropriate). MCOs are also incentivized to ensure that services in the community are sufficient to meet the person’s needs since they are at financial risk for the higher cost NF placement.
In this year’s survey, states were also asked to identify the most significant rebalancing challenges they currently face. Among states that responded, the challenges most frequently cited included lack of affordable and accessible housing, gaps in community-based provider capacity (especially in rural areas) and/or direct care workforce shortages, reimbursement challenges (e.g., rising and/or more favorable rates paid to nursing facilities compared to HCBS providers and the need to risk adjust rates as patterns of utilization change), and the expiration of the Money Follows the Person (MFP) program.
LTSS Direct Care Workforce
Many states are struggling to find sufficient numbers of trained direct care workers to meet the demand for services, including the demand for care in home and community-based settings.63 ,64 Low wages, few benefits, limited opportunities for career advancement, inadequate training, and high rates of worker injury are factors that also contribute to a workforce shortage and high workforce turnover among paid LTSS direct care workers. The National Center for Health Workforce Analysis projects that demand for direct care workers (including nursing assistants, home health aides, personal care aides, and psychiatric assistants/aides) could grow by 48% between 2015 and 2030, growth that is expected to far exceed the available workforce.65
To address LTSS direct care workforce shortages and turnover, increasingly states are reporting implementing wage increases and workforce development activities (Exhibit 13). In FY 2018, 15 states reported implementing wage increases for Medicaid-reimbursed direct care workers, while 24 states report implementing wage increases in FY 2019 (14 states in both years). In addition, 12 states had direct care workforce development strategies (e.g., recruiting, training, credentialing) in place in FY 2018, and 10 states reported expanding or implementing new workforce development strategies in FY 2019 (Exhibit 13).
| Exhibit 13: Strategies to Address LTSS Direct Care Workforce Shortages & Turnover | |||
| Fiscal Year | # of States | States | |
| Wage Increases | 2018 | 15 | AZ, CA, CO, CT, MA, MD, MI, MT, NH, NY, TN, UT, VT, WA, WI |
| 2019 | 24 | AZ, CA, CO, CT, DE, HI, IL, MA, MD, MI, MN, MT, NC, NJ, NY, OK, OR, TN, UT, VA, VT, WA, WI, WV | |
| Workforce Development(including recruiting, training, credentialing etc.) | In PlaceFY 2018 | 12 | AZ, CA, CT, MA, NH, NY, OR, PA, SC, TN, WA, WI |
| New/Expanded FY 2019 | 10 | AR, AZ, IA, MN, NC, PA, TN, VT, WA, WI | |
LTSS Direct Care Workforce Initiatives – State Examples
- Arizona requires Medicaid MCOs to incorporate and monitor a workforce development plan as a component of its network development and management plan, with the Medicaid program partnering with the LTSS industry to determine workforce development priorities.
- North Carolina is expanding workforce options by adopting a new live-in support service that allows a caregiver to move into a beneficiary’s home or for the beneficiary to move into the caregiver’s home.
- Tennessee is using federal State Innovation Model (SIM) test grant funding to create new education and training curriculum for direct care workers, where individuals will be able to earn college credit, complete a post-secondary certificate, and apply credits toward a new Associate’s degree.
HCBS Benefit Changes
More states reported actions to add or enhance HCBS benefits than states reporting actions to reduce or restrict HCBS benefits in FY 2018 and FY 2019. HCBS benefits include those in Section 1915(c) or Section 1115 waivers, under Section 1915(i) authority or Section 1915(k) authority (“Community First Choice” or “CFC”), PACE, and state plan personal care services, home health services, or private duty nursing. Eighteen states in FY 2018 and 26 states in FY 2019 reported a wide variety of HCBS benefit additions or expansions (Exhibit 14). Most HCBS benefit changes reported involve the addition of HCBS services to existing waiver or state plan programs. Examples of HCBS services added by states include new housing-related services or embedded post-MFP community transition services in HCBS authorities; changes to increase access to respite services or to provide training for family, consumers, and unpaid caregivers; enhanced transportation services; and pest eradication services.
A few states implemented new HCBS programs in FY 2018 or FY 2019. In FY 2018, Idaho added a new 1915(i) state plan program for children to offer respite and person-specific planning supports. Maryland added two new 1915(c) waivers to provide family and community support for individuals with intellectual and developmental disabilities (I/DD). In FY 2019, California will implement a new 1915(i) program that will add housing access, family support, and other services for individuals with I/DD. Rhode Island proposes to add a Section 1915(k) Community First Choice program, and Alaska plans to add a new Section 1915(k) program. Seven states reported adding new PACE sites in both FY 2018 and FY 2019. Nine additional states will add new PACE sites in FY 2019 (Exhibit 14).
| Exhibit 14: HCBS Benefit Enhancements or Additions | ||||
| Benefit | FY 2018 | FY 2019 | ||
| HCBS Enhancements or Additions to Existing HCBS Authority | 12 States | CA, LA, MA, MI, MN, OH, PA, RI, SC, TX, UT, VA | 15 States | CO, HI, ID, IN, MA, MI, NC, ND, NM, NY, OH, PA, SD, TN, VA |
| New Section 1915(c), (i), or (k) | 2 States | ID, MD | 3 States | AK, CA, RI |
| New PACE Sites Added | 7 States | CA, MI, NJ, NY, OR, PA, WA | 16 States | AR, CA, CO, DC, DE, FL, IN, MI, NC, ND, NJ, NY, OR, PA, TX, WA |
Rhode Island Proposed Section 1115 HCBS
Rhode Island is proposing to add additional preventive HCBS for target populations to further reduce or prevent the use of high cost services under its Section 1115 waiver renewal. Examples of new preventive services the state is proposing include home stabilization, peer support, chore services, and personal emergency response systems. The state also proposes to add services to its core HCBS, including but not limited to career planning, community transition, home stabilization, and training and counseling services for unpaid caregivers.
Three states in FY 2018 (Missouri, Montana, and Oregon) and two states in FY 2019 (DC and Montana) implemented or plan to implement benefit changes that will reduce services under HCBS authorities. States reported targeted restrictions, noting they are being introduced to meet budget neutrality requirements or to reflect changes in available state funding. For example, DC proposes restricting the number of personal care assistance hours available under its Section 1915(i) elderly and persons with physical disabilities state plan option, and Montana is eliminating several services from its Section 1915(c) waivers for individuals with I/DD and those with severe disabling mental illness.66
Money Follows the Person and Housing Supports
Money Follows the Person (MFP) is a federal grant program, enacted under the Deficit Reduction Act of 2005 and extended through September 2016 by the Affordable Care Act, which operated in 44 states.67 ,68 Enhanced federal funding under MFP has supported the transition of over 75,151 individuals from institutional to home and community-based long-term care settings as of December 2016.69 This includes the transition of older adults, individuals with physical disabilities, individuals with mental illness, and individuals with intellectual and developmental disabilities. Although states are developing sustainability plans and completing tasks to close the current MFP grant program, states can use unexpended MFP grant funds through the end of federal FY 2020. However, a few states reported in this year’s survey that they have already exhausted their MFP grants.
Although many states are still developing sustainability plans and making determinations about whether and which services may continue, 30 states identified specific housing-related services that they plan to continue after MFP funding expires. With MFP resources, many states have offered new housing-related services, incorporated housing expertise within the Medicaid program to increase the likelihood of successful community living for persons who need supports, and engaged in strategic activities to assist in identifying and securing housing resources for individuals who choose HCBS.70 In this year’s survey, states were asked to describe housing-related services that will continue (under SPA or waiver authority) after the MFP funding expires.71 The most common services that states expect to continue are transition or relocation services (e.g., case management, coverage for one-time set up costs etc.) and services designed to help individuals locate and maintain housing in the community (e.g., tenancy supports, housing coordination, or supported housing).
About half of MFP-funded states anticipate they will have to discontinue services or administrative activities due to the expiration of MFP funding. States identified a wide range of services and key administrative functions that they expect to discontinue. Examples of services some states may discontinue include intensive transition case management, supportive living services, community transition/housing relocation services, transitional behavioral health supports, and residential environmental modifications. Examples of administrative functions some states may discontinue include statewide housing coordinator, local housing specialists, transition and outreach workers, options counseling, and assistance for individuals to access Section 811 vouchers.
Capitated Managed Long-Term Services and Supports (MLTSS)
As of July 1, 2018, almost half of states (24 states) covered LTSS through one or more of the following types of capitated managed care arrangements:
- Medicaid MCO covering Medicaid acute care and LTSS (20 states)
- PHP covering only Medicaid LTSS (6 states)
- MCO arrangement for dual eligible beneficiaries covering Medicaid and Medicare acute care and Medicaid LTSS services in a single, financially aligned contract under the federal Financial Alignment Demonstration (FAD) (9 states)
Of the 24 states that reported using one or more of these MLTSS models, nine states reported using two models, and one state (New York) reported using all three. Of the states with capitated MLTSS, 17 offered some form of MLTSS plan on a statewide basis for at least some LTSS populations as of July 1, 2018 (Table 10). Almost every MLTSS state includes both institutional and HCBS in the same contractual arrangement, while three states (California, Michigan, and Tennessee) report that this varies by MLTSS arrangement.
Nine states offered an MCO-based FAD (California, Illinois, Massachusetts, Michigan, New York, Ohio, Rhode Island, South Carolina, and Texas) as of July 1, 2018.72 The FAD model involves a three-way contract between an MCO, Medicare, and the state Medicaid program.73 ,74 Four states have requested an extension of the FAD beyond the current end date of the demonstration at the time of the survey (California, Massachusetts, Ohio, and South Carolina). Virginia closed its FAD at the end of calendar year 2017.
Many states encourage improved coordination and integration of services for the dually eligible population under MCO arrangements outside of the FAD. Massachusetts and Minnesota operate an administrative alignment demonstration (with no financial alignment) for some dually eligible beneficiaries. Nine states75 reported that they require Medicaid-contracting MCOs to be Medicare Dual Eligible Special Needs Plans (D-SNP)76 or Fully Integrated Dual Eligible (FIDE) Special Needs Plans77 in some or all MLTSS models offered in the state, creating an opportunity for improved coordination and integration for beneficiaries. Five states78 reported that they encourage MCOs to be a D-SNP or a FIDE-SNP.
MLTSS Enrollment
For geographic areas where MLTSS operates, this year’s survey asked whether, as of July 1, 2018, certain populations were enrolled in MLTSS on a mandatory or voluntary basis or were always excluded. On the survey, states selected from “always mandatory,” “always voluntary,” “varies,” or “always excluded” for the following dually eligible and non-dually eligible populations: seniors, persons with I/DD, and nonelderly persons with physical disabilities. Dual eligible and non-dual eligible seniors were most likely to be enrolled on a mandatory basis followed closely by persons with physical disabilities. Dual and non-dual persons with I/DD were most likely to be excluded from MLTSS enrollment. No state offering MLTSS always excludes full benefit dual eligible seniors or persons with physical disabilities from MLTSS enrollment (Exhibit 15).
| Exhibit 15: MLTSS Enrollment by Populations (# of States) | ||||||
| Non-Dual Eligibles | Dual Eligibles | |||||
| Seniors | Persons w/ Physical Disabilities | Persons w/ I/DD | Seniors | Persons w/ Physical Disabilities | Persons w/I/DD79 | |
| Always mandatory | 16 | 15 | 7 | 18 | 17 | 7 |
| Always voluntary | 1 | 2 | 3 | 3 | 4 | 4 |
| Varies | 3 | 3 | 8 | 3 | 3 | 8 |
| Always excluded | 4 | 4 | 6 | 0 | 0 | 4 |
MLTSS Population Changes
In FY 2018, Pennsylvania implemented MLTSS, with a plan to phase-in statewide over time, and Virginia ended its FAD but adopted statewide MLTSS for a wider population, including dual eligible individuals. Also, one state, Arkansas, reported that it will implement a capitated model of MLTSS for the first time in FY 2019 when it plans to adopt a global payment approach for its new “PASSE” program that provides comprehensive services, including personal care and other HCBS specialty services, for individuals who have the need for an intensive level of community based behavioral health or developmental disabilities services. While the state will exempt individuals who are receiving services under the DD waiver from this model, individuals who are on the waiting list for waiver services will be enrolled, as well as individuals who reside in private ICF-IDs.
In total, two states expanded the geographic reach of MLTSS in FY 2018 and five states are expanding MLTSS geographically in FY 2019 (Exhibit 16). Also, four states added previously excluded populations to MLTSS arrangements in FY 2018 and five states will add previously excluded populations in FY 2019. In all states except for South Carolina (where the state is expanding voluntary enrollment options under its FAD to all Medicare Advantage enrolled seniors statewide), the new populations covered are subject to mandatory enrollment. Rhode Island reported ending its MLTSS contract for individuals who opt out of the FAD, effective September 2018. After that date, individuals who opt out of the FAD will return to Medicaid fee-for-service.
| Exhibit 16: MLTSS Population Expansions, FY 2018 and FY 2019 | ||
| FY 2018 | FY 2019 | |
| Geographic Expansions | ID, MA | ID, IL, MA, PA, SC |
| New Population Groups Added | FL, ID, NY, VA | ID, NY, OH, PA, SC |
| Implementing an MLTSS program for the First Time | PA | AR |
Table 9: Long-Term Care Actions to Serve More Individuals in Community Settings in all 50 States and DC, FY 2018 and FY 2019
| States | Sec. 1915(c)or Sec. 1115 HCBS Waiver | Sec. 1915(i) HCBS State Plan Option | Sec. 1915(k) “Community First Choice” Option | Building Rebalancing Incentives into MLTSS | PACE(* indicatesnew sites) | Close/ Downsize Institution | Total States with HCBS Expansions | |||||||
| 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | |
| Alabama | X | X | X | X | ||||||||||
| Alaska | X | X | X | |||||||||||
| Arizona | X | X | X | X | ||||||||||
| Arkansas | X | X | X* | X | X | |||||||||
| California | X | X | X | X | X | X | X | X* | X* | X | X | X | X | |
| Colorado | X | X | X | X | X | X* | X | X | ||||||
| Connecticut | X | X | X | X | X | X | X | X | X | |||||
| DC* | X | X | X | X | X | X | X | X* | X | X | ||||
| Delaware | X* | X | ||||||||||||
| Florida | X | X | X | X | X* | X | X | |||||||
| Georgia | X | X | X | X | ||||||||||
| Hawaii | X | X | X | X | ||||||||||
| Idaho | X | X | X | X | X | X | ||||||||
| Illinois | X | X | X | X | X | X | ||||||||
| Indiana | X | X | X | X | X | X* | X | X | ||||||
| Iowa | X | X | X | X | X | X | X | X | ||||||
| Kansas | X | X | X | X | ||||||||||
| Kentucky | ||||||||||||||
| Louisiana | X | X | X | X | ||||||||||
| Maine | X | X | X | X | ||||||||||
| Maryland | X | X | X | X | X | X | X | X | X | |||||
| Massachusetts | X | X | X | X | ||||||||||
| Michigan | X | X | X | X | X | X* | X* | X | X | |||||
| Minnesota | ||||||||||||||
| Mississippi | X | X | X | X | X | X | ||||||||
| Missouri | X | X | ||||||||||||
| Montana | X | X | X | X | X | X | X | |||||||
| Nebraska | X | X | X | X | ||||||||||
| Nevada | X | X | X | X | X | |||||||||
| New Hampshire | X | X | X | X | ||||||||||
| New Jersey | X | X | X* | X* | X | X | ||||||||
| New Mexico | X | X | X | X | X | X | X | |||||||
| New York | X | X | X | X | X | X | X | X | X* | X* | X | X | X | X |
| North Carolina | X* | X | ||||||||||||
| North Dakota | X | X | X* | X | X | X | ||||||||
| Ohio | X | X | X | X | X | X | X | X | X | |||||
| Oklahoma | X | X | X | X | X | X | ||||||||
| Oregon | X | X | X | X | X* | X* | X | X | ||||||
| Pennsylvania | X | X | X | X | X* | X* | X | X | X | X | ||||
| Rhode Island | X | X | X | X | X | |||||||||
| South Carolina | X | X | X | X | X | X | X | X | ||||||
| South Dakota | X | X | X | X | ||||||||||
| Tennessee | X | X | X | X | X | X | ||||||||
| Texas | X | X | X | X | X | X | X | X | X* | X | X | |||
| Utah | X | X | X | X | ||||||||||
| Vermont | X | X | X | X | ||||||||||
| Virginia | X | X | X | X | X | X | X | |||||||
| Washington | X | X | X | X | X* | X* | X | X | X | X | ||||
| West Virginia | X | X | X | X | ||||||||||
| Wisconsin | X | X | X | X | X | X | X | |||||||
| Wyoming | X | X | X | X | X | X | ||||||||
| Totals | 40 | 40 | 12 | 14 | 8 | 9 | 17 | 18 | 16 | 22 | 10 | 7 | 46 | 48 |
| NOTES: “1915(c) or Sec. 1115 Waiver” actions include: adopting new waivers; adding and filling more waiver slots; or filling more waiver slots. Actions under “1915(i) and 1915(k)” include adding new 1915(i) or 1915(k) SPAs or serving more individuals through existing 1915(i) or 1915(k) SPAs. Actions under PACE include more individuals served in existing and/or new PACE sites, with an * indicating which states expect new sites in FY 2018 or FY 2019. NY noted they will add one or more PACE sites in FY 2018 and FY 2019 but also indicated enrollment in PACE has declined. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||||||||
Table 10: Capitated MLTSS Models in all 50 States and DC, as of July 1, 2018
| States | Medicaid MCO | PHP | Financial Alignment Demonstration(FAD) for Duals | Any MLTSS | Statewide |
| Alabama | |||||
| Alaska | |||||
| Arizona | X | X | X | ||
| Arkansas | |||||
| California | X | X | X | ||
| Colorado | |||||
| Connecticut | |||||
| Delaware | X | X | X | ||
| DC | |||||
| Florida | X | X | X | ||
| Georgia | |||||
| Hawaii | X | X | X | ||
| Idaho | X | X | |||
| Illinois | X | X | X | ||
| Indiana | |||||
| Iowa | X | X | X | ||
| Kansas | X | X | X | ||
| Kentucky | |||||
| Louisiana | |||||
| Maine | |||||
| Maryland | |||||
| Massachusetts | X | X* | X | ||
| Michigan | X | X | X | X | |
| Minnesota* | X* | X | X | ||
| Mississippi | |||||
| Missouri | |||||
| Montana | |||||
| Nebraska | |||||
| Nevada | |||||
| New Hampshire | |||||
| New Jersey | X | X | X | ||
| New Mexico | X | X | X | ||
| New York | X | X | X | X | X |
| North Carolina | X | X | X | ||
| North Dakota | |||||
| Ohio | X | X | X* | ||
| Oklahoma | |||||
| Oregon | |||||
| Pennsylvania | X | X | |||
| Rhode Island | X | X | X | X | |
| South Carolina | X | X | |||
| South Dakota | |||||
| Tennessee | X | X | X | X | |
| Texas | X | X | X | X | |
| Utah | |||||
| Vermont | |||||
| Virginia | X | X | X | ||
| Washington | |||||
| West Virginia | |||||
| Wisconsin | X | X | X | X | |
| Wyoming | |||||
| Totals | 20 | 6 | 9 | 24 | 17 |
| NOTES: States were asked whether they cover long-term services and supports through any of the following managed care (capitated) arrangements as of July 1, 2018: Medicaid MCO (MCO covers Medicaid acute + Medicaid LTSS); PHP (covers only Medicaid LTSS); MCO arrangement for dual eligibles under the Financial Alignment Demonstration (Medicaid MCO covers Medicaid and Medicare acute + Medicaid LTSS). *ID operates a PHP that covers LTSS in conjunction with an Medicare Advantage plan in selected counties and expects to expand to new counties in FY 2018 and FY 2019. *MA operates a FAD and a separate administrative alignment-only demonstration for dually eligible beneficiaries. *MN operates an administrative alignment-only demonstration for dually eligible beneficiaries using an MCO arrangement. *OH offers a Medicaid MCO (MCO offers Medicaid acute + Medicaid LTSS) only in those counties where the FAD is offered; dually eligible seniors who opt out of the FAD must enroll in this Medicaid MCO model for Medicaid services. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | |||||
Report: Provider Rates And Taxes
Key Section Findings
Provider rate changes are often tied to the economy. In FY 2018 and FY 2019, with stable economic conditions in most states, more states made or are planning provider rate increases compared to restrictions. This holds true across provider types, except for inpatient hospital rates (inpatient hospital rate restrictions are primarily rate freezes, which are counted as restrictions in this report). Further, the number of states that reported at least one rate restriction in FY 2019 is the smallest number since FY 2008. All states except Alaska rely on provider taxes and fees to fund a portion of the non-federal share of the costs of Medicaid. Two states indicate plans for new provider taxes in FY 2019, including Virginia that plans a new hospital provider tax to finance state costs of the newly adopted Medicaid expansion. Over half of MCO states (21 of 39) require MCO payments for some or all types of providers to follow percent or level changes made in comparable FFS rates. Twenty-seven states reported that their MCO contracts include rate floors for some provider types, and five states reported they had minimum MCO payment requirements for all types of Medicaid providers.
What to watch:
- As enrollees are predominantly in MCOs, the significance of changes in FFS payment rates is difficult to assess without a better understanding of how changes in FFS rates affect changes in MCO rates paid to providers.
- Twenty-nine states have at least one provider tax that is at or above 5.5% of net patient revenues (close to the maximum safe harbor threshold of 6%). Therefore, federal action to lower that threshold as proposed in the past would have financial implications for many states.
Tables 11 through 13 provide complete listings of Medicaid provider rate changes and provider taxes and fees in place in FY 2018 and FY 2019.
Provider Rates
Provider rate changes are often tied to the economy. During economic downturns and budget shortfalls, states often turn to rate restrictions to contain costs, and during periods of recovery and revenue growth, states are more likely to increase rates. This report examines rate changes across major provider categories: inpatient hospitals, nursing facilities, MCOs, outpatient hospitals, primary care physicians, specialists, dentists, home and community-based services (HCBS), and pharmacy dispensing fees. States were asked to report aggregate rate changes for each provider category in their FFS programs.
The number of states that made or are planning rate increases exceeds the number implementing or planning rate restrictions in both FY 2018 and FY 2019. In FY 2018, almost every state implemented rate increases for at least one category of providers (49 states), while fewer implemented rate restrictions (38 states) (Figure 7 and Table 11). For FY 2019, the number of states with at least one implemented or planned rate increase (47 states) is greater than the number of states with at least one implemented or planned rate restriction (31 states) (Figure 7 and Table 12). The number of states that reported at least one rate restriction in FY 2019 is the smallest number since FY 2008.
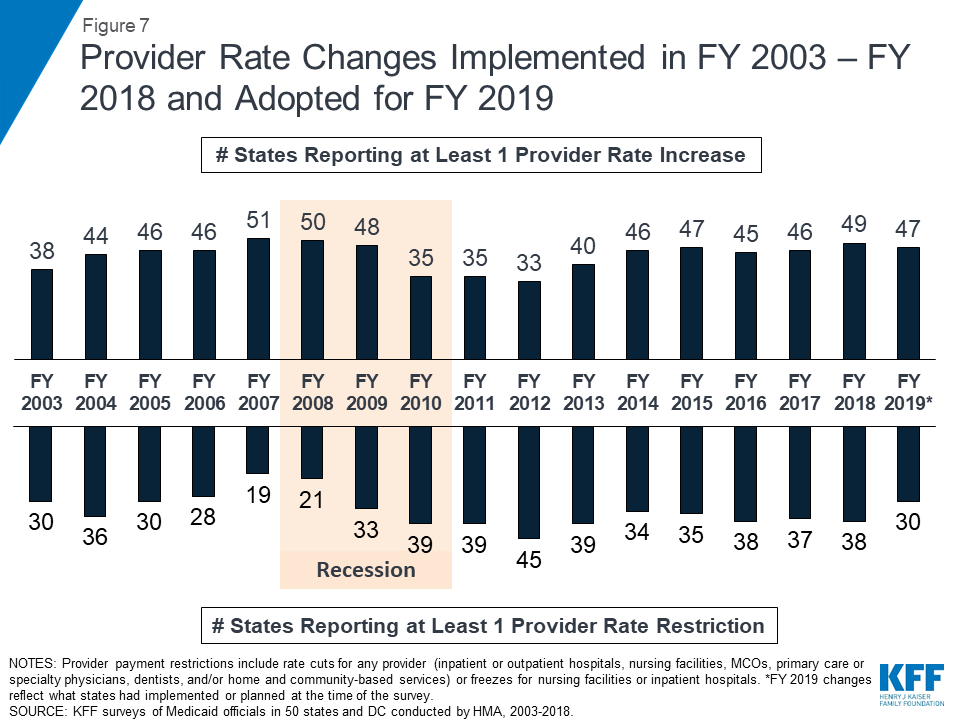
The number of states with rate increases exceeds the number of states with restrictions in FY 2018 and FY 2019 across all major categories of providers, with the exception of rates for inpatient hospital services (Figure 8 and Tables 11 and 12). For the purposes of this report, cuts or freezes in rates for inpatient hospitals and nursing facilities are counted as restrictions.80 Most of the restrictions of inpatient hospital rates are rate freezes. Three states in FY 2018 and three states in FY 2019 had implemented or planned cuts to inpatient hospital rates. While three states cut nursing facility rates in FY 2018, no states indicate plans to cut nursing facility rates in FY 2019.
The number of states planning to increase nursing facility rates in FY 2019 (40 states) is greater than the number of states increasing those rates in FY 2018 (34 states). HCBS providers were also among those most likely to receive rate increases (32 states in FY 2018 and 31 states in FY 2019) (Figure 8).
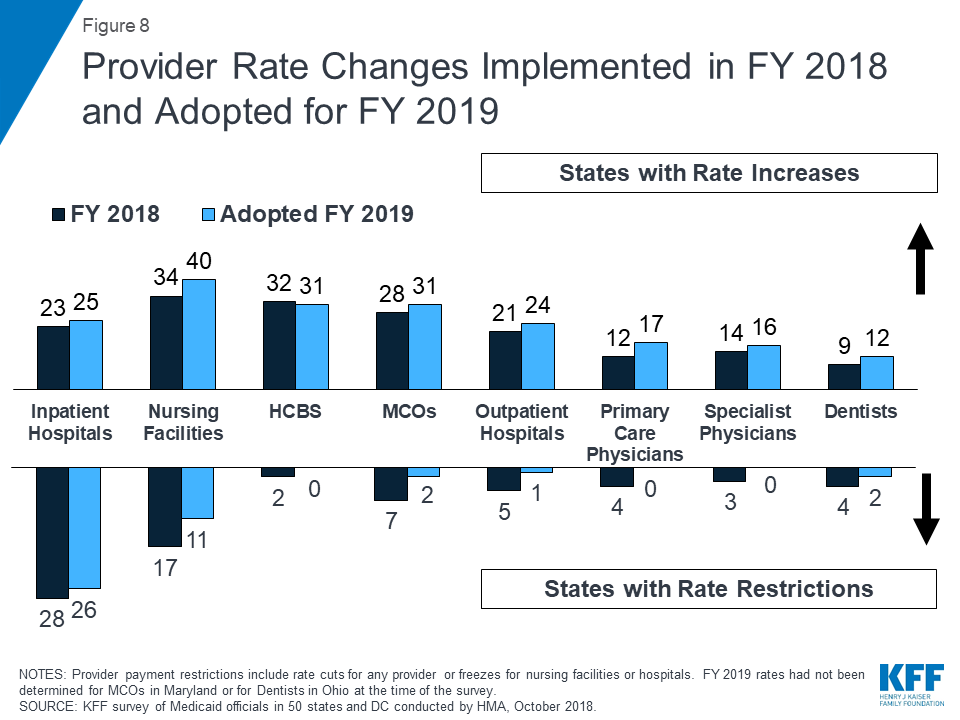
State authority to adjust capitation payments for MCOs is limited by the federal requirement that states pay actuarially sound rates. In FY 2018 and FY 2019, the majority of the 39 states with Medicaid MCOs either implemented or planned increases in MCO rates. While seven states reported MCO rate cuts in FY 2018, only two states plan to cut MCO rates in FY 2019.81
MCO Rate Requirements
Over half of MCO states require MCOs to change provider payment rates in accordance with FFS payment rate changes. In many states, MCOs make most of the Medicaid payments to providers. This year’s survey asked states to report whether they require their MCOs to make changes to their provider payments that follow percent or level changes in FFS rates. Of the 39 states with MCOs, 18 states indicated that they had no such requirement, 19 states have such a requirement for some provider types, and two states (Louisiana and Mississippi) required MCOs to make these changes for all types of Medicaid providers.
Most MCO states mandate minimum provider reimbursement rates in their MCO contracts. Of the 39 MCO states, seven indicated that they had no rate floors, 27 states indicated that they had rate floors for some provider types, and five states said they had minimum MCO payment requirements for all Medicaid provider types. Among states with rate floors for some provider types, the most commonly mentioned providers were long term care providers (nursing facilities and home and community-based service providers), community health centers (federally qualified health centers and rural health centers), and various providers of behavioral health services.
Provider Taxes and Fees
Provider taxes are an integral source of Medicaid financing. In this year’s survey, states reported continuing or increased reliance on provider taxes and fees to fund a portion of the non-federal share of Medicaid costs in FY 2018 and FY 2019. At the beginning of FY 2003, 21 states had at least one provider tax in place. Over the next decade, a majority of states imposed new taxes or fees and increased existing tax rates and fees to raise revenue to support Medicaid. By FY 2013, all but one state (Alaska) had at least one provider tax or fee in place.82 In FY 2018, 36 states, including DC, had three or more provider taxes in place (Figure 9).

Very few states made or are making any changes to their provider tax structure in FY 2018 or FY 2019. The most common Medicaid provider taxes in place in FY 2018 were taxes on nursing facilities (44 states), followed by taxes on hospitals (42 states) and taxes on intermediate care facilities for people with intellectual disabilities (36 states) (Table 13). Two states reported plans to add new taxes in FY 2019. Virginia is implementing a new hospital tax to fund the state’s share of Medicaid expansion costs, and California is implementing a new tax on Ground Emergency Medical Transportation (GEMT or ambulance).
Eleven states report planned increases to one or more provider taxes in FY 2019, while six states report provider tax decreases. In addition, 29 states reported at least one provider tax that is at or above 5.5% of net patient revenues, which is close to the maximum federal safe harbor threshold of 6%. Federal action to lower that threshold, as has been proposed in the past, would therefore have financial implications for many states.
Twelve states report that they have taxes on MCOs as of FY 2018. Federal Medicaid law was changed83 effective July 1, 2009 to restrict the use of Medicaid provider taxes on managed care organizations such as HMOs. Prior to that date, states could apply a provider tax to Medicaid HMOs that did not apply to MCOs more broadly and could use that revenue to match Medicaid federal funds. In recent years, several states have implemented new MCO taxes that tax member months rather than premiums and that meet the federal statistical requirements for broad-based and uniform taxes. As a result, the number of MCO taxes has increased in recent years. In addition to the 12 states reporting MCO taxes, some states have implemented taxes on health insurers more broadly that generate revenue for their Medicaid programs.
An increasingly common provider tax is a tax on Ground Emergency Medical Transportation, or an ambulance tax. As noted above, California is implementing such a tax in FY 2019, bringing the number of states with an ambulance tax to eight states.
Table 11: Provider Rate Changes in all 50 States and DC, FY 2018
| States | Inpatient Hospital | Outpatient Hospital | Primary Care Physicians | Specialists | Dentists | MCOs | Nursing Facilities | HCBS | Pharmacy Dispensing Fee | Total | ||||||||||
| Rate Change | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – |
| Alabama | X | — | — | X | X | X | X | |||||||||||||
| Alaska | X | X | X | X | X | — | — | X | X | |||||||||||
| Arizona | X | X | X | X | X | X | X | X | X | |||||||||||
| Arkansas | X | — | — | X | X | X | ||||||||||||||
| California | X | X | X | X | X | X | X | X | X | X | ||||||||||
| Colorado | X | X | X | X | X | X | X | X | ||||||||||||
| Connecticut | X | X | — | X | X | X | ||||||||||||||
| Delaware | X | X | X | X | X | X | X | X | X | |||||||||||
| DC | X | X | X | X | X | X | X | |||||||||||||
| Florida | X | X | X | X | X | X | X | X | ||||||||||||
| Georgia | X | X | X | X | X | X | X | X | ||||||||||||
| Hawaii | X | X | X | X | X | X | X | |||||||||||||
| Idaho | X | X | X | X | — | — | X | X | X | X | ||||||||||
| Illinois | X | X | X | X | X | |||||||||||||||
| Indiana | X | — | X | X | X | X | X | |||||||||||||
| Iowa | X | X | X | X | X | X | ||||||||||||||
| Kansas | X | X | X | X | X | X | X | X | X | |||||||||||
| Kentucky | X | X | X | X | X | X | ||||||||||||||
| Louisiana | X | X | X | X | X | X | ||||||||||||||
| Maine | X | — | — | X | X | X | X | X | ||||||||||||
| Maryland | X | X | X | X | X | X | X | X | ||||||||||||
| Massachusetts | X | X | X | X | X | X | X | X | X | |||||||||||
| Michigan | X | X | X | X | X | X | X | |||||||||||||
| Minnesota | X | X | X | X | X | X | X | X | ||||||||||||
| Mississippi | X | X | X | X | X | X | X | X | ||||||||||||
| Missouri | X | X | X | X | X | X | X | X | X | X | X | |||||||||
| Montana | X | X | X | X | X | — | — | X | X | X | X | X | ||||||||
| Nebraska | X | X | X | X | X | X | ||||||||||||||
| Nevada | X | X | X | X | X | X | X | X | X | |||||||||||
| New Hampshire | X | X | X | X | NR | X | X | |||||||||||||
| New Jersey | X | X | X | X | X | X | X | |||||||||||||
| New Mexico | X | X | X | X | ||||||||||||||||
| New York | X | X | X | X | X | X | ||||||||||||||
| North Carolina | X | — | — | X | X | X | ||||||||||||||
| North Dakota | X | X | X | X | X | |||||||||||||||
| Ohio | X | X | X | X | X | X | ||||||||||||||
| Oklahoma | X | — | — | X | X | |||||||||||||||
| Oregon | X | X | X | X | X | X | X | X | ||||||||||||
| Pennsylvania | X | X | X | X | X | X | X | |||||||||||||
| Rhode Island | X | X | X | X | X | NR | X | X | ||||||||||||
| South Carolina | X | X | X | X | X | X | X | |||||||||||||
| South Dakota | X | — | — | X | X | X | X | |||||||||||||
| Tennessee | X | X | X | X | X | X | X | |||||||||||||
| Texas | X | X | X | X | X | |||||||||||||||
| Utah | X | X | X | X | X | X | ||||||||||||||
| Vermont | X | X | X | — | — | X | X | X | X | |||||||||||
| Virginia | X | X | X | X | X | |||||||||||||||
| Washington | X | X | X | X | ||||||||||||||||
| West Virginia | X | X | X | X | X | X | ||||||||||||||
| Wisconsin | X | X | X | X | X | X | X | X | ||||||||||||
| Wyoming | X | — | — | X | X | X | X | |||||||||||||
| Totals | 23 | 28 | 21 | 5 | 12 | 4 | 14 | 3 | 9 | 4 | 28 | 7 | 34 | 17 | 32 | 2 | 12 | 1 | 49 | 38 |
| NOTES: “+” refers to provider rate increases and “-” refers to provider rate restrictions. MCOs: Managed care organizations. HCBS: Home and community-based services. For the purposes of this report, provider rate restrictions include cuts to rates for physicians, dentists, outpatient hospitals, managed care organizations, HCBS, and pharmacy dispensing fees as well as both cuts or freezes in rates for inpatient hospitals and nursing facilities. There are 12 states that did not have Medicaid MCOs in operation in FY 2018; they are denoted as “—” in the MCO column. NR: State did not report. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||||||||||||||
Table 12: Provider Rate Changes in all 50 States and DC, FY 2019
| States | Inpatient Hospital | Outpatient Hospital | Primary Care Physicians | Specialists | Dentists | MCOs | Nursing Facilities | HCBS | Pharmacy Dispensing Fee | Total | ||||||||||
| Rate Change | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – |
| Alabama | X | — | — | X | X | X | X | |||||||||||||
| Alaska | X | X | X | X | X | — | — | X | X | X | ||||||||||
| Arizona | X | X | X | X | X | X | X | X | ||||||||||||
| Arkansas | X | — | — | X | X | X | ||||||||||||||
| California | X | X | X | X | X | X | X | X | X | X | ||||||||||
| Colorado | X | X | X | X | X | X | X | X | X | |||||||||||
| Connecticut | X | — | — | X | X | |||||||||||||||
| Delaware | X | X | X | X | X | X | X | X | X | X | ||||||||||
| DC | X | X | X | X | X | X | X | X | X | |||||||||||
| Florida | X | X | X | X | X | X | X | |||||||||||||
| Georgia | X | X | X | X | X | X | X | |||||||||||||
| Hawaii | X | X | X | X | X | X | ||||||||||||||
| Idaho | X | X | X | X | — | — | X | X | X | X | ||||||||||
| Illinois | X | X | X | X | ||||||||||||||||
| Indiana | X | X | X | |||||||||||||||||
| Iowa | X | X | X | X | X | X | ||||||||||||||
| Kansas | X | X | X | X | X | X | ||||||||||||||
| Kentucky | X | X | X | X | X | X | ||||||||||||||
| Louisiana | X | X | X | X | X | |||||||||||||||
| Maine | X | — | — | X | X | X | X | |||||||||||||
| Maryland | X | X | X | X | TBD | TBD | X | X | X | |||||||||||
| Massachusetts | X | X | X | X | X | X | ||||||||||||||
| Michigan | X | X | X | X | X | X | X | |||||||||||||
| Minnesota | X | X | X | X | X | X | X | X | ||||||||||||
| Mississippi | X | X | X | X | X | X | X | |||||||||||||
| Missouri | X | X | X | X | X | X | X | X | X | X | ||||||||||
| Montana | X | X | X | X | X | — | — | X | X | X | X | |||||||||
| Nebraska | X | X | X | |||||||||||||||||
| Nevada | X | X | X | X | X | X | ||||||||||||||
| New Hampshire | X | X | X | NR | X | X | ||||||||||||||
| New Jersey | X | X | X | X | X | X | X | X | X | |||||||||||
| New Mexico | X | X | X | X | X | X | X | |||||||||||||
| New York | X | X | X | X | X | X | ||||||||||||||
| North Carolina | X | — | — | X | X | X | ||||||||||||||
| North Dakota | X | X | X | X | X | |||||||||||||||
| Ohio | X | X | TBD | TBD | X | X | X | X | ||||||||||||
| Oklahoma | X | X | X | X | X | — | — | X | X | X | X | |||||||||
| Oregon | X | X | X | |||||||||||||||||
| Pennsylvania | X | X | X | X | X | X | ||||||||||||||
| Rhode Island | X | X | X | X | X | NR | X | X | ||||||||||||
| South Carolina | X | X | X | X | X | X | ||||||||||||||
| South Dakota | X | X | X | X | X | — | — | X | X | X | ||||||||||
| Tennessee | X | X | X | X | X | |||||||||||||||
| Texas | X | X | X | X | X | X | ||||||||||||||
| Utah | X | X | X | X | X | X | ||||||||||||||
| Vermont | X | X | X | — | — | X | X | X | ||||||||||||
| Virginia | X | X | X | X | X | |||||||||||||||
| Washington | X | X | X | X | X | X | ||||||||||||||
| West Virginia | X | X | X | X | X | |||||||||||||||
| Wisconsin | X | X | X | X | X | TBD | TBD | X | ||||||||||||
| Wyoming | X | — | — | X | X | X | X | |||||||||||||
| Totals | 25 | 26 | 24 | 1 | 17 | 0 | 16 | 0 | 12 | 2 | 31 | 2 | 40 | 11 | 31 | 0 | 7 | 0 | 47 | 31 |
| NOTES: “+” refers to provider rate increases and “-” refers to provider rate restrictions. MCOs: Managed care organizations. HCBS: Home and community-based services. For the purposes of this report, provider rate restrictions include cuts to rates for physicians, dentists, outpatient hospitals, managed care organizations, HCBS, and pharmacy dispensing fees as well as both cuts or freezes in rates for inpatient hospitals and nursing facilities. There are 12 states that did not have Medicaid MCOs in operation in FY 2019; they are denoted as “–” in the MCO column. TBD: At the time of the survey, calendar year 2019 MCO rates had not been set for Maryland, rates for dentists were in development in Ohio, and Wisconsin was considering changes to pharmacy dispensing fees. NR: State did not report. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||||||||||||||
Table 13: Provider Taxes in Place in all 50 States and DC, FY 2018 and FY 2019
| States | Hospitals | Intermediate Care Facilities | Nursing Facilities | Other | ||||
| 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | 2018 | 2019 | |
| Alabama | X | X | X | X | X | X | ||
| Alaska | ||||||||
| Arizona | X | X | X | X | ||||
| Arkansas | X | X | X | X | X | X | ||
| California | X | X | X | X | X | X | X | X* |
| Colorado | X | X | X | X | X | X | ||
| Connecticut | X | X | X | X | X | X | X | X |
| Delaware | X | X | ||||||
| DC | X | X | X | X | X | X | X | X |
| Florida | X | X | X | X | X | X | ||
| Georgia | X | X | X | X | ||||
| Hawaii | X | X | X | X | ||||
| Idaho | X | X | X | X | X | X | ||
| Illinois | X | X | X | X | X | X | ||
| Indiana | X | X | X | X | X | X | ||
| Iowa | X | X | X | X | X | X | ||
| Kansas | X | X | X | X | ||||
| Kentucky | X | X | X | X | X | X | X* | X* |
| Louisiana | X | X | X | X | X | X | X* | X* |
| Maine | X | X | X | X | X | X | X | X |
| Maryland | X | X | X | X | X | X | X | X |
| Massachusetts | X | X | X | X | X | X | ||
| Michigan | X | X | X | X | X | X | ||
| Minnesota | X | X | X | X | X | X | X | X |
| Mississippi | X | X | X | X | X | X | X | X |
| Missouri | X | X | X | X | X | X | X* | X* |
| Montana | X | X | X | X | X | X | X | X |
| Nebraska | X | X | X | X | ||||
| Nevada | X | X | ||||||
| New Hampshire | X | X | X | X | ||||
| New Jersey | X | X | X | X | X | X | X* | X* |
| New Mexico | X* | X* | ||||||
| New York | X | X | X | X | X | X | X* | X* |
| North Carolina | X | X | X | X | X | X | ||
| North Dakota | X | X | ||||||
| Ohio | X | X | X | X | X | X | X | X |
| Oklahoma | X | X | X | X | X | X | ||
| Oregon | X | X | X | X | X | X | ||
| Pennsylvania | X | X | X | X | X | X | X* | X* |
| Rhode Island | X | X | X | X | X | X | ||
| South Carolina | X | X | X | X | ||||
| South Dakota | X | X | ||||||
| Tennessee | X | X | X | X | X | X | X* | X* |
| Texas | X | X | X | X | ||||
| Utah | X | X | X | X | X | X | X | X |
| Vermont | X | X | X | X | X | X | X* | X* |
| Virginia | X | X | X | |||||
| Washington | X | X | X | X | X | X | ||
| West Virginia | X | X | X | X | X | X | X* | X* |
| Wisconsin | X | X | X | X | X | X | ||
| Wyoming | X | X | X | X | ||||
| Totals | 42 | 43 | 36 | 36 | 44 | 44 | 26 | 26 |
| NOTES: This table includes Medicaid provider taxes as reported by states. Some states also have premium or claims taxes that apply to managed care organizations and other insurers. Since this type of tax is not considered a provider tax by CMS, these taxes are not counted as provider taxes in this report. (*) has been used to denote states with multiple “other” provider taxes. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||
Report: Benefits And Copayments
Key Section Findings
A total of 19 states expanded or enhanced covered benefits in FY 2018 and 24 states plan to add or enhance benefits in FY 2019. The most common benefit enhancements reported were for mental health/substance use disorder (SUD) services (including waiver of the IMD exclusion for SUD treatment). A handful of states reported expansions related to dental services, telemonitoring/telehealth, physical or occupational therapies, and screening and home visiting services for pregnant women. Eight states reported new or increased copayments and nine states reported policies to eliminate or reduce a copay requirement for FY 2018 or FY 2019.
What to watch:
- Medicaid continues to play an important role in addressing the opioid epidemic and more broadly in connecting Medicaid beneficiaries to behavioral health services. The SUPPORT Act, expected to be signed into law as this report was being finalized, creates a state option to cover IMD services for up to 30 days in a year for non-elderly adults with an SUD and codifies the 2016 Medicaid Managed Care Final Rule provision allowing “in lieu of” IMD coverage for up to 15 days in a month. Going forward, it will be important to follow the impact of this legislation as well as trends and innovations in how states use Medicaid to increase access to behavioral health services.
Tables 14 and 15 provide a complete listing of Medicaid benefit changes for FY 2018 and FY 2019. Table 16 provides a list of states that reported copayment actions for FY 2018 and FY 2019.
Benefit Changes
The number of states reporting new benefits and benefit enhancements continues to significantly outpace the number of states reporting benefit cuts and restrictions. Nineteen states reported new or enhanced benefits in FY 2018, and 24 states are adding or enhancing benefits in FY 2019. Few states reported benefit cuts or restrictions – four in FY 2018 and six in FY 2019 (Figure 10 and Table 14).
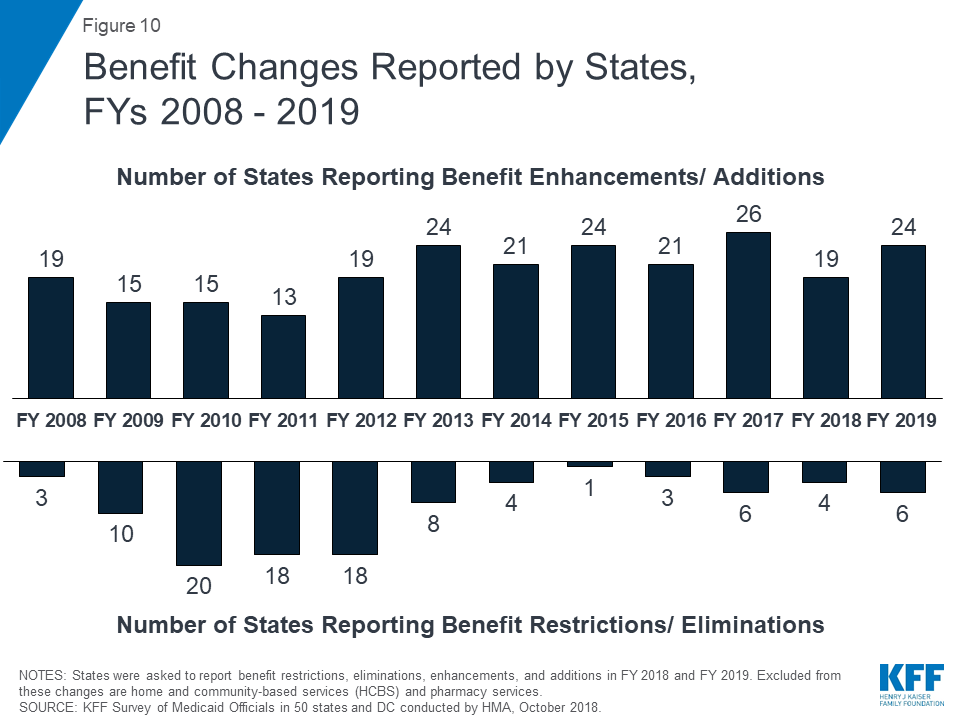
The most common benefit enhancements reported were for mental health and substance use disorder (SUD) services. Exhibit 17 also highlights states implementing other select benefit enhancements for dental, telemonitoring/telehealth, physical and occupational therapies, screening and home visiting services for pregnant women and children, and alternative therapies.
| Exhibit 17: Select Categories of Benefit Enhancements or Additions | ||||
| Benefit | FY 2018 | FY 2019 | ||
| Mental Health/Substance Use Disorder Services | 9 States | IN, MD, MA, NE, OH, RI, UT, VA, WV | 18 States | AK, DC, HI, IL, IN, KS, KY, MD, NC, NH, NJ, NM, RI, SD, TN, TX, WI, WV |
| Dental Services | 3 States | AZ, CA, UT | 2 States | IL, MD |
| Telemonitoring/ Telehealth Services | 4 States | IN, MD, NY, SC | 1 State | TX |
| Therapy Services (PT, OT) | 3 States | AZ, CO, WI | 1 State | NY |
| Screening and Home Visiting Services for Pregnant Women and/or Children | 3 States | CO, TX, VT | 2 States | IL, NM |
| Alternative Therapies (e.g., Chiropractic and Acupuncture) | 2 States | IN, OH | 1 State | MO |
Similar to our findings in last year’s budget survey, a number of states continue to report expanded mental health and/or SUD services. Many of these expansions are state initiatives to use Medicaid funds for services provided in institutions for mental disease (IMDs) under approved or pending Section 1115 waivers. These expansions include states responding to July 2015 CMS guidance84 stating that states can request federal funding for SUD services delivered to nonelderly adults in IMDs through Section 1115 demonstration waivers, as well as revised November 2017 guidance85 that continues to allow states to seek Section 1115 waivers to pay for SUD services provided in IMDs.
While IMD waivers approved under the previous administration were contingent on coverage of services across the care continuum, recently approved IMD waivers generally do not address coverage of community-based SUD services. Also, the SUPPORT Act,86 which was expected to be signed into law as this report was being finalized, would create a state plan option from October 1, 2019 to September 30, 2023 to cover IMD services for up to 30 days in a year for non-elderly adults with an SUD. The SUPPORT Act also would codify the 2016 Medicaid Managed Care Final Rule provision allowing “in lieu of” IMD coverage for up to 15 days in a month.
Other non-IMD mental health and SUD service expansions that states reported include expanding access to screening and intervention services and supporting recovery with new services such as peer supports. States also continue to increase access to naloxone and medication assisted treatment (MAT) services. See the “Opioid Harm Reduction Strategies” section of this report for details on these initiatives.
Other noteworthy benefit expansions include:
- Neonatal Abstinence Syndrome (NAS) Treatment Services: In FY 2018, West Virginia became the first state to receive CMS SPA approval to finance NAS services using a bundled payment for providers outside the hospital inpatient setting. The incidence of NAS is directly related to the nation’s opioid epidemic and involves infant withdrawal symptoms due to in utero exposure to certain substances.87 West Virginia’s NAS benefit package includes pharmacological and non-pharmacological interventions to holistically treat the withdrawal symptoms, which can include tremors, seizures, and vomiting. CMS highlighted West Virginia’s approach in a June Informational Bulletin88 as a model that pays an all-inclusive rate for neonatal abstinence treatment professional services and other ancillary services in a pediatric residential center specializing in NAS treatment. In addition, the SUPPORT Act89 would create a new State Plan option, effective upon enactment, to provide inpatient or outpatient residential pediatric recovery centers services for infants under age 1 with NAS and their families.
- Community Health Workers: Both Indiana and South Dakota reported plans to cover services provided by Community Health Workers (CHWs). Indiana began covering CHW services on July 1, 2018, adopting the American Public Health Association’s definition of a CHW. For services to be covered, the CHW must be certified by a recognized organization, employed by a Medicaid-enrolled provider, and have delivered services under their supervision. The CHW is part of the health care team and provides patient education, facilitates communication when cultural factors may be a barrier to care, promotes healthy behaviors, and provides direct preventive services or services intended to slow the progression of chronic disease.90
- Diabetes Prevention: Two states reported plans to cover Diabetes Prevention Program services in FY 2019. Diabetes Prevention Programs aim to delay or prevent the onset of type 2 diabetes with targeted health behavior interventions. California will start covering these services for beneficiaries diagnosed with prediabetes in FY 2019. New Jersey is also adding coverage of Diabetes Prevention Program services in FY 2019, as well as diabetes self-management education.
- Enhanced Screening, Identification, and Other Support Services for New Mothers: In FY 2018, Colorado and Texas started covering depression screenings for mothers in the first 12 months of a child’s life. In Texas, the screening is provided through the child’s benefit package as part of a well child visit. In FY 2019, New Mexico will pilot a home visiting program as part of its pending Section 1115 waiver that will focus on prenatal care, postpartum care, and early childhood development. Home visits will include patient education, skill building, screenings for risk factors (including depression and substance misuse), breast feeding support and education, and child developmental screenings. Illinois also plans to cover home visiting services for new mothers and/or at risk children in FY 2019, targeting women who give birth to babies born with withdrawal symptoms.
Most benefit restrictions in FY 2018 or FY 2019 are narrowly targeted. Benefit restrictions reflect the elimination of a covered benefit, benefit caps, or the application of utilization controls for existing benefits. The most common benefit restrictions limited dental coverage (Alaska, Connecticut, Iowa, Kentucky, Nevada, and Oklahoma) or implemented new prior authorization requirements (Colorado and Nevada). Other notable benefit restrictions that are pending CMS approval include proposals in New Mexico and Utah to eliminate Early and Periodic Screening, Diagnostic, and Treatment (EPSDT) coverage for individuals ages 19 and 20 in FY 2019 and Kentucky’s proposals to eliminate non-emergency medical transportation (NEMT) services for the Medicaid expansion group and eliminate NEMT for methadone services for all non-pregnant adults in FY 2019 (Table 15).
Copayments
Federal law limits cost-sharing for people with income below 100% FPL to “nominal” amounts (defined in federal regulations), with higher amounts allowed for beneficiaries at higher income levels. Certain groups are exempt from cost-sharing, including mandatory eligible children, pregnant women, most children and adults with disabilities, people residing in institutions, and people receiving hospice care. In addition, certain services are exempt from cost-sharing: emergency services, preventive services for children, pregnancy-related services, and family planning services. Also, total Medicaid premiums and cost-sharing for a family cannot exceed 5% of the family’s income on a quarterly or monthly basis.91
Most state Medicaid programs require beneficiary copayments, but to varying degrees. Thirteen states reported changes to copayment requirements in either FY 2018, FY 2019, or both years. Details about state actions related to copayments can be found in Table 16 and key changes are described below.
Eight states reported new or increased copayment requirements for FY 2018 or FY 2019. Key changes include:
- Five states (Colorado, Kentucky, Maine,92 Massachusetts, and New Mexico) reported new or increased copayments for non-emergency use of a hospital emergency department (ED). These changes are part of pending Section 1115 waiver requests in Kentucky, Maine, and New Mexico.
- Colorado, New Mexico, and Utah are adding or increasing pharmacy copayments. Colorado, Michigan, and Utah reported increased copayments for hospital outpatient services.
Nine states reported policies that eliminate or reduce a copayment requirement for some or all covered populations in FY 2018 or FY 2019. Key changes include:
- One state (Indiana)93 decreased copays in FY 2018 for non-emergency use of the ED to the state plan amount ($8). The state previously imposed graduated copays of up to $25 for non-emergency ED use under Section 1115 waiver authority.
- New Mexico is eliminating copayments for behavioral health services for working disabled adults and Michigan is eliminating behavioral health copays for all beneficiaries.
- Delaware is eliminating copayments for naloxone, Massachusetts is eliminating copayments for SUD treatment, aspirin, and statin drugs, and South Carolina is eliminating copayments for a subset of prescription drug classes deemed to be of the highest value.
TABLE 14: BENEFIT CHANGES IN ALL 50 STATES AND DC, FY 2018 AND FY 2019
| States | FY 2018 | FY 2019 | ||
| Enhancements/Additions | Restrictions/Eliminations | Enhancements/Additions | Restrictions/Eliminations | |
| Alabama | ||||
| Alaska | X | X | ||
| Arizona | X | |||
| Arkansas | ||||
| California | X | X | ||
| Colorado | X | X | X | |
| Connecticut | X | |||
| Delaware | ||||
| DC | X | |||
| Florida | ||||
| Georgia | X | |||
| Hawaii | X | |||
| Idaho | ||||
| Illinois | X | |||
| Indiana | X | X | ||
| Iowa | X | |||
| Kansas | X | |||
| Kentucky | X | X | ||
| Louisiana | X | |||
| Maine | ||||
| Maryland | X | X | ||
| Massachusetts | X | |||
| Michigan | ||||
| Minnesota | ||||
| Mississippi | X | |||
| Missouri | X | |||
| Montana | ||||
| Nebraska | X | |||
| Nevada | X | X | X | |
| New Hampshire | X | |||
| New Jersey | X | |||
| New Mexico | X | X | ||
| New York | X | X | ||
| North Carolina | X | |||
| North Dakota | ||||
| Ohio | X | |||
| Oklahoma | X | |||
| Oregon | ||||
| Pennsylvania | ||||
| Rhode Island | X | X | ||
| South Carolina | X | |||
| South Dakota | X | |||
| Tennessee | X | |||
| Texas | X | X | ||
| Utah | X | X | ||
| Vermont | X | |||
| Virginia | X | |||
| Washington | ||||
| West Virginia | X | X | ||
| Wisconsin | X | X | ||
| Wyoming | ||||
| Totals | 19 | 4 | 24 | 6 |
NOTES: States were asked to report benefit restrictions, eliminations, enhancements, and additions in FY 2018 and FY 2019. Home and community-based services (HCBS) and pharmacy benefit changes are excluded from this table. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||
Table 15: States Reporting Benefit Actions Taken in FY 2018 and FY 2019[endnote 375798-11]
State | Fiscal Year | Benefit Changes |
| Alabama | 2019 | Children (nc): Add coverage of Applied Behavior Analysis (ABA) services for individuals under age 21 (October 1, 2018). |
| Alaska | 2018 | Adults (-): Recategorize emergent and enhanced dental service codes, and eliminate coverage of other codes. |
| 2019 | Children (nc): Add coverage of Applied Behavior Analysis (ABA) services under the EPSDT benefit. All (+) Pending Sec. 1115 Waiver: Begin phasing in a revised and expanded behavioral health services benefit package (TBD). | |
| Arizona | 2018 | Non-LTSS Adults (+): Add a $1,000 per year benefit for emergency dental services (October 1, 2017). Non-LTSS Adults (+): Add coverage of outpatient occupational therapy services (October 1, 2017). |
| California | 2018 | All (nc): Reaffirm coverage of non-emergency medical transportation as provided in state law (July 1, 2017). Adults (+): Fully restore coverage for dental services (January 1, 2018). Children (nc): Expand coverage of Behavioral Health Treatment (BHT) to individuals under age 21 without a diagnosis of Autism Spectrum Disorder (ASD) (March 1, 2018). |
| 2019 | Adults (+): Add Diabetes Prevention Program (DPP) for individuals diagnosed with prediabetes who meet qualifying criteria (January 1, 2019). | |
| Colorado | 2018 | Pregnant Women (+): Add coverage of up to three postpartum depression screenings in the first year following a child’s birth (July 1, 2017). Children (+): Restore coverage of routine circumcisions as an elective benefit (July 1, 2017). Adults (+): Add coverage for physical therapy/occupational therapy services above the 12-hour cap with prior authorization (November 1, 2017). |
| 2019 | All (+): Add coverage of 12-month supply of birth control pills, after an initial three-month dispensing period (January 1, 2019). All (+): Expand non-emergency medical transportation (NEMT) services benefit to meet urgent transportation needs (January 1, 2019). All (-): Implement prior authorization requirements for certain medical benefits and physician administered drugs (January 1, 2019). All (-): Implement a comprehensive hospital admission review program (January 1, 2019). | |
| Connecticut | 2018 | Adults (-): Apply $1,000 annual cap on coverage for dental services, with exception for medical necessity (January 1, 2018). |
| District of Columbia | 2019 | All (+): Add coverage of Clubhouse peer support services for individuals with a mental health diagnosis living in the community (TBD). |
| Georgia | 2018 | Children (nc): Add coverage for Autism Spectrum Disorders (ASD) services for individuals under the age of 21 (January 1, 2018). |
| 2019 | All (+): Add coverage of Emergency Medical Services (EMS) transportation to a non-hospital destination and treatment without transport (July 1, 2018). Pregnant Women (+): Add coverage for group prenatal care services/CenteringPregnancy (October 1, 2018). | |
| Hawaii | 2019 | Adults (+) Pending Sec. 1115 Waiver: Expand mental health and substance abuse benefits including addition of intensive case management and tenancy supports for beneficiaries classified as chronically homeless (TBD). |
| Illinois | 2019 | Adults (+): Restore adult dental benefit (July 1, 2018). All (+) Approved Sec. 1115 Waiver: Add coverage of residential and inpatient treatment for individuals with substance use disorder at institutions for mental disease (IMD) under a statewide pilot (January 1, 2019). All (+) Approved Sec. 1115 Waiver: Expand coverage of the following behavioral health services through pilot programs approved under a Section 1115 waiver: clinically managed residential withdrawal management; substance use disorder case management services; peer recovery support services; crisis intervention services; and supported employment services (January 1, 2019). Pregnant women and children (+) Approved Sec. 1115 Waiver: Cover evidence-based home visiting services under a pilot program, including postpartum home visits and child home visits to postpartum mothers who gave birth to a baby born with withdrawal symptoms (January 1, 2019). Children (+) Approved Sec. 1115 Waiver: Add coverage of intensive in-home clinical and support services under a pilot program to support and stabilize a child/youth in their home or home-like setting (January 1, 2019). Children (+) Approved Sec. 1115 Waiver: Add coverage of respite services under a pilot program to provide families scheduled relief to help prevent stressful situations (January 1, 2019). |
| Indiana | 2018 | Adults (+): Add coverage of chiropractic spinal manipulation for Healthy Indiana Plan (HIP) Plus enrollees (January 1, 2018). Adults (+) Approved Sec. 1115 Waiver: Expand coverage of opioid use disorder and substance use disorder treatment services to include inpatient substance use treatment at institutions for mental disease (IMD) (February 1, 2018). All (+) Approved Sec. 1115 Waiver: Add coverage for short-term low-intensity and high-intensity residential treatment for opioid use disorder and substance use disorder in settings of all sizes, including IMDs (March 1, 2018). All (+): Revise coverage for telemedicine services, including an elimination of the distance requirement between distant and originating sites (April 1, 2018). |
| 2019 | All (+): Add coverage for certain services provided by community health workers, including but not limited to patient education, health promotion, and facilitation of cultural brokering between an individual and their health care team (July 1, 2018). All (+): Expand coverage for peer support recovery, crisis intervention, and intensive outpatient behavioral health services (TBD). | |
| Iowa | 2019 | Adults (-): Apply a $1,000 annual maximum to dental benefits, excluding preventive services and dentures (September 1, 2018). |
| Kansas | 2019 | Adults (+) Pending Sec. 1115 Waiver: Add coverage of inpatient behavioral health services at publicly-owned and non-public institutions for mental disease (IMD) for Medicaid managed care enrollees (January 1, 2019). |
| Kentucky | 2018 | All (nc): Expand non-emergency medical transportation services to include travel to pharmacies (July 1, 2017). |
| 2019 | Adults (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Change access to enhanced benefits, such as vision and dental, such that individuals must access through My Rewards Account (TBD). All (+): Approved Sec. 1115 Waiver: Add coverage of substance use disorder treatment services at institutions for mental disease (IMD) under pilot program (TBD). Expansion Adults (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Waive coverage of NEMT services (TBD). Adults (-) Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering: Waive coverage of NEMT services for methadone treatment, with exceptions for children under age 21 (EPSDT), former foster care youth, and pregnant women (TBD). | |
| Louisiana | 2018 | All (+): Remove home health visit limits (January 20, 2018). Family Planning Eligibility Group (+): Remove family planning services visit limit (March 20, 2018). |
| Maryland | 2018 | Adults (+) Approved Sec. 1115 Waiver: Add coverage of substance use disorder residential treatment services in ASAM Level 3.3, 3.5, and 3.7 settings (July 1, 2017). All (+): Add coverage of remote patient monitoring for beneficiaries who meet qualifying medical criteria (January 1, 2018). |
| 2019 | Adults (nc): Add coverage of audiology services and hearing aids (July 1, 2018). Adults (+) Approved Sec. 1115 Waiver: Add coverage of substance use disorder residential treatment services in ASAM Level 3.1 settings (January 1, 2019). Dual Eligibles (+): Implement pilot for coverage of adult dental services (January 1, 2019). | |
| Massachusetts | 2018 | All (+) Approved Sec. 1115 Waiver: Add coverage of recovery support navigator services and recovery coach services (March 1, 2018). |
| Michigan | 2018 | Adults (nc): Add coverage of hearing aids for adults age 21 and older (September 1, 2018). |
| Mississippi | 2019 | Children (+): Add coverage of pharmacist-administered vaccines for children ages 10 to 18 years old (TBD). |
| Missouri | 2019 | All (+): Add coverage of acupuncture and chiropractic services in lieu of an opioid prescription as an alternative pain management strategy (February 2019). |
| Nebraska | 2018 | All (+): Add coverage of nutrition services (July 1, 2017). All (+): Add coverage of peer support services (July 1, 2017). |
| Nevada | 2018 | All (-): Implement a prior authorization requirement for hospice services (July 1, 2017). Children (-): Limit coverage of orthodontia services (July 1, 2017). All (+): Add coverage of gender dysphoria services (January 1, 2018). All (+): Add coverage of medical nutrition therapy services (January 1, 2018). All (+): Add coverage of 12-month supply of birth control pills (January 1, 2018). Adults (+): Add coverage of podiatry services (January 1, 2018). All (nc): Expand definition of covered durable medical equipment services (June 26, 2018). |
| 2019 | All (-): Establish basic skills training services tiers (TBD). | |
| New Hampshire | 2019 | Adults (+) Approved Sec. 1115 Waiver: Add coverage of substance use disorder treatment services at institutions for mental disease (IMD) (TBD). Expansion Adults (nc) Pending Sec. 1115 Waiver: Align alternative benefit package with standard Medicaid benefits as state terminates the current Premium Assistance Program and transitions Medicaid expansion beneficiaries into the state’s Medicaid managed care delivery system (January 1, 2019). |
| New Jersey | 2019 | All (+): Eliminate prior authorization requirements for nicotine replacement therapies (July 1, 2018). Adults (+) Approved Sec. 1115 Waiver: Add coverage of short-term residential services for substance use disorder and withdrawal management services at an institution for mental disease (IMD) (July 1, 2018). Adults (+) Approved Sec. 1115 Waiver: Add coverage of long-term residential services for substance use disorder at an institution for mental disease (IMD) (October 1, 2018). Children (nc): Expand coverage of Autism Spectrum Disorder (ASD) services statewide (January 1, 2019). All (+): Add coverage of Diabetes Prevention Program services and self-management education (TBD). |
| New Mexico | 2019 | All (+) Pending Sec. 1115 Waiver: Add coverage of behavioral health services, substance use disorder treatment in adult residential treatment centers, expanded Opioid Treatment Program services, expanded provider types for Comprehensive Community Support Services, and teaming crisis treatment services (January 1, 2019). All (+): Add coverage of Screening, Brief Intervention, and Referral to Treatment (SBIRT) services. All (+) Pending Sec. 1115 Waiver: Add coverage of inpatient services in an institution for mental disease (IMD) for beneficiaries with severe mental illness or a substance use disorder (January 1, 2019). Children (-) Pending Sec. 1115 Waiver: Waive coverage of Early and Periodic Screening, Diagnosis, and Treatment (EPSDT) services for 19- and 20-year-olds (January 1, 2019). Pregnant Women (+) Pending Sec. 1115 Waiver: Pilot home visiting program that focuses on prenatal care, postpartum care, and early childhood development (January 1, 2019). Adults (+) Pending Sec. 1115 Waiver: Add coverage of pre-tenancy and tenancy support services to adults who are Seriously Mentally Ill (January 1, 2019). |
| New York | 2018 | All (+): Add coverage of digital breast tomosynthesis (DBT) screening services (September 1, 2017 for fee-for-service and November 1, 2017 for Medicaid managed care). All (+): Add coverage of continuous glucose monitoring devices for beneficiaries with Type 1 diabetes (November 1, 2017 for fee-for-service and January 1, 2018 for Medicaid managed care). Children (+): Add coverage of pasteurized donor human breast milk for infants <1500 grams (December 1, 2017 for fee-for-service and February 15, 2018 for Medicaid managed care). |
| 2019 | All (+): Expand coverage of physical therapy services by increasing cap from 20 visits to 40 visits for eligible beneficiaries (July 1, 2018). All (+): Add limited infertility benefit for beneficiaries between the ages of 21 to 44 (TBD, pending CMS approval). | |
| North Carolina | 2019 | Adults (+): Add coverage of adult vision services (January 1, 2019). Adults (+) Pending Sec. 1115 Waiver: Add coverage of substance use disorder services in institutions for mental disease (IMD) (TBD). |
| North Dakota | 2018 | All (nc): Update and clarify the services covered under the Rehabilitation Services benefit category, and the provider types who may render the service (April 1, 2018). |
| Ohio | 2018 | All (+): Expand coverage of behavioral health services to include assertive community treatment for adults, family counseling, intensive home-based treatment for youth at risk of out-of-home placement, and primary care services delivered by a behavioral health provider (January 1, 2018). All (+): Expand provider types who may provide acupuncture services to treat pain (January 1, 2018). |
| Oklahoma | 2018
| Adults (-): Limit cap on Targeted Case Management services from 25 units per month to 16 units per year (September 1, 2017). Adults (-): Limit coverage of tooth extractions to emergency services only (September 14, 2017). Adults (-): Eliminate coverage of non-mandatory over-the-counter drugs (October 1, 2017). |
| Rhode Island | 2018 | Adults (+) Approved Sec. 1115 Waiver: Add coverage of Recovery Navigation Program services for individuals with substance use disorder. |
| 2019 | Adults (+) Approved Sec. 1115 Waiver: Add coverage of peer recovery specialist services for individuals with substance use disorder. Adults (+) Pending Sec. 1115 Waiver: Add coverage of residential mental health and substance use disorder treatment services in institutions for mental disease (IMD) (TBD). All (+) Pending Sec. 1115 Waiver: Add coverage for a Behavioral Health Link triage center to support crisis stabilization and short-term treatment for individuals experiencing a mental health or substance use disorder crisis (TBD). | |
| South Carolina | 2018 | All (+): Expand coverage of telemedicine services (July 1, 2017). Children (nc): Add Autism Spectrum Disorder (ASD) services to the State Plan for eligible beneficiaries up to age 21 (July 1, 2017). |
| South Dakota | 2019 | Adults (+): Expand coverage of substance use disorder treatment services to all adults (SUD services were previously only covered for pregnant women) (July 1, 2018). All (+): Expand definition of mental health practitioners (January 1, 2019). All (+): Add coverage of services provided by community health workers (April 1, 2019). |
| Tennessee | 2019 | Adults (+) Pending Sec. 1115 Waiver: Add coverage of inpatient and residential substance use disorder treatment services in institutions for mental disease (IMD) (TBD). |
| Texas | 2018 | Children (+): Add coverage for one postpartum depression screening for mother during infant’s Texas Health Steps medical visit during the first year (July 1, 2018). |
| 2019 | Children (+): Expand coverage of telemedicine services to occupational therapy and speech-language pathology provided in a school-based setting (TBD). Adults (+): Add coverage of peer specialist services for adults with mental health conditions or substance use disorders (TBD). | |
| Utah | 2018 | All (+): Add coverage of Screening, Brief Intervention, and Referral to Treatment (SBIRT) services for beneficiaries age 13 and older (July 1, 2017). People with Disabilities (+): Add coverage of dental services for individuals with disabilities (July 1, 2017). Adults (+) Approved Sec. 1115 Waiver: Add coverage of residential treatment services in an institution of mental disease (IMD) for individuals with a substance use disorder (November 9, 2017). |
| 2019 | Children (-) Pending Sec. 1115 Waiver: Waive coverage of Early and Periodic Screening, Diagnosis, and Treatment (EPSDT) services for certain 19 and 20 year olds (TBD). | |
| Vermont | 2018 | All (+): Add coverage of in-home lactation consultations (June 1, 2018). |
| Virginia | 2018
| All (+): Add coverage for peer support services for beneficiaries with serious mental illness and/or substance use disorders (for all members other than family planning-only beneficiaries) (July 1, 2017). Limited Adult Coverage Group (+) Approved Sec. 1115 Waiver: Expand coverage of residential addiction recovery and treatment services for beneficiaries with serious mental illnesses and/or substance use disorders in the GAP waiver population (October 1, 2017). |
| West Virginia | 2018 | Children (+): Add coverage of neonatal abstinence syndrome (NAS) treatment services (October 31, 2017). All (+) Approved Sec. 1115 Waiver: Add coverage of screening, brief intervention, and referral to treatment (SBIRT) services (January 14, 2018). |
| 2019 | All (+) Approved Sec. 1115 Waiver: Add coverage of substance use disorder services, including adult residential treatment services, peer recovery support services, and withdrawal management services (July 1, 2018). | |
| Wisconsin | 2018 | Children (+): Redesign targeted case management services for children with complex medical conditions (September 1, 2017). All (+): Eliminate prior authorization requirements for therapy services evaluations and re-evaluations (January 1, 2018). Children (+): Add coverage of pharmacist-administered vaccines for children ages six to 18 years old (January 1, 2018). Children (+): Eliminate prior authorization requirements for environmental lead investigation services (March 1, 2018). |
| 2019 | Adults (+) Pending Sec. 1115 Waiver: Add coverage of residential substance use disorder treatment services in institutions for mental disease (IMD) for managed care and fee-for-service populations (TBD). |
table 16: Copayment actions taken in the 50 states and dc, FY 2017 and 2018
State | Fiscal Year | Copayment Changes |
| Colorado | 2018 | Increase (all non-exempt eligibility groups): Increase pharmacy copayment to $3.00 per prescription (January 1, 2018). Increase (all non-exempt eligibility groups): Double the hospital outpatient copayments (January 1, 2018). Increase (all non-exempt eligibility groups): Increase emergency room copayment for non-emergency events (January 1, 2018). |
| Delaware | 2018 | Decrease (LTSS population): Treatment of pre-eligibility medical expenses in determining post eligibility cost of care contribution for LTSS population; “look-back” period expanded from 30 days to 90 days. Potential to reduce the monthly “patient pay” amount (July 1, 2017). |
| 2019 | Elimination (all eligibility groups): Eliminate copayment for naloxone (July 1, 2018). | |
| Indiana | 2018 | Decrease (for HIP 2.0 expansion population): Eliminating the graduated copayment for non-emergent ER use and replaced with a flat $8 copay (February 1, 2018) (Approved Sec. 1115 Waiver). |
| Kentucky | 2019 | Increase (Expansion Adults and Parents/Caretakers): MCOs will not be authorized to waive copayments that apply in fee-for-service (TBD). New (Expansion Adults and Parents/Caretakers): Incentive account funds deducted for non-emergent use of the ER (Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering). New (Expansion Adults and Parents/Caretakers): Incentive account funds deducted for missed appointments (Sec. 1115 Waiver Approval Set Aside by Court, CMS Reconsidering). |
| Maine | 2019 | New (all non-exempt groups): Impose a copayment of $10 on all populations for non-emergent use of the ED (dual eligibles, those in institutions and a few other groups are exempt) (Pending Sec. 1115 Waiver). |
| Massachusetts | 2019 | Elimination (all groups): Eliminate pharmacy copays for SUD treatment, aspirin, and statin drugs (TBD). Elimination (Persons with income at or below 50% FPL): Eliminate all copayments for those with income below 50% FPL (TBD). Increase (adults above 50% FPL); Implement new copayments for specialty services and non-emergent use of the emergency room (TBD). Decrease (all groups): Implementing a 2% cost-sharing cap for those below 150% FPL and 5% for those above 150% FPL (TBD). |
| Michigan | 2019 | Increase (all non-exempt groups): Increase outpatient hospital copay from $1 to $2 (October 1, 2018). Elimination (all groups): Eliminate cost sharing for mental health and substance use disorder services (October 1, 2018). |
| Montana | 2018 | Increase (expansion population with incomes from 51% to 138% FPL): Premium credit of 2% eliminated and members now pay point of service copayments (January 1, 2018) (Approved Sec. 1115 Waiver). |
| New Hampshire | 2019 | Decrease (adult enrollees with incomes between 100% and 133% FPL): Cost sharing will be aligned with standard Medicaid (January 1, 2019) (Approved Sec. 1115 Waiver). |
| New Jersey | 2018 | Decrease (nursing facility residents): Personal needs allowance was increased from $35 to $50 per month (July 1, 2017). |
| New Mexico | 2018 | Elimination (for Working Disabled Adults): Eliminated copayments for behavioral health services (January 1, 2018). |
| 2019 | New (for most populations): Copays for brand-name prescriptions when there is a less expensive generic equivalent medicine available (March 1, 2019). New (for most populations): Copays for non-emergent use of the emergency department (March 1, 2019) (Pending Sec. 1115 Waiver). New (for most populations): A fee of $5 for missing 3 or more appointments in a calendar year (March 1, 2019) (Pending Sec. 1115 Waiver). Elimination (for Working Disabled Adults): Eliminate most copayments (January 1, 2019). | |
| South Carolina | 2019 | Elimination (all populations): Copayments eliminated for a subset of prescription drug classes deemed to be of the highest value. |
| Utah | 2018 | Neutral (all but children and pregnant women): Change inpatient copayments from $220 per year to $75 per stay (October 1, 2017). Increase (all but children and pregnant women): Increase outpatient copayments (July 1, 2017). Increase (all but children and pregnant women): Increase pharmacy copayment from $2 to $4 per prescription (July 1, 2017). |
Report: Pharmacy And Opioid Strategies
Key Section Findings
Most states identified specialty and high-cost drugs (individually or in general) as the most significant cost driver of Medicaid pharmacy spending. Twenty-two states in FY 2018 and 19 states in FY 2019 reported new or enhanced pharmacy cost containment initiatives, especially initiatives to generate greater rebate revenue and implementation of new utilization controls (e.g., prior authorization requirements, step therapy, other clinical criteria, or dose optimization). Thirty-five of 39 MCO states reported that the pharmacy benefit was “generally carved-in,” unchanged from 2017. Of these 35 states, the majority reported requirements that MCOs have uniform clinical protocols (31 states) or uniform preferred drug lists (PDLs) (17 states) in place for one or more drugs as of the end of FY 2019.
In FY 2018, all states report FFS pharmacy management strategies targeted at opioid harm reduction including quantity limits (50 states); clinical criteria claim system edits (48 states); step therapy (39 states), and other prior authorization requirements (44 states). Somewhat fewer states (32) reported requirements in place for Medicaid prescribers to check their states’ Prescription Drug Monitoring Program (PDMP) before prescribing opioids to a Medicaid patient. Of the 35 states that used MCOs to deliver pharmacy benefits, 26 reported that they required MCOs to follow some or all of their FFS pharmacy management policies for opioids.
What to watch:
- For FY 2019, several states noted the emerging cost challenge posed by gene therapies and CAR-T cell therapies, which are designated by CMS as covered outpatient drugs.
- States continue to increase access to Medication Assisted Treatment (MAT) for opioid use disorder, and 38 states reported coverage of methadone in FY 2018. Many states reported experiencing challenges related to access to MAT, especially in rural areas. Looking ahead, the SUPPORT Act requires state Medicaid programs to cover all FDA-approved MAT drugs from October 2020 through September 2025.
Table 17 provides additional details on Medicaid FFS pharmacy benefit management strategies for opioids.
Prescription Drug Utilization and Cost Control Initiatives
Under federal law, once a manufacturer enters into a rebate agreement with the Secretary of HHS, states must generally cover (with limited exceptions) all drugs produced by that manufacturer. Because approximately 600 manufacturers currently have rebate agreements,94 states cover nearly all FDA-approved drugs. As pharmacy expenditure growth became a greater Medicaid budget concern in the late 1990’s and early 2000’s, most states implemented aggressive pharmacy cost containment strategies, including preferred drug lists (PDLs), supplemental rebate programs, state maximum allowable cost programs, and prior authorization policies linked to clinical criteria. While these programs have matured, they are also subject to ongoing updates and refinements as states respond to changes, especially new product offerings, in the pharmaceutical marketplace. In recent years, however, a disproportionate increase in prescription drug costs relative to overall spending has heightened state attention on pharmacy reimbursement and coverage policies. In this year’s survey, states reported a variety of actions in FY 2018 and FY 2019 to refine and enhance their pharmacy programs, including actions to react to new and emerging specialty and high-cost drug therapies.
PHARMACY COST DRIVERS
Specialty and high-cost drugs remain the biggest cost driver of pharmacy spending growth in most states. This year’s survey asked states to identify the biggest cost drivers that affected growth in total pharmacy spending95 (federal and state) in FY 2018 and projected for FY 2019. Consistent with the results of prior surveys in recent years, most states again identified specialty and high-cost drugs (individually or in general) as the most significant pharmacy cost driver. While several states noted that the cost of hepatitis C antivirals had recently come down due to the market entry of a competitor drug, a significant number of states specifically identified this drug class as a major cost driver. Other drug classes frequently cited as major cost drivers include hemophilia factor and oncology, mental health, and HIV/AIDS drugs. For FY 2019, several states also noted the emerging cost challenge posed by gene therapies and immunotherapies like “CAR-T” (Chimeric Antigen Receptor-T cell) therapies,96 designated by CMS as covered outpatient drugs. For example, the first gene therapy approved by the Food and Drug Administration in 2017 to treat congenital blindness was priced at $850,000 ($425,000 per eye).97 One state noted that gene therapies and CAR-T cell therapies currently under development could have a “huge” impact on states in coming years.
States also reported facing a variety of challenges in controlling pharmacy costs. A majority of states noted the burden of covering high-cost drugs (including gene therapies and CAR-T cell therapies) or increasing drug prices generally. Other challenges cited by several states include:
- The federal requirement to cover all rebateable drugs – despite the cost and even when evidence of clinical effectiveness or safety is poor
- State law limitations, including those that shield certain drugs or drug classes from utilization management efforts
Other challenges mentioned include limited administrative resources to provide clinical oversight to implement evidence-based coverage policies; the difficulty of appropriately accounting for the cost of new and emerging drugs within actuarially sound capitation rates; claims system constraints that make it difficult to add clinical utilization edits; and a lack of transparency related to PBM pricing policies and rebate collections.
Pharmacy Cost Containment Actions in FY 2018 and FY 2019
Almost all states had prescription drug cost containment policies (including prior authorization requirements and PDLs) in place prior to FY 2018, and most are constantly refining and updating these policies. While states were not asked to report every refinement or routine change in this year’s survey, 22 states in FY 2018 and 19 states in FY 2019 reported newly implementing or making changes to a wide variety of cost containment initiatives in the area of prescription drugs. The most frequently cited action was new or expanded initiatives to generate greater rebate revenue (including PDL expansions) (eight states in both FY 2018 and FY 2019). These initiatives include the following notable actions in three states:
- New York implemented a new state law in FY 2018 that applies a cap on Medicaid drug expenditures as a separate component of the global state Medicaid spending cap that the state has had in place since 2011. If the state determines that drug spending will exceed the annual growth limit, the Commissioner of the Department of Health may identify and refer drugs to the Drug Utilization Review (DUR) Board for additional review and recommendations regarding appropriated supplemental rebates. At the time of the survey, the DUR Board had taken action to recommend a supplemental rebate target amount for one drug,98 and negotiations between the state and the drug’s manufacturer were ongoing.
- Oklahoma became the first state in the nation to receive CMS approval to pursue value-based supplemental rebate agreements with pharmaceutical manufacturers. The first contract took effect in August 2018 and relates to adherence and persistency for an injectable atypical anti-psychotic.
- West Virginia reported that its July 1, 2017 MCO pharmacy carve-out (applicable to point-of-sale pharmacy benefits but not drugs covered as a medical benefit in an inpatient or outpatient setting), resulted in lower administration costs and increased federal and supplemental rebate collections. West Virginia also reported plans to expand supplemental rebates to certain additional diabetic supplies in FY 2019.
Seven states in both FY 2018 and FY 2019 also reported the application of new or expanded utilization controls (e.g., prior authorization requirements, step therapy, other clinical criteria, or dose optimization). Other frequently cited newly implemented or expanded pharmacy cost containment actions were:
- Ingredient cost reductions: six states in FY 2018 and one state in FY 2019 reported reductions in certain ingredient cost reimbursements, and one state ended reimbursement for non-rebatable products and implemented system changes to recognize 340B pricing. Conversely, in FY 2019, Arizona reported plans to negotiate with CMS to obtain better reimbursement for high-cost specialty drugs for Medicaid enrollees utilizing Indian Health Service (IHS) facilities.
- Medication therapy management, case management, or adherence programs: these programs were implemented or expanded by five states in FY 2018 and four in FY 2019.
Managed Care’s Role in Delivering Pharmacy Benefits
Since the passage of the ACA, states have been able to collect rebates on prescriptions purchased by managed care organizations (MCOs) operating under capitated arrangements. As a result, many states have chosen to “carve-in” the pharmacy benefit to their managed care benefits. As more states have enrolled additional Medicaid populations into managed care arrangements over time, MCOs have played an increasingly significant role in administering the Medicaid pharmacy benefit. In this year’s survey, states with MCO contracts were asked whether pharmacy benefits were covered under those contracts as of July 1, 2018. Of the 39 states contracting with comprehensive risk-based MCOs, 35 states reported that the pharmacy benefit was “generally carved-in (with possible exceptions),” unchanged from FY 2017.
Among the states that carved drugs into MCOs, several reported carve-outs for selected drug classes. The most commonly reported carved-out drugs were hemophilia clotting factor reported by at least eight states, hepatitis C antivirals reported by at least six states, mental health drugs reported by at least five states, HIV drugs reported by at least three states, and selected substance use disorder (SUD) treatment drugs reported by at least three states. At least nine states noted carve-outs for other selected high-cost drugs, and some of these states cited the challenge of accurately developing capitation rates when new high-cost drugs enter the market with no available historical utilization data. Washington reported that a state workgroup was currently working to define “high-cost drugs” and establish consistent policies (that would begin in FY 2020) for drug carve-ins and carve-outs that would also include policies for pass-through payments for drugs that are carved-out.
Consistent with last year’s survey results, four states (Missouri, Tennessee, West Virginia, and Wisconsin) reported that the pharmacy benefit was “generally carved-out.” While Wisconsin noted that pharmacy was carved into its Family Care Partnership program (an integrated health and long-term care program for frail elderly and people with disabilities), the state noted that this program had a very small enrollment (approximately 3,300 as of June 201899 ) and that all other Wisconsin Medicaid enrollees received their pharmacy benefit through the FFS delivery system.
States with MCOs are moving to align MCO pharmacy policies with FFS policies. Prior reports show that nearly all states use prior authorization and PDLs in FFS programs. This year’s survey asked whether MCOs were required (in FY 2018) or would be required (in FY 2019) to adhere to uniform clinical protocols (state prescribed medical necessity criteria) for one or more drugs or a uniform PDL (state prescribed requirements for designating a specified drug product as either preferred, meaning covered without the need to obtain prior authorization, or non-preferred). This means that to the extent states impose these policies in FFS, the same policies would apply in managed care. The survey also asked whether MCO contracts included risk-sharing provisions for one or more drugs (e.g., risk corridors, risk pools, reinsurance, etc.) (Exhibit 18).
| Exhibit 18: Managed Care Pharmacy Policies | |||||||
| Policy | In Place in FY 2018 | FY 2019 Changes | |||||
| New | Expanded | ||||||
| Uniform Clinical Protocols(1 or more drugs) | 30 States | AZ, CA, DC, DE, GA, HI, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MS, ND, NE, NJ, NM, NY, OH, OR, PA, RI, SC, TX, VA, WA | 1 States | UT | 6 States | KY, LA, OH, SC, VA, WA | |
| Uniform PDL(1 or more drug classes) | 14 States | AZ, DE, FL, IA, KS, MA, MN, MS, ND, NE, OR, TX, VA, WA | 3 States | LA, OH, UT | 2 States | VA, WA | |
| Risk-sharing(for 1 or more drugs) | 14 States | AZ, DE, HI, IN, KS, MA, NM, NV, OH, OR, PA, RI, VA, WV | 2 States | FL, UT | 2* States | IN, VA | |
| *Delaware reported plans to remove hepatitis C antivirals from its risk pool in CY 2019. | |||||||
Uniform clinical protocols and PDL requirements for MCOs reported by states were often limited to one or a few specific drug classes. Hepatitis C antivirals were the most commonly mentioned drug class targeted by uniform clinical protocols (reported by California, DC, Georgia, Hawaii, Illinois, Kentucky, Maryland, Minnesota, New Mexico, Oregon, Rhode Island, and Virginia) and were also reported as a specific focus of uniform PDL requirements in Massachusetts, Minnesota and Oregon. Four states also reported uniform protocols in place or planned for opioids and/or Medication Assisted Treatment drugs (Kentucky, New York, South Carolina and Virginia). Of the five states that reported plans to implement or expand a uniform PDL in FY 2019, one state (Washington) specifically noted plans to phase-in a comprehensive uniform PDL while two states (Louisiana and Virginia) reported that their uniform PDLs would be limited to certain therapeutic classes.
Strategies reported by states to mitigate or share financial risk with MCOs for certain high cost drugs included selected drug carve-outs (mentioned above), “kick payments,”100 risk corridors, and risk pools, and were most commonly applied to hepatitis C antivirals and hemophilia clotting factor, but in some cases were applied to other high cost drugs (Indiana and Kansas), drugs above a certain dollar threshold (Hawaii), and cystic fibrosis drugs (Pennsylvania). Florida, for example, reported plans to add a new prescribed high-risk drug pool to its CY 2019 MCO contracts. While the pool parameters were still under negotiation at the time of the survey, the state indicated that it is planning to withhold a portion of the capitation payment to fund the pool, which will be paid out to the MCOs at a later date based on utilization. Two states also reported interest in value-based purchasing approaches for drugs: Virginia reported plans to explore value-based purchasing agreements (and other risk-sharing opportunities for high-cost drugs), and West Virginia reported that payments for certain physician-administered cancer treatments are made only if the patient is in remission 30 days after treatment.
Opioid Harm Reduction Strategies
According to the U.S. Department of Health and Human Services, 2.1 million people in the United States have an opioid use disorder and 11.5 million people misuse prescription opioids as of 2016.101 The Centers for Disease Control and Prevention (CDC) indicates the number of drug overdose deaths continues to increase, and the majority (around 66%) involve an opioid (prescription opioids, synthetic opioids, and heroin).102 The number of opioid overdose deaths is five times higher than it was in 1999, with 115 people dying from opioid-related drug overdoses each day.103 The opioid epidemic was declared a nationwide public health emergency on October 26, 2017.104
Medicaid plays an important role in addressing the epidemic, covering 4 in 10 people with opioid use disorder in 2016 and providing access to a range of treatment services.105 These expansions include states responding to July 2015 CMS guidance106 stating that states can request federal funding for substance use disorder services delivered to nonelderly adults in Institutions for Mental Disease (IMDs) through Section 1115 demonstration waivers, as well as revised November 2017 guidance107 that continues to allow states to seek Section 1115 waivers to pay for SUD services provided in IMDs. Both state Medicaid director letters set out parameters for states to obtain Section 1115 waivers to test using federal Medicaid funds to provide short-term inpatient and residential SUD treatment services in IMDs.108
As this report was being finalized, the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities (SUPPORT) Act was expected to be signed into law. While very broad in scope, the SUPPORT Act109 contains a number of provisions related to Medicaid’s role110 in helping states provide coverage and services to people who need SUD treatment, particularly those needing opioid use disorder (OUD) treatment. These provisions include new authority to cover services in IMDs for up to 30 days in a year for non-elderly adults with a SUD from October 2019 through September 2023, a requirement that state Medicaid programs cover all FDA-approved MAT drugs as well as counseling and behavioral therapy services from October 2020 through September 2025, authority for new demonstrations to help states increase Medicaid SUD provider capacity, and a prohibition on states terminating Medicaid eligibility for individuals under age 21 or former foster care youth up to age 26 while incarcerated.
Medicaid Pharmacy Benefit Management Strategies
This year’s survey asked states to report Medicaid pharmacy benefit management strategies for preventing opioid-related harms that were in place in FY 2018 for FFS and changes to these strategies planned for FY 2019. Specifically, the survey asked about the following strategies: opioid quantity limits,111 clinical criteria claim system edits112 (subject to prior authorization (PA) override), step therapy PA criteria,113 other PA requirements for opioids, and requirements that prescribers check the state’s Prescription Drug Monitoring Program (PDMP) before prescribing opioids.114 All states and DC reported having at least one of these opioid-focused pharmacy management policies in FFS in place in FY 2018, and more than three-fourths of states (40 states) plan to take at least one action in FY 2019 to newly implement or increase opioid controls through one of these strategies. Some states also identified early successes in their current initiatives, such as reducing the number of opioids prescribed. See Exhibit 19 and Table 17 for details on states implementing or expanding these controls.
| Exhibit 19: States Implementing Opioid-Focused Pharmacy Benefit Management Strategies in FFS | |||
| Strategy | In Place in FY 2018(# of states) | FY 2019 (# of states) | |
| New | Expanded | ||
| Quantity Limits | 50 | 1 | 25 |
| Clinical criteria claim system edits (subject to Prior Authorization override) | 48 | 0 | 30 |
| Step Therapy PA criteria | 39 | 1 | 5 |
| Other Prior Authorization | 44 | 2 | 15 |
| Required Use of Prescription Drug Monitoring Programs By Prescribers | 32 | 4 | 4 |
Many states reported changes in clinical criteria claims system edits, limits on days supply, and/or cumulative morphine milligram equivalent (MME) limits and utilization controls for FY 2019. At least four states reported changes targeted to a narrow population, such as more restrictive quantity limits for children or when the opioid prescription is written by a dentist. Thirty-six states reported that they either have a legislative mandate or other policy that requires prescribers to check the state PDMP prior to prescribing opioids in place or will be implementing this type of policy by the end of FY 2019. To strengthen the effectiveness of a PDMP requirement, Oklahoma reports the ability to impose sanctions for non-compliance and New Mexico will be establishing recoupment authority for prescribers who do not check the database.
Specific trends in pharmacy management strategies recently adopted or planned include but are not limited to the following:
- Prior authorization, step therapy, and/or other requirements/utilization controls for long acting opioids
- Prior authorization, claims system edits, and/or other utilization controls related to co-prescribing or concurrent use of benzodiazepines
- Expanded pharmacy and/or prescriber profiling, alerts or education
- Policies to encourage or require non-opioid or non-pharmacologic treatment for pain prior to prescribing an opioid
- Pain management contract for chronic pain patients
- SBIRT screening, patient education regarding risks, and contraception counseling for women of childbearing age
A majority of states that use MCOs to deliver pharmacy benefits require or partially require MCOs to follow the state’s FFS pharmacy benefit management policies for opioids. Of the 35 states with MCOs that deliver pharmacy benefits, 17 states responded “yes” to a survey question asking whether MCOs were required to follow the state’s FFS pharmacy benefit management policies for opioids as of July 1, 2018, and 9 states responded “yes, in part.”115 Of the nine states answering “yes in part,” most reported some level of flexibility for MCOs to establish their own PDL and/or coverage criteria. At least two states require that MCOs be no more restrictive in their prior authorization or other criteria than FFS, while other states reported ongoing efforts to develop a more uniform, comprehensive strategy across their FFS and managed care delivery systems.
Looking ahead, beginning in October 2019 the SUPPORT Act116 would require states to have drug utilization review safety edits in place for opioid refills and monitor concurrent prescribing of opioids and other drugs. Medicaid MCOs would be required to have these processes in place as well. The SUPPORT Act also requires each state to have Medicaid providers check the state’s PDMP before prescribing controlled substances and offers enhanced federal matching funds for implementation activities if states have agreements with contiguous states for providers to access these programs.
Medication-Assisted Treatment coverage and access
The ACA requires state Medicaid programs to provide SUD treatment coverage for their ACA expansion populations, but does not specify which SUD services must be included. This requirement, however, has bolstered states’ work to respond to the opioid epidemic. The standard of care for opioid use disorder is medication-assisted treatment (MAT), which combines psychosocial treatment with medication.117 Compared to psychosocial treatment alone, MAT is associated with greater adherence to treatment, decreased opioid use, and reduced likelihood of overdose fatalities.118 There are three medications used as part of MAT for opioid use disorder: methadone, buprenorphine, and both oral and extended-release injectable naltrexone.119 All state Medicaid programs cover at least one MAT medication, and most cover all three.120 ,121
About three-quarters of states reported coverage of methadone in FY 2018. State Medicaid programs are less likely to cover methadone than buprenorphine or naltrexone.122 ,123 In this year’s survey, thirty-eight states reported coverage of methadone in FY 2018.124 Two states reported plans to add coverage for methadone in FY 2019 (Kentucky and Louisiana) and three states reported that methadone coverage was under consideration (Nebraska, North Dakota, and South Carolina). Six states (Alabama, Idaho, Kansas, Oklahoma, Tennessee, and Wyoming) reported no coverage or plans to add coverage for methadone.
In this year’s survey, states identified a range of challenges related to access to MAT. Many states reported lack of waivered physicians to prescribe buprenorphine and a need for additional opioid treatment programs, especially in rural areas. Shortages of ancillary behavioral health providers contributes to the problem, since MAT must be provided as part of a comprehensive treatment plan that includes counseling and social support services. Seven states identified cash-only methadone clinics that do not participate in Medicaid as a barrier to access, and one state noted that these clinics also pose quality concerns. A few states reported challenges related to philosophical differences among providers or populations served (such as preference for an abstinence-based approach), lack of awareness about MAT, and low reimbursement rates. Other states identified challenges when MAT medication is covered as a medical benefit versus a pharmacy benefit. For example, some clinics are hesitant to “buy and bill” for injectable MAT treatments and prefer the medication be dispensed by a pharmacy. One state cited local zoning ordinances as a barrier to increasing the number of available opioid treatment programs.
Although this year’s survey did not ask directly about initiatives to address these challenges, states identified a handful of new initiatives and strategies related to MAT. Multiple states are leveraging technology or telemedicine to increase access, including Indiana University’s Opioid ECHO project to expand the number of trained MAT prescribers in the state. Tennessee reports working in close partnership with its MCOs to develop a statewide MAT provider network and uniform clinical guidelines for effective MAT treatment. Several states mentioned using Substance Abuse and Mental Health Services Administration (SAMHSA) Opioid State Targeted Response (STR) grant funding to support their efforts to increase access to MAT treatment. For example, Arizona is using STR grant funds to establish 24/7 opioid treatment on demand Centers of Excellence and Medication Units in rural hospitals in the state. Other states identified increasing MAT reimbursement rates or removing prior authorization requirements on different MAT therapies to promote access.
The SUPPORT Act expands access to MAT drugs and includes funding to address provider capacity. Looking ahead, the SUPPORT Act,125 expected to be signed into law as this report was being finalized, would require state Medicaid programs to cover all FDA-approved MAT drugs as well as counseling and behavioral therapy services from October 2020 through September 2025, unless a state certifies to the Secretary’s satisfaction that statewide implementation is infeasible due to provider shortages. The Act also would authorize new demonstrations to help states increase Medicaid SUD provider capacity. It would allow for 18-month planning grants, totaling $50 million, for 10 states, giving preference to those with average or higher SUD prevalence, particularly opioid use disorder. Up to five of these states would receive enhanced federal matching funds for Medicaid SUD treatment services during the 36-month waiver implementation.
Table 17: Medicaid FFS Pharmacy Benefit Management Strategies for Opioids in all 50 States and DC, in Place in FY 2018 and Actions Taken in FY 2019
| States | Opioid Quantity Limits | Clinical Edits in Claim System | Opioid Step Therapy Requirements | Other Prior Authorization Requirements for Opioids | Required use of Prescription Drug Monitoring Programs | Any Opioid Management Strategies | ||||||
| In place FY 2018 | New/Exp FY 2019 | In place FY 2018 | New/Exp FY 2019 | In place FY 2018 | New/Exp FY 2019 | In place FY 2018 | New/Exp FY 2019 | In place FY 2018 | New/Exp FY 2019 | In place FY 2018 | New/Exp FY 2019 | |
| Alabama | X | X | X | X | X | X | X | |||||
| Alaska | X | X | X | X | X | X | X | X | X | X | X | |
| Arizona | X | X | X | X | X | X | X | X | ||||
| Arkansas | X | X | X | X | X | X | X | X | X | X | X | |
| California | X | X | X | X | ||||||||
| Colorado | X | X | X | X | X | X | X | |||||
| Connecticut | X | X | X | X | X | X | X | X | ||||
| Delaware | X | X | X | X | X | X | X | X | X | X | ||
| DC | X | X | X | X | X | X | X | |||||
| Florida | X | X | X | X | X | X | X | X | X | X | X | X |
| Georgia | X | X | X | X | X* | X | X | |||||
| Hawaii | X | X* | X | X | ||||||||
| Idaho | X | X | X | X | X | X | X | |||||
| Illinois | X | X | X | X | X | |||||||
| Indiana | X | X | X | X | X | X | X | X* | X | X | ||
| Iowa | X | X | X | X | X | X | X | X | X | X | ||
| Kansas | X* | X | X | X* | X* | X | X | |||||
| Kentucky | X | X | X | X | X | X | X | X | X | X | X | |
| Louisiana | X | X | X | X | X | X | ||||||
| Maine | X | X | X | X | X | X | ||||||
| Maryland | X | X | X | X | X | |||||||
| Massachusetts | X | X | X | X | X | X | X | X | X | |||
| Michigan | X | X | X | X | X | X | X | X | X | |||
| Minnesota | X | X | X | X | X | X | X | X | ||||
| Mississippi | X | X | X | X | X | X | X | |||||
| Missouri | X | X | X | X | X | X | ||||||
| Montana | X | X | X | X | X | X | X | X | ||||
| Nebraska | X | X | X | X | X | X | X | X | ||||
| Nevada | X | X | X | X | X | |||||||
| New Hampshire | X | X | X | X | X | X | X | X | X | X | ||
| New Jersey | X | X | X | X | X | X | ||||||
| New Mexico | X | X | X | X | X | |||||||
| New York | X | X | X | X | X | X | X | X | X | |||
| North Carolina | X | X | X | X | X | X | ||||||
| North Dakota | X | X | X | X | X | X | X | X | ||||
| Ohio | X | X | X | X | X | X | X | X | X | |||
| Oklahoma | X | X | X | X | X | X | X | X | X | X | X | |
| Oregon | X | X | X | X | X | X | X* | X | X | X | ||
| Pennsylvania | X | X | X | X | X | X | X | |||||
| Rhode Island | X | X | X | X | X | X | ||||||
| South Carolina | X | X | X | X | X | X | X | X | ||||
| South Dakota | X | X | X | X | X | X | X | X | X | |||
| Tennessee | X | X | X | X | X | X | ||||||
| Texas | X | X | X | X | X | X | X | |||||
| Utah | X | X | X | X | X | X | X* | X | X | |||
| Vermont | X | X | X | X | X | X | X | X | X | |||
| Virginia | X | X | X | X | X | X | ||||||
| Washington | X | X | X | X | ||||||||
| West Virginia | X | X | X | X | X | X | X | |||||
| Wisconsin | X | X | X | X | X | X | X | X | X | |||
| Wyoming | X | X | X | X | X | X | X | |||||
| Totals | 50 | 26 | 48 | 30 | 39 | 6 | 44 | 17 | 32 | 8 | 51 | 40 |
| NOTES: States were asked to report whether they had select pharmacy benefit management strategies in place in their FFS programs in FY 2018, and/or had plans to adopt or expand these strategies in FY 2019. “*” indicates that a policy was newly adopted in FY 2019, meaning that the state did not have any policy in that category/column in place in FY 2018. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2018. | ||||||||||||
Report: Challenges And Priorities In Fy 2019 And Beyond Reported By Medicaid Directors And Conclusion
States reported a wide variety of priorities for FY 2019 and beyond, including implementing managed care, payment and delivery system reform initiatives; undertaking major information technology system procurements and upgrades; amending or pursuing new Section 1115 demonstration waivers; continuing to tackle the opioid epidemic; and managing their Medicaid budgets.
Over two-thirds of states reported improving quality and focusing on health outcomes as a key priority. Consistent with survey findings in recent years, most states are continuing to develop and implement initiatives to improve the quality of care and patient health outcomes while containing costs. States are doing this through managed care expansions, reforms, and improvements; value-based purchasing initiatives; and other delivery system reforms. In addition, a number of states mentioned addressing the opioid epidemic and expanding the availability of SUD treatment as top priorities (sometimes through Section 1115 demonstration waivers mentioned below).
A number of states mentioned implementation or pursuit of new Section 1115 demonstration waivers or waiver amendments as key priorities beyond 2019. Section 1115 Medicaid demonstration waivers provide states an avenue to test new approaches in Medicaid that differ from federal program rules. While previous sections of this report capture Section 1115 waiver-related policy actions implemented in FY 2018 or planned for implementation in FY 2019, the survey also asked states whether they are planning program changes under Section 1115 authority that would be implemented after FY 2019. The most frequently reported waiver concepts for implementation after FY 2019 address behavioral health services and/or the IMD exclusion, followed by waivers that would implement work and community engagement requirements. Many waivers require significant administrative time and resources to develop, negotiate with CMS, and implement. Waivers also often necessitate system changes (MMIS and/or eligibility), contracting with new support vendors, MCO coordination (including contract amendments), outreach and engagement of members, providers, and other stakeholders, state regulatory changes, and other administrative tasks. For additional details on pending or approved Section 1115 waivers, see the KFF Medicaid Waiver Tracker.
Continuing to tackle the opioid epidemic is another key priority for states in FY 2019 and beyond. New federal legislation expected to be signed into law as this report was being finalized, the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities (SUPPORT) Act, contains a number of provisions related to Medicaid’s role in helping states provide coverage and services to people who need SUD treatment,126 particularly those needing opioid use disorder (OUD) treatment. These provisions include the ability to use federal Medicaid funds for services in “institutions for mental disease” (IMDs) for nonelderly adults for up to 30 days from October 1, 2019 to September 30, 2023; required coverage of all FDA-approved drugs for medication-assisted treatment (MAT) from October 2020 through September 2025; required suspension of Medicaid eligibility for individuals under age 21 or former foster care youth up to age 26 while incarcerated, and restoration of coverage upon release; creation of new demonstrations to help states increase Medicaid SUD provider capacity; and tighter prescription drug oversight.
As in the past, a significant number of states reported information technology systems projects currently underway or planned as high priorities. These are usually related to Medicaid Management Information Systems (MMIS) procurements and eligibility system upgrades and replacements. A few states commented on the need to redesign their MMIS procurements to meet new federal MMIS architecture “modularity” requirements, which are intended to promote the reuse of technical solutions among states, minimize customization and configuration needs, and increase vendor competition. States also commented on the need for system improvements or enhancements to better support other program objectives related, for example, to delivery system reform and value-based purchasing, quality improvement, provider and MCO monitoring, data analytics, and cost control strategies.
States noted that a number of federal regulations pose challenges for Medicaid agencies. Medicaid agencies must comply with ongoing changes in federal and state laws and regulations. Passage of the ACA in 2010, for example, was followed by years of administrative efforts and initiatives to implement the coverage expansions and other Medicaid policy changes required by the ACA and its related regulations. This year’s survey asked states to describe any notable expected administration effects or challenges of recent or anticipated federal regulations. Key findings include:
- States most frequently noted challenges related to the implementation timeline for the electronic visit verification system as required under the 21st Century Cures Act (although the compliance deadline was recently extended from January 1, 2019 to January 1, 2020 for personal care services and to January 1, 2023 for home health).
- Several states cited compliance challenges with the Access Rule, which requires states to develop and periodically update Access Monitoring Review Plans and to perform access reviews when FFS reimbursement cuts are proposed.
- Some states noted challenges with the provider enrollment and screening requirements in the Medicaid managed care regulation.
- Some states cited ongoing challenges with the HCBS Settings rule that establishes requirements for the qualities of settings that are eligible for reimbursement as Medicaid HCBS.
- A few states also pointed to challenging budget implications of the Home Health Rule, as it requires documentation of a face-to-face encounter between a certifying physician and a home health beneficiary and also expands the definition of medical equipment and supplies that are covered under the home health benefit.
Since the survey was fielded, the administration issued proposed rules related to changes in federal “public charge” policies that govern how the use of public benefits may affect individuals’ ability to enter the U.S. or adjust to legal permanent resident (LPR) status (i.e., obtain a “green card”). In anticipation of these regulations, a few states mentioned challenging potential effects including concerns that the anticipated policy changes would further burden the state’s safety net and public health system by depressing Medicaid and CHIP enrollment or result in fear of accessing services, which could increase uncompensated care costs or the frequency of adverse labor and delivery events.
Conclusion
State actions in FY 2018 and FY 2019 show that the Medicaid program is constantly evolving in response to federal policy changes, the economy, and state budget and policy priorities. With less economic stress, more states reported expansions or enhancements to provider rates and benefits (including expansions for community based long-term services and supports and behavioral health services) as well as a focus on improvements in outcomes and value through delivery system reforms and requirements imposed on managed care plans. On the other hand, consistent with policies promoted by the Trump administration, an increasing number of states are pursuing demonstration waivers that include provisions that could result in enrollment declines such as work requirements and retroactive eligibility elimination or restriction. As states continue to work to tackle the opioid epidemic, new federal legislation (the SUPPORT Act) could help states provide coverage and services to people who need SUD treatment. Looking ahead, the trajectory of the economy, the direction of federal policies around Medicaid Section 1115 waivers, and the outcomes of state and federal elections in November 2018 will be factors that continue to shape Medicaid in FY 2019 and beyond.
Methods
The Kaiser Family Foundation (KFF) commissioned Health Management Associates (HMA) to survey Medicaid directors in all 50 states and the District of Columbia to identify and track trends in Medicaid spending, enrollment, and policy making. This is the 18th annual survey, each conducted at the beginning of the state fiscal year from FY 2002 through FY 2019. Additionally, eight mid-fiscal year surveys were conducted during state fiscal years 2002-2004 and 2009-2013, when a large share of states were considering mid-year Medicaid policy changes due to state budget and revenue shortfalls. Findings from previous surveys are referenced in this report when they help to highlight current trends. Archived copies of past reports are available on the following page.127
The KFF/HMA Medicaid survey on which this report is based was conducted from June through September 2018. The survey instrument (in the Appendix) was designed to document policy actions in place in FY 2018 and implemented or adopted for FY 2019 (which began for most states on July 1, 2018).128 The survey captures information consistent with previous surveys, particularly for eligibility, provider payment rates, benefits, long-term care, and managed care, to provide some trend information. Each year, questions are added or revised to address current issues.
Medicaid directors and staff provided data for this report in response to a written survey and a follow-up telephone interview. The survey was sent to each Medicaid director in June 2018. All 50 states and DC completed surveys and participated in telephone interview discussions between July and September 2018.129 The telephone discussions are an integral part of the survey to ensure complete and accurate responses and to record the complexities of state actions.
The survey does not attempt to catalog all Medicaid policies in place for each state. The focus is on changes in Medicaid policy and new initiatives that are planned for FY 2018. Experience has shown that adopted policies are sometimes delayed or not implemented for reasons related to legal, fiscal, administrative, systems, or political considerations, or due to delays in approval from CMS. Policy changes under consideration without a definite decision to implement are not included in the survey. The District of Columbia is counted as a state for the purposes of this report; the counts of state policies or policy actions that are interspersed throughout this report include survey responses from the 51 “states” (including DC). Given differences in the financing structure of their programs, the U.S. territories were not included in this analysis but a separate survey was fielded and results will be released in another report.
Glossary
Acronym Glossary
AAC – Actual Acquisition Cost
ACA – Affordable Care Act
ACO – accountable care organization
ASO – Administrative Services Organization
APCD – all-payer claims database
APM – alternative payment model
BH – behavioral health
CDC – The Centers for Disease Control and Prevention
CFC – Community First Choice
CHIP – Children’s Health Insurance Program
CHIPRA – Children’s Health Insurance Program Reauthorization Act of 2009
CMS – The Centers for Medicare and Medicaid Services
CON – Certificate of Need
CSHCNs – children with special health care needs
DBM – dental benefit manager
D-SNP – Medicare Dual Eligible Special Needs Plans
DSRIP – Delivery System Reform Incentive Program
DUR – drug utilization review
EAC – Estimated Acquisition Cost
ECHO, Project – Extension for Community Healthcare Outcomes
ED – emergency department
EPSDT – Early and Periodic Screening, Diagnostic, and Treatment
FAD – Financial Alignment Demonstration
FDA – Food and Drug Administration
FFS – fee-for-service
FFY – federal fiscal year
FIDE-SNP – Fully Integrated Dual Eligible Special Needs Plans
FPL – federal poverty level
FQHC – federally qualified health center
FY – state fiscal year
GED – general educational development or diploma
HSA – health savings account
HCBS – home and community-based services
HEDIS – Healthcare Effectiveness Data and Information Set
HIT – health information technology
ICF-ID – intermediate care facilities for individuals with intellectual disabilities
I/DD – intellectual and developmental disabilities
IEP – individualized education program
IMD – institutions for mental diseases
LTSS – long-term services and supports
MAGI – modified adjusted gross income
MAT – medication-assisted treatment
MCO – managed care organization
MED – morphine equivalent dose
MFP – Money Follows the Person (federal grant program)
MH – mental health
MLTSS – managed long-term services and supports
MLR – medical loss ratio
MME – morphine milligram equivalent
MMIS – Medicaid Management Information System
NADAC – National Average Drug Acquisition Costs
NCQA – National Committee for Quality Assurance
NEMT – non-emergency medical transportation
NF – nursing facility
OT – occupational therapy
OUD – opioid use disorder
P4P – pay for performance
PA – prior authorization
PACE – Programs of All-Inclusive Care for the Elderly
PCCM – primary care case management
PCMH – patient-centered medical home
PDL – preferred drug list
PDMP – Prescription Drug Monitoring Program
PHP – prepaid health plan
PIP – performance improvement projects
PMPM – per-member per-month
PT – physical therapy
RHC – rural health center
SAMHSA – Substance Abuse and Mental Health Services Administration
SBIRT – Screening, Brief Intervention, and Referral to Treatment
SED – serious emotional disturbance
SIM – State Innovation Models federal grant program
SMI – serious mental illness
SNAP – Supplemental Nutrition Assistance Program
SPA – State Plan Amendment
SSI – supplemental security income
SUD – substance use disorder
TPL – third party liability
VBP – value-based purchasing
WIC – Special Supplemental Nutrition Program for Women, Infants, and Children
Appendix
Survey Instrument
Download the Survey (.pdf)
Endnotes
- Gene therapy is used to treat or prevent genetic diseases by seeking to augment, replace or suppress one or more mutated genes with functional copies. CAR T-cell therapy is a form of immunotherapy that uses specially altered T cells (part of the immune system) collected from the patient to fight cancer. ↩︎
- MaryBeth Musumeci and Jennifer Tolbert, Federal Legislation to Address the Opioid Crisis: Medicaid Provisions in the SUPPORT Act (Washington, DC: Kaiser Family Foundation, October 2018), https://modern.kff.org/medicaid/issue-brief/federal-legislation-to-address-the-opioid-crisis-medicaid-provisions-in-the-support-act/. ↩︎
- Centers for Medicare and Medicaid Services, National Health Expenditure Projections 2017 – 2026 (Washington, DC: Centers for Medicare and Medicaid Services, February 2018), https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/NationalHealthAccountsProjected.html. ↩︎
- National Association of State Budget Officers, States Finalize Fiscal 2019 Budgets, (NASBO, July 2018), https://www.nasbo.org/resources/communityblogs. ↩︎
- National Conference of State Legislatures, FY 2019 State Budget Status, (NCSL, August 2018), http://www.ncsl.org/research/fiscal-policy/fy-2019-budget-status.aspx. ↩︎
- National Association of State Budget Officers, Summary: Fall 2017 Fiscal Survey of the States (National Association of State Budget Officers, December 2017), https://www.nasbo.org/reports-data/fiscal-survey-of-states. ↩︎
- National Association of State Budget Officers, States Target Surpluses to Rainy Day Funds, Other Priorities after Fiscal 2018 Revenues Exceed Estimates (National Association of State Budget Officers Budget Blog, July 2018), http://budgetblog.nasbo.org/budgetblogs/blogs/brian-sigritz/2018/07/30/states-target-surpluses-to-rainy-day-funds-other-p. ↩︎
- Kaiser Family Foundation, 50-State Medicaid Budget Survey Archives (Washington, DC: Kaiser Family Foundation, October 2017), https://modern.kff.org/medicaid/report/medicaid-budget-survey-archives/. ↩︎
- Responses for North Dakota reflect information gathered during a telephone interview in early September 2018 and related research. ↩︎
- State fiscal years begin on July 1 except for these states: NY on April 1; TX on September 1; AL, MI and DC on October 1. ↩︎
- Brian Neale letter to state Medicaid directors, January 11, 2018, https://www.medicaid.gov/federal-policy-guidance/downloads/smd18002.pdf ↩︎
- Kaiser Family Foundation, Medicaid Waiver Tracker: Which States Have Approved and Pending Section 1115 Medicaid Waivers? (Washington, DC: Kaiser Family Foundation, accessed September 2018), https://modern.kff.org/medicaid/issue-brief/section-1115-medicaid-demonstration-waivers-a-look-at-the-current-landscape-of-approved-and-pending-waivers/. ↩︎
- In this report, work requirement policies are counted based on the initial date of implementation rather than the date on which the first coverage terminations will occur. ↩︎
- Medicaid statute requires that Medicaid coverage for most eligibility groups include coverage for a period of 90 days prior to the date of the application for medical assistance. ↩︎
- MaryBeth Musumeci, Elizabeth Hinton, and Robin Rudowitz, Approved Changes in Indiana’s Section 1115 Medicaid Waiver Extension (Washington, DC: Kaiser Family Foundation, February 2018), https://modern.kff.org/medicaid/issue-brief/approved-changes-in-indianas-section-1115-medicaid-waiver-extension/. ↩︎
- The member can reenroll within 90 days from the end of the expired benefit period if they submit the requested redetermination information. However, after the 90-day period, the member is required to wait another three months, or six months from the initial date of disenrollment, until their next open enrollment before being permitted to reenroll in HIP. Indiana has also proposed a work requirement, but that provision would not be effective until FY 2019. ↩︎
- MaryBeth Musumeci, Elizabeth Hinton, and Robin Rudowitz, Proposed Medicaid Section 1115 Waivers in Maine and Wisconsin (Washington, DC: Kaiser Family Foundation, updated August 2017), https://modern.kff.org/medicaid/issue-brief/proposed-medicaid-section-1115-waivers-in-maine-and-wisconsin/. ↩︎
- Maine Question 2, Medicaid Expansion Initiative, (Ballotpedia, 2017), https://ballotpedia.org/Maine_Question_2,_Medicaid_Expansion_Initiative_(2017). ↩︎
- Nevada also implemented this option but did so using Children’s Health Insurance Program (CHIP) funds and therefore is not counted in this report. ↩︎
- Jennifer Ryan, Lucy Pagel, Katy Smali, Samantha Artiga, Robin Rudowitz, and Alexandra Gates, Connecting the Justice-Involved Population to Medicaid Coverage and Care: Findings from Three States (Washington, DC, Kaiser Commission on Medicaid and the Uninsured, June 2016), https://modern.kff.org/medicaid/issue-brief/connecting-the-justice-involved-population-to-medicaid-coverage-and-care-findings-from-three-states/. ↩︎
- Positive changes from the beneficiary’s perspective that were counted in this report are denoted with (+). Negative changes from the beneficiary’s perspective that were counted in this report are denoted with (-). Reductions to Medicaid eligibility pathways in response to the availability of other coverage options (including Marketplace or Medicaid expansion coverage) were denoted as (#). ↩︎
- New premiums are denoted as (New). Changes to premium policies that have a neutral impact from the beneficiary’s perspective are denoted as (Neutral). ↩︎
- This table captures eligibility and premium changes that states have implemented or plan to implement in FY 2018 or 2019, including changes that are part of pending Section 1115 waivers. For pending waivers, only provisions planned for implementation before the end of FY 2019 (according to the state or waiver application documents) are counted in this table. Waiver provisions in pending waivers that states plan to implement in FY 2020 or after are not counted here. ↩︎
- A court order issued on June 29, 2018 vacated the CMS approval of the Kentucky HEALTH waiver and remanded it to CMS for reconsideration regarding how the waiver would meet the medical assistance objectives of the Medicaid statute. At the time of the survey, the waiver remained under consideration at CMS. ↩︎
- After failing to meet the SPA submission deadline (April 3, 2018), Maine’s Governor complied with a Maine Supreme Judicial Court order to submit an expansion SPA on September 4, 2018. However, he also sent a letter to the federal government asking CMS to reject the SPA. Expansion has not yet been implemented as of the time of this survey. ↩︎
- Massachusetts’ pending amendment would remove an existing waiver provision that allows it to enroll expansion adults and other populations in coverage during a 90-day provisional eligibility period while income verification is pending. ↩︎
- Connecticut does not have capitated managed care arrangements, but does carry out many managed care functions, including ASO arrangements, payment incentives based on performance, intensive care management, community workers, educators, and linkages with primary care practices. ↩︎
- California has a small PCCM program operating in LA County for those with HIV. South Carolina uses PCCM authority to provide care management services to approximately 200 medically complex children, but is not counted as a PCCM program for purposes of this report. ↩︎
- Julia Paradise and MaryBeth Musumeci, CMS’s Final Rule on Medicaid Managed Care: A Summary of Major Provisions (Washington, DC: Kaiser Family Foundation, June 2016), http://files.kff.org/attachment/CMSs-Final-Rule-on-Medicaid-ManagedCare. ↩︎
- The general effective date of the final rule is July 5, 2016, although individual provisions of the rule take effect at different times. ↩︎
- National Association of Medicaid Directors, Biweekly Update, August 14, 2018, available at: https://medicaiddirectors.org/covered-in-this-newsletter-is-cms-drug-rebate-guidance-notice-cbo-report-on-growth-medicaid-managed-care-and-state-job-openings/. ↩︎
- Brian Neale, Medicaid Managed Care Regulations with July 1, 2017 Compliance Dates, (Center for Medicaid and CHIP Services Informational Bulletin, June 2017), https://www.medicaid.gov/federal-policy-guidance/downloads/cib063017.pdf. ↩︎
- North Dakota’s total MCO penetration rate estimated as approximately 22% based on data provided in the North Dakota Department of Human Services Quarterly Budget Insight, July 2017 – June 2018, available at http://www.nd.gov/dhs/info/pubs/docs/qtrly-budget-insight-july17-june2018.pdf. ↩︎
- Centers for Medicare and Medicaid Services, Medicaid & CHIP Monthly Application, Eligibility Determinations, and Enrollment Reports, (Washington, DC: Centers for Medicare and Medicaid Services, May 2018), https://www.medicaid.gov/medicaid/program-information/medicaid-and-chip-enrollment-data/report-highlights/index.html. ↩︎
- Illinois reported the MCO penetration rate for all beneficiaries but did not report penetration rates for the individual eligibility categories. ↩︎
- The five Medicaid expansion states without risk-based managed care were Alaska, Arkansas, Connecticut, Montana, and Vermont. ↩︎
- Two other states (Colorado and Massachusetts) reported covering less than 75% MCO penetration for this group. Illinois reported the MCO penetration rate for all beneficiaries but did not report penetration rates for the individual eligibility categories. ↩︎
- 81 FR 27497, available at: https://www.gpo.gov/fdsys/granule/FR-2016-05-06/2016-09581. ↩︎
- In the rule, CMS formalized its policy around “in lieu of,” which is an authority that a number of states were using to cover stays in IMDs prior to this rule. Some of these states must now adapt policies to meet the 15-day requirement, which may have fiscal and programmatic implications for these states. ↩︎
- 28 states answered “yes” for FYs 2018 and 2019: AZ, CO, DC, DE, FL, GA, HI, IA, IL, IN, KY, LA, MA, MI, MN, NJ, NM, NV, OH, OR, PA, RI, TN, TX, UT, VA, WA, and WI. 3 states (MO, SC, WV) plan to start using this authority in FY 2019. CA, MD, MS, NE and NH reported “no” and 3 MCO states – KS, NY, ND – did not provide a response. ↩︎
- U.S. Congress, House, HR 6, 115th Congress (2017-2018)., September 28, 2018, Substance Use–Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act https://www.congress.gov/115/bills/hr6/BILLS-115hr6eah.pdf ↩︎
- In April 2016, CMS issued a final rule on managed care in Medicaid and CHIP that provided a framework of plan standards and requirements designed to improve the quality, performance, and accountability of these programs. The current administration, however, is expected to release revised Medicaid managed care regulations for public comment. ↩︎
- National Association of Medicaid Directors, Medicaid Value-Based Purchasing: What Is It & Why Does It Matter? (Washington, DC: National Association of Medicaid Directors, January 2017), http://medicaiddirectors.org/wp-content/uploads/2017/01/Snapshot-2-VBP-101_FINAL.pdf. ↩︎
- For more information on the State Innovation Models (SIM) initiative, see: https://innovation.cms.gov/initiatives/state-innovations/. ↩︎
- CMS, through the Health Care Payment Learning and Action Network, developed an APM Framework to create a common framework for measuring progress toward VBP. Category 1 includes fee-for-service strategies with no link to payment quality; Category 2 includes fee-for-service strategies with a link of payment to quality and value; Category 3 includes alternative payment models built on fee-for-service architecture; and Category 4 includes population-based payment. Information found at https://innovation.cms.gov/initiatives/Health-Care-Payment-Learning-and-Action-Network. ↩︎
- Ibid. ↩︎
- Centers for Medicare and Medicaid Services, CMS’ Accountable Health Communities Model selects 32 participants to serve as local “hubs”, (Baltimore, MD: Centers for Medicare and Medicaid Services, April 2017), https://www.cms.gov/Newsroom/MediaReleaseDatabase/Press-releases/2017-Press-releases-items/2017-04-06.html. ↩︎
- Arkansas reported plans to implement an MCO program for the first time in FY 2019. ↩︎
- CMCS Information Bulletin, Medicaid Managed Care Regulations with July 1, 2017 Compliance Dates (Baltimore, MD: Centers for Medicare and Medicaid Services, June 30, 2017), https://www.medicaid.gov/federal-policy-guidance/downloads/cib063017.pdf. ↩︎
- Six states reported that they had no MLR specified in MCO contracts as of July 1, 2018 (GA, HI, NH, TN, TX and WI) but are monitoring MLR reporting by plans. Tennessee and Texas both noted that they rely on a methodology that controls for excess MCO profits. ↩︎
- One of the 28 states reporting a PHP arrangement that is not included in Exhibit 12 is Alabama, which reported having a PHP for maternity care. ↩︎
- National Committee on Quality Assurance, “Patient-Centered Medical Home Recognition,” http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx. ↩︎
- Kaiser Commission on Medicaid and the Uninsured, Medicaid Delivery System and Payment Reform: A Guide to Key Terms and Concepts (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, June 2015), https://modern.kff.org/medicaid/fact-sheet/medicaid-delivery-system-and-payment-reform-a-guide-to-key-terms-and-concepts/. ↩︎
- Kaiser Commission on Medicaid and the Uninsured, Medicaid Delivery System and Payment Reform: A Guide to Key Terms and Concept (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, June 2015), https://modern.kff.org/medicaid/fact-sheet/medicaid-delivery-system-and-payment-reform-a-guide-to-key-terms-and-concepts/. ↩︎
- Samantha Artiga, Robin Rudowitz, Jennifer Tolbert, Julia Paradise, and Melissa Majerol, Findings from the Field: Medicaid Delivery Systems and Access to Care in Four States in Year Three of the ACA (Washington, DC, Kaiser Commission on Medicaid and the Uninsured, September 2016), https://modern.kff.org/report-section/findings-from-the-field-medicaid-delivery-systems-and-access-to-care-in-four-states-in-year-three-of-the-aca-issue-brief/. ↩︎
- Alexandra Gates, Robin Rudowitz, and Jocelyn Guyer, An Overview of Delivery System Reform Incentive Payment (DSRIP) Waivers (Washington, DC, Kaiser Commission on Medicaid and the Uninsured, September 2014), https://modern.kff.org/report-section/findings-from-the-field-medicaid-delivery-systems-and-access-to-care-in-four-states-in-year-three-of-the-aca-issue-brief/. ↩︎
- In this report, Oregon’s Coordinated Care Organization (CCO) program is counted as an MCO program, but not as an ACO program, consistent with its CMS designation and the state’s survey response. According to the state, “A coordinated care organization is a network of all types of health care providers (physical health care, addictions and mental health care and sometimes dental care providers) who have agreed to work together in their local communities to serve people who receive health care coverage under the Oregon Health Plan (Medicaid).” (Oregon Health Authority website accessed at: http://www.oregon.gov/oha/HPA/Pages/CCOs-Oregon.aspx.) ↩︎
- Consumer Assessment of Healthcare Providers and Systems ↩︎
- Jack Hoadley, Karina Wagnerman, Joan Alker, and Mark Holmes, Medicaid in Small Towns and Rural America: A Lifeline for Children, Families, and Communities, Georgetown University Center for Children and Families and the University of North Carolina, NC Rural Health Research Program, (Washington D.C., June 2017), https://ccf.georgetown.edu/wp-content/uploads/2017/06/Rural-health-final.pdf. ↩︎
- Project ECHO (Extension for Community Health Outcomes) increases access to specialty treatment in rural and underserved areas by using telehealth to link front-line clinicians with specialist mentors at an academic medical center or hub. ↩︎
- The Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities (SUPPORT) Act, expected to be signed into law as this report was being finalized, contains a number of provisions related to Medicaid’s role in helping states provide coverage and services to people who need substance use disorder (SUD) treatment, particularly those needing opioid use disorder (OUD) treatment. For example, the Act includes new authority to cover IMD services for up to 30 days in a year for persons with an SUD. ↩︎
- Steve Eiken, Kate Sredl, Brian Burwell, and Angie Amos, Medicaid Expenditures for Long-Term Services and Supports in FY 2016 (IAP: Medicaid Innovation Accelerator Program: IBM Watson Health May 2018), https://www.medicaid.gov/medicaid/ltss/downloads/reports-and-evaluations/ltssexpenditures2016.pdf. ↩︎
- Molly O’Malley Watts, MaryBeth Musumeci, and Petry Ubri, Medicaid Section 1115 Managed Long-Term Services and Supports Waivers: A Survey of Enrollment, Spending an Program Policies, (Washington, DC: Kaiser Family Foundation, January 2017), http://modern.kff.org/medicaid/report/medicaid-section-1115-managed-long-term-services-and-supports-waivers-a-survey-of-enrollment-spending-and-program-policies/. ↩︎
- U.S. Senate Commission on Long-Term Care, Report to the Congress, (U.S. Senate Commission on Long-Term Care, September 2013), https://www.gpo.gov/fdsys/pkg/GPO-LTCCOMMISSION/pdf/GPO-LTCCOMMISSION.pdf. ↩︎
- US Department of Health and Human Services, Long-Term Services and Supports: Direct Care Worker Demand Projections 2015-2030 (Health Resources and Services Administration Bureau of Health Workforce, US Department of Health and Human Services, March 2018), https://bhw.hrsa.gov/sites/default/files/bhw/nchwa/projections/hrsa-ltts-direct-care-worker-report.pdf ↩︎
- In FY 2019, Montana is eliminating live-in caregiver services and children’s case management under the 1915(c) waiver for individuals with I/DD and eliminating occupational therapy, dietician/nutrition services, overnight support, and companion services from the Severe Disabling Mental Illness (SDMI) waiver. ↩︎
- After September 2016, with CMS approval, states can continue to transition eligible individuals through 2018 and expend remaining MFP funds through federal FY 2020. ↩︎
- Oregon is not included in this count. The state terminated its MFP program, effective June 30, 2015. ↩︎
- Rebecca Coughlin, Johanna Ward, Noelle Denny-Brown, et al. Final Report: Money Follows the Person Demonstration: Overview of State Grantee Progress, January to December 2016, (Centers for Medicare and Medicaid Services, September 2017), https://www.medicaid.gov/medicaid/ltss/downloads/money-follows-the-person/2016-cross-state-report.pdf. ↩︎
- Most of these states are using current Section 1915(c) waivers that provide community transition services and environmental modifications for seniors, individuals with physical disabilities and/or individuals with intellectual or developmental disabilities, and some states offer housing coordinators or other search services to assist waiver beneficiaries. ↩︎
- In June 2015, CMS issued an Informational Bulletin to clarify when and how Medicaid reimburses for certain housing-related activities, including individual housing transition services, individual housing and tenancy sustaining services, and state-level housing-related collaborative activities. CMS’s intent was to assist states in designing on-going benefits that support community integration for seniors, individuals with disabilities, and individuals experiencing chronic homelessness. Many of the services outlined in CMS’s Informational Bulletin were initially developed under the auspices of MFP… ↩︎
- This count does not include states that have/had managed FFS FADs. For more information see: https://www.cms.gov/Medicare-Medicaid-Coordination/Medicare-and-Medicaid-Coordination/Medicare-Medicaid-Coordination-Office/FinancialAlignmentInitiative/ManagedFeeforServiceModel.html. ↩︎
- The Affordable Care Act (ACA) authorized the Secretary of Health and Human Services to implement the Financial Alignment Initiative to allow state-administered demonstration projects to improve the integration and coordination of services for individuals who are covered under both Medicare and Medicaid. This population, as a group, experiences high rates of hospitalization and use of LTSS and is, on average, a high need, high cost population. See: https://www.cms.gov/Medicare-Medicaid-Coordination/Medicare-and-Medicaid-Coordination/Medicare-Medicaid-Coordination-Office/FinancialAlignmentInitiative/FinancialModelstoSupportStatesEffortsinCareCoordination.html.. ↩︎
- Kaiser Commission on Medicaid and the Uninsured, Health Plan Enrollment in the Capitated Financial Alignment Demonstrations for Dual Eligible Beneficiaries (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, August 2016), https://modern.kff.org/medicaid/fact-sheet/health-plan-enrollment-in-the-capitated-financial-alignment-demonstrations-for-dual-eligible-beneficiaries/. ↩︎
- Arizona, Idaho, Minnesota, New Mexico, Pennsylvania, Tennessee, Texas, Virginia and Wisconsin. ↩︎
- Dual Eligible Special Needs Plans (D-SNPs) enroll beneficiaries who are entitled to both Medicare and Medicaid and offer the opportunity to better coordinate benefits among Medicare and Medicaid. For more information see: https://www.cms.gov/Medicare/Health-Plans/SpecialNeedsPlans/DualEligibleSNP.html. ↩︎
- Fully Integrated Dual Eligible SNPs were created by Congress in Section 3205 of the Affordable Care Act to promote full integration and coordination of Medicaid and Medicare benefits for dual eligible beneficiaries by a single managed care organization. They must have a MIPPA compliant contract with a State Medicaid Agency that includes coverage of specified primary, acute and long-term care benefits and services under risk-based financing. For more information see: https://www.cms.gov/Medicare/Health-Plans/SpecialNeedsPlans/DualEligibleSNP.html#s3. ↩︎
- Delaware, Florida, Iowa, Massachusetts, and New Jersey. ↩︎
- Rhode Island did not provide response to enrollment policy for dual eligible persons with I/DD. ↩︎
- Historically, Medicaid reimbursement for hospitals and nursing homes was cost-based, automatically reflecting incurred cost increases. When rates for these providers are frozen, such annual increases do not occur; hence for this report, rate freezes are counted as restrictions. ↩︎
- Maryland was not able to report MCO rate changes for FY 2019 because rate development was not complete. ↩︎
- Some states also have premium or claims taxes that apply to managed care organizations and other insurers. Since this type of tax is not considered a provider tax by CMS, these taxes are not counted as provider taxes in this report. ↩︎
- The Deficit Reduction Act of 2005 modified section 1903(w)(7)(A) of the Social Security Act. This statute and the implementing regulations eliminated states’ ability to tax only Medicaid MCOs. ↩︎
- Centers for Medicare and Medicaid Services, New Service Delivery Opportunities for Individuals with a Substance Use Disorder (Baltimore, MD: CMS, July 2015), https://www.medicaid.gov/federal-policy-guidance/downloads/SMD15003.pdf. ↩︎
- Centers for Medicare and Medicaid Services, Strategies to Address the Opioid Epidemic (Baltimore, MD: CMS, November 2017), https://www.medicaid.gov/federal-policy-guidance/downloads/smd17003.pdf. ↩︎
- U.S. Congress, House, HR 6, 115th Congress (2017-2018)., September 28, 2018, Substance Use–Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act https://www.congress.gov/115/bills/hr6/BILLS-115hr6eah.pdf ↩︎
- Centers for Medicare and Medicaid Services, Neonatal Abstinence Syndrome: A Critical Role for Medicaid in the Care of Infants (Baltimore, MD: CMS, June 2018), https://www.medicaid.gov/federal-policy-guidance/downloads/cib060818.pdf. ↩︎
- Centers for Medicare and Medicaid Services, Neonatal Abstinence Syndrome: A Critical Role for Medicaid in the Care of Infants (Baltimore, MD: CMS, June 2018), https://www.medicaid.gov/federal-policy-guidance/downloads/cib060818.pdf. ↩︎
- U.S. Congress, House, HR 6, 115th Congress (2017-2018), September 28, 2018, Substance Use–Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act https://www.congress.gov/115/bills/hr6/BILLS-115hr6eah.pdf ↩︎
- Indiana Health Coverage Programs, IHCP bulletin, IHCP adds coverage of community health worker services (Indiana Health Coverage Programs, May 2018), http://provider.indianamedicaid.com/ihcp/Bulletins/BT201826.pdf. ↩︎
- Julia Paradise, Medicaid Moving Forward (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, March 2015), https://modern.kff.org/health-reform/issue-brief/medicaid-moving-forward/. ↩︎
- MaryBeth Musumeci, Elizabeth Hinton, and Robin Rudowitz, Proposed Medicaid Section 1115 Waivers in Maine and Wisconsin (Washington, DC: Kaiser Family Foundation, updated August 2017), https://modern.kff.org/medicaid/issue-brief/proposed-medicaid-section-1115-waivers-in-maine-and-wisconsin/. ↩︎
- MaryBeth Musumeci, Robin Rudowitz, and Elizabeth Hinton, Approved Changes in Indiana’s Section 1115 Medicaid Waiver Extension (Washington, DC: Kaiser Family Foundation, February 2018), https://modern.kff.org/medicaid/issue-brief/approved-changes-in-indianas-section-1115-medicaid-waiver-extension/. ↩︎
- CMS Medicaid Drug Rebate Program website: https://www.medicaid.gov/medicaid/prescription-drugs/medicaid-drug-rebate-program/index.html. ↩︎
- Katherine Young, Robin Rudowitz, Rachel Garfield, and MaryBeth Musumeci, Medicaid’s Most Costly Outpatient Drugs (Washington, DC: Kaiser Family Foundation, July 2016), https://modern.kff.org/medicaid/issue-brief/medicaids-most-costly-outpatient-drugs/. ↩︎
- Gene therapy is used to treat or prevent genetic diseases by seeking to augment, replace or suppress one or more mutated genes with functional copies. CAR T-cell therapy is a form of immunotherapy that uses specially altered T cells (part of the immune system) collected from the patient to fight cancer. ↩︎
- Richard Mark Kirkner, Gene Therapy: Must Sky-High Prices ‘Come on Down’ Before the Price Is Right?, (Managed Care Magazine, July 2, 2018; https://www.managedcaremag.com/archives/2018/7/gene-therapy-must-sky-high-prices-come-down-price-right. ↩︎
- New York State Medicaid Drug Utilization Review (DUR) Board Meeting Summary for April 26, 2018; https://www.health.ny.gov/health_care/medicaid/program/dur/meetings/2018/04/summary_durb.pdf. ↩︎
- Wisconsin Department of Health Services, Family Care, Family Care Partnership, and PACE Enrollment Data, (Wisconsin Department of Health Services, July 2018), https://www.dhs.wisconsin.gov/familycare/reports/enrollmentdata.pdf. ↩︎
- A “kick payment” is a supplemental payment over and above the capitation payment made to the MCO for beneficiaries utilizing a specified set of services or having a certain condition. ↩︎
- Substance Abuse and Mental Health Services Administration (SAMHSA), 2016 National Survey on Drug Use and Health: Detailed Tables (Rockville, MD: SAMHSA, September 2017), https://www.samhsa.gov/data/sites/default/files/NSDUH-DetTabs-2016/NSDUH-DetTabs-2016.pdf ↩︎
- “Understanding the Epidemic,” Centers for Disease Control and Prevention, accessed on September 2, 2018, https://www.cdc.gov/drugoverdose/epidemic/index.html. ↩︎
- “Understanding the Epidemic,” Centers for Disease Control and Prevention, accessed on September 2, 2018, https://www.cdc.gov/drugoverdose/epidemic/index.html. ↩︎
- U.S. Department of Health and Human Services, Determination That a Public Health Emergency Exists (HHS, October 26, 2017), https://www.hhs.gov/sites/default/files/opioid%20PHE%20Declaration-no-sig.pdf. ↩︎
- Kaiser Family Foundation, Medicaid’s Role in Addressing the Opioid Epidemic (Washington, DC: Kaiser Family Foundation, February 2018), https://modern.kff.org/infographic/medicaids-role-in-addressing-opioid-epidemic/. ↩︎
- Centers for Medicare and Medicaid Services, New Service Delivery Opportunities for Individuals with a Substance Use Disorder (Baltimore, MD: CMS, July 2015), https://www.medicaid.gov/federal-policy-guidance/downloads/SMD15003.pdf. ↩︎
- Centers for Medicare and Medicaid Services, Strategies to Address the Opioid Epidemic (Baltimore, MD: CMS, November 2017), https://www.medicaid.gov/federal-policy-guidance/downloads/smd17003.pdf. ↩︎
- Neither letter addresses the use of federal Medicaid funds for IMD mental health services. ↩︎
- U.S. Congress, House, HR 6, 115th Congress (2017-2018)., September 28, 2018, Substance Use–Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act https://www.congress.gov/115/bills/hr6/BILLS-115hr6eah.pdf ↩︎
- MaryBeth Musumeci and Jennifer Tolbert, Federal Legislation to Address the Opioid Crisis: Medicaid Provisions in the SUPPORT Act (Washington, DC: Kaiser Family Foundation, October 2018), https://modern.kff.org/medicaid/issue-brief/federal-legislation-to-address-the-opioid-crisis-medicaid-provisions-in-the-support-act/. ↩︎
- Several states mentioned plans to implement quantity limits based on a “morphine equivalent dose” (MED), which is the amount of opioid prescription drugs, converted to a common “standard” unit (milligrams of morphine). For example, both 60 mg of oxycodone (approximately 2 tablets of oxycodone sustained-release 30 mg) and approximately 20 mg of methadone (4 tablets of methadone 5 mg) are equal to 90 MMEs (morphine milligram equivalents). ↩︎
- “Clinical edits” are clinically-based claims adjudication rules that a claims system will follow when processing a pharmacy claim. ↩︎
- Step therapy prior authorization criteria involves requiring the use of another agent or therapy prior to the use of a specific opioid. ↩︎
- Prescription Drug Monitoring Programs (PDMPs) are state-run electronic databases that are valuable tools for addressing prescription drug diversion and abuse. Currently, except for Missouri, every state and the District of Columbia operates a PDMP. ↩︎
- In this year’s survey, Illinois did not report whether MCOs are required to follow Medicaid fee-for-service policies related to opioids and pharmacy benefit management. ↩︎
- U.S. Congress, House, HR 6, 115th Congress (2017-2018)., September 28, 2018, Substance Use–Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act https://www.congress.gov/115/bills/hr6/BILLS-115hr6eah.pdf ↩︎
- Substance Abuse and Mental Health Services Administration, “Medication-Assisted Treatment (MAT),” (Substance Abuse and Mental Health Services Administration, last updated 02/07/2018), https://www.samhsa.gov/medication-assisted-treatment. ↩︎
- The Pew Charitable Trusts, Medication-Assisted Treatment Improves Outcomes for Patients With Opioid Use Disorder, (Washington, DC: The Pew Charitable Trusts, November 2016), http://www.pewtrusts.org/en/research-and-analysis/fact-sheets/2016/11/medication-assisted-treatment-improves-outcomes-for-patients-with-opioid-use-disorder. ↩︎
- Substance Abuse and Mental Health Services Administration, “Medication-Assisted Treatment (MAT),” Substance Abuse and Mental Health Services Administration, last updated 02/07/2018, https://www.samhsa.gov/medication-assisted-treatment. ↩︎
- Kathleen Gifford et al., Medicaid Moving Ahead in Uncertain Times: Results from a 50-State Medicaid Budget Survey for State Fiscal Years 2017 and 2018 (Washington, DC: Kaiser Family Foundation, October 2017), https://modern.kff.org/medicaid/report/medicaid-moving-ahead-in-uncertain-times-results-from-a-50-state-medicaid-budget-survey-for-state-fiscal-years-2017-and-2018/. ↩︎
- Naltrexone does not carry abuse or diversion potential, and any provider licensed to prescribe medications can prescribe naltrexone. However, to prescribe or dispense buprenorphine, physicians must obtain a “waiver”. This process involves 1) registering with the Drug Enforcement Administration (DEA) to dispense controlled substances; 2) certifying intent to treat no more than 30 patients at one time in the first year; and 3) receipt of required training or certification. Physicians may apply to increase the allowable patient caseload, and if approved may treat up to 100 patients in their first year and up to 275 patients in subsequent years. Methadone may only be dispensed by opioid treatment programs certified by the Substance Abuse and Mental Health Services Administration (SAMHSA). Opioid treatment programs may also dispense buprenorphine. ↩︎
- Kathleen Gifford et al., Medicaid Moving Ahead in Uncertain Times: Results from a 50-State Medicaid Budget Survey for State Fiscal Years 2017 and 2018 (Washington, DC: Kaiser Family Foundation, October 2017), https://modern.kff.org/medicaid/report/medicaid-moving-ahead-in-uncertain-times-results-from-a-50-state-medicaid-budget-survey-for-state-fiscal-years-2017-and-2018/. ↩︎
- On last year’s budget survey, all 49 states that responded to a question about coverage of each of the MAT drugs reported coverage of buprenorphine and both oral and injectable naltrexone, but fewer states reported coverage of methadone. ↩︎
- In this year’s survey, Illinois and Arkansas did not report whether their state covers Methadone to treat opioid use disorders. ↩︎
- U.S. Congress, House, HR 6, 115th Congress (2017-2018)., September 28, 2018, Substance Use–Disorder Prevention that Promotes Opioid Recovery and Treatment (SUPPORT) for Patients and Communities Act https://www.congress.gov/115/bills/hr6/BILLS-115hr6eah.pdf ↩︎
- MaryBeth Musumeci and Jennifer Tolbert, Federal Legislation to Address the Opioid Crisis: Medicaid Provisions in the SUPPORT Act (Washington, DC: Kaiser Family Foundation, October 2018), https://modern.kff.org/medicaid/issue-brief/federal-legislation-to-address-the-opioid-crisis-medicaid-provisions-in-the-support-act/. ↩︎
- Kaiser Family Foundation, 50-State Medicaid Budget Survey Archives, (Washington, DC: Kaiser Family Foundation, October 2017), https://modern.kff.org/medicaid/report/medicaid-budget-survey-archives/. ↩︎
- State fiscal years begin July 1 except for these states: NY on April 1; TX on September 1; AL, MI and DC on October 1. ↩︎
- Responses for North Dakota reflect information gathered during a telephone interview in early September 2018 and related research. ↩︎