5 Key Facts About Medicaid Prescription Drugs
Medicaid is the primary program providing comprehensive health and long-term care to low-income people, including access to prescription drugs to treat acute problems and manage ongoing chronic conditions, covering about one in five people in the United States. In recent years, Medicaid spending on prescription drugs has grown substantially, in part due to the emergence of new, high-cost drugs, including GLP-1s and cell and gene therapies that treat, and sometimes cure, rare diseases. At the same time, a more tenuous fiscal climate coupled with federal funding cuts and policy changes in the 2025 reconciliation law have put pressure on state Medicaid programs. As a result, both states and the federal government continue to prioritize the management of rising pharmacy costs. There have been several recent Trump administration prescription drug initiatives, including new payment models, that could help combat rising costs for state Medicaid programs, though questions remain about their impact. While lower prices for state Medicaid programs through the new models could result in reduced Medicaid prescription drug spending and potentially expanded coverage of certain drugs, the extent of the savings and how states or manufacturers will respond remain unclear. To provide context for emerging debates about federal actions to address prescription drug costs, this issue brief highlights five key facts about Medicaid prescription drug coverage, payment, and administration.
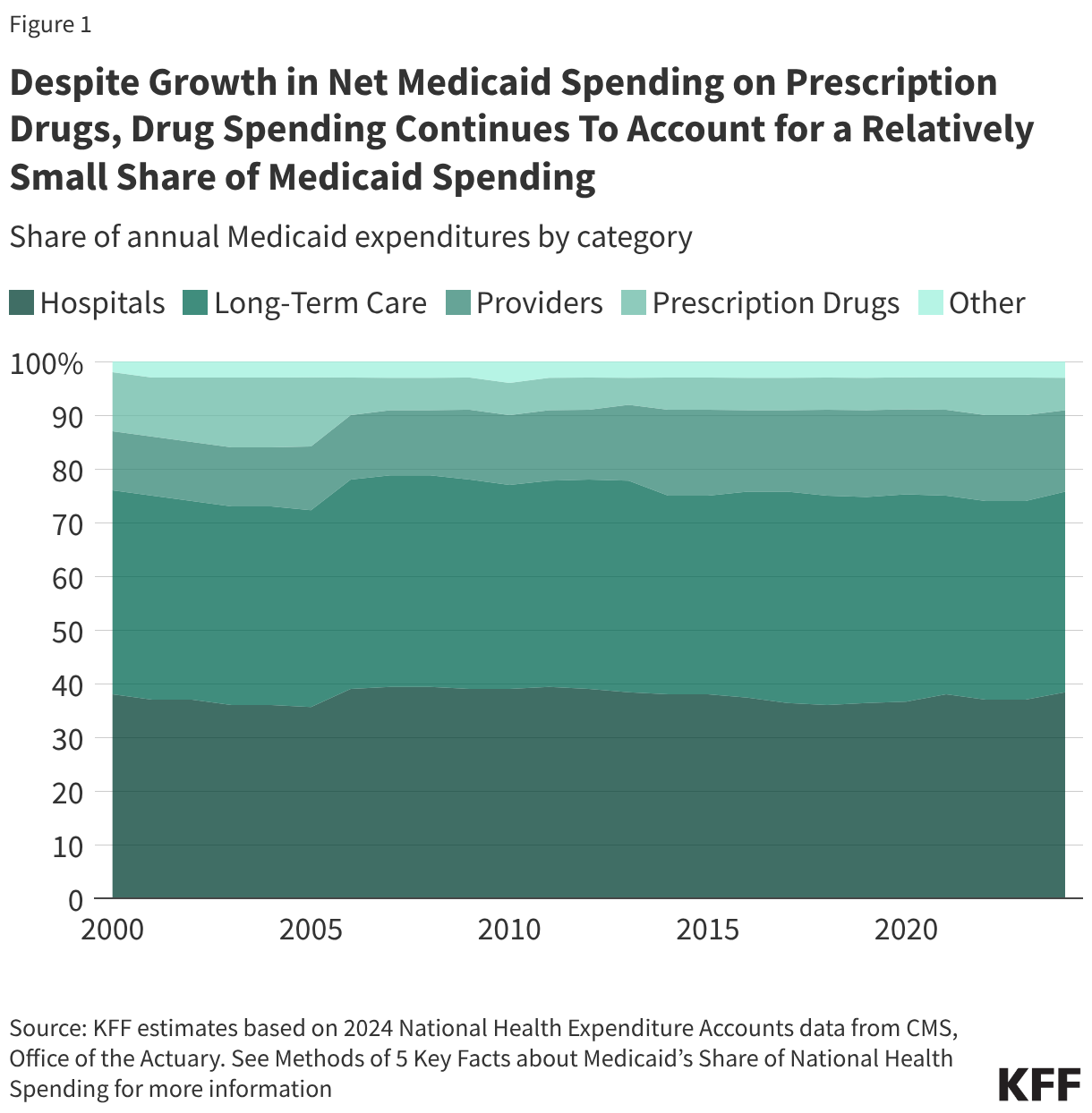
1. Despite growth in net Medicaid spending on prescription drugs, drug spending continues to account for a relatively small share of Medicaid spending.
Drug spending accounts for a relatively small share of overall Medicaid spending. The percent of Medicaid spending on prescription drugs was 6% in 2024, substantially smaller than Medicaid’s spending on hospitals (38%), long-term care (37%), and providers (15%) (Figure 1). Following the enactment of the Medicare prescription drug benefit in 2006 — which shifted some drug spending for dually-eligible beneficiaries to the federal government — the percent of Medicaid spending on prescription drugs has remained relatively stable between 5% and 7%. Despite this, management of prescription drug expenditures continues to be a focus area at both the state and federal levels. Net spending on prescription drugs increased by 46% between federal fiscal year (FY) 2019 and FY 2024, which is a similar rate to growth in Medicaid spending overall (52%) over the period. The emergence of new, high-cost drugs can put pressure on state budgets, and states reported experiencing several substantial Medicaid budget pressures including increasing pharmacy costs in KFF’s latest annual Medicaid budget survey.
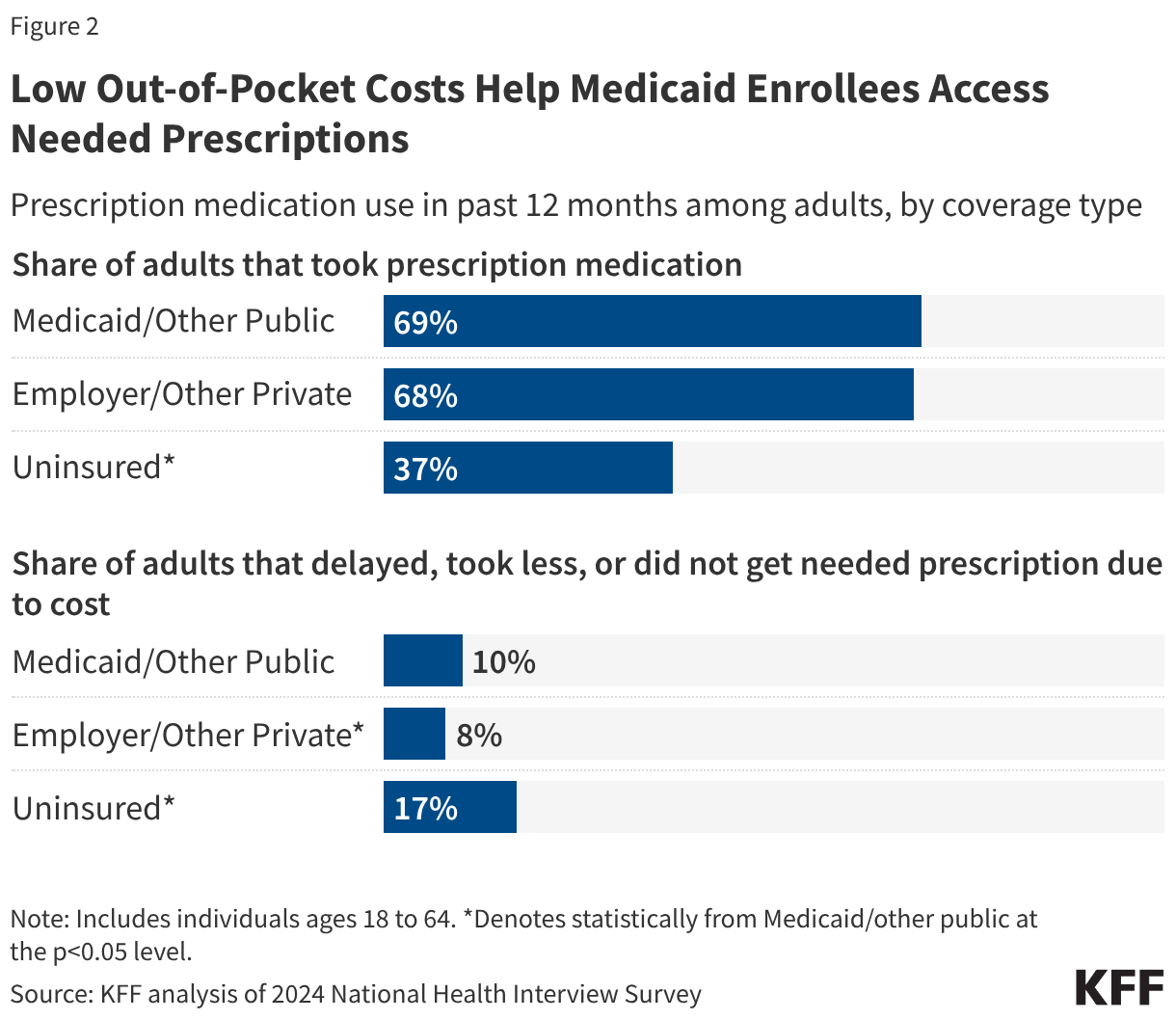
2. Low out-of-pocket costs help Medicaid enrollees access needed prescriptions.
Federal law limits out-of-pocket costs for Medicaid enrollees to nominal amounts. Since people on Medicaid must have low incomes to qualify for the program, Medicaid is designed to provide access to prescription drugs with little cost to enrollees. Federal rules limit beneficiary cost-sharing or copays to up to $4 for preferred drugs and $8 for non-preferred drugs for individuals with incomes at or below 150% of the federal poverty level (FPL) and slightly higher for those with higher incomes. Some Medicaid populations (e.g. most children under 18, pregnant women, etc.) are exempt from cost-sharing requirements. Within those limits, states have flexibility to establish their cost sharing levels, though fewer than half of states required prescription drug cost-sharing for non-exempt enrollees as of July 1, 2023.
Medicaid coverage helps adults avoid cost-related prescription medication rationing or delays. Over two-thirds of Medicaid-enrolled adults took prescription medication in the past year, which is similar to the share of adults with private insurance (68%) but much higher than the share of uninsured adults (37%) (Figure 2). Uninsured adults (17%) were also more likely than Medicaid-enrolled adults (10%) and privately insured adults (8%) to report delaying filling, taking less, or not getting a needed prescription at all due to cost. Medicaid adults report cost-related rationing or delays at slightly higher rates than privately insured adults (10% compared with 8%), likely because Medicaid covers a predominantly low-income population. While prescription out-of-pocket costs in Medicaid are limited to nominal amounts, even low amounts may still be prohibitive for some families. Recently proposed federal prescription drug initiatives may affect overall Medicaid spending for prescription drugs, but they are not expected to have an impact on Medicaid enrollee affordability.
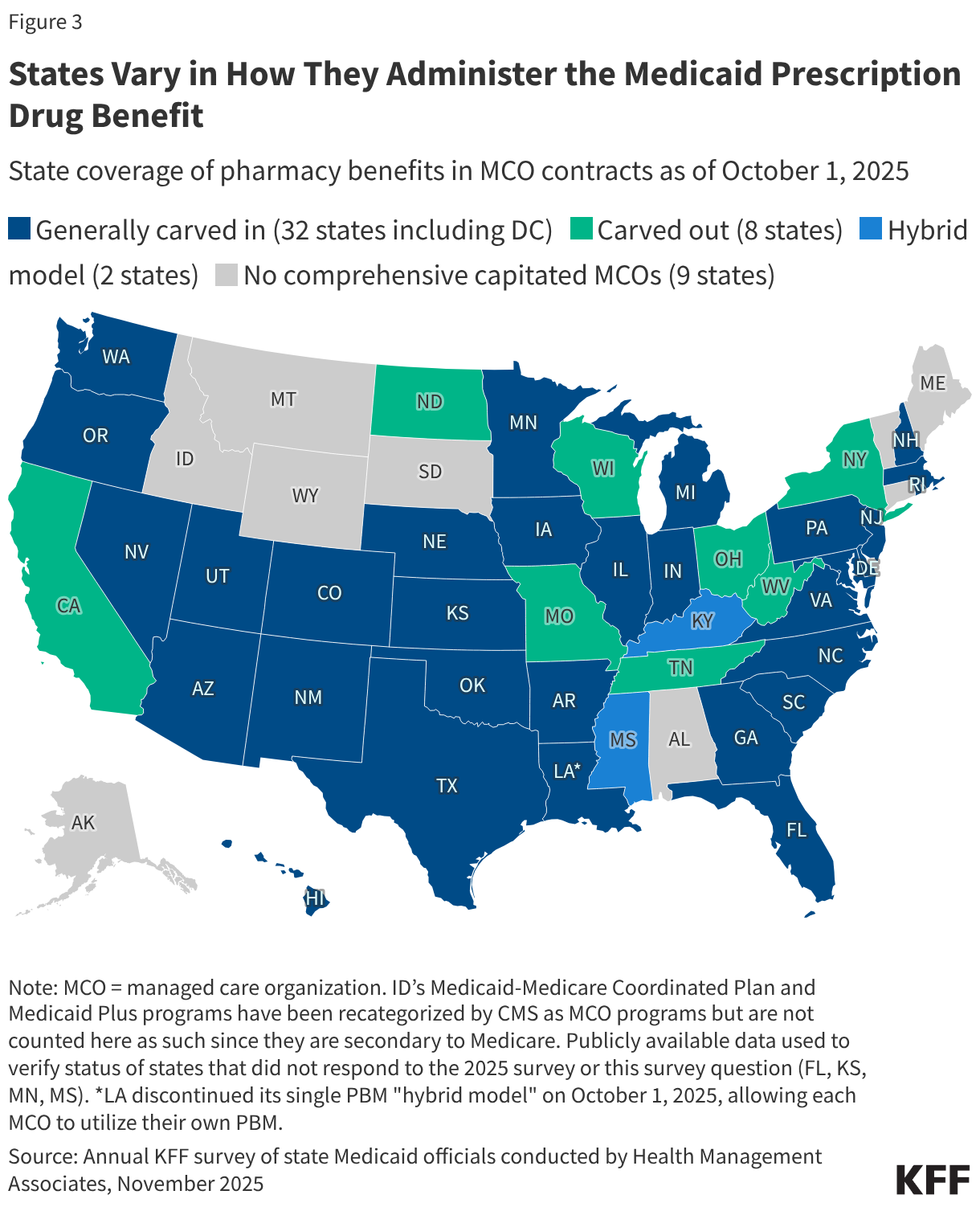
3. States vary in how they administer the prescription drug benefit.
While not required by Medicaid statute, all state Medicaid programs cover prescription drugs, though states administer the benefit in different ways. Within federal rules regarding payment and medical necessity requirements, states have flexibility to administer and manage the pharmacy benefit within their Medicaid programs. Capitated managed care is now the dominant way in which states deliver services, including prescription drugs, to Medicaid enrollees. As of July 2025, only eight out of 42 states that contract with managed care organizations (MCOs) “carved out” prescription drug coverage from managed care and delivered the pharmacy benefit through fee-for-service (FFS) (Figure 3). For the other MCO states, the pharmacy benefit is included in the overall capitation rate paid to the plan, though as a MCO risk mitigation strategy or for other reasons, states may also carve out one or more specific drugs (or drug classes) from MCO capitation payments. While most Medicaid prescription drugs are covered through the pharmacy benefit, some drugs, including outpatient physician-administered drugs or drugs provided as part of a hospital stay, are covered through the medical benefit or both, depending on how the drug is dispensed, administered, and billed.
States and MCOs often contract with external vendors like pharmacy benefit managers (PBMs) to manage or administer the pharmacy benefit. PBMs may perform a variety of administrative and clinical services for Medicaid programs (e.g., negotiating supplemental rebates with drug manufacturers, adjudicating claims, monitoring utilization, overseeing and formulating preferred drug lists, etc.) and are used in both FFS and managed care settings. States have increased their reliance on PBMs over the years, with 33 states reporting contracting with a PBM to administer their FFS pharmacy benefit as of July 1, 2023. However, PBMs have faced increased scrutiny in recent years as more states adopt reforms to increase transparency and improve oversight. Congress also included Medicare and employer health plan PBM reforms in the Consolidated Appropriations Act, 2026, though Medicaid PBM provisions were not included.
4. Medicaid payments for prescription drugs are determined by a complex set of policies.
Total Medicaid spending for a given outpatient drug is based on the amount paid to the pharmacy less rebates received from the manufacturer. Pharmacies negotiate prices and purchase drugs from manufacturers or wholesalers and then fill prescriptions for Medicaid enrollees. In return, the pharmacy receives payment from the state Medicaid agency based on the ingredient cost of the drug and professional dispensing fees, plus any copays paid by the enrollee (Figure 4). States set policies on dispensing fees and, within federal guidelines, enrollee cost-sharing, while federal regulations guide FFS payment levels for outpatient drug ingredient costs (physician-administered drugs or those received in an inpatient setting follow a different set of payment guidelines). If the state delivers the pharmacy benefit through managed care, MCOs reimburse the pharmacy, usually through a PBM. MCOs have more flexibility to set payment rates, though they must set payment rates sufficient to guarantee enrollee access. The final cost to Medicaid is then offset by any rebates received under the federal Medicaid Drug Rebate Program (MDRP) and state-negotiated supplemental rebates (if any).
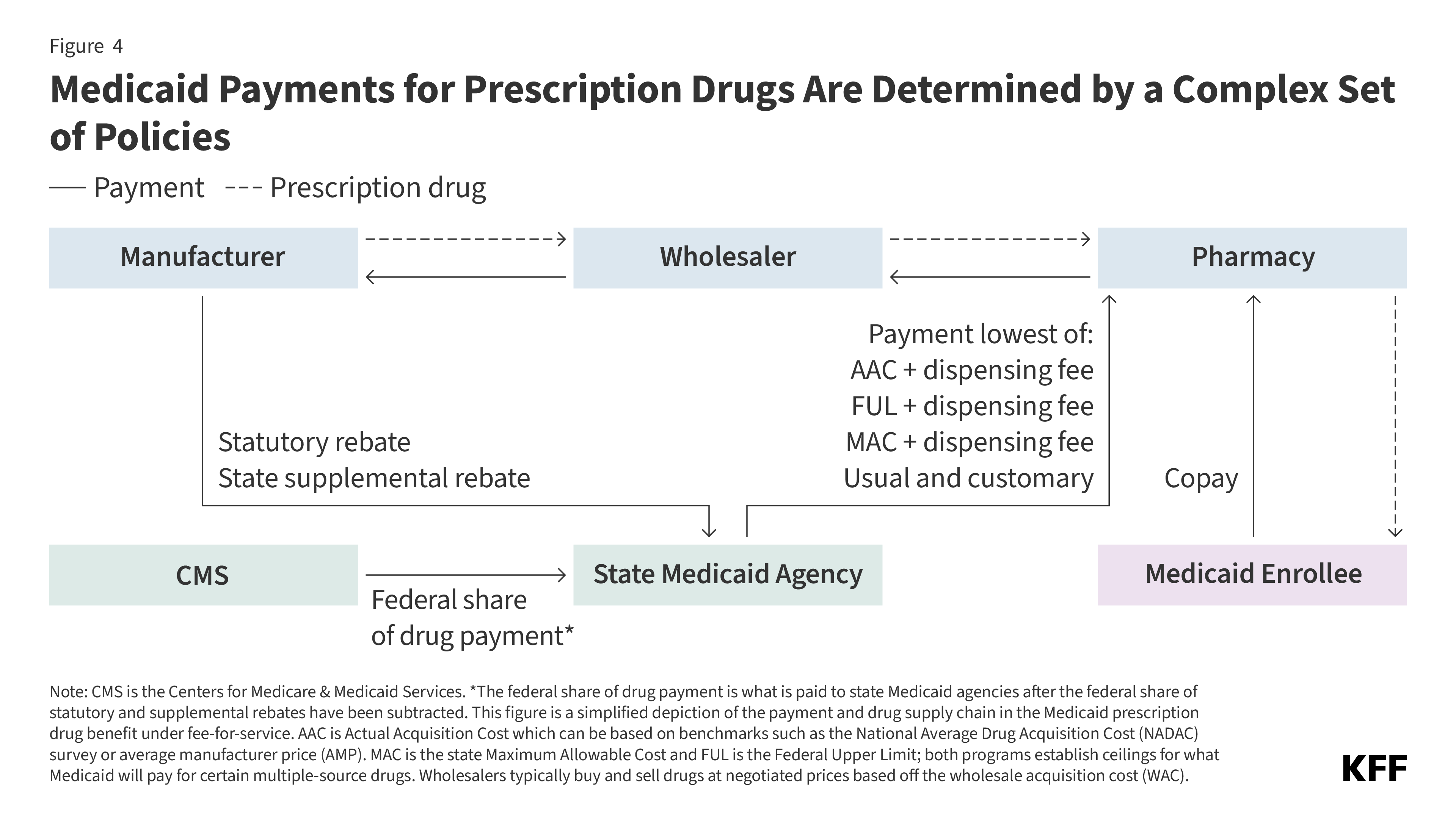
Under FFS, state Medicaid programs reimburse pharmacies for prescription drugs based on the ingredient costs for the drug and a dispensing fee for filling the prescription (Figure 4). The ingredient cost must reflect the actual acquisition cost (AAC) or the price to the pharmacy of acquiring the drug from a manufacturer or wholesaler (see Box 1 for a list of key Medicaid drug pricing terms and their definitions). States have some flexibility to determine AAC, which can include using the National Average Drug Acquisition Cost (NADAC) survey or average manufacturer price (AMP) as a benchmark. With the exception of some multiple-source drugs for which there are additional limits, federal regulations require Medicaid programs to reimburse pharmacies based on the lesser of:
- (1) AAC plus a professional dispensing fee or
- (2) the pharmacy’s “usual and customary charge” to the public.
For certain multiple-source drugs, AAC is capped at the federal upper limits (FUL) or state maximum allowable costs (MAC). Overall, depending on the drug and state, reimbursement is typically set at the lesser of:
- (1) the state’s AAC formula plus dispensing fee,
- (2) the FUL plus dispensing fee,
- (3) the state MAC plus dispensing fee, or
- (4) the pharmacy’s usual and customary charge to the public.
These limits ensure Medicaid is paying a reasonable market price for prescription drugs. The dispensing fee is intended to cover reasonable pharmacy costs associated with filling a prescription. States establish dispensing fees, and they can vary by type of pharmacy or drug. The ingredient and dispensing fee cost are considered together when assessing the adequacy of a state’s drug payment, and the total payment must meet federal requirements.
Manufacturers who want their drugs covered by Medicaid are required to rebate a portion of drug payments under the MDRP, and in return, Medicaid must cover almost all FDA-approved drugs produced by those manufacturers. The rebate formula is set in statute, varies by type of drug (brand or generic), and is the same regardless if drugs are purchased through FFS or managed care. For most brand name drugs, the rebate is the greater of either 23.1% of average manufacturer price (AMP), which is the average price paid to drug manufacturers, or the difference between AMP and “best price”, which is the lowest available price to any wholesaler, retailer, or provider, excluding certain government programs. The best price provision ensures that Medicaid gets the lowest drug prices (with some exceptions), and a majority of brand drug rebates are based on best price. For generic drugs, the rebate amount is 13% of AMP, and there is no best price provision. The rebate calculation also includes an additional inflationary component, which requires additional rebates if a drug’s price rises faster than inflation, and as of January 1, 2024, there is no longer a cap on the total rebate amount if a drug’s price increases quickly over time. The rebates are paid to the states quarterly, and shared between the states and federal government based on the state’s federal medical assistance percentage or “FMAP” to offset the total cost of prescription drug payments. Drugs not dispensed by the pharmacy but received in other settings, including physician-administered drugs, can be eligible for rebates under the MDRP if they meet the definition of a “covered outpatient drug,” generally meaning a prescription drug that is FDA approved from a rebating manufacturer and identified separately on a claim for payment.
In addition to federal statutory rebates, most states negotiate with manufacturers for supplemental rebates. As of September 2025, 48 states and DC had supplemental rebate agreements in place. States often use placement on a preferred drug list (PDL) as leverage to negotiate supplemental rebates with manufacturers (see Box 2). Some states have also formed multi-state purchasing pools to increase negotiating power and/or negotiated value-based arrangements (VBAs), which increase supplemental rebates if a drug does not perform as expected. In addition, Medicaid managed care plans (or typically PBMs on behalf of the plan) may negotiate their own supplemental rebate agreements with manufacturers, which then, when passed on to the plan, may result in lower capitation rates. In Medicaid and in general, there are many opaque aspects to the pricing of prescription drugs, and much of the data is proprietary, which makes it difficult to understand which prescription drugs and parts of the supply chain are the spending drivers.
Box 1: Key Terms in Medicaid Drug Pricing
AAC: Actual acquisition cost is the state Medicaid agency’s determination of pharmacy providers’ actual prices paid to acquire drug products from a specific manufacturer. AAC is the current Medicaid benchmark to set payment for drug ingredients.
AMP: Average manufacturer price is the average price paid to the manufacturer by wholesalers and retail community pharmacies that purchase drugs directly from the manufacturer. AMP is used to calculate drug rebates under the MDRP.
Best price: The best price is the lowest available price to any wholesaler, retailer, or provider, excluding certain government programs like the 340B drug pricing program and the health program for veterans.
Brand drug: A brand drug can be a single source or innovator multiple source drug. Brand drugs are produced under an original drug application through the Food and Drug Administration (FDA) and use a proprietary, trademark-protected name.
Dispensing fee: The dispensing fee covers costs in excess of the ingredient cost of a covered outpatient drug and intended to cover reasonable pharmacy costs associated with filling a prescription.
FUL: The federal upper limit sets a reimbursement limit for some multiple source drugs; calculated as 175% AMP.
Generic drug: Generic drugs are produced by multiple manufacturers and are therapeutically equivalent to their brand name counterpart.
MAC: Maximum allowable cost is a reimbursement limit set by some states for multiple source drugs in addition to the FUL.
Multiple source drug: A multiple source drug is a drug that is produced by multiple manufacturers or, under the MDRP, a drug with at least one other product that is therapeutically equivalent (or has a generic equivalent available).
NADAC: The national average drug acquisition cost is intended to be a national average of the prices at which pharmacies purchase a prescription drug from manufacturers or wholesalers, including some rebates. NADAC can be used to calculate AAC.
Single source drug: Single source drugs are produced under an original drug application through the FDA through a single manufacturer and have patent protection
WAC: Wholesale acquisition cost represents manufacturers’ published catalog, or list, price for sales of a drug (brand-name or generic) to wholesalers. However, in practice, discounts are negotiated and the full WAC is not what wholesalers or pharmacies pay for drugs.
Wholesaler: Wholesalers act as an intermediary between drug manufacturers and pharmacies, purchasing drugs from manufacturers and then storing and selling them to pharmacies. Most prescription drugs in the U.S. are distributed via a wholesaler. Wholesalers typically buy and sell drugs at negotiated prices based off the WAC.
5. While states must cover nearly all drugs under the MDRP, states use an array of payment strategies and utilization controls to manage prescription drug expenditures.
While rebates through the MDRP are a key tool to manage prescription drug expenditures, states also use an array of payment strategies and utilization controls. Under the MDRP, state Medicaid programs must cover nearly all of a rebating manufacturer’s FDA-approved drugs, essentially creating an open formulary. There is a small group of drugs that can be excluded from coverage, including drugs used for weight loss, though some states have opted to cover GLP-1s for obesity treatment or weight loss. The open formulary facilitates greater access to prescription drugs for enrollees but can also limit states’ ability to control drug costs through restrictive formularies. Instead, states use several tools to manage pharmacy expenditures and ensure safe use of medications. This includes innovative payment strategies such as VBAs, subscription models, or participating in recent federal drug payment models as well as tools such as preferred drug lists (PDLs), prior authorization linked to clinical criteria, quantity limits, and more to manage utilization (see Box 2). MCOs may apply differing utilization controls and medical necessity criteria unless the state’s MCO contract specifies otherwise. Utilization controls can also differ for physician-administered drugs covered under the medical benefit, and states have identified a number of challenges managing utilization and spending of drugs under the medical benefit in particular.
States continually develop, update, and expand their payment strategies and utilization controls. KFF’s 2025 Medicaid budget survey found that most responding states reported at least one new or expanded initiative to contain prescription drug costs, including participating in CMS’s cell and gene therapy access model or other initiatives related to VBAs, or implementing other policy changes related to maximizing rebates, expanding utilization controls, or oversight. Many of the cost containment initiatives reported specifically targeted high-cost specialty drugs, which are contributing to increases in Medicaid drug spending. At the federal level, there have been several recent prescription drug initiatives, including new drug payment models, that could help combat rising costs for state Medicaid programs, though questions remain about the implementation and impact of the deals.
Box 2: Common State Medicaid Utilization Management Strategies
Prior authorization: Prior authorization is one of the primary tools states have used to manage the utilization of prescription drugs. Prior authorization requires prescribers to obtain approval from the state Medicaid agency (or its contractor) before a particular drug can be dispensed. State prior authorization processes in FFS and managed care must adhere to federal requirements, though prior authorization has come under scrutiny in recent years for hindering patient access to care.
Preferred drug list (PDL): A PDL is a list of outpatient drugs states encourage providers to prescribe over others. Often, drugs on PDLs are cheaper or include drugs for which a manufacturer has provided supplemental rebates. A state may require prior authorization for a drug not on a preferred drug list or attach higher co-pays, creating incentives for a provider to prescribe a drug on the PDL when possible.
Step therapy: States can require an enrollee to use a lower-cost drug at the start of treatment and only allow enrollees to “step up” to other, higher-cost drugs once the lower-cost treatment(s) are proven ineffective.
Prescription or quantity limits: States may limit the number of prescriptions an enrollee may access without prior authorization, which may be a limit on the total number of prescriptions per month or limit on the number of brand drugs. States are also authorized under federal law to set minimum or maximum numbers of pills or doses per prescription, as well as the number of refills.
Medication therapy management (MTM): MTM is often provided by pharmacists and is intended to ensure the best therapeutic outcomes for patients by addressing issues of polypharmacy (using multiple medications at the same time), preventable adverse drug events, medication adherence, and medication misuse.
Drug utilization review (DUR) boards and pharmacy & therapeutics (P&T) committees: Drug utilization review programs are required by federal law and must establish standards to ensure prescriptions are appropriate, medically necessary, and unlikely to lead to adverse medical results. DUR programs must also include evaluation for problems like duplicate prescriptions, incorrect dosage, and clinical misuse. To establish a PDL, federal law requires a state Medicaid agency to establish a committee of physicians and pharmacists to inform the development of the PDL, review drugs, and develop coverage decisions. In many states these activities are performed by a pharmacy and therapeutics (P&T) committee, though states also have the option to use their DUR board to fill this role. The composition, structure, and operations of P&T committees and how responsibilities are split between DUR boards and P&T committees varies by state.