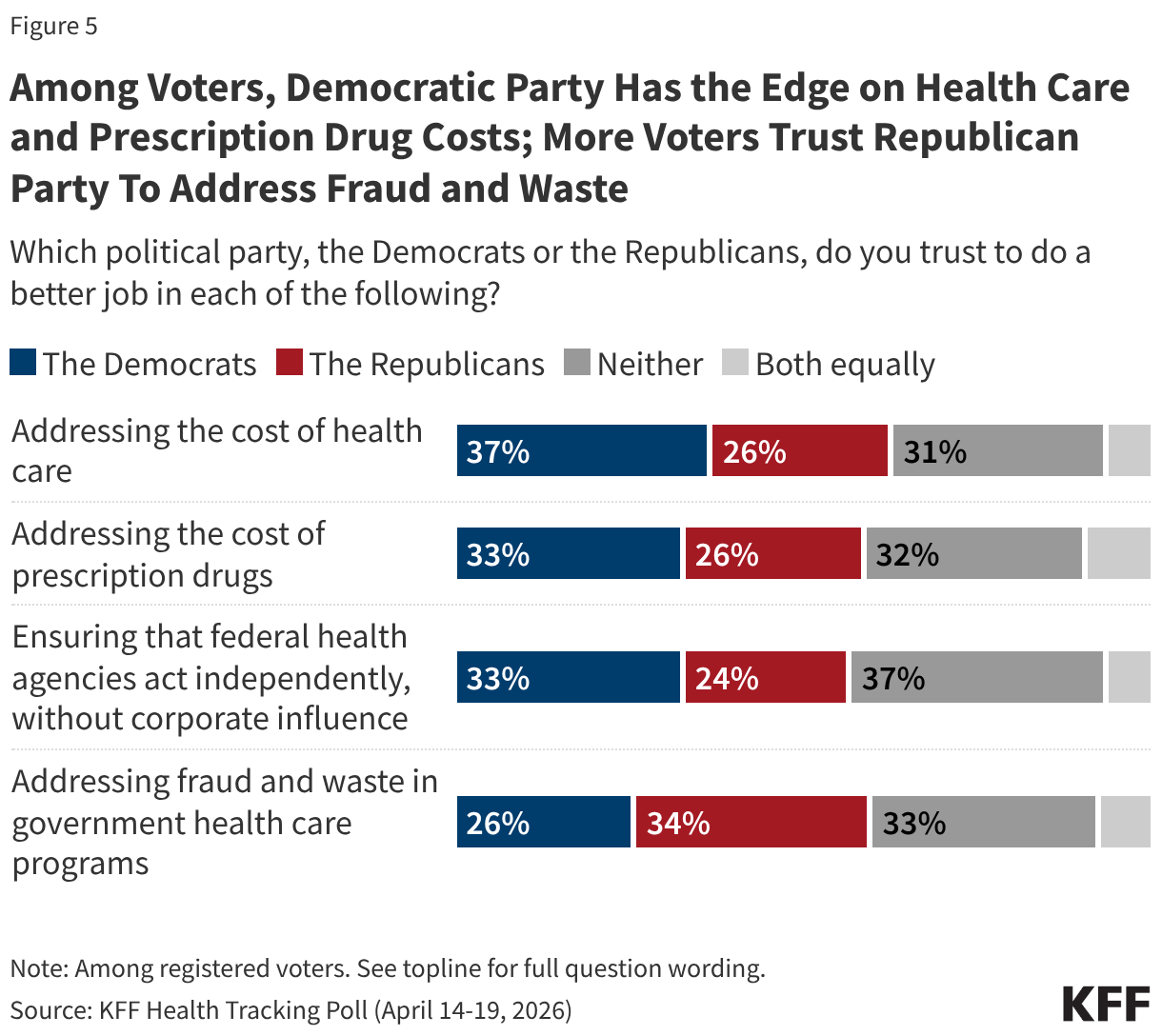
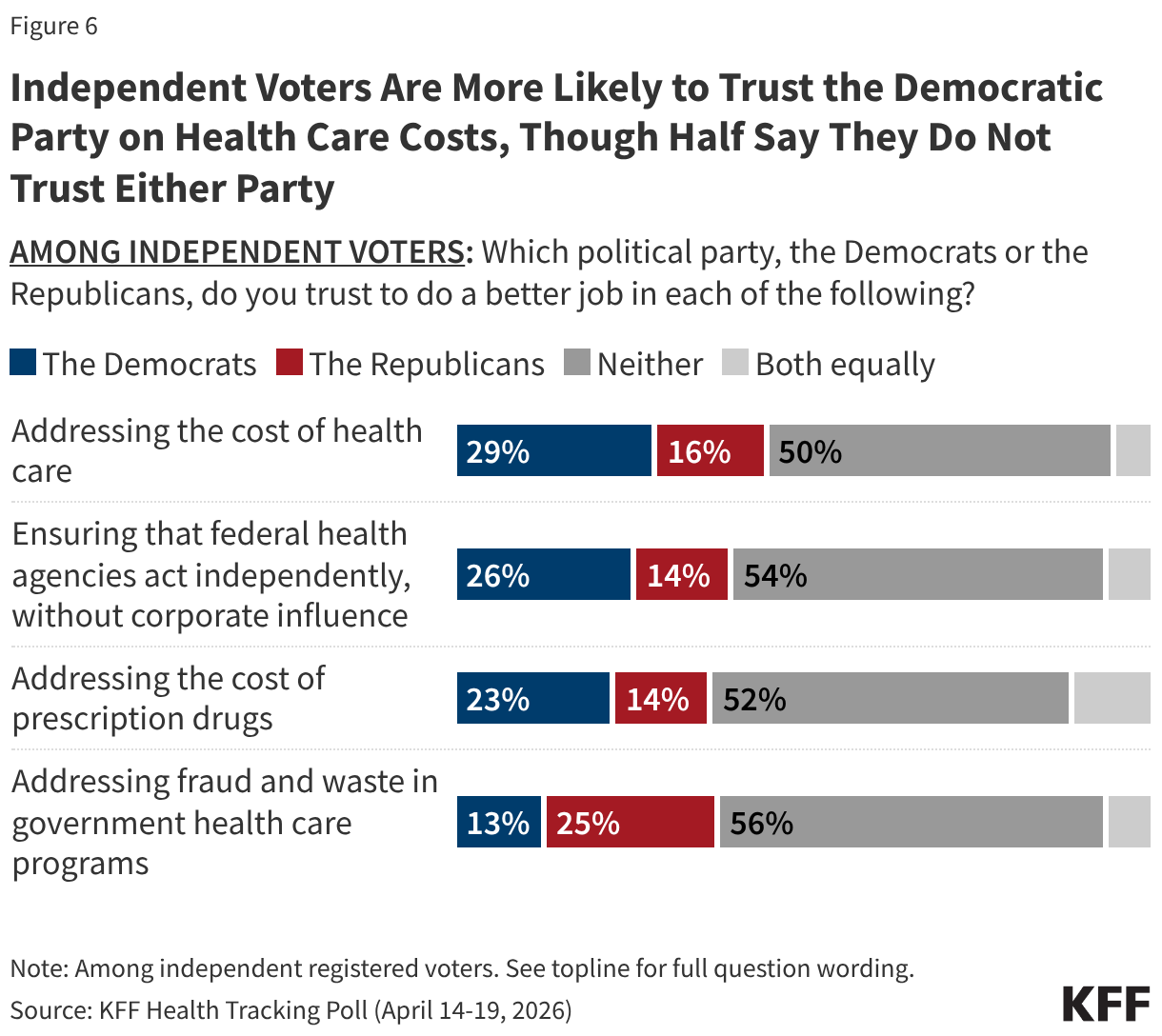
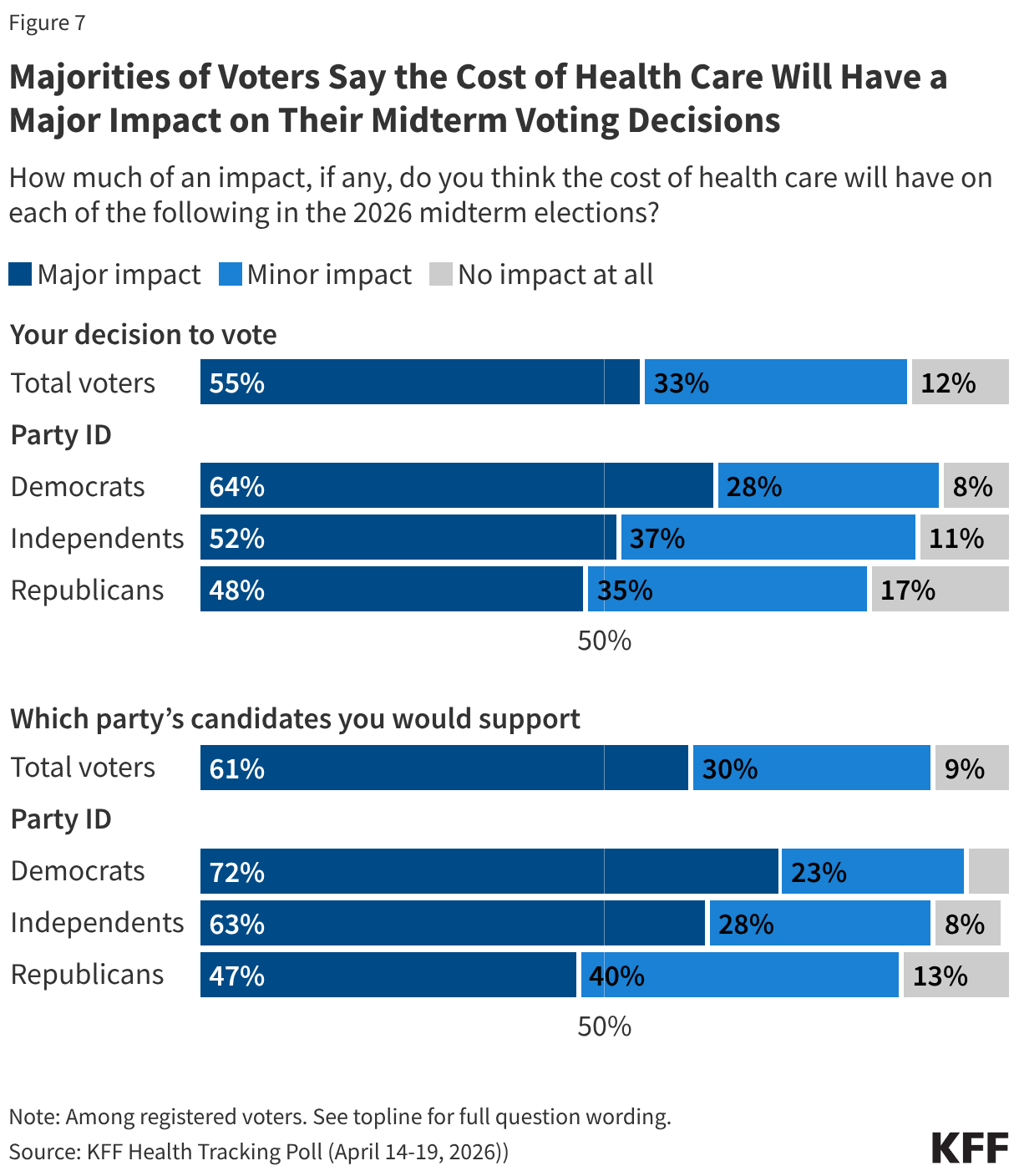
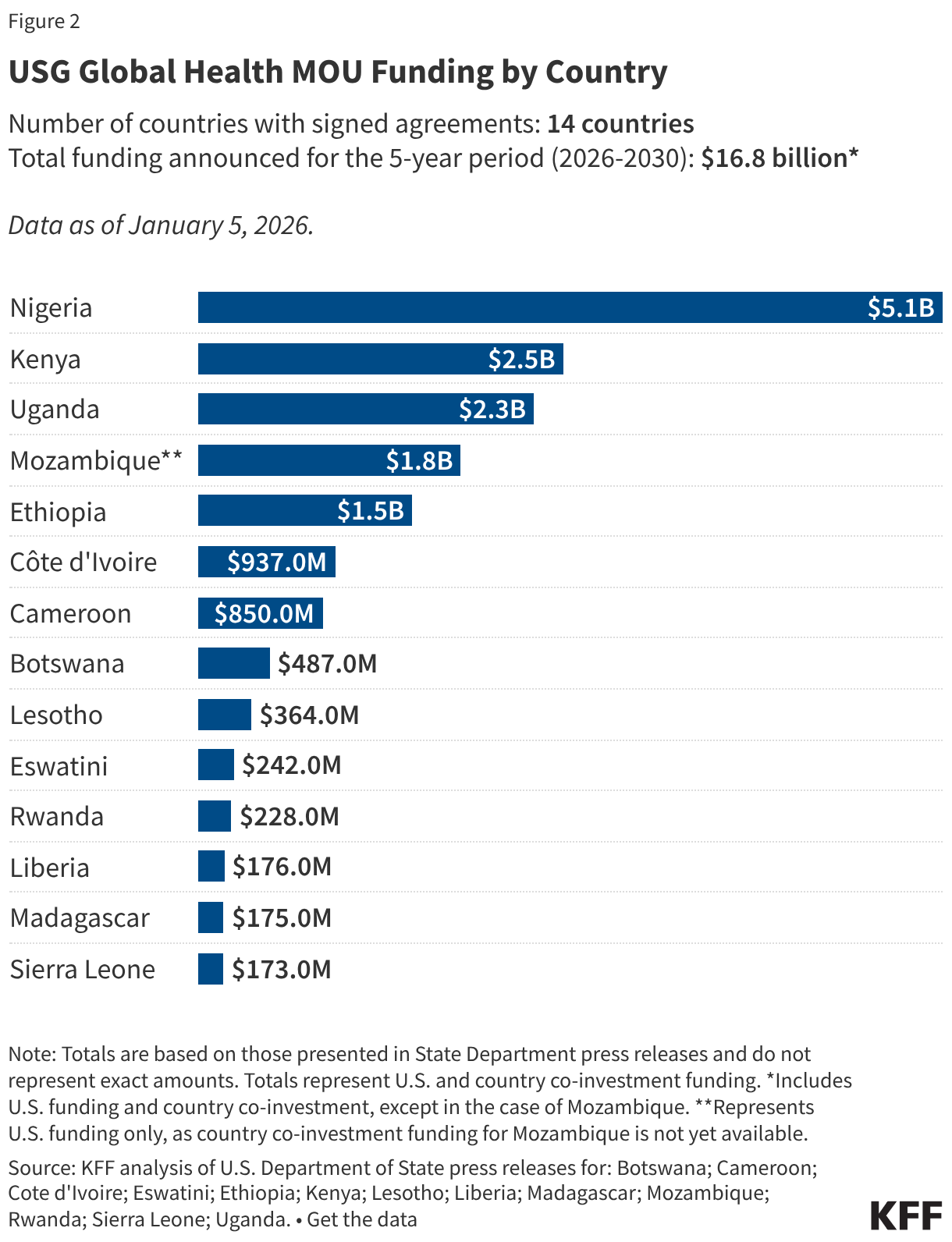
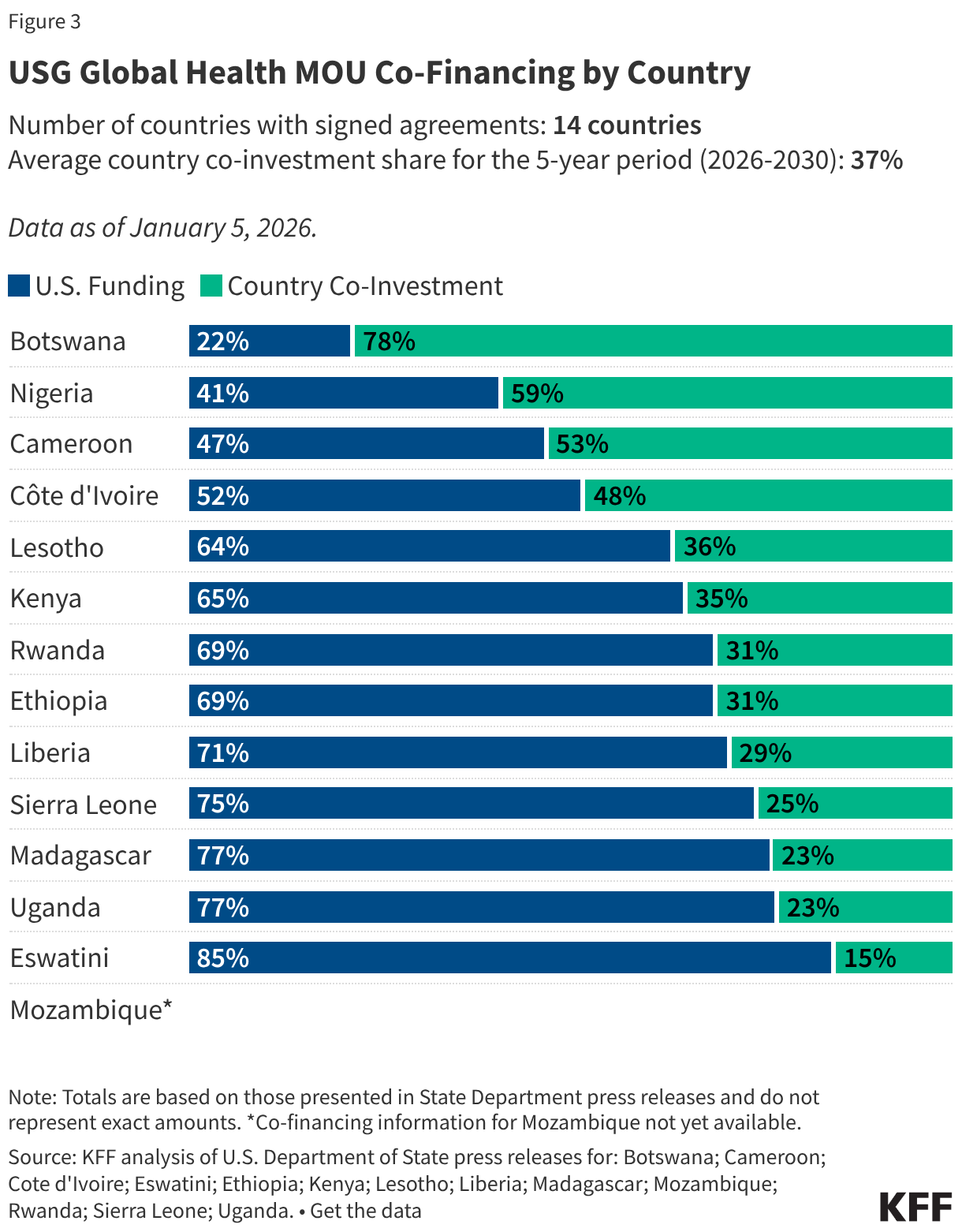
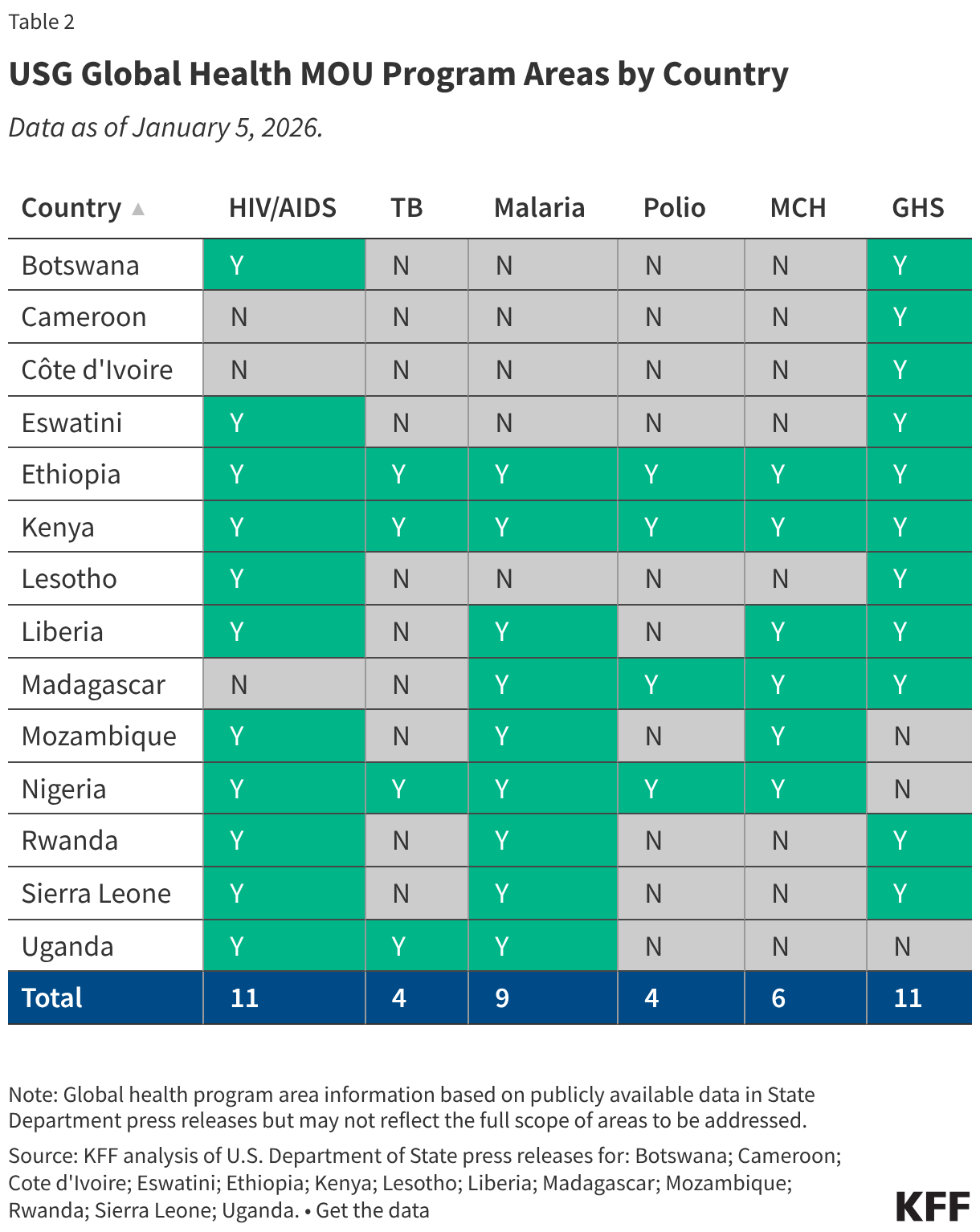
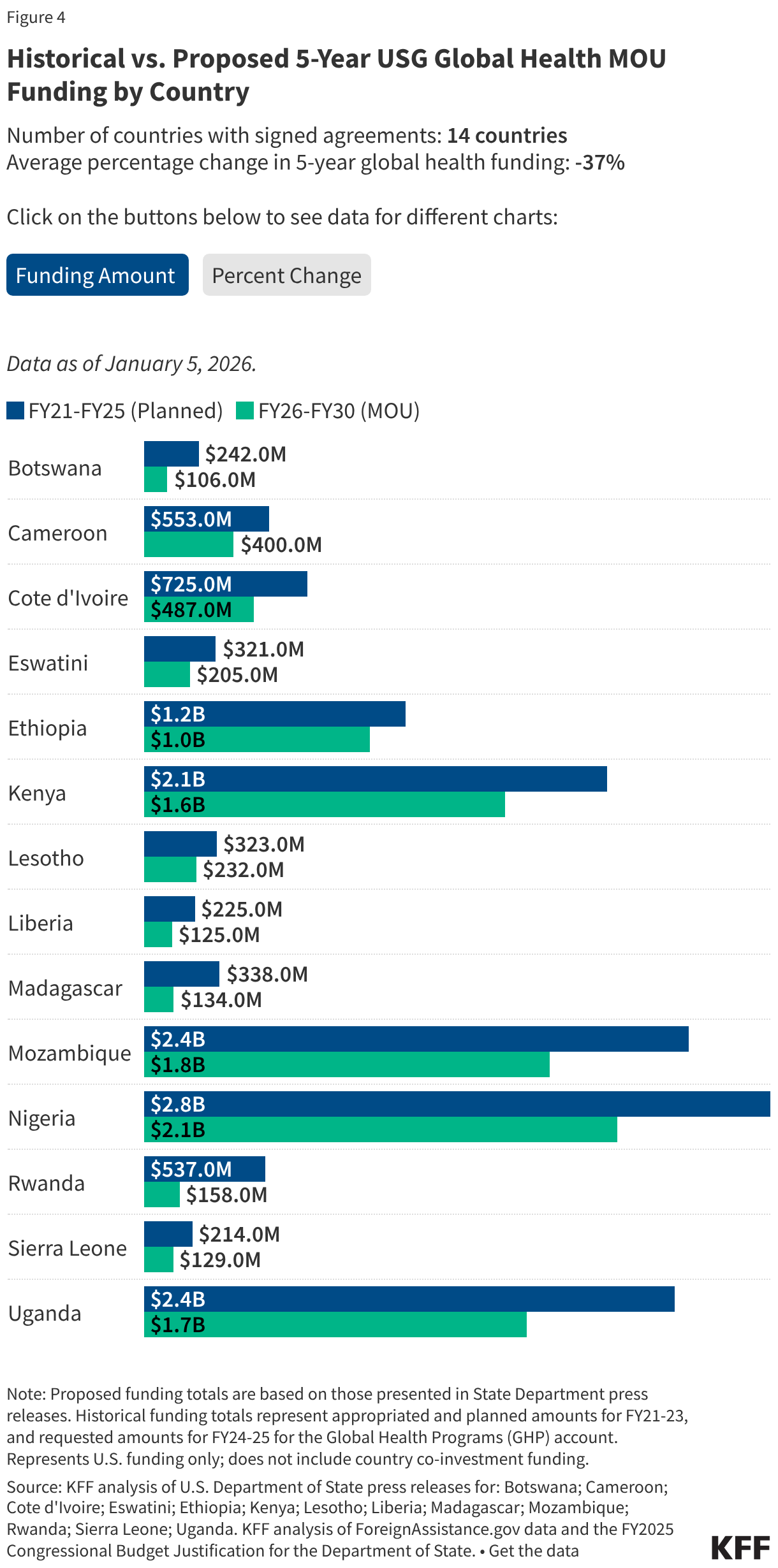
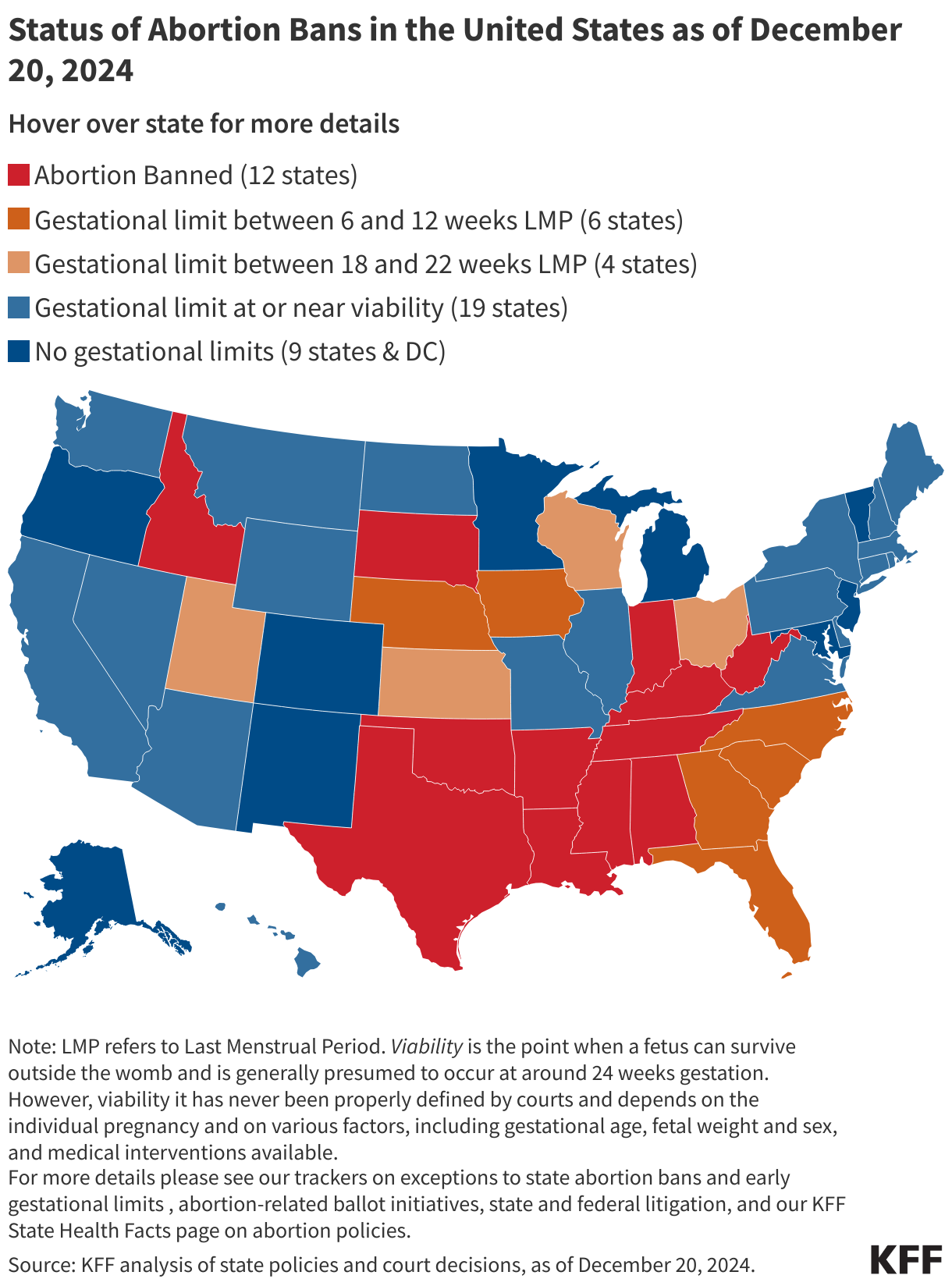
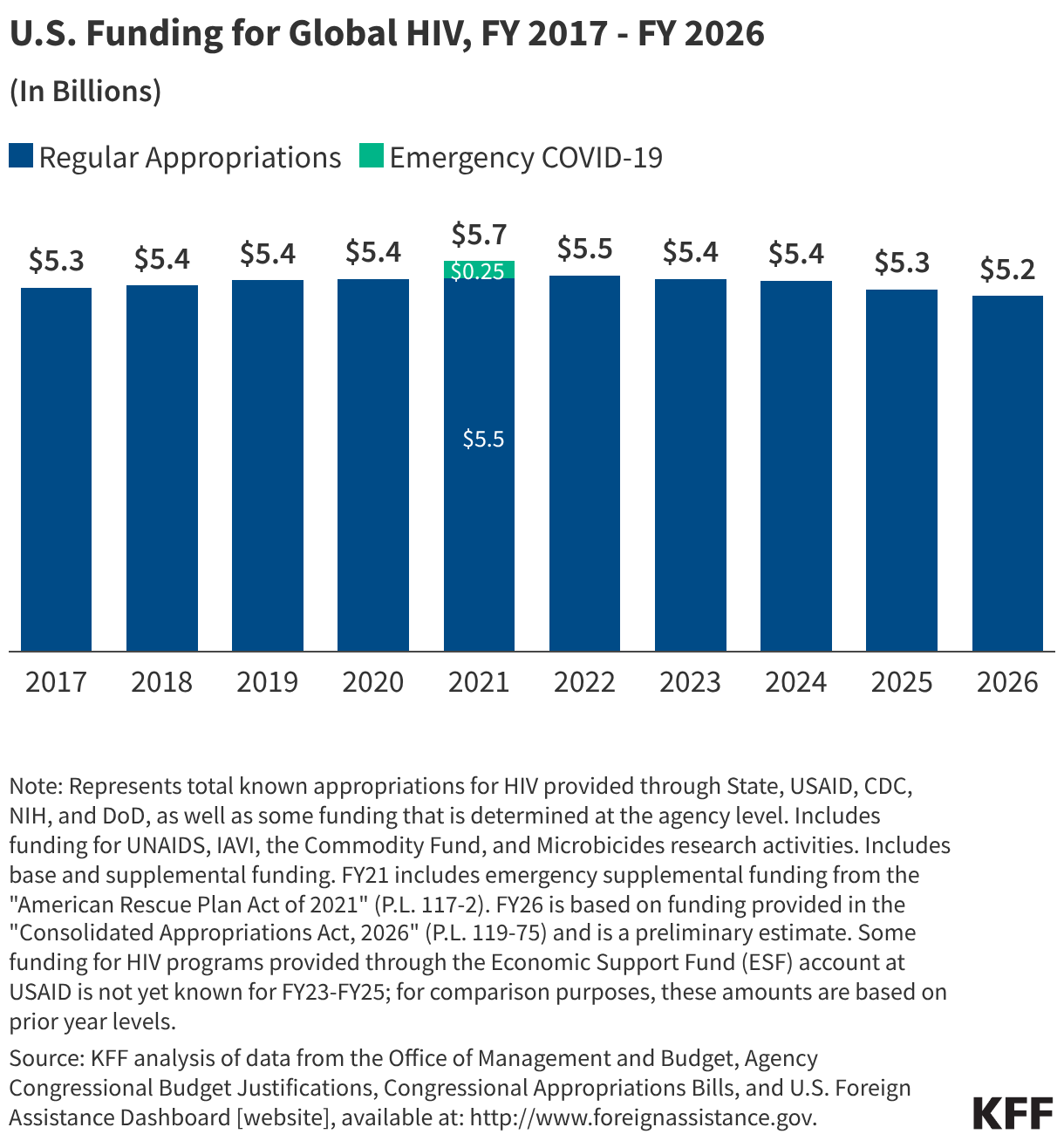
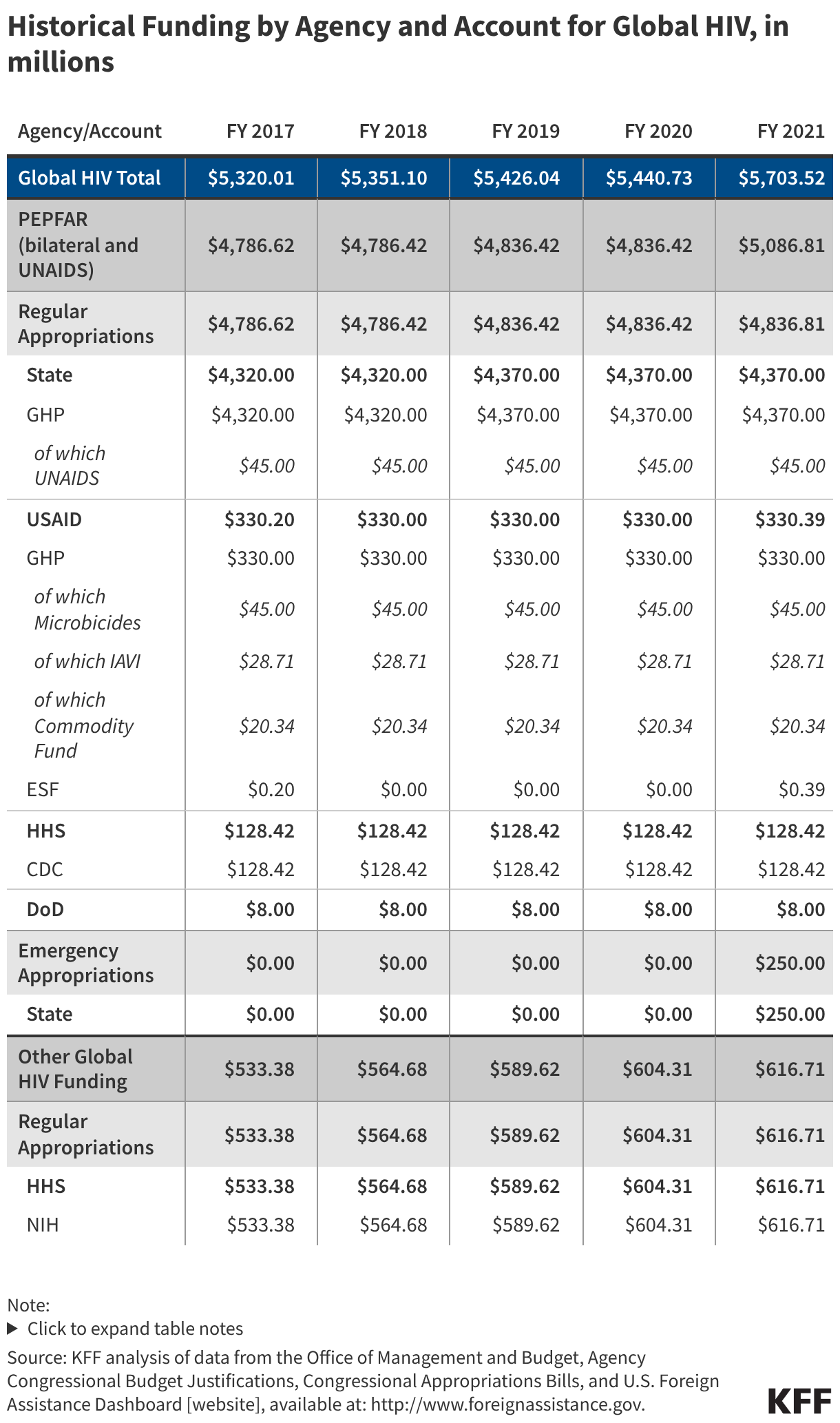
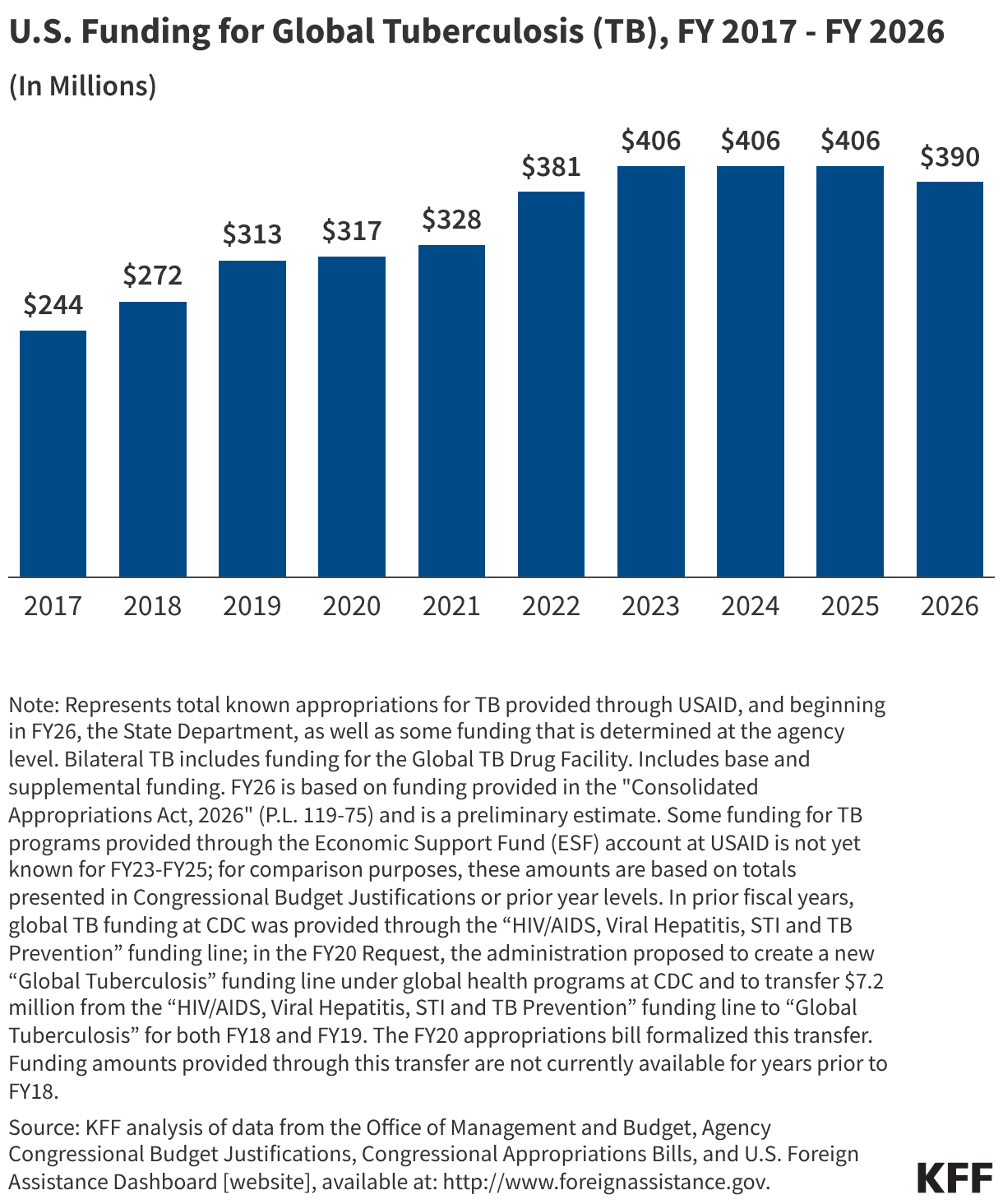
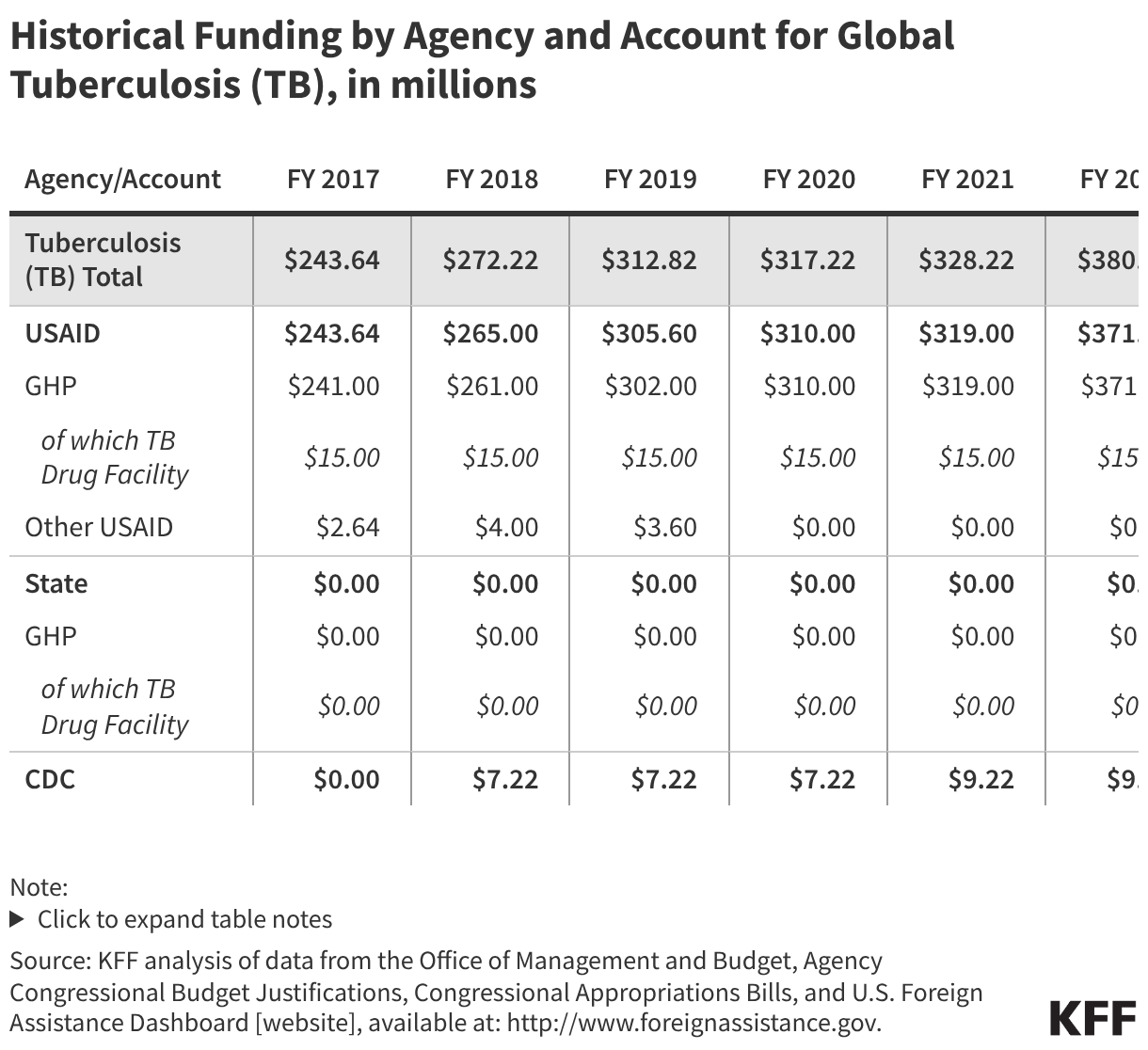
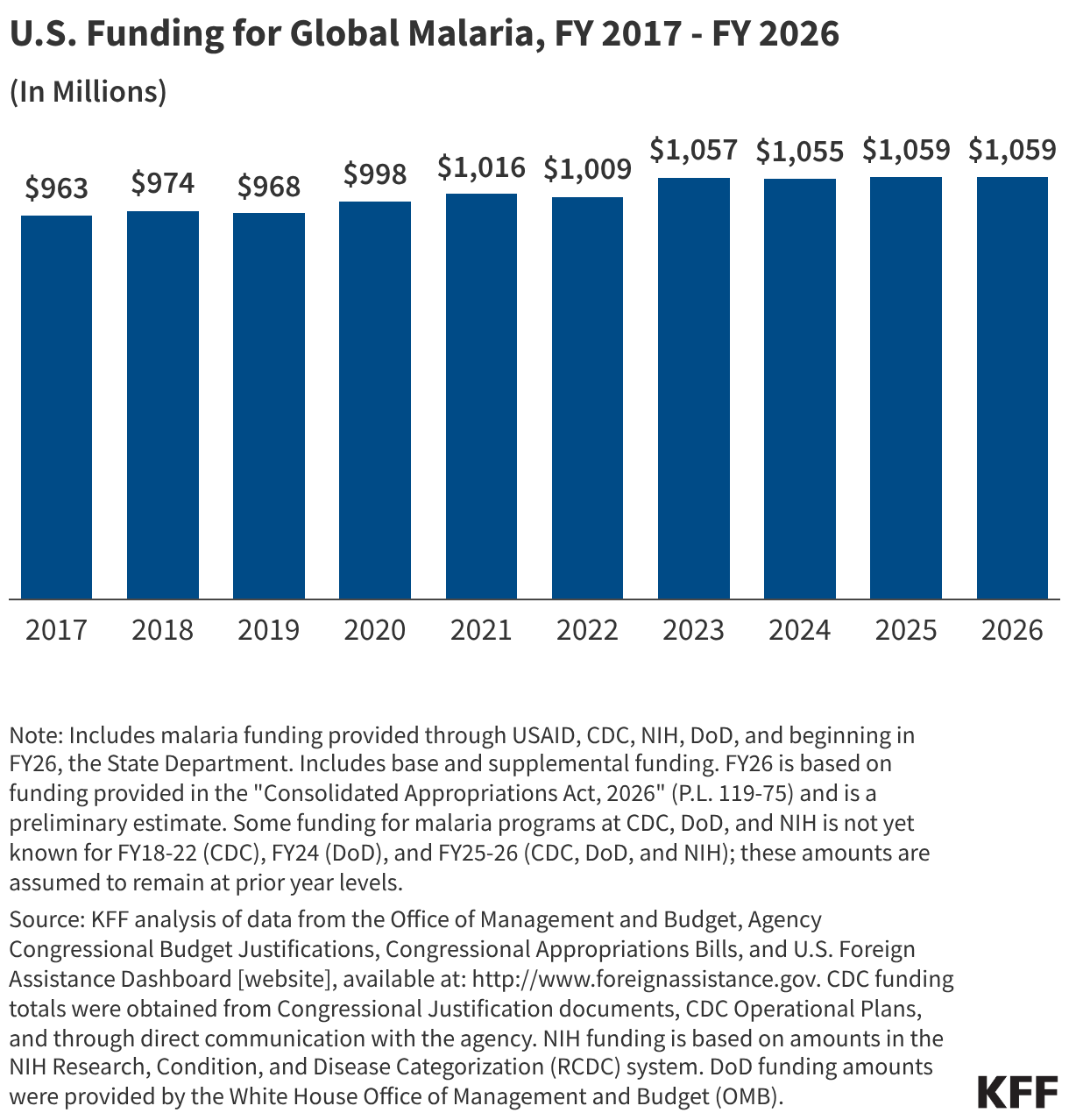
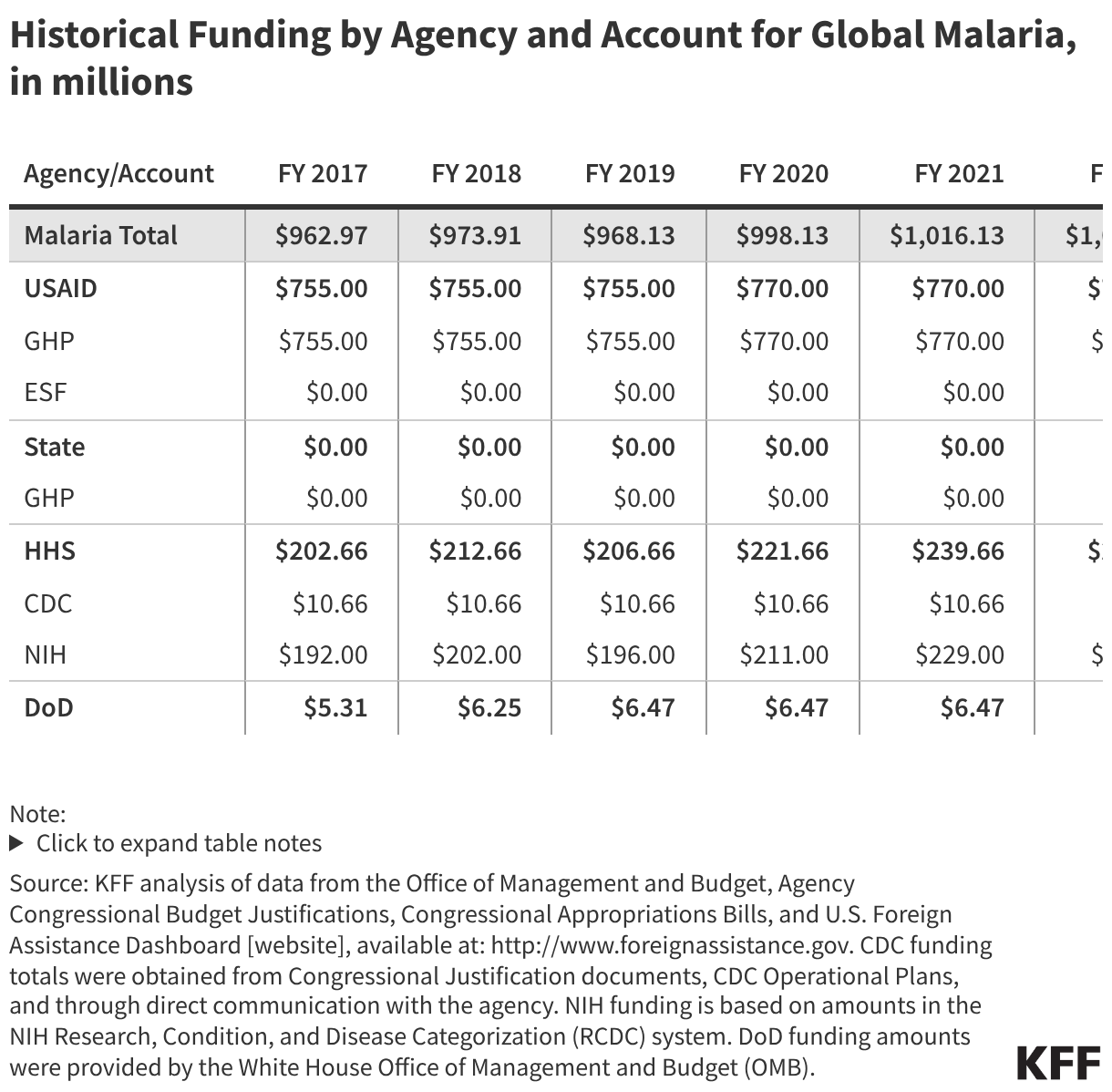
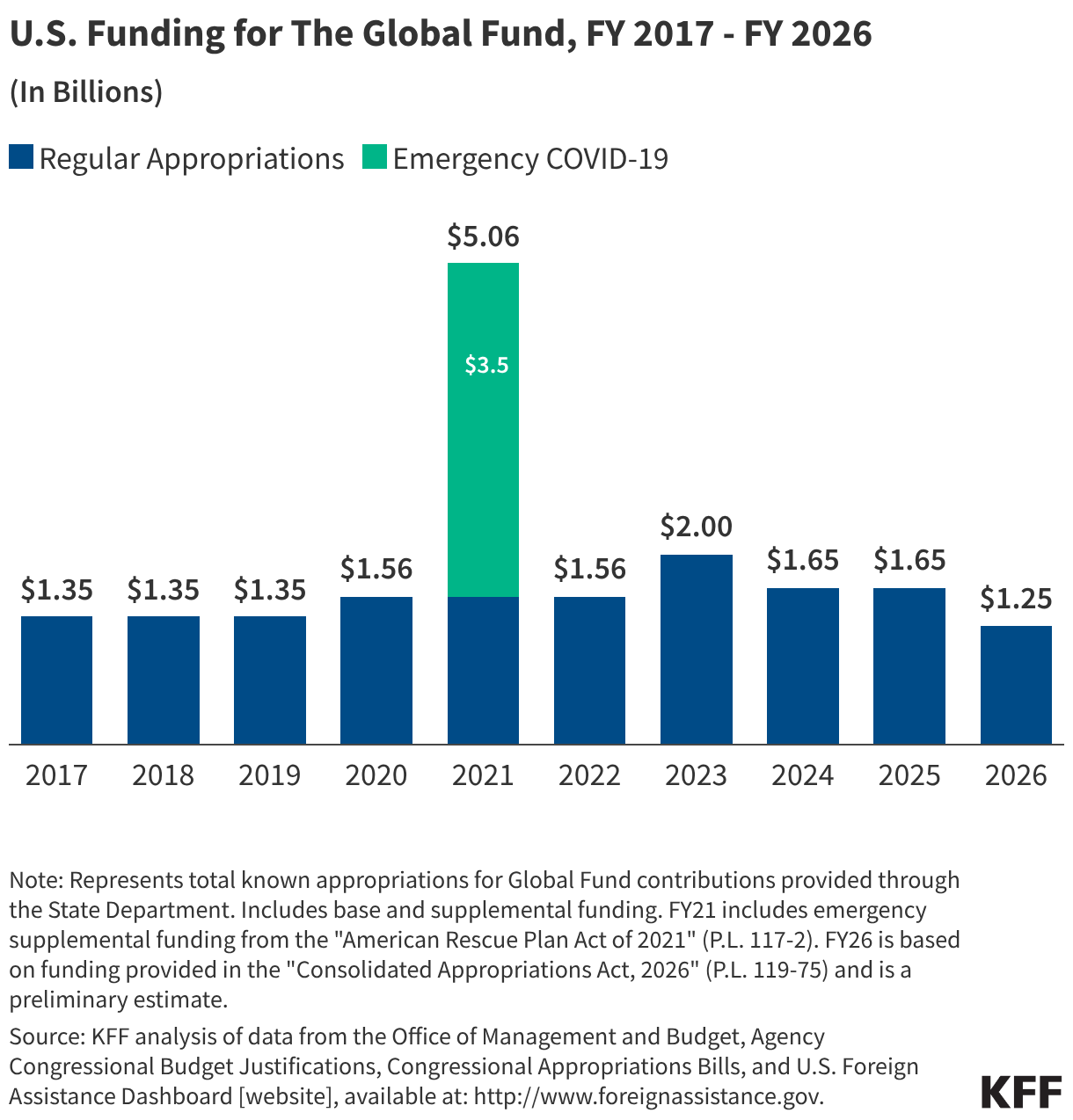
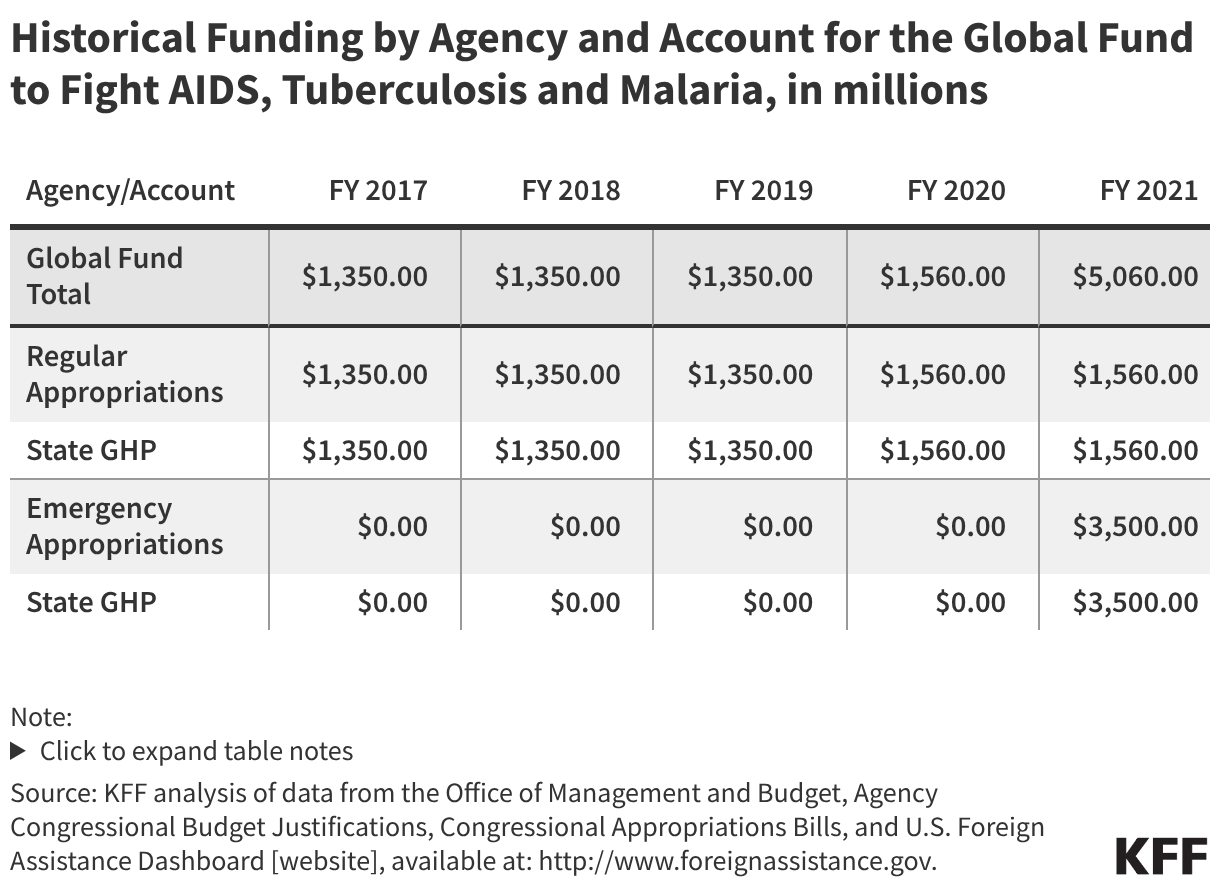
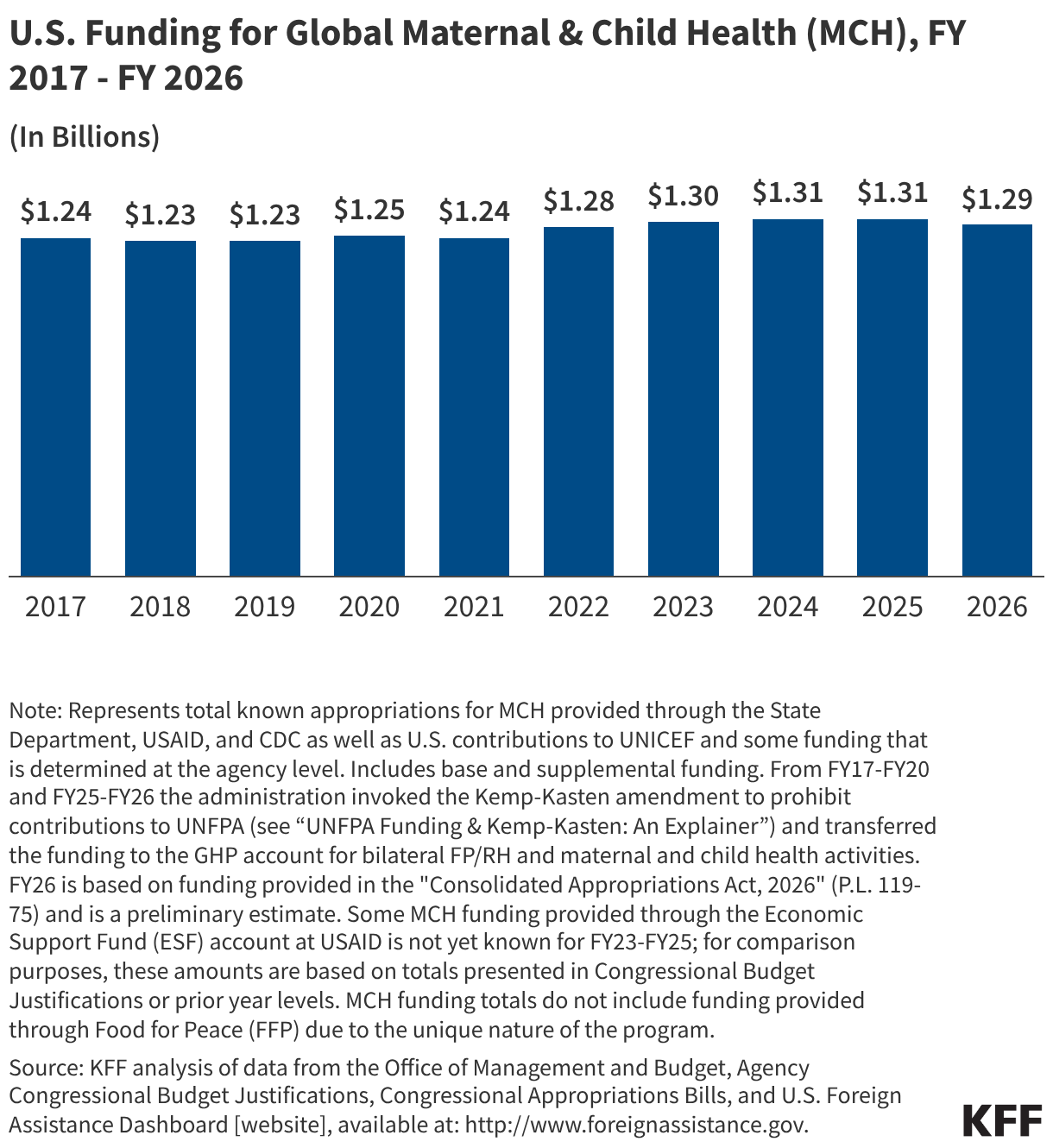
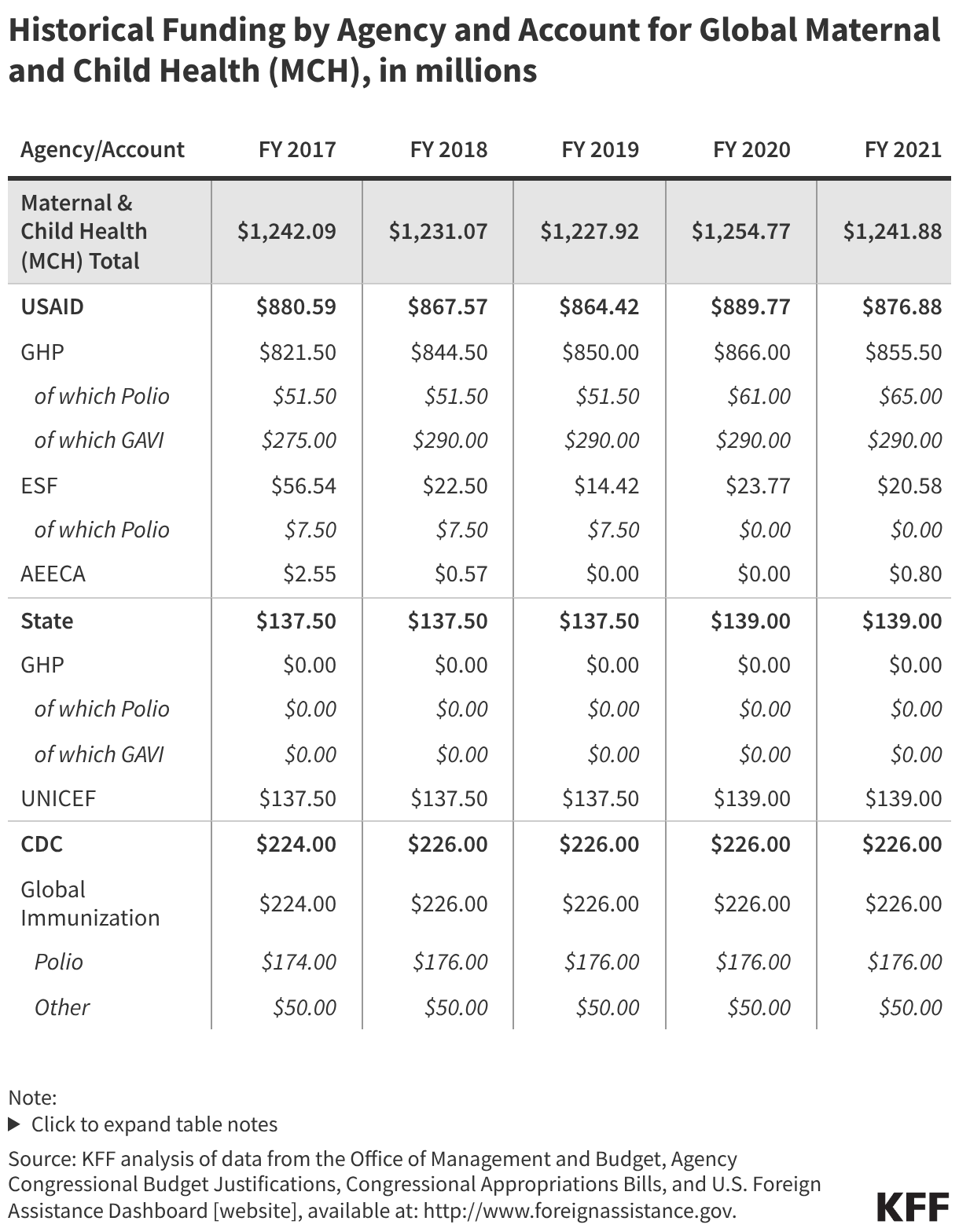
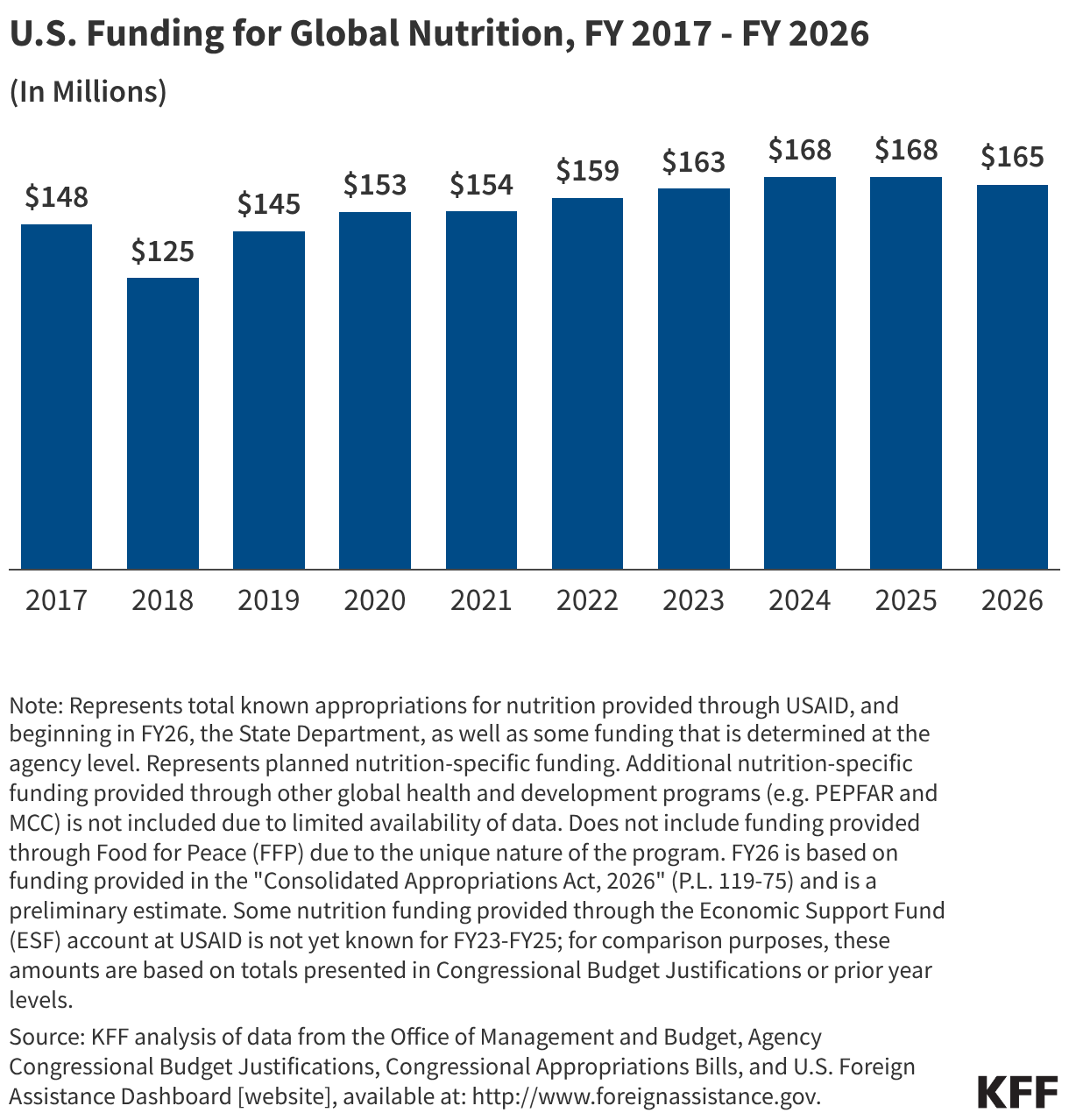
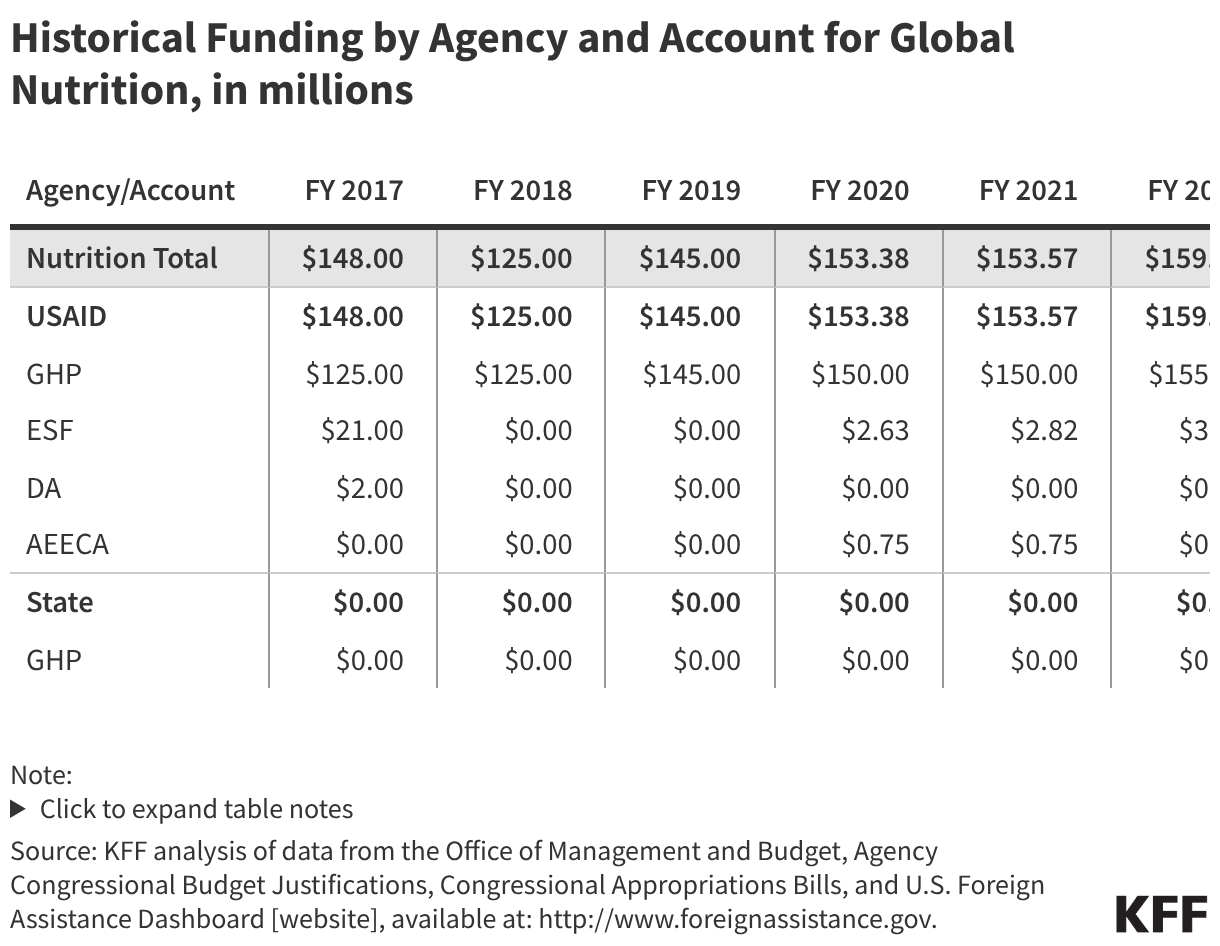
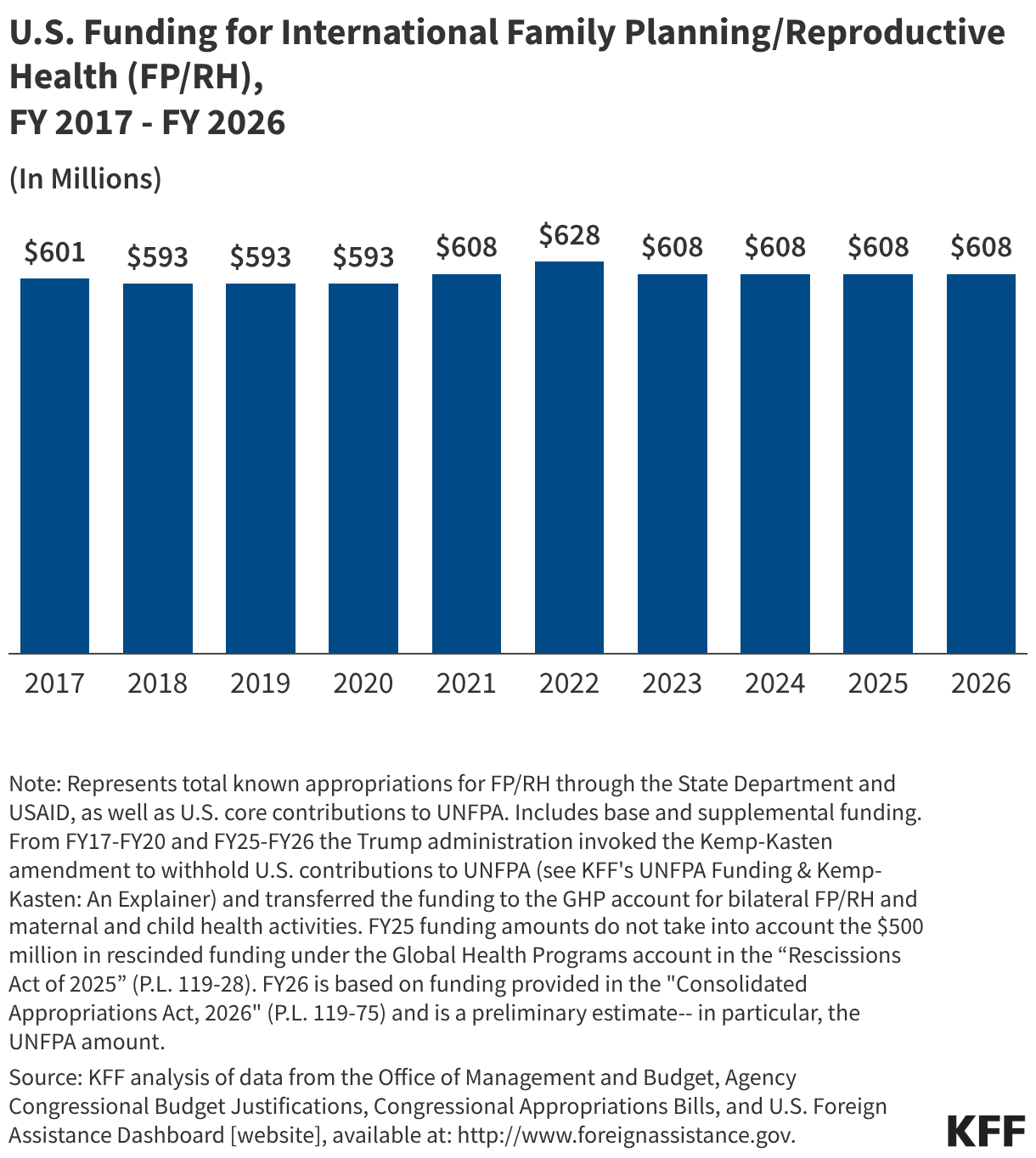
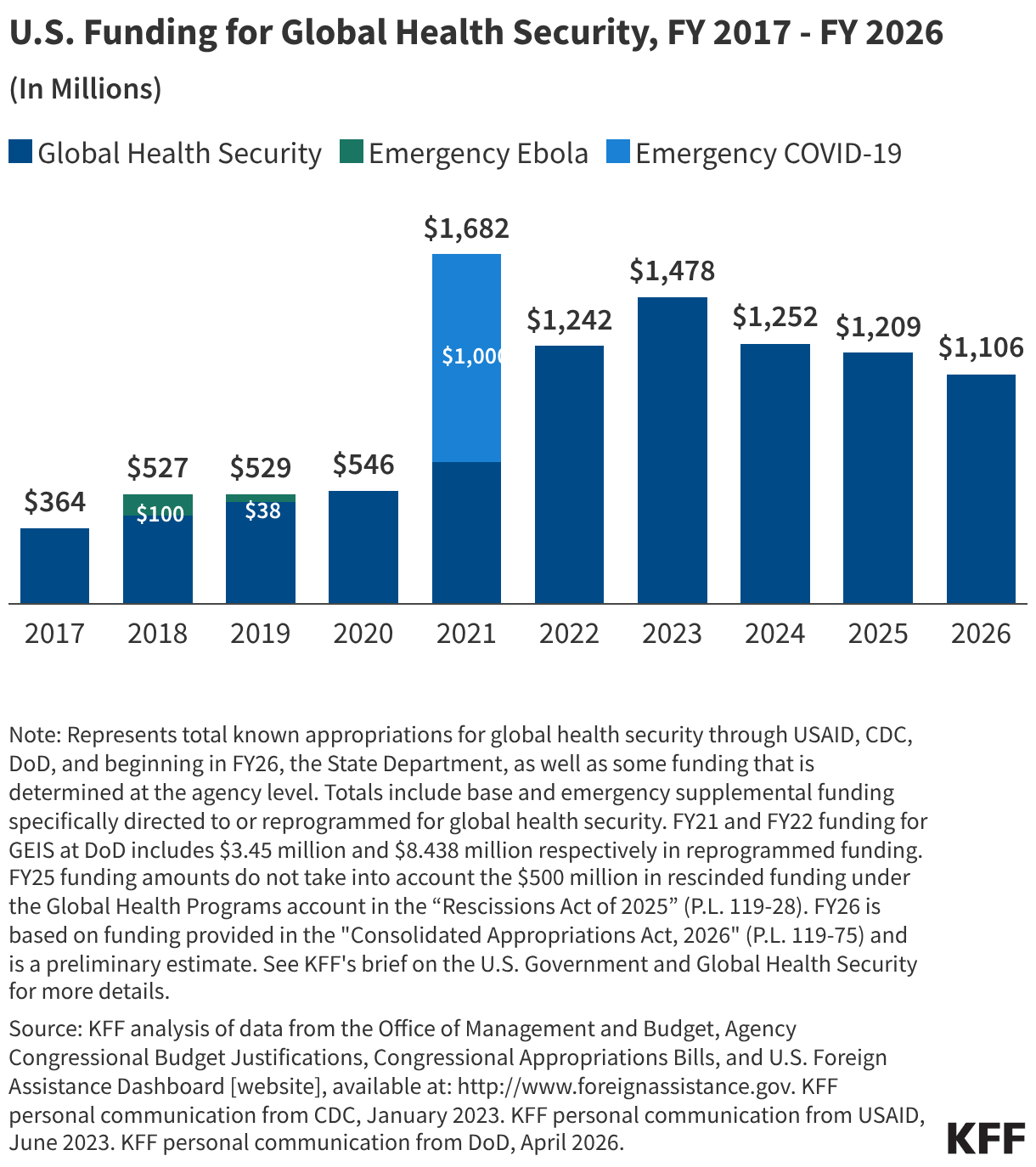
Since the start of the second Trump administration, the President’s Emergency Plan for AIDS Relief (PEPFAR), the U.S. global HIV/AIDS program credited with saving 26 million lives, has undergone significant changes and disruptions as part of a broader foreign aid review. Recent changes include: a temporary stop work order and eventual limits to what services could be continued; the cancellation of numerous PEPFAR awards; and a reorganization of U.S. global health programs, including the launch of a new “America First Global Health Strategy” which is anchored to bilateral agreements with countries, a focus on frontline commodities and services, and a shift from disease-specific programming to a more integrated approach. While modeling estimates and field surveys have provided some information about the potential impact of these changes and disruptions, there has been limited data available for such assessments. PEPFAR’s flagship data platform has historically posted financial and program level results, including from PEPFAR’s Monitoring, Evaluation, and Reporting (MER) system (MER was launched more than a decade ago). However, the data platform was temporarily removed in early 2025 and, when restored, it did not include any program data from FY 2025.
On April 17, 2026, the State Department released Fiscal Year (FY) 2025 fourth quarter (Q4) MER data (covering the July 1 to September 30, 2025 period), providing the first program-level data made available since the changes of last year. Data for quarters 1-3 were not released, which, per the State Department, is due to data reporting and implementation challenges due to the changes. To provide a snapshot of PEPFAR results after these changes, and given this limitation, this analysis compares PEPFAR’s FY 2025 Q4 results to Q4 results from the previous four fiscal years for a subset of MER indicators. By comparing the same time period for each fiscal year, this approach helps to capture seasonal or other reporting fluctuations that could occur. However, it does not allow for an assessment of disruptions or other changes that may have occurred for the full FY 2025 period.1
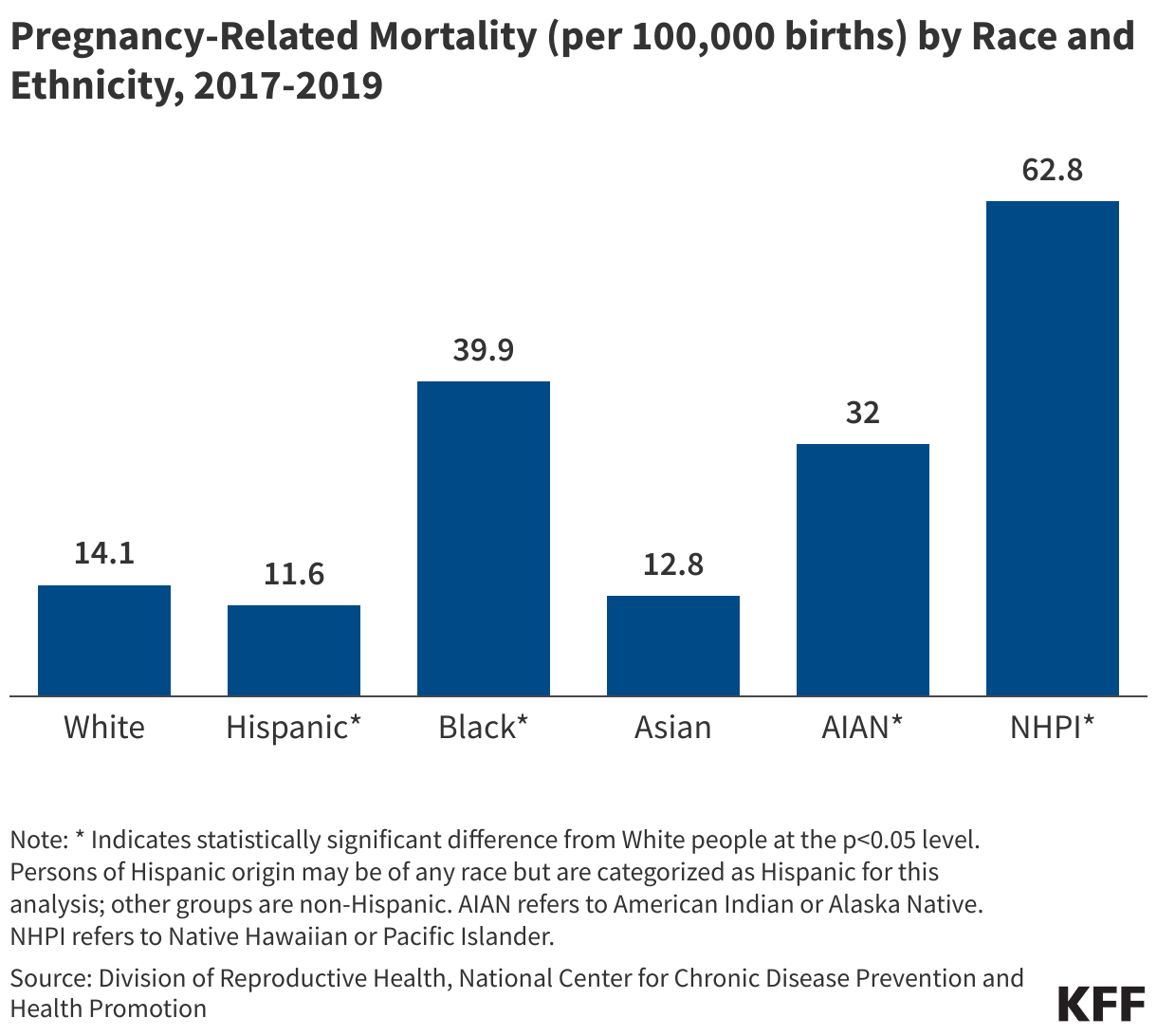
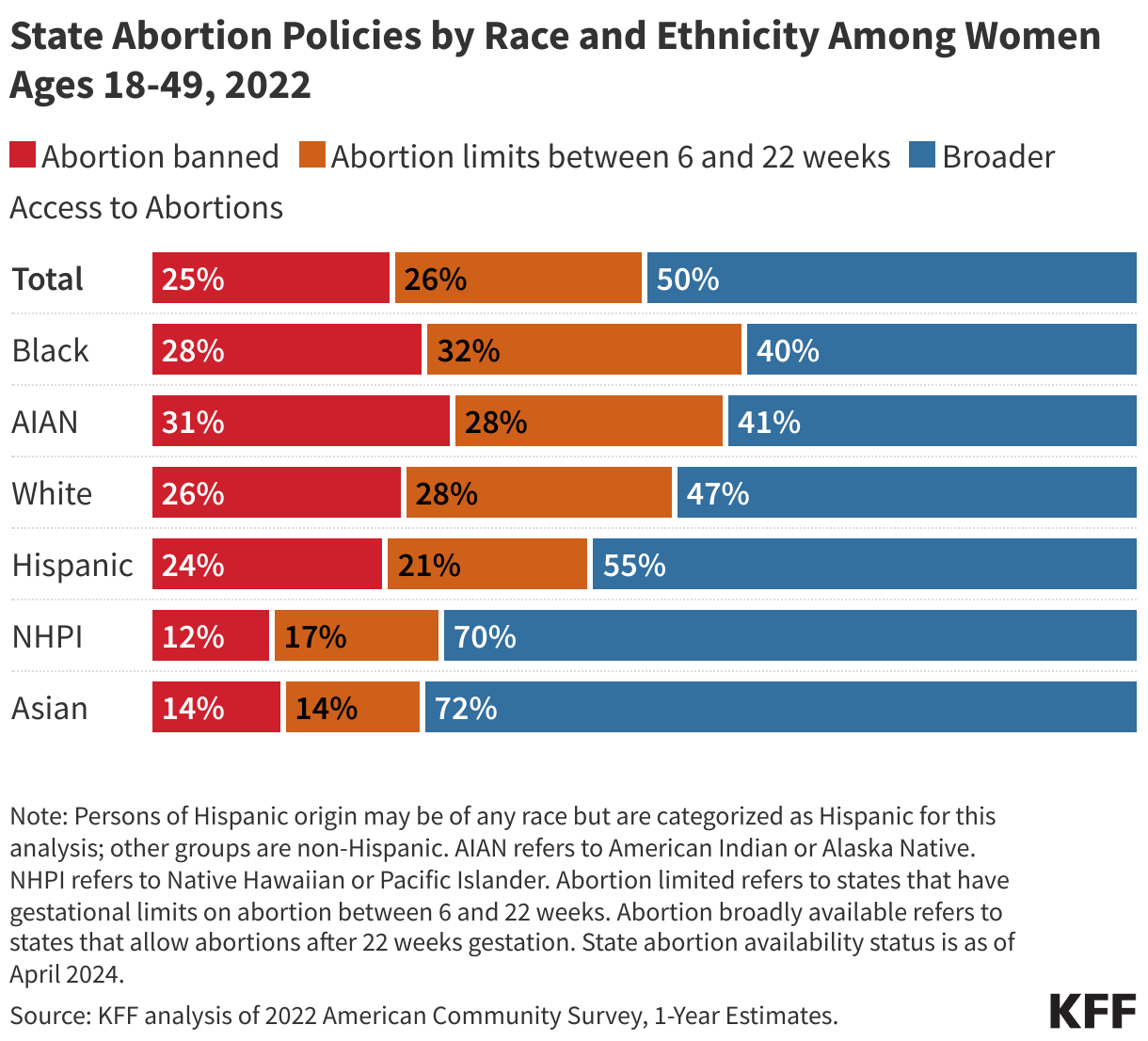
While these data are limited (see box on methodology and data limitations) and provide only a snapshot view, they nevertheless provide insights into understanding how PEPFAR is performing following changes by the Trump administration. Overall, the data show that for some indicators, progress declined in FY 2025 Q4, including support for prevention services such as pre-exposure prophylaxis (PrEP) and the DREAMS program for adolescent girls and young women, both of which were significantly scaled back by the administration. Reduced access to prevention could lead to increases in new HIV infections in the future. There was also a drop in the number of people with HIV newly enrolled on antiretroviral therapy (ART), an important measure of access. At the same time, without more complete data, there is ambiguity in some of the indicators. For example, both the number of HIV tests conducted and the number testing positive for HIV fell, which could represent an actual decline in new infections or simply a decline in access to testing. Finally, there are areas where progress has been maintained or potentially improved, including the total number of people with HIV on ART, which was stable, and an increase in the number of people living with both HIV and TB who are receiving ART. Going forward, the future of transparent PEPFAR data monitoring and reporting remains uncertain, as it’s unclear whether or not these data updates will continue to be provided given the shift in the U.S. global health strategy to country governments and from disease-specific programming to a more integrated approach. Without such data, it will be difficult to understand the implications and outcomes of these significant changes.
Findings
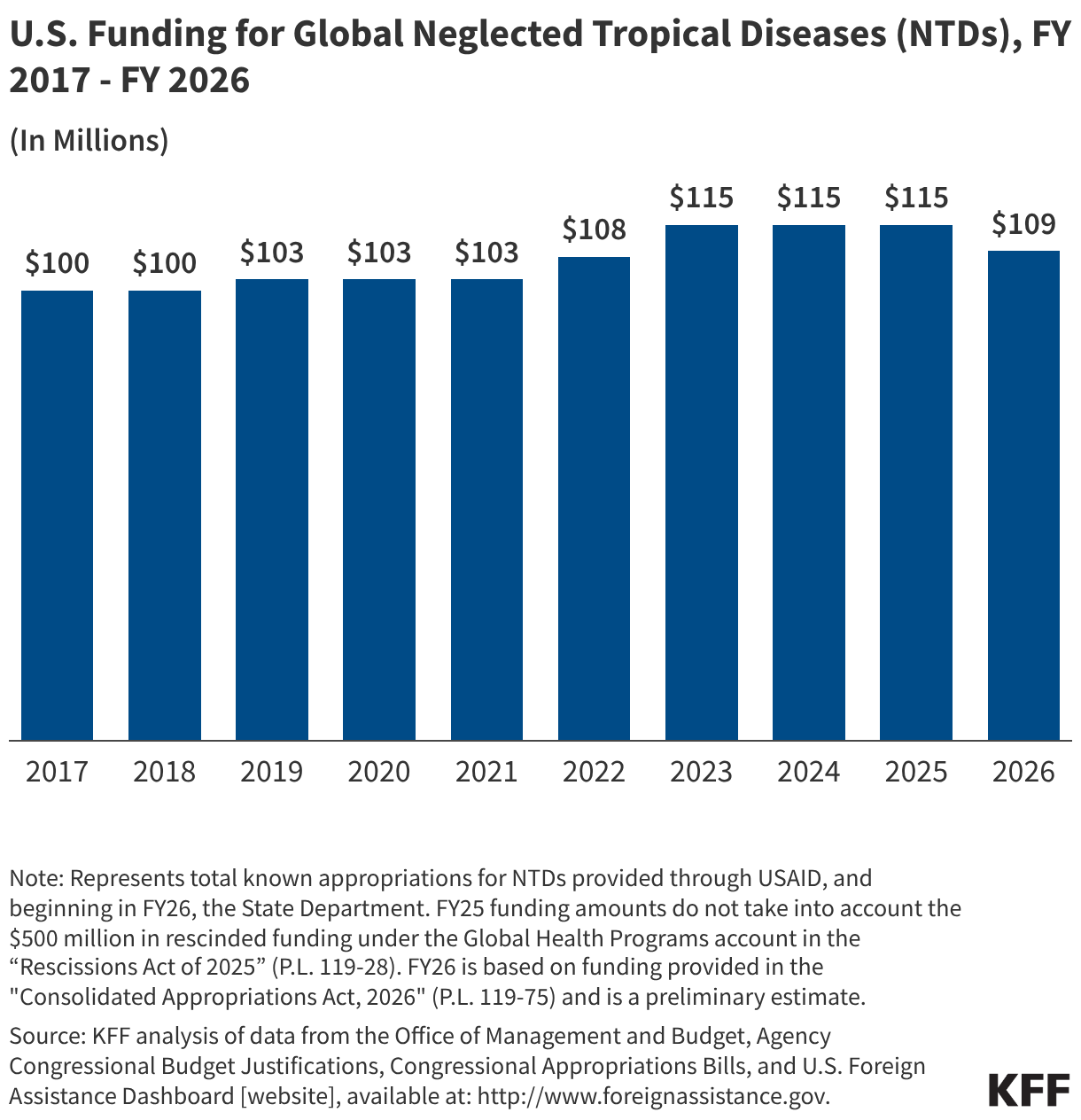
PEPFAR’s Q4 treatment results over the period were mixed. While there were some stable results, particularly for the number of people receiving ART, others dropped, such as those newly enrolled on ART.
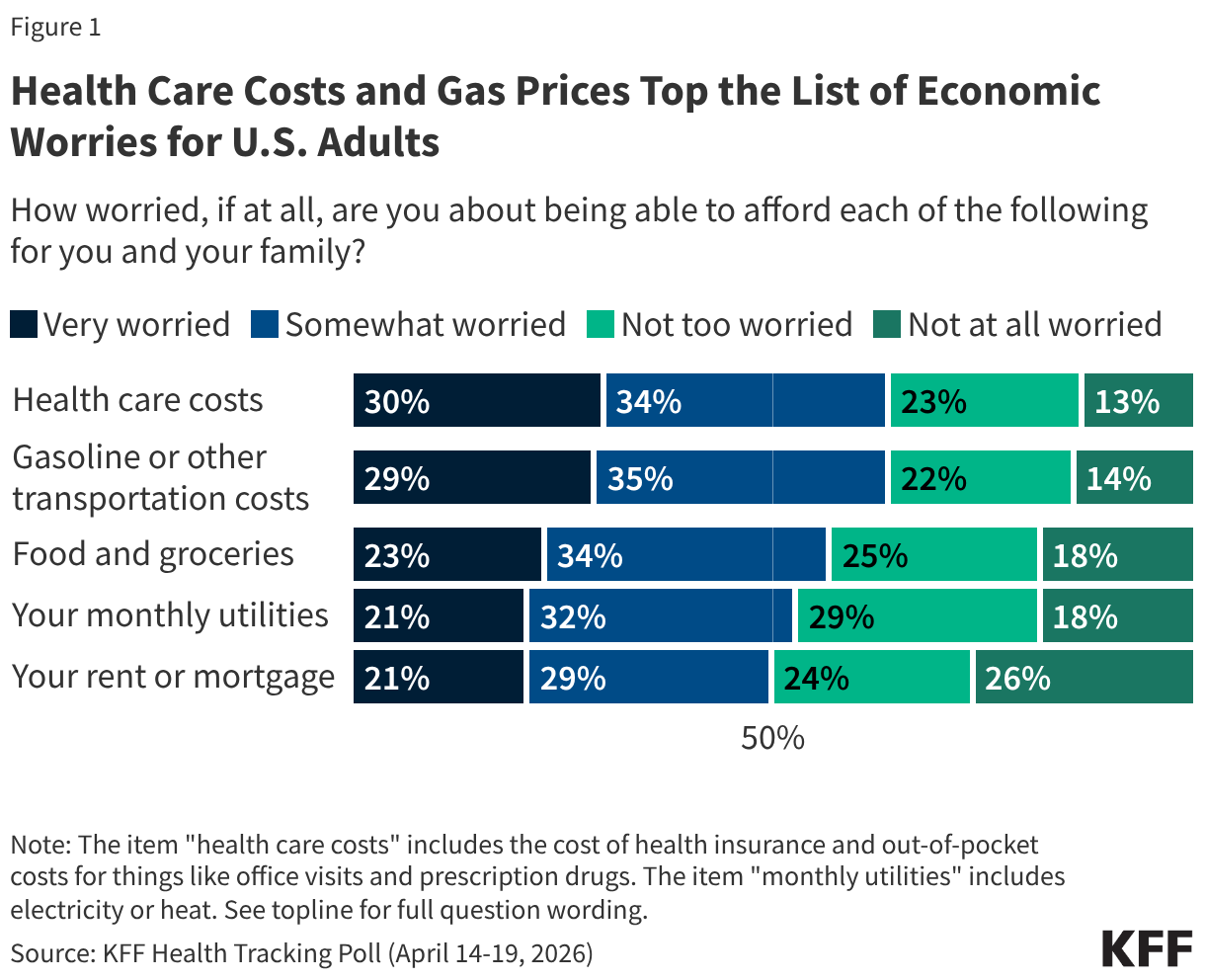
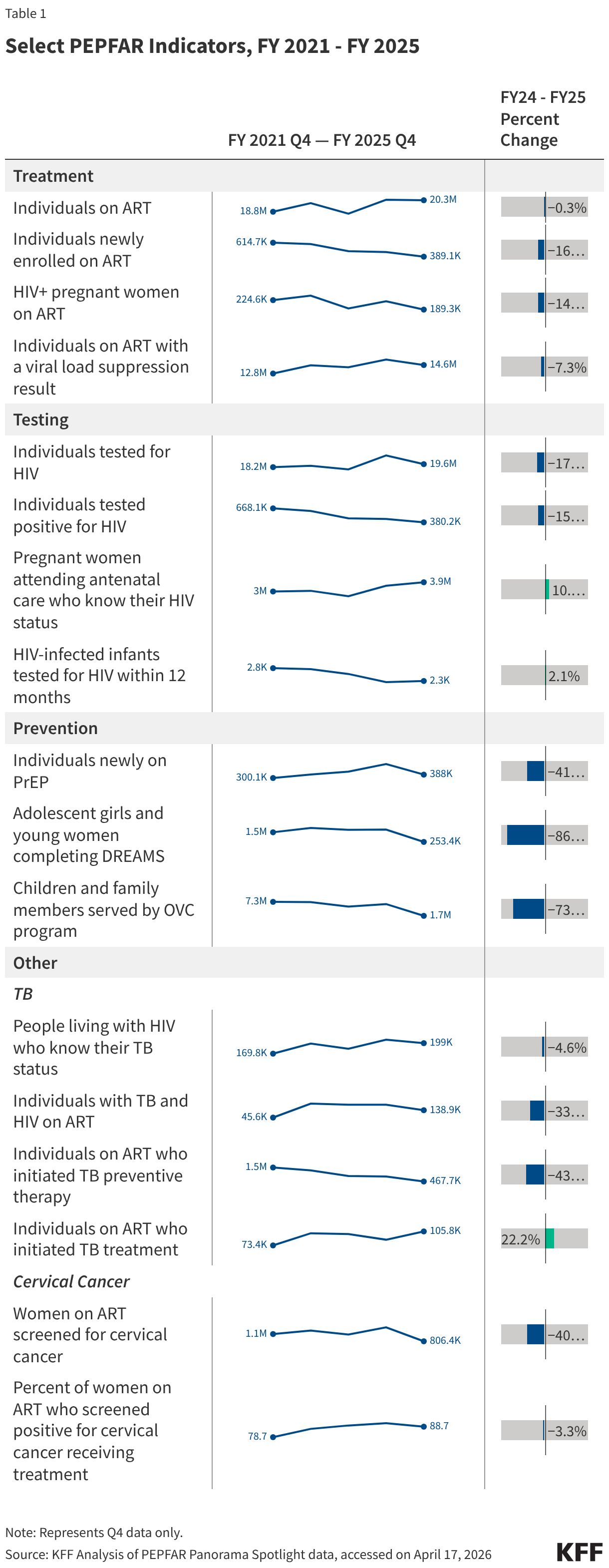
- The number of individuals with HIV on antiretroviral therapy (ART) remained relatively stable in FY 2025 Q4 compared to FY 2024 Q4 (20.3 million2 compared to 20.4 million) and was higher than the prior fiscal years.
- At the same time, the number newly enrolled on ART in FY 2025 Q4 was the lowest over the period, including 16% lower than FY 2024 Q4 (389.1K compared to 463.5K). This is part of a broader decline over the past five years but one of the steepest year-to-year quarter declines.
- Similarly, the number of pregnant women testing positive for HIV and receiving ART in FY 2025 Q4 was also the lowest over the period, including a 14% decline compared to FY 2024 Q4 (189.3K compared to 220.7K).
- Finally, individuals on ART with documented viral load suppression (VLS) declined in FY 2025 Q4 by 7% compared to FY 2024 Q4 (14.6 million compared to 15.8 million), but was higher than the prior fiscal years.
HIV testing and diagnostic results were mixed in FY 2025 Q4 compared to FY 2024 Q4, although there were fluctuations over the five-year period.
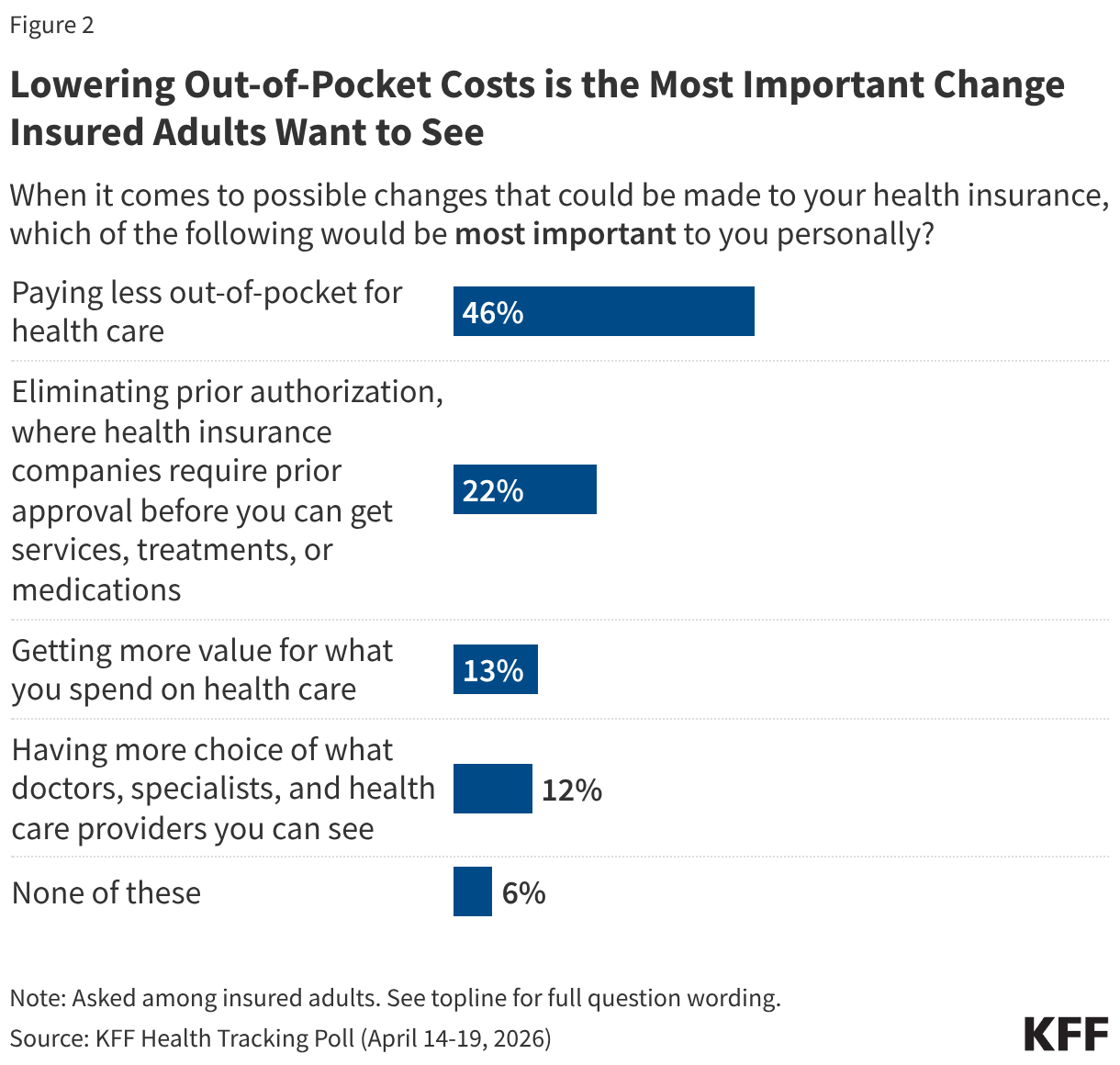
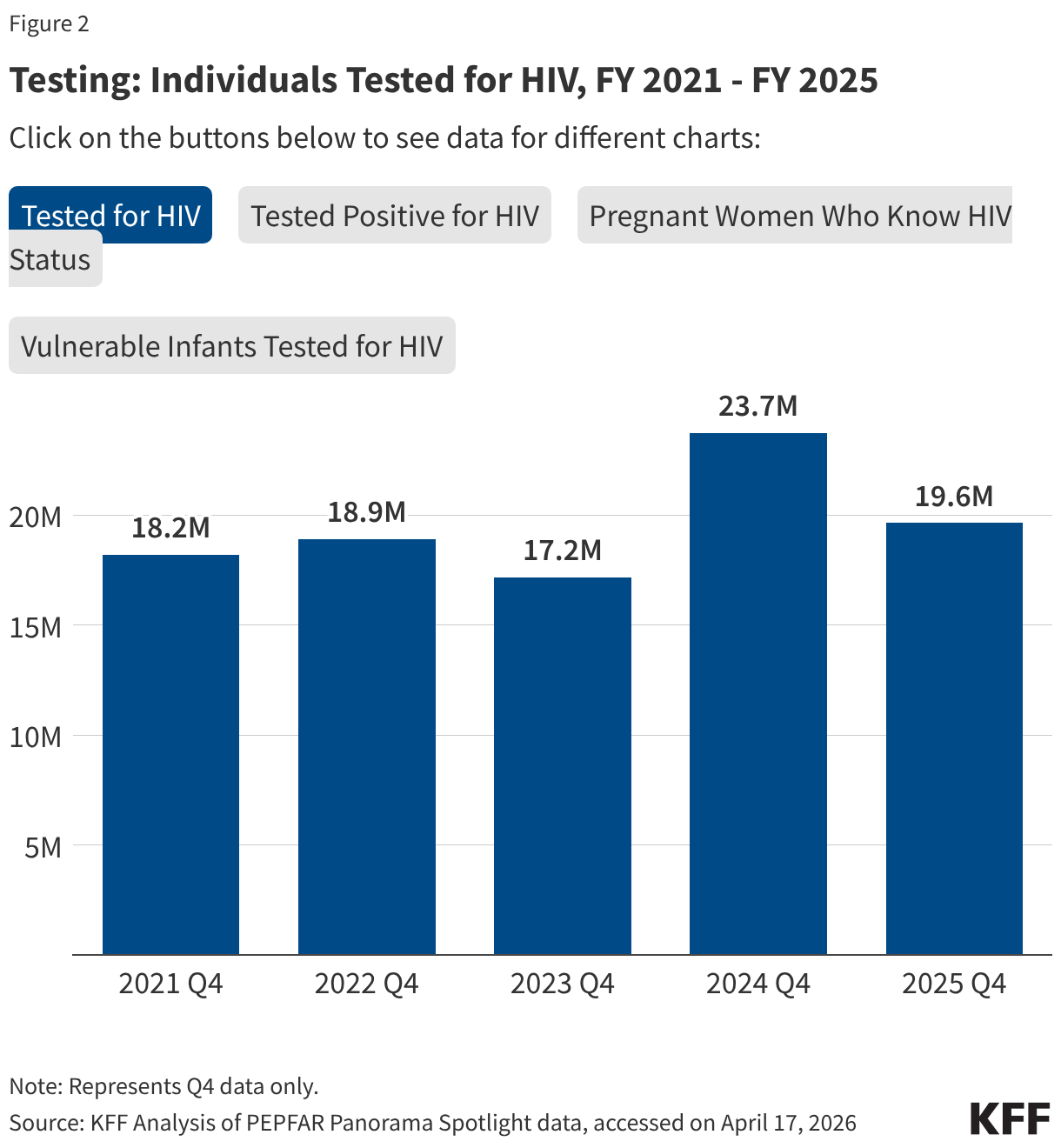
- Both the number of individuals tested for HIV and the number testing positive fell. The number of individuals tested fell by 17% in FY 2025 Q4 compared to FY 2024 Q4 (19.6 million compared to 23.7 million), though was still higher than in prior years. The number testing positive similarly fell by 15% in FY 2025 Q4 compared to FY 2024 Q4 (380.2K compared to 449.8K), continuing a downward decline and reaching the lowest number over the period.
- Despite these drops, the number of pregnant women attending antenatal care who know their HIV status in FY 2025 Q4 was highest over the period, including a 10% increase compared to FY 2024 Q4 (3.9 million compared to 3.6 million).
- In addition, the number of HIV-infected infants with an HIV diagnostic sample collected within the first year increased slightly in FY 2025 Q4 compared to FY 2024 Q4 (2,272 compared to 2,225) but was lower than prior years.
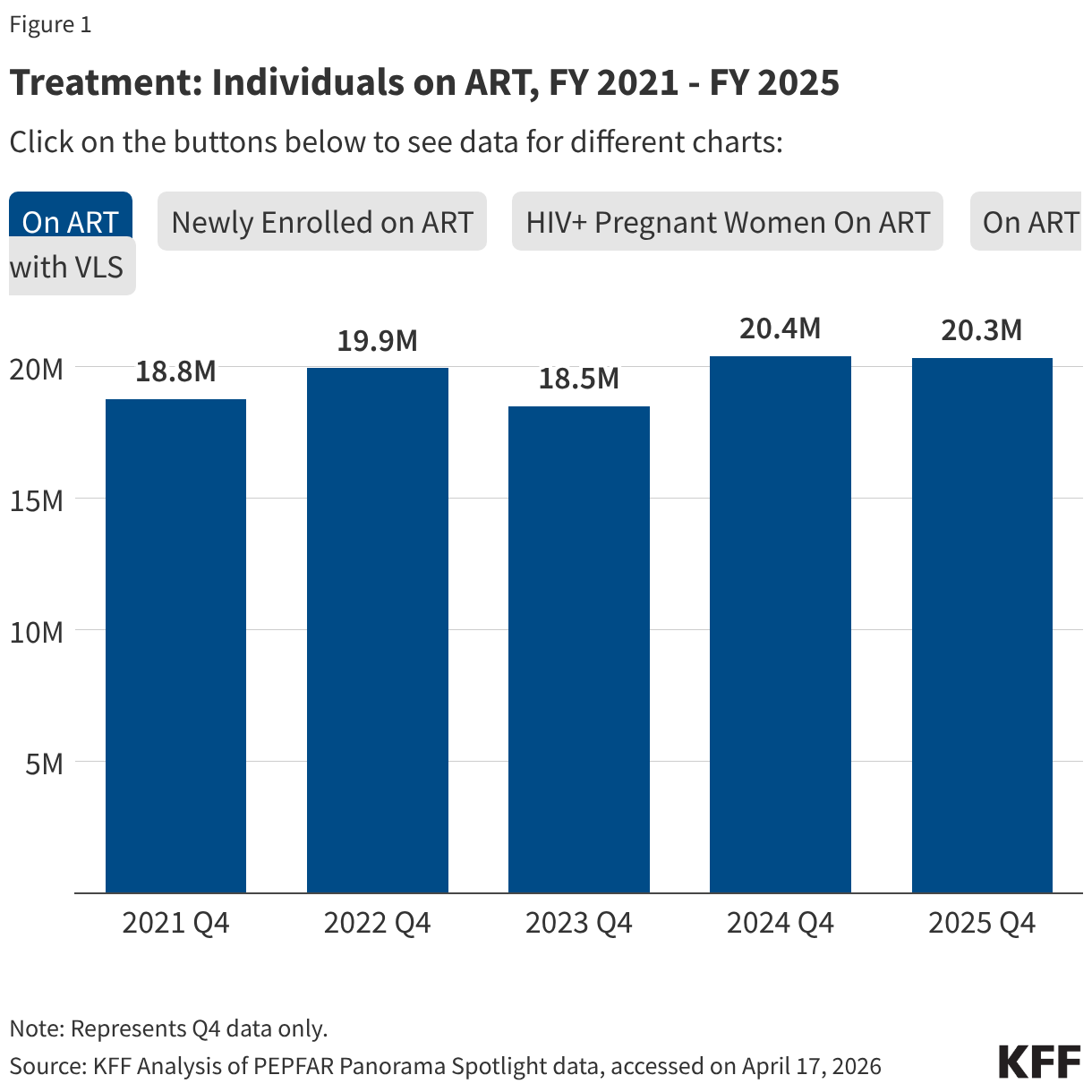
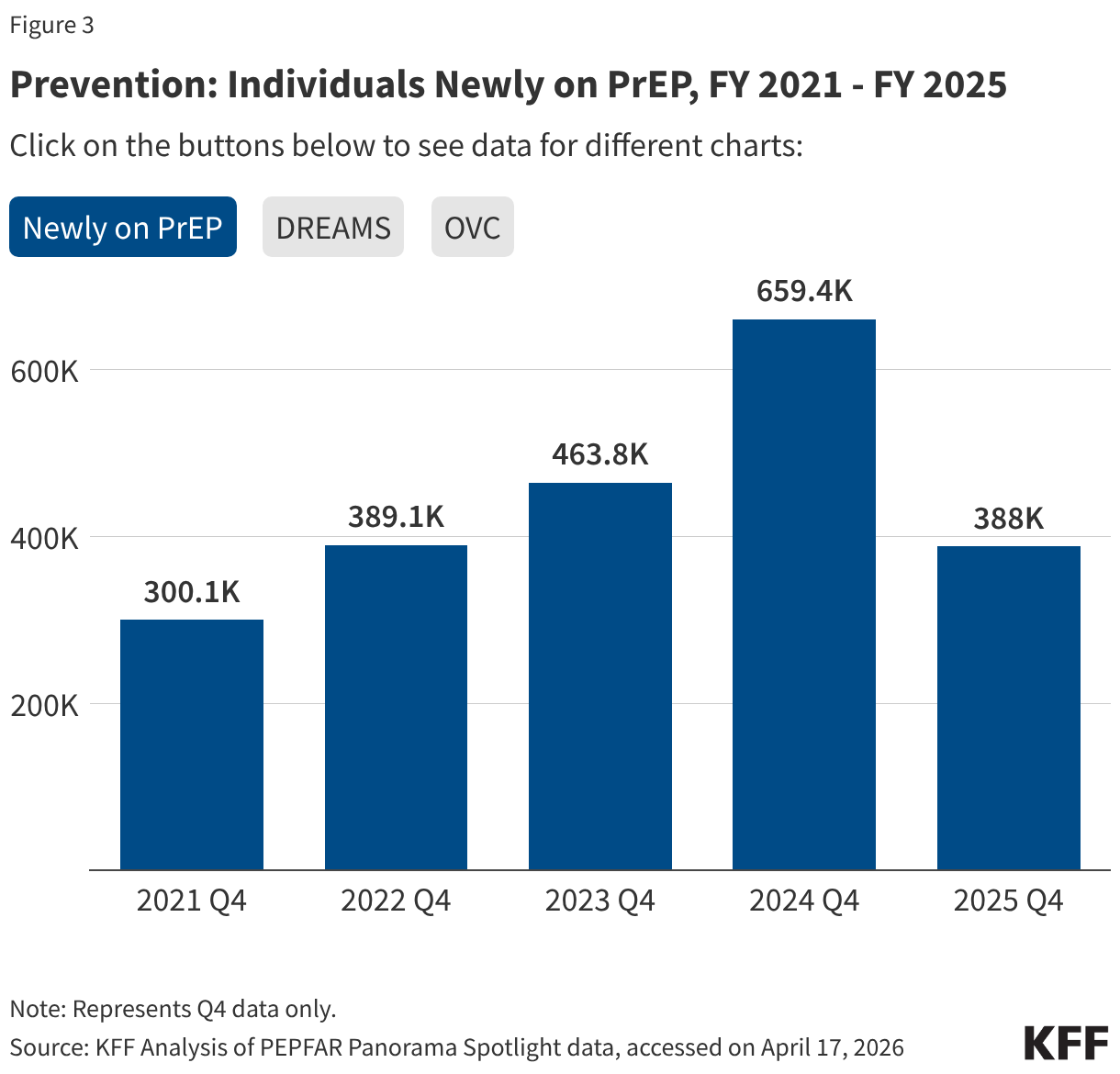
Key PEPFAR prevention results saw large declines, including for PrEP.
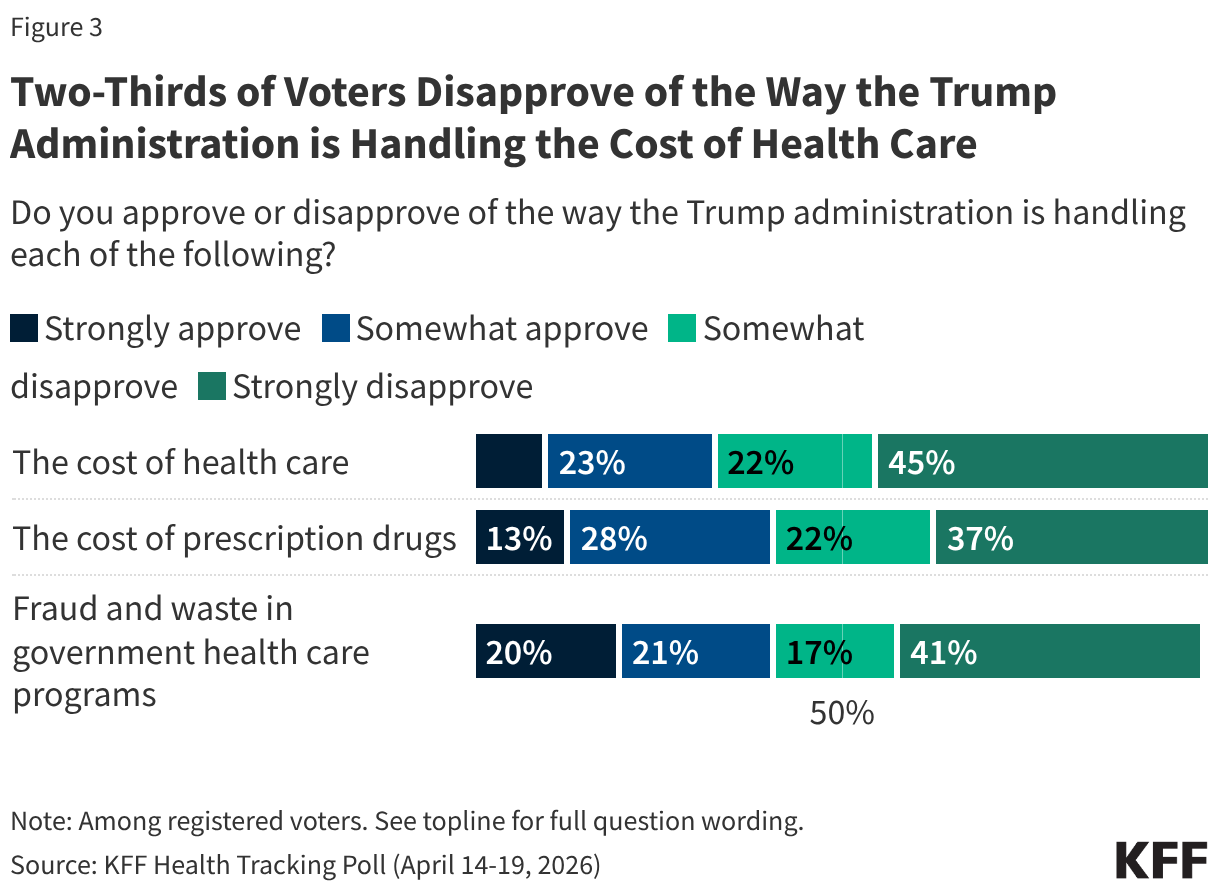
- The number of individuals who newly initiated PrEP declined by 41% in FY 2025 Q4 compared to FY 2024 Q4 (388K compared to 659.4K), falling to FY 2022 levels. Access to PrEP with PEPFAR support had increased steadily in recent years, before this drop.
- Also declining significantly was the number of adolescent girls and young women (AGYW) who completed the DREAMS package of prevention services in FY 2025 Q4 compared to FY 2024 Q4 (a drop of 86%, from 1.9 million to 253.4k), its lowest level over the period and less than a quarter of those served in the prior years analyzed. DREAMS had been a major PEPFAR initiative to address the drivers of high HIV incidence rates among AGYW, including gender-based violence, gender inequality, poverty, and inadequate access to education. These broader services are no longer being prioritized by PEPFAR.
- Similarly, the number of children and family members served by the Orphans and Vulnerable Children (OVC) program declined significantly in FY 2025 Q4 compared to FY 2024 Q4 (1.7 million compared to 6.5 million), falling to its lowest level in the last five years, and less than a quarter of those served in Q4 of 2022.
- PEPFAR eliminated reporting on key and priority populations (those who were marginalized or faced particular barriers to HIV services, including men who have sex with men, people in prison, displaced persons or mobile communities, and others) reached with prevention and other interventions. Reporting on voluntary medical male circumcision (VMMC) was also eliminated.
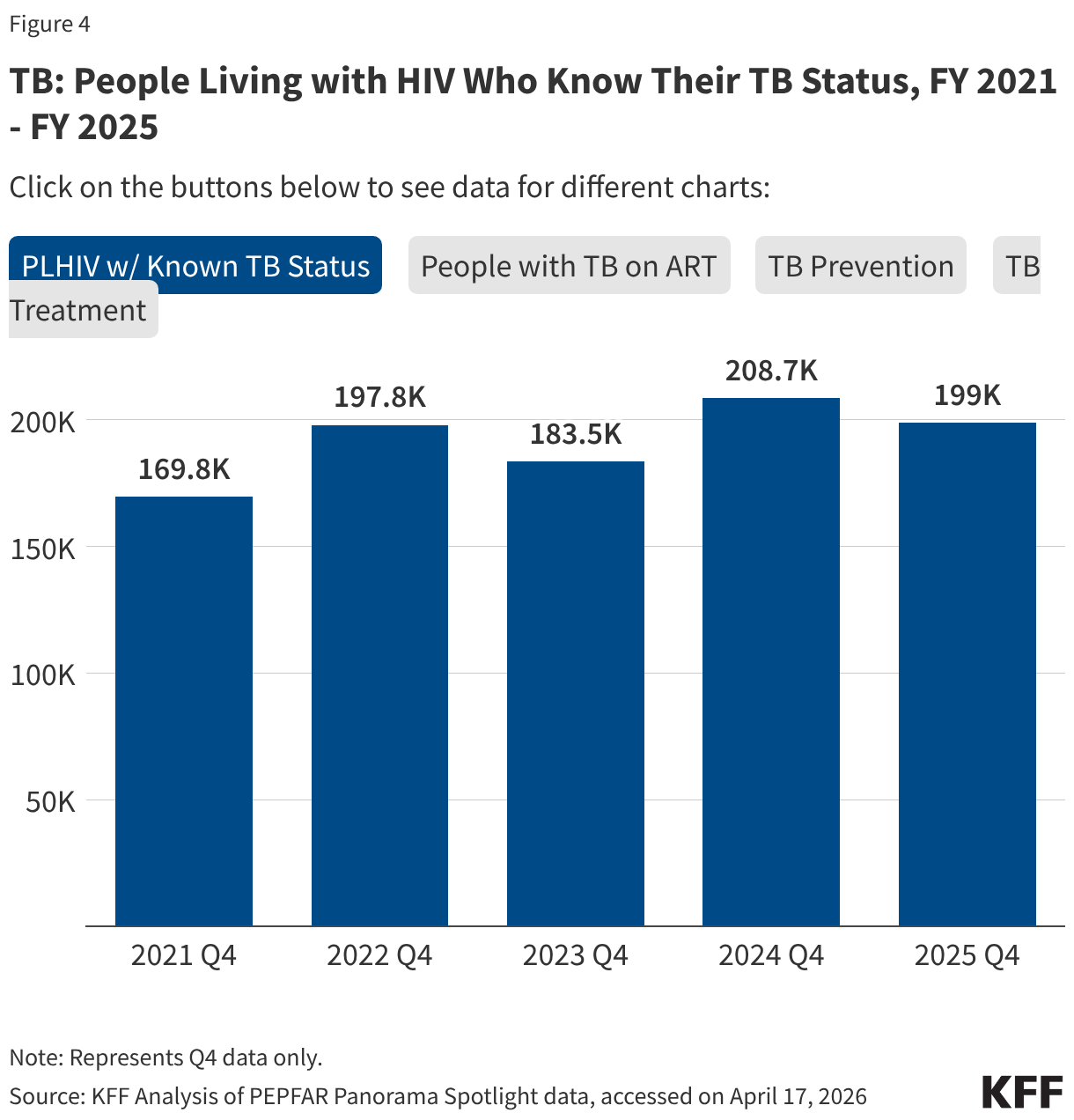
PEPFAR has also provided significant support to address HIV and TB co-infection, and risk of TB among those with HIV. These results were varied.
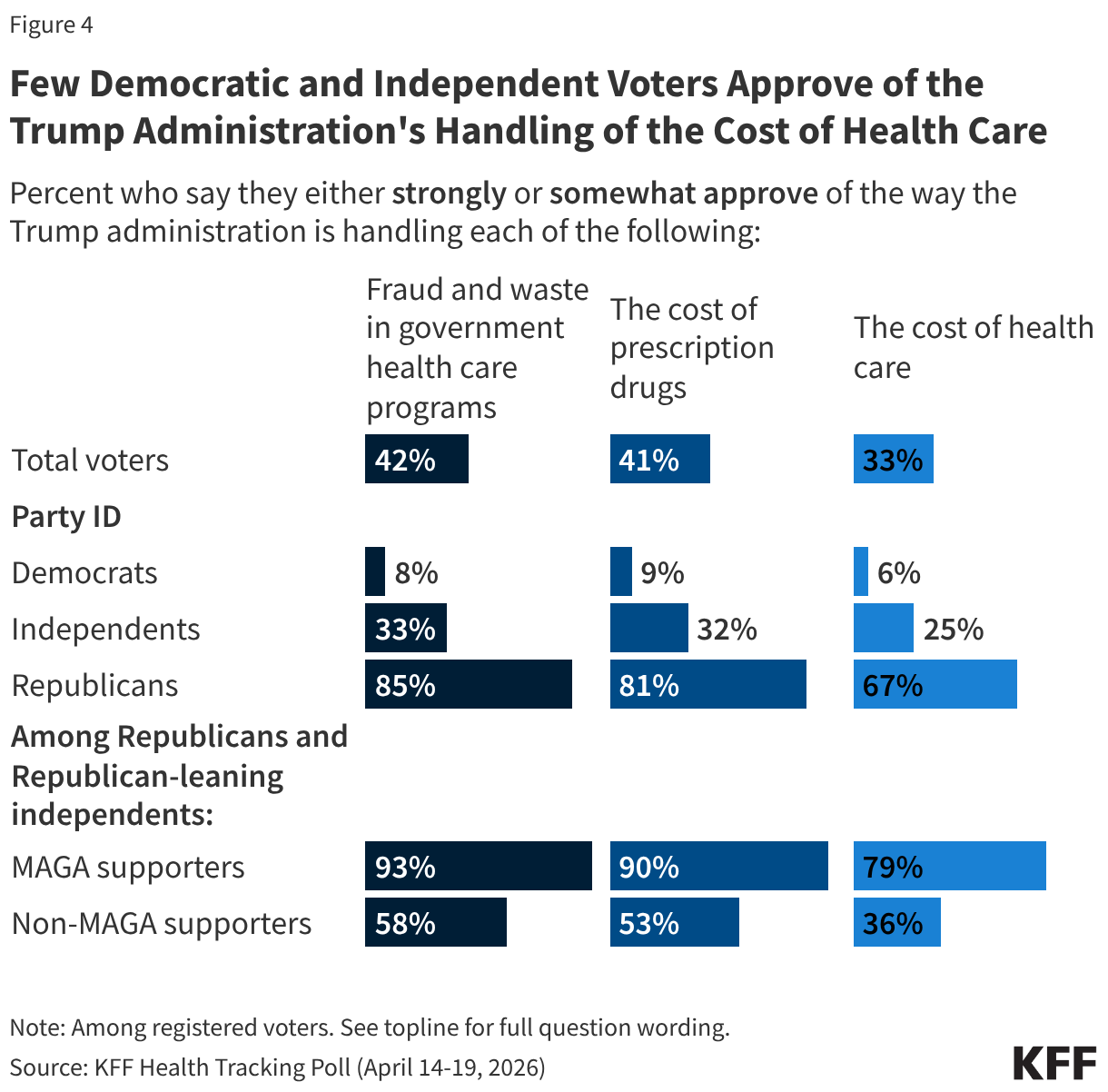
- The number of individuals living with HIV who know their TB status slightly declined in FY 2025 Q4 compared to FY 2024 Q4 (199k compared to 208.7k) but was higher than prior years.
- The number of individuals living with HIV and TB who are receiving ART declined considerably in FY 2025 Q4, compared to previous fiscal years, including a 33% drop compared to FY 2024 (138.9k compared to 208.1k).
- Similarly, the number of individuals receiving ART who initiated TB prevention therapy declined in FY 2025 Q4 (467.7k compared to 823k), reaching its lowest level over the period and continuing a declining trend. The number for FY 2025 Q4 was a third of the number in FY 2021.
- At the same time, the number of individuals receiving ART who initiated TB treatment increased in FY 2025 Q4 compared to FY 2024 Q4, to its highest level over the period.
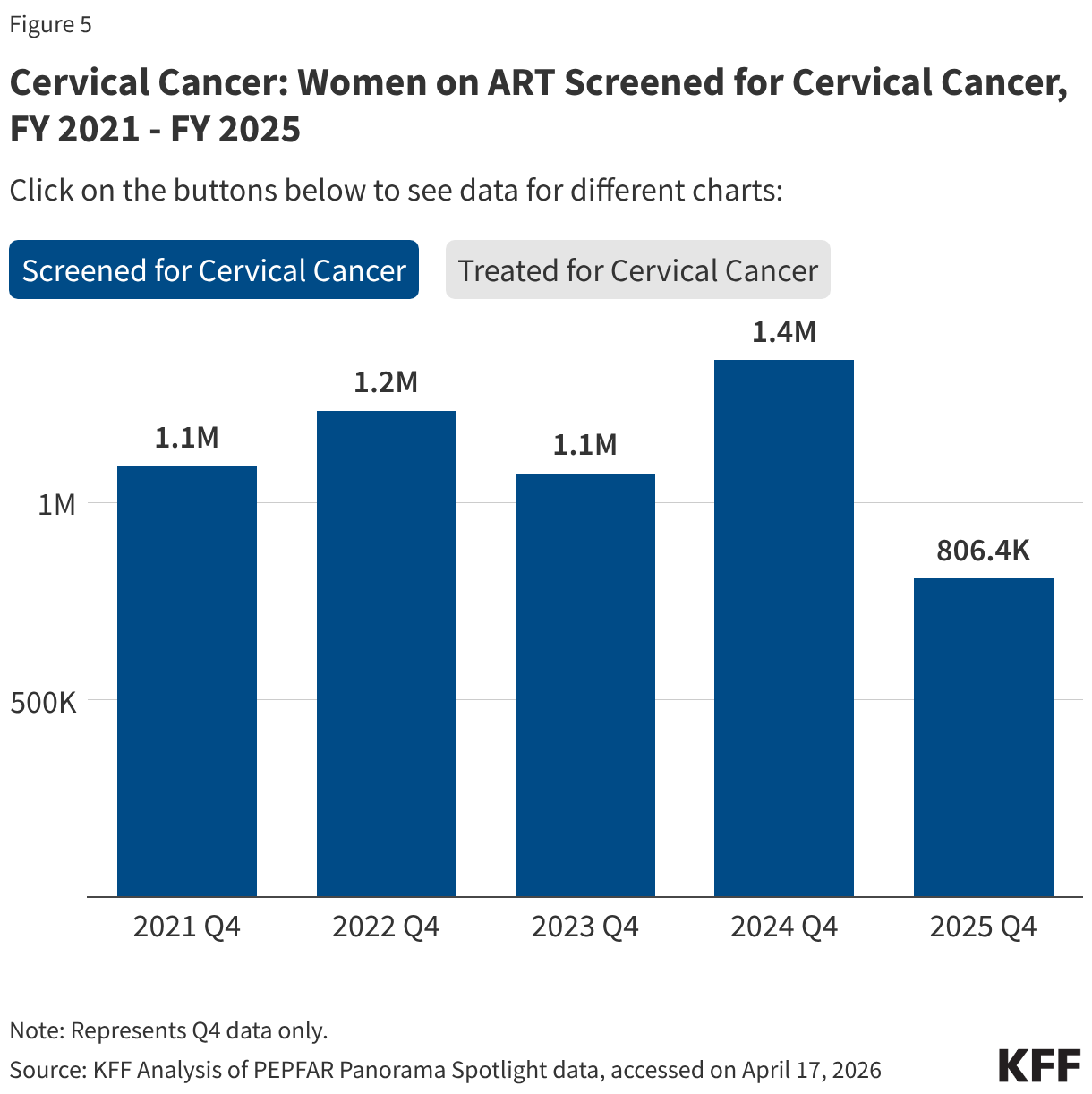
PEPFAR has, for many years, also worked to address the elevated risk of cervical cancer among women living with HIV. While screening for cervical cancer declined, the share receiving treatment for cervical cancer held steady.
- The number of women living with HIV and receiving ART who were screened for cervical cancer declined in FY 2025 Q4 compared to FY 2024 Q4 (806.4K compared to 1.4 million), its lowest level over the period.
- Of the women living with HIV and receiving ART and screened positive for cervical cancer, the share receiving treatment was only slightly below the prior period (89% in FY 2025 Q4 compared to 92% in FY 2024 Q4).
Methods and Data Limitations
Data represent KFF and Boston University analysis of PEPFAR monitoring, evaluation and reporting (MER) datasets from the PEPFAR Panorama Spotlight for quarter 4 of fiscal years 2021-2025, with a particular focus on changes between FY 2024 and FY 2025 (all countries that reported data in FY 2024 Q4 also reported data in FY 2025 Q4). Data were accessed on April 17, 2026.
Reporting for several indicators included in this analysis was changed from “required” to “optional” at some point for FY 2025 (current and prior reference guides for MER reporting can be found here). These indicators include:
- Adolescent girls and young women completing DREAMS (AGYW_PREV)
- Children and family members served by the OVC program (OVC_SERV)
- Individuals with TB and HIV receiving ART (TB_ART)
- Individuals on ART who initiated TB preventive therapy (TB_PREV)
- Women on ART screened for cervical cancer (CXCA_SCRN)
- Percentage of women on ART who screened positive for cervical cancer receiving treatment (CXCA_TX)
Because of these reporting requirement changes, declines in the data may reflect actual declines in services, or reduced reporting since participants are no longer required to track these metrics.
Jen Kates and Anna Rouw are with KFF. Allyala Nandakumar is with Boston University.