Potential Savings for Medicare Part D Enrollees Under Proposals to Add a Hard Cap on Out-of-Pocket Spending
Medicare Part D, the outpatient prescription drug benefit for Medicare beneficiaries, provides coverage above a catastrophic threshold for high out-of-pocket drug costs, but there is no cap on total out-of-pocket drug costs that beneficiaries pay each year. Part D enrollees are required to pay 5% of their total drug costs in the catastrophic phase unless they qualify for Part D Low-Income Subsidies (LIS). In 2021, the catastrophic threshold is set at $6,550 in out-of-pocket drug costs, which includes what beneficiaries themselves pay and the value of the manufacturer discount on the price of brand-name drugs in the coverage gap (sometimes called the “donut hole”), which counts towards this amount. This lack of a hard out-of-pocket spending cap can expose Part D enrollees to thousands of dollars in out-of-pocket costs if they take several costly medications or even just one expensive drug.
President Biden has endorsed adding a hard cap on out-of-pocket Medicare Part D prescription drug spending, and this proposed change has also been included in legislation sponsored by policymakers on both sides of the aisle, including H.R. 3, which passed the U.S. House of Representatives in December 2019 and was recently reintroduced in the 117th Congress; H.R. 19, the House GOP prescription bill (a similar version was introduced in the Senate); bipartisan legislation that passed out of the Senate Finance Committee in the 116th Congress (S. 2543); and other legislation. Under H.R. 3, out-of-pocket drug spending under Part D would be capped at $2,000 (beginning in 2024), while under the GOP drug price legislation and the 2019 Senate Finance bill, the cap would be set at $3,100 (beginning in 2022); under each of these proposals, the out-of-pocket cap excludes the value of the manufacturer price discount. A lower cap would help more beneficiaries and provide more out-of-pocket savings than a higher cap, but could mean higher costs for the federal government, plans, and drug manufacturers, depending on the specific features included in these Part D benefit redesign proposals.
To inform discussions about the potential impact of this proposal, in a previous KFF analysis, we analyzed how many Part D enrollees without low-income subsidies exceeded the catastrophic coverage threshold annually and over multiple years, taking into account both beneficiary out-of-pocket spending and the value of the manufacturer discount, and we found that 1.5 million enrollees did so in 2019, and close to 3 million did so between 2015 and 2019. In this analysis, we focus on the potential impact of different out-of-pocket spending caps in terms of how many beneficiaries would be affected and how much they could save. We analyze how many beneficiaries paid more than $2,000 or $3,100 out of their own pockets for their medications in 2019 (excluding the value of manufacturer discounts they may have received), and the magnitude of potential savings for beneficiaries had these caps been in place in 2019. We also analyze the individual drugs for which Part D enrollees incurred average annual out-of-pocket costs in 2019 above the amount of proposed caps. The analysis is based on 2019 Part D claims data (the most current year available) for Part D enrollees without low-income subsidies (LIS) from the Centers for Medicare & Medicaid Services Chronic Conditions Data Warehouse (see Methods for details).
How many Medicare Part D enrollees incurred out-of-pocket drug costs above $2,000 and $3,100 in 2019?
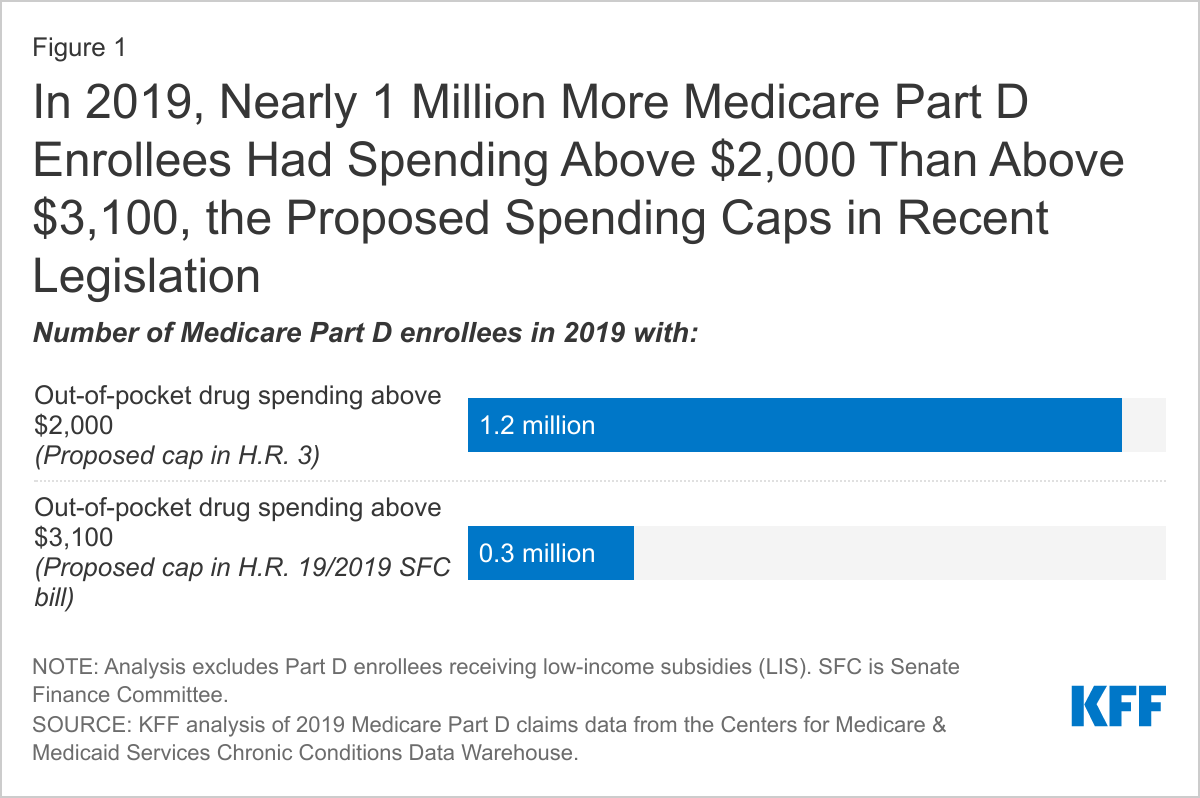
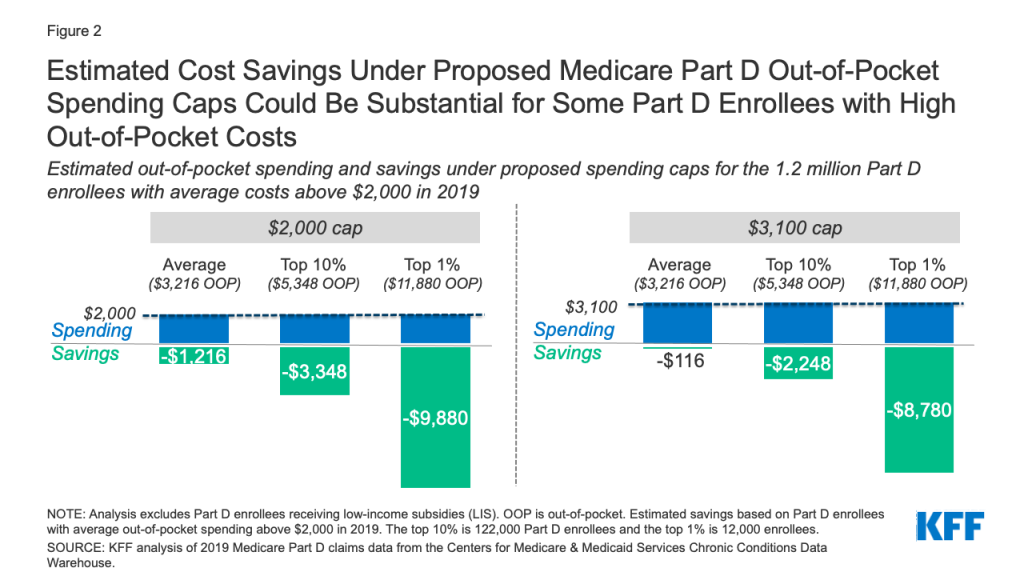
- In 2019, nearly 1 million more Part D enrollees incurred out-of-pocket costs for their medications above $2,000, the proposed out-of-pocket spending limit in H.R. 3, than above $3,100, the proposed out-of-pocket spending limit in the GOP drug legislation and the 2019 Senate Finance Committee bill (Figure 1). Overall, 1.2 million Part D enrollees in 2019 incurred annual out-of-pocket costs for their medications above $2,000, while 0.3 million spent more than $3,100 out of pocket.
- The number of Medicare Part D enrollees who have annual out-of-pocket costs greater than $2,000 or $3,100 in a future year, when a proposed cap could be implemented, is likely to exceed our estimates that are based on 2019 claims data, considering enrollment growth, rising drug prices for existing drugs, and the availability of new, higher-priced medications covered by Part D. Moreover, while adding an out-of-pocket cap to Part D may affect a relatively small number of enrollees in any given year, it would help a larger share and number over time, as our previous analysis showed.
What is the magnitude of potential savings for Part D enrollees with out-of-pocket costs above $2,000 or $3,100 based on proposed spending caps?
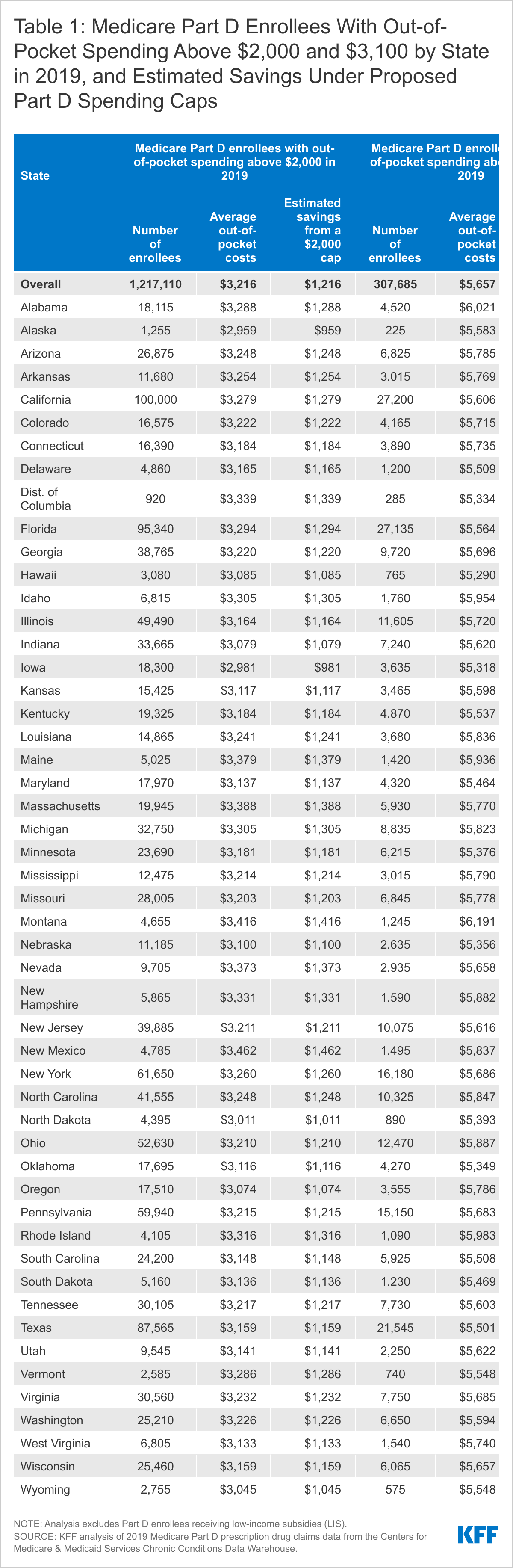
- As expected, a $2,000 cap on out-of-pocket spending would generate larger savings than a $3,100 cap. Average out-of-pocket spending was $3,216 among the 1.2 million Part D enrollees with out-of-pocket spending above $2,000 in 2019. These enrollees would have saved $1,216, or 38% of their annual costs, on average, if a $2,000 cap had been in place in 2019, but only $116, or 4%, under a $3,100 cap (Figure 2). (See Table 1 for estimates of the number of Medicare Part D enrollees with out-of-pocket spending above $2,000 and $3,100 in 2019 by state, and estimated savings under proposed Part D spending caps.)
- Medicare Part D enrollees with higher-than-average out-of-pocket costs could save substantial amounts with an out-of-pocket spending cap. For example, the top 10% of beneficiaries (122,000 enrollees) with average out-of-pocket costs for their medications above $2,000 in 2019 – who spent at least $5,348 – would have saved $3,348 (63%) in out-of-pocket costs with a $2,000 cap and $2,248 (42%) with a $3,100 cap. The top 1% of beneficiaries with average out-of-pocket costs above $2,000 (12,000 enrollees) – who spent nearly $12,000 or more – would have saved $9,880 (83%) with a $2,000 cap and $8,780 (74%) with a cap of $3,100.
How many and which drugs had average out-of-pocket costs in 2019 above proposed spending caps?
- In 2019, there were 154 drugs where Medicare Part D enrollees incurred average annual out-of-pocket costs for that one drug alone greater than $2,000, including 108 drugs where average annual out-of-pocket costs exceeded $3,100.
- While some of these high-priced drugs are treatments for rare diseases that are taken by a relatively small number of Part D enrollees, the out-of-pocket cost for individual patients taking these drugs can be substantial. For example, average out-of-pocket spending was $42,440 for Strensiq, which treats a rare metabolic disease called hypophosphatasia; $15,108 for Takhzyro, a treatment for hereditary angioedema; and $13,090 for Firdapse, a treatment for Lambert-Eaton myasthenic syndrome (LEMS), a rare muscle disease. It is important to note that these spending estimates do not include additional out-of-pocket costs that users of these 154 drugs incurred for other medications, so the total out-of-pocket cost burden in 2019 for users of these drugs was likely higher, suggesting that the savings associated with proposed caps on out-of-pocket spending would be even greater than the amount associated with a given relatively high-priced drug.
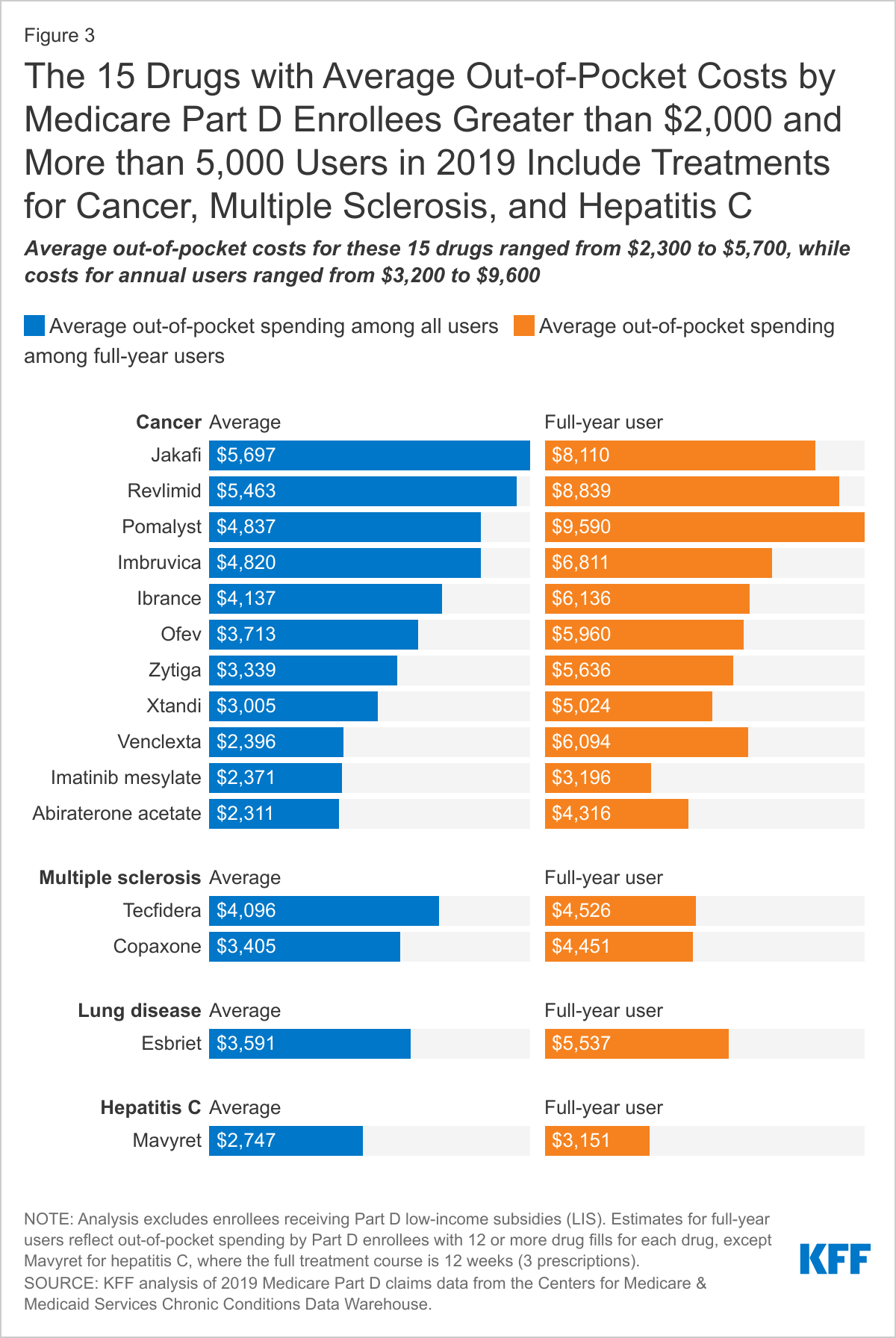
- Most of these relatively high-priced drugs were used by fewer than 1,000 non-LIS Part D enrollees in 2019, but 15 drugs were used by at least 5,000 enrollees, and these include drugs to treat cancer, multiple sclerosis (MS), and hepatitis C. For most of these drugs, average out-of-pocket costs in 2019 were well over $2,000, and in many cases well over $3,100 (Figure 3).
- Average out-of-pocket spending for these 15 drugs in 2019 ranged from $2,300 for abiraterone acetate, a prostate cancer drug used by 14,000 non-LIS enrollees, to $5,700 for Jakafi, a treatment for blood cancer used by 8,000 enrollees.
- Part D enrollees who used one of these 15 drugs for an entire year spent substantially more than the average user. Average spending tends to understate spending incurred by people who take high-priced drugs for an entire year, since the average includes beneficiaries who begin taking medication after the first of the year (for example, those with a new diagnosis mid-year) as well as those who stop taking a drug at some point during the year for various reasons (such as switching medications, or upon their death). For example:
- Medicare Part D enrollees who used the cancer drug Revlimid for the entire year in 2019 spent nearly $9,000 out of pocket for this drug alone, two-thirds more than the average user, who only filled 7 prescriptions.
- Beneficiaries who used the MS drug Tecfidera for the full year in 2019 spent around $4,500 out of pocket for this drug, 31% more than the average user, who filled 9 prescriptions.
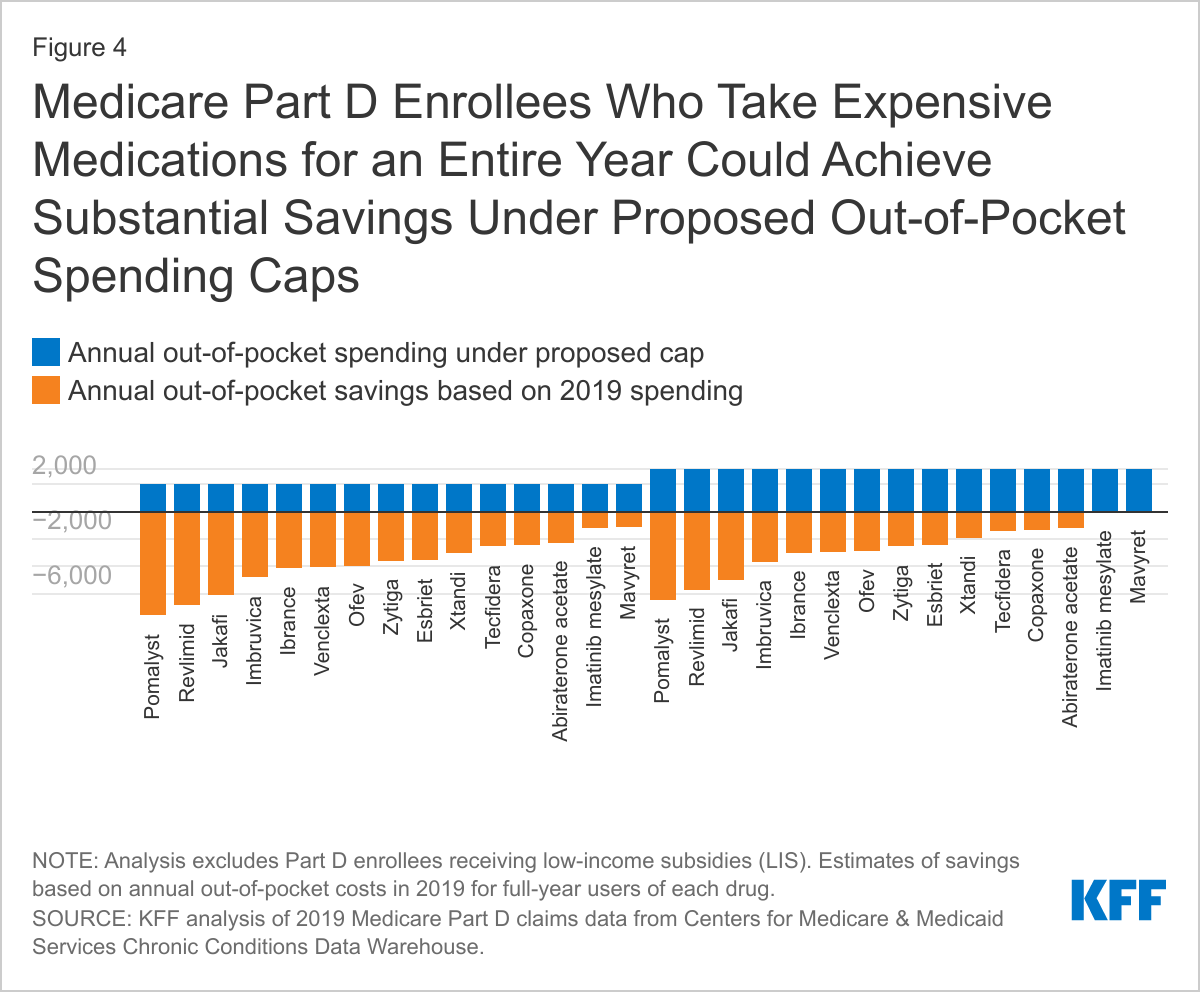
- Beneficiaries with higher-than-average out-of-pocket costs, including those who take expensive medications for an entire year, could achieve substantial savings under proposed spending caps. For example, Part D enrollees who took Revlimid for the entire year in 2019 would have seen savings of close to $7,000 under a $2,000 spending cap, and more than $5,700 with a $3,100 cap in 2019 (Figure 4) – not including potential savings from other drugs they may also have been taking.
Discussion
Our analysis shows that close to 1 million more Medicare Part D enrollees would have had their out-of-pocket costs capped in 2019 under a $2,000 out-of-pocket drug spending limit (as under H.R. 3) than a $3,100 limit (as under the GOP bill and the 2019 Senate Finance Committee bill). Under either cap, however, savings could be considerable for Part D enrollees who take high-cost medications for conditions such as cancer and MS. Deciding on the level of the cap involves tradeoffs, with more enrollees benefitting and higher out-of-pocket cost savings from a lower cap, but with the potential for higher spending by the federal government, plans, and drug manufacturers, depending on the specific features included in the Part D benefit redesign proposal.
While proposed legislation to cap out-of-pocket costs under Medicare Part D would help beneficiaries who take several costly medications or even just one high-priced drug, these proposals would not cap drug spending for expensive physician-administered injectable and infused medications that are covered under Medicare Part B. These drugs are subject to a 20% coinsurance, with no cap on out-of-pocket costs. While many Medicare beneficiaries have supplemental coverage, such as employer-sponsored retiree benefits or Medigap, to help pay their share of costs, nearly 6 million beneficiaries lack supplemental coverage and another 26 million are enrolled in Medicare Advantage plans and typically face 20% coinsurance for Part B drugs up to their plan’s out-of-pocket maximum.
For example, a Medicare beneficiary who takes aducanumab, the new Alzheimer’s drug priced at $56,000 annually, would face cost-sharing liability of more than $11,000 in a year, according to KFF analysis, unless they have supplemental insurance. Medicare Advantage enrollees would have a portion of their out-of-pocket costs for this drug covered but would need to pay out of pocket up to their plan’s limit for Medicare Part A and B benefits ($7,550 in 2021 for in-network and $11,300 for in-network and out-of-network combined).
The number of Medicare Part D enrollees who have annual out-of-pocket costs greater than $2,000 or $3,100 in a future year, when a proposed cap could be implemented, is likely to exceed our estimates that are based on actual claims data for 2019, considering enrollment growth, rising drug prices for existing drugs, and the availability of new, higher-priced medications covered by Part D. These estimates also do not reflect the interactive effects of other provisions being considered in current prescription drug legislation, such as allowing the federal government to negotiate drug prices or Part B and Part D drug price inflation caps, which would also affect out-of-pocket drug spending.
Adding an out-of-pocket cap to Part D would protect Part D enrollees with high drug costs, which may affect only a small share of enrollees in any given year but a larger share over time, including those who have persistently high drug costs over multiple years and others who have high costs in one year but not over time. The outcome of current discussions in Congress about prescription drug legislation has implications for the affordability of prescription drugs among Medicare beneficiaries.
Juliette Cubanski and Tricia Neuman are with KFF. Anthony Damico is an independent consultant.
This work was supported in part by Arnold Ventures. We value our funders. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.
Methods
This analysis is based on 2019 Medicare Part D claims data from the Centers for Medicare & Medicaid Services Chronic Conditions Data Warehouse for Part D enrollees who are not receiving low-income subsidies (LIS). We exclude Part D enrollees receiving full low-income subsidies because they face only modest cost-sharing amounts before the catastrophic coverage phase and no cost sharing for catastrophic coverage, as well as those receiving partial low-income subsidies, who pay 15% coinsurance before the catastrophic coverage phase and modest copayments of no more than $3.70 for generics and $9.20 for brands in the catastrophic phase.
For this analysis, we estimated the number of Part D enrollees without low-income subsidies who had average annual out-of-pocket spending for all the medications they took in 2019 above $2,000 and $3,100, as well as the specific drugs where non-LIS Part D enrollees incurred average annual out-of-pocket costs above $2,000 and $3,100 in 2019. Except where noted, we define a Medicare beneficiary as a full-year user when they either have 12 or more drug fills (generally 30-day supplies) or when their annualized prescription medication quantity received exceeds 360 days.