State Approaches to Addressing the Opioid Epidemic: Findings from a Survey of State Medicaid Programs
Opioid overdoses rose to 81,051 in 2022, a slight increase from 80,411 in 2021, but a substantial 63% jump from 2019 (49,860). The sharp rise in recent years is mainly due to fentanyl, as the illicit supply of this drug has risen substantially. Notably, rises in overdose deaths have been particularly sharp among people of color and young people. Adding to these data, recent KFF polling shows that 29% of adults say either they or a family member have experienced an opioid addiction. Three medications for opioid use disorder (MOUD) are FDA approved: methadone, buprenorphine, and naltrexone. Under the SUPPORT Act (major legislation passed in 2018 to address the opioid epidemic) these medications must be covered by Medicaid through 2025.1 Many provisions of the SUPPORT Act expired in September 2023 and Congress continues to debate legislation to reauthorize some expiring provisions and adopt new policies to address the opioid epidemic. There are a variety of efforts at the state level to address the epidemic. State Medicaid programs play a particular role in the opioid epidemic, covering nearly 40% of people with opioid use disorder (OUD).
To better understand how states are using Medicaid to help address the opioid epidemic, the 23rd annual Medicaid budget survey, conducted by KFF and Health Management Associates (HMA) asked about the following specific strategies adopted or planned by state Medicaid officials for state fiscal years (SFYs) 2023 and 2024 to address the opioid epidemic: removing prior authorization for buprenorphine (a medication treatment for OUD), reimbursing for the initiation of buprenorphine treatment via telehealth, covering over-the-counter (OTC) Narcan, and implementing other initiatives. Key takeaways include:
- Most states have removed prior authorization requirements for certain buprenorphine treatments, but other barriers remain and are complicated by the fentanyl epidemic. Prior authorization requirements for standard buprenorphine treatment have decreased substantially in recent years, but other hurdles, such as counseling mandates or prior authorization requirements for higher doses—potentially necessary for treating people who used fentanyl—still remain.
- Most Medicaid programs cover buprenorphine induction via telehealth, but this policy is dependent on evolving federal regulations. Over three-quarters of states provide fee-for-service (FFS) reimbursement for buprenorphine induction over telehealth, but the future of this policy is subject to evolving federal regulations on prescribing controlled substances via telehealth.
- At least one-third of states have or plan to add OTC Narcan to FFS Medicaid OTC formularies. One-third of states commented on plans or actions to add over the counter Narcan to their state’s FFS formularies. Increased accessibility may also be impacted by effective Medicaid communication about OTC reimbursement policies and procedures.
- A number of states are pursuing options to add pre-release Medicaid coverage for incarcerated populations. At the time of the survey, two states secured CMS approval to provide Medicaid coverage to incarcerated individuals before release, including OUD treatment. As of January 2024, two states (California and Washington) have obtained CMS approval, and 15 other states have waivers pending.
Opioid Use Disorder Treatment without Prior Authorization
Medicaid programs must cover medications to treat OUD, like buprenorphine and methadone, but the actual access to these medications may vary for a variety of reasons, including utilization management strategies such as prior authorization (PA) that vary across Medicaid FFS programs and managed care organizations (MCO). In states without PA, patients may have longer buprenorphine treatment episodes, and Medicare data suggests that removing PA can reduce hospital and emergency department use. Although a number of states have enacted laws prohibiting PA for medications to treat OUD, many have not, leaving PA policy decisions up to the state Medicaid programs and sometimes their contracted health plans.
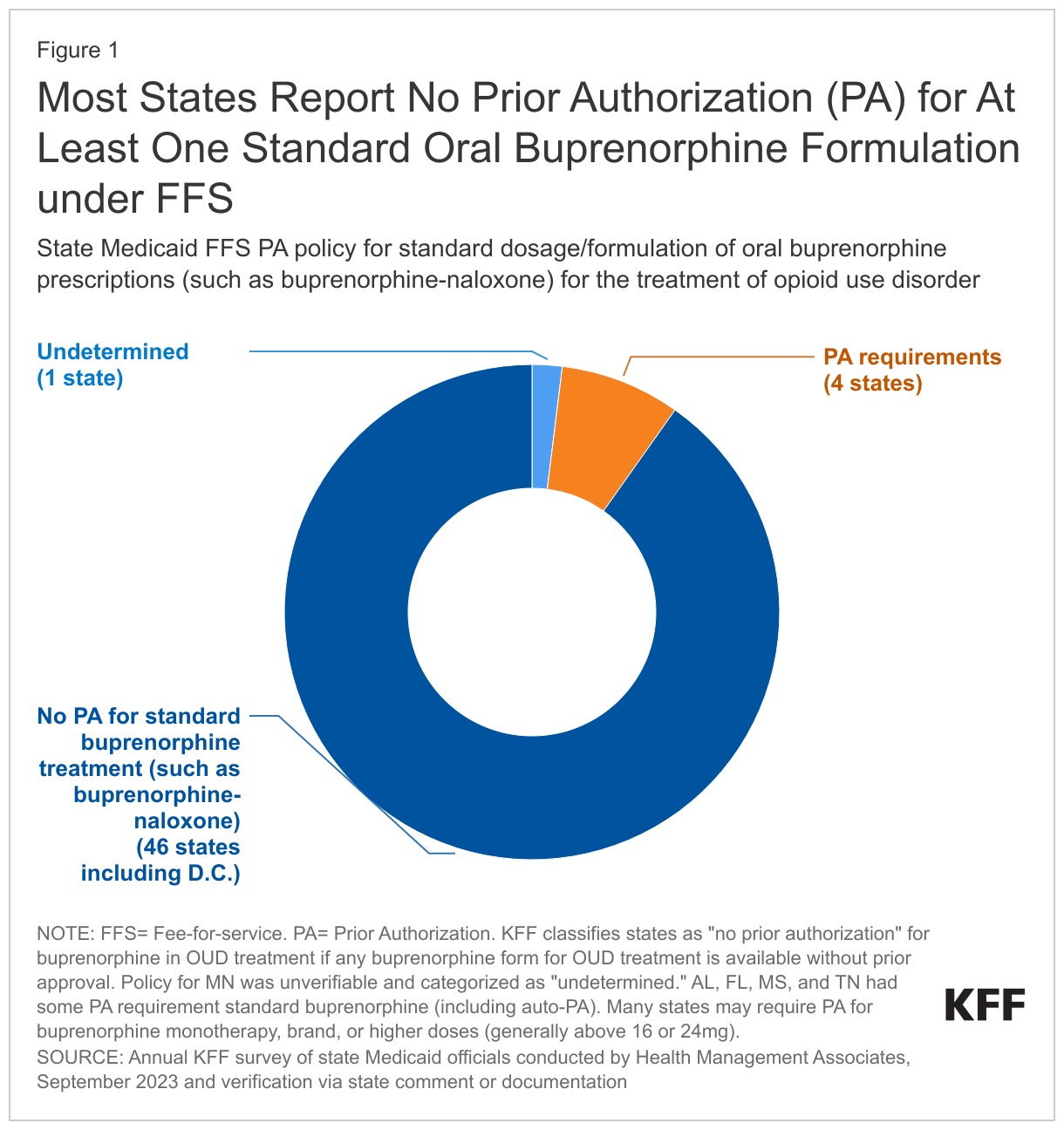
As of SFY 2024, most states no longer require prior authorization for at least one standard formulation of buprenorphine medication therapy under fee-for-service (FFS), reflecting a policy shift in recent years. As of SFY 2024, nearly all states cover at least one standard formulation and dosage of oral buprenorphine, such as buprenorphine-naloxone, without PA (Figure 1).2 Just a few years ago, in 2018, MACPAC reported that 30 states required PA for buprenorphine-naloxone, but as of SFY 2024, only a small number of Medicaid programs still have that requirement for at least one standard formulation or dosage. Many states impose PA requirements on non-standard forms of buprenorphine, such as higher doses or buprenorphine monotherapy, and PA requirements may vary across Medicaid managed care organizations within a state. Categorizing prior authorization policy is complex due to rapidly evolving policies and differences in prior authorization requirements that can vary across dosing, formulations, and populations.3
Even though most states removed prior authorization requirements for certain buprenorphine treatments, enrollees may still encounter ongoing and emerging barriers, including policies that have not yet adapted to the spread of the more potent fentanyl. While many states removed prior authorization for specific buprenorphine formulations, they often maintain prior authorization for higher doses. Yet, some physicians report that higher dosing may be necessary for patients with a history of fentanyl use, and research shows that higher dosing has implications for treatment retention. Beyond prior authorization policies, some states impose other criteria such as urine screening requirements, quantity limits, and counseling mandates—additional restrictions that may impact access to or retention in care. Provider reimbursement rates may also play a role in attracting workforce to provide services.
Coverage of Buprenorphine Treatment Initiation via Telehealth
During the COVID-19 Public Health Emergency (PHE), the Drug Enforcement Administration (DEA) temporarily relaxed federal regulations to allow patients to start buprenorphine treatment for OUD via telehealth, eliminating the initial in-person visit requirement. This change boosted access and retention in care without increasing buprenorphine-related overdoses, and may have also helped to mitigate workforce shortages. While most states expanded telehealth coverage for behavioral health services in both FFS and managed care programs during the PHE, the extent of telehealth coverage for buprenorphine treatment varies by state, as Medicaid programs individually set their telehealth policies, as long as they are aligned with federal and state policy. Recently, the DEA, jointly with the Department of Health and Human Services, authorized a second temporary extension of COVID-19 telemedicine flexibilities, which continues the pandemic-era policies that allow controlled substance prescribing without an initial in-person visit through the end of 2024.
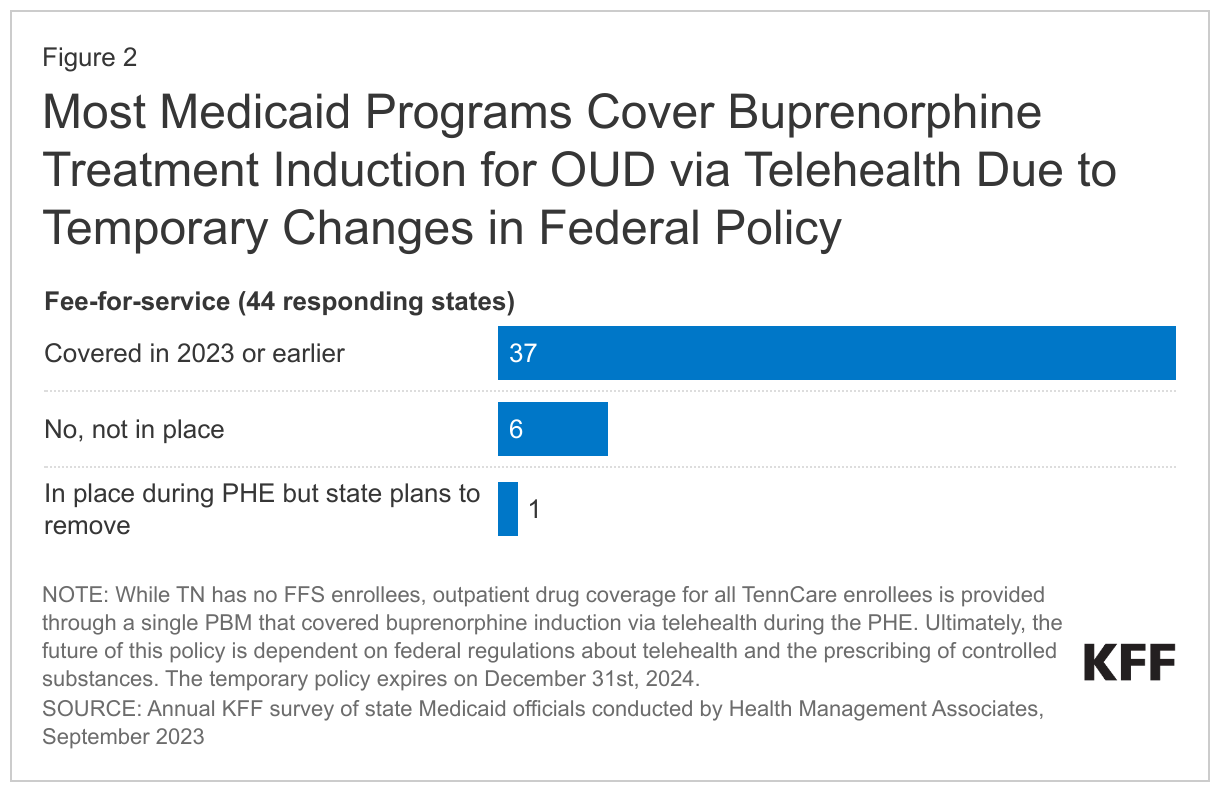
Over three-quarters of states reported covering buprenorphine induction via telehealth under fee-for-service in SFY 2023. However, six states did not allow this coverage, and one state reported plans to discontinue coverage. The continuation of this policy is dependent on federal regulations pertaining to telehealth and prescribing of controlled substances. During the pandemic, federal regulations were relaxed to facilitate treatment access, allowing buprenorphine to be prescribed via telehealth without an in-person visit--a temporary measure set to expire on December 31st 2024. However, state policies can still mandate in-person visits, potentially complicating treatment access in those states.
Coverage of Over-the-Counter Narcan
Naloxone, commonly known as Narcan, a life-saving medication that can reverse opioid overdoses, is now available over the counter as a nasal spray. While the FDA has approved two versions of OTC Narcan, only one is currently available in pharmacies. Its price tag, roughly $45 for a 2-dose, 4mg nasal spray, may be unaffordable for many who lack OTC insurance coverage including Medicaid enrollees in states that have not added OTC Narcan to their Medicaid OTC formularies and/or required their MCOs to do so. To obtain federal matching funds, federal Medicaid law also requires Medicaid enrollees to obtain a prescription for covered OTC products – another hurdle that may impede access. The most recent KFF budget survey asked states about their strategies to enhance enrollee access to OTC Narcan and many states offered comments regarding their coverage or planned coverage of OTC Narcan.
About one-third of states reported that they had added or planned to add OTC Narcan to their state’s OTC formularies. This is likely an undercount of states currently covering OTC Narcan as not all states chose to specifically comment on their Medicaid coverage policy. In addition to adding OTC Narcan to their formularies, Massachusetts commented that Narcan is exempt from Medicaid copay requirements and California mentioned the state’s Naloxone Distribution Project ships free naloxone directly to eligible entities such as law enforcement, schools, community organizations, and city, county, and Tribal organizations. Other states mentioned standing order policies or other initiatives that make it easier for pharmacists to dispense Narcan. Although these policies might make it easier for enrollees to access OTC Narcan, factors such as clear enrollee communication about the reimbursement for OTC Narcan, where Narcan is placed in stores, and Narcan access for family members may also play a role in its overall accessibility.
Other State Initiatives
States were asked about other Medicaid initiatives to combat the opioid epidemic.
The most frequently mentioned initiative was the pursuit of Section 1115 Demonstration Waiver authority to cover OUD services for inmates prior to release. At the time of the survey, two states obtained CMS approval (California and Washington) and 14 states had waiver requests under review for these demonstration projects under section 1115, which permits Medicaid programs to provide services before inmate release (including OUD treatment). As of January 2024, two states have approvals and an additional 15 states have re-entry waiver requests under CMS review. Following release from prison, individuals face a significantly higher risk of overdose and death, primarily from opioids, with a study in one state showing a 40 times greater risk of opioid overdose in the first two weeks after release compared to the general public. In states that expanded Medicaid, most individuals recently released from prison meet income and eligibility requirements for Medicaid coverage, but the Medicaid Inmate Exclusion Policy under federal Medicaid law prohibits federal dollars from paying for health care services of inmates in most cases. CMS guidance requires that states seeking pre-release waivers must cover medication-assisted treatment, but other details vary.
States reported other initiatives; some that are specific to Medicaid and others that are broader:
- Expanding access to services by removing copays, expanding provider types, or adjusting reimbursement. For example, Maryland added coverage of peer recovery support services for SUD services provided at specific locations and Indiana began to reimburse Opioid Treatment Programs using a weekly bundled rate, rather than a per diem rate. More broadly, Medicaid programs are continuing to broaden coverage for mental health and substance use disorder services. In FY 2023 and 2024, more than a third of states reported benefit enhancements or additions and about three-quarters of states reported increases or plans to increase, the fee-for-service payment rates for at least one type of behavioral health provider.
- Innovative programs or solutions. Some states are testing out new delivery methods or innovative programs. For example, Delaware submitted a waiver amendment to allow coverage for contingency management Texas is continuing to provide services under the Maternal Opioid Misuse (MOM) model to provide treatment services to pregnant women with OUD. Often, state efforts involve collaborations that span across several state agencies. For example, Arizona is working to expand access to mobile opioid treatment programs to bring treatment to those in underserved areas and is partnering with other state agencies to coordinate an opioid response plan. Vermont has a longstanding approach, Hub and Spoke model, to address OUD that spans multiple agencies.
- Alternative opioid treatment or safe opioid prescribing. Other states are working to reduce reliance on prescriptions opioids. For example, Idaho and Alaska reported focusing on safe opioid prescribing policies and Pennsylvania reported revising prior authorization guidelines for opioid prescriptions. Missouri’s health plans offer a program intended to help provide alternatives to opioids for the treatment of chronic pain.
Looking ahead
Despite recent increased access to OUD treatment, a notable treatment gap remains as one-third of diagnosed Medicaid enrollees are not receiving treatment. This issue is particularly acute among Black people, youth, and those with disabilities, where substantial disparities in access to MOUD persist. Behavioral health workforce shortages, prevalent across the continuum of behavioral health services and provider types, are also present in Medicaid-- along with a lack of provider diversity and few culturally competent care options. Medicaid programs in many states are addressing these challenges by implementing strategies to address workforce shortages and by using managed care contracts to address and reduce disparities, with some efforts specifically focused on reducing disparities in substance use disorders. Regulatory authorities recently made permanent the pandemic-era policy that allows opioid treatment patients to take home methadone doses, previously dispensed only onsite.
Both the House and Senate are engaged in reauthorizing the recently expired SUPPORT Act, though the outcome remains uncertain. The House passed a bill to reauthorize and extend the Act, focusing on reauthorizing programs and expanding support for prevention and treatment, workforce initiatives, and programs to address trauma. The House bill also includes several Medicaid-specific measures, such as making permanent the requirement that states cover medications for opioid use disorder, such as buprenorphine and methadone. Other Medicaid specific provisions include making the state plan amendment option to allow payment for treatment in institutions of mental disease (IMD) permanent, preventing Medicaid disenrollment due to incarceration, and expanding the CMS substance use disorder data book to include mental health conditions. It also proposes classifying Xylazine as a controlled substance and reevaluating the drug scheduling for medications that combine buprenorphine and naloxone (generally considered lower risk). The Senate will soon debate a separate version of the SUPPORT Act reauthorization, although the full details are not yet available. The Senate HELP Committee also marked up another bill aimed at broadening access to methadone by extending prescribing privileges to qualified practitioners outside of federally registered opioid treatment programs and allowing dispensing at pharmacies. Further, the FEND off Fentanyl Act, a bill that has recently been attracting attention, focuses on reducing the illicit supply of fentanyl.
Broader state-level opportunities and challenges are at play in efforts to address the opioid epidemic. States are deciding how to use opioid settlement funds, but questions about state spending decisions and transparency have been raised. While some states and counties are using the funds to invest in treatment for uninsured, expand treatment infrastructure, and increase access to naloxone, some counties have made more questionable decisions--such as using the settlement funds to plug holes in debt or purchase police patrol cars. Meanwhile, innovative state approaches to address the opioid epidemic have also emerged. For example, Pennsylvania built a “warm handoff” program, which offers direct referrals to SUD treatment for overdose survivors, enhancing access to care. Similarly, to combat overdoses, some states are investing funds in installing vending machines stocked with Narcan. Despite these efforts, some state policies may present obstacles. For example, state laws that treat fentanyl test strips as drug paraphernalia and a lack of protections for individuals who prescribe or administer Narcan during an overdose may hinder progress in reducing opioid overdoses.
Medicaid unwinding may lead to loss or interruptions in coverage for people who depend on Medicaid for OUD treatment, potentially disrupting their access to necessary treatment and increasing the risk of overdose and death. The recent resumption of Medicaid renewals following a three-year pandemic halt – ‘Medicaid unwinding’ – has led to millions individuals losing Medicaid coverage, primarily due to procedural reasons. Many people who lose Medicaid have alternative insurance options. However, for those who rely on Medicaid to cover OUD treatment, the loss of Medicaid coverage could disrupt their access to ongoing OUD treatment. Such a loss may disrupt treatment and increase overdose risks, especially in the midst of the ongoing fentanyl crisis that is recently further complicated by emerging threats like Nitazenes and Xylazine.
This brief draws on work done under contract with Health Management Associates (HMA). Kathleen Gifford is an expert on state Medicaid programs and a Principal at HMA.
- According to a recent OIG report, Hawaii, South Dakota, and Wyoming were granted an exception from the SUPPORT requirement to cover all FDA-approved MOUD. However, the report found that all these programs covered or reimbursed buprenorphine or naltrexone in 2021. ↩︎
- While TN has no FFS enrollees, outpatient drug coverage for all TennCare enrollees is provided through a single PBM that imposes reduced buprenorphine PA requirements on prescribers participating in the state's "BESMART" medication assisted treatment (MAT) provider network. PA may be required for providers outside of this network. ↩︎
- KFF considers states as having "no prior authorization" for buprenorphine products used in OUD treatment if they offer at least one type of buprenorphine medication (brand or generic, solo or combined with naloxone) without prior authorization requirements, despite possible age or dose limits. Other studies on buprenorphine prior authorization might differ by lookup dates, formulations, and other methodological differences, leading to differences in reported counts across studies. ↩︎