The Ryan White HIV/AIDS Program: The Basics
Key Facts
- The Ryan White HIV/AIDS Program, first enacted in 1990, is the largest federal programdesigned specifically for people with HIV, serving over half of all those diagnosed. It is a discretionary grant program dependent on annual appropriations from Congress.
- It is the nation’s safety net program for people with HIV, providing outpatient HIV care, treatment, and support services to those without health insurance and filling in gaps in coverage and assisting with costs for those with insurance limitations.
- Most Ryan White clients are low-income, male, people of color, and half are gay and bisexual men and other men who have sex with men.
- The program is the third largest sourceof federal funding for HIV care in the U.S., following Medicare and Medicaid and is the largest source of HIV discretionary funding. Funding is distributed to states/territories, cities, and HIV organizations in the form of grants. In FY 2024, the Ryan White HIV/AIDS Program was funded at $2.6 billion, which includes continued funding for the federal “Ending the HIV Epidemic” (EHE) initiative, created by President Trump during his first term.
- While the Ryan White Program has a long history of bipartisanship, the Trump administration has indicated that it will seek to eliminate the EHE, including its funding, end one part of the Ryan White program, and move the remainder of Ryan White into a new HHS agency as part of a larger departmental reorganization.
Overview
The Ryan White HIV/AIDS Program (Ryan White), the largest federal program designed specifically for people with HIV in the United States, serves over half of people in the U.S. diagnosed with HIV. First enacted in 1990, Ryan White has played an increasingly significant role as the number of people living with HIV has grown over time and people with HIV are living longer. It provides outpatient care and support services to individuals and families affected by HIV, functioning as the “payer of last resort,” by filling in the gaps for those who have no other source of coverage or face coverage limits or cost barriers. Multiple “parts” of the program (described below) can purchase health insurance on behalf of clients, which is often less expensive than paying for drugs alone and offers broader health coverage (which is different from when the program pays directly for medical costs as those must relate specifically to HIV).
The program has been reauthorized by Congress four times since it was first created (1996, 2000, 2006, and 2009) and each reauthorization has made adjustments to the program. The current authorization lapsed in FY 2013, but the program has continued to be funded through the annual appropriations process as there is no “sunset” provision or end date attached to the legislation. The program is currently administered by the HIV/AIDS Bureau (HAB) at the Health Resources and Services Administration (HRSA) of the Department for Health and Human Services (HHS), which provides funding to state, local, and community-based grantees to provide HIV services across the country.
The Ryan White Program has been a central component of the federal government’s Ending the HIV Epidemic (EHE) initiative, launched in 2019 under the first Trump Administration. New EHE funding has allowed the program to serve new people and brought others who had fallen out of care, back in. However, questions have been raised about whether the Trump administration will support the EHE going forward.
Clients
More than half a million Ryan White clients received at least one medical, health, or related support service through the program in 2022, with many receiving multiple types of services:
- More than half (59%) had incomes at or below the federal poverty level (FPL) (which in 2022 was $13,590 for a single person or $27,750 for a family of four); 28% had incomes between 101% and 250% FPL.
- Nearly one-fifth (18%) were uninsured, a decrease from 28% in 2013, prior to major coverage expansions under the Affordable Care Act (ACA). Most clients (82%) have some form of insurance coverage: Medicaid is the primary payer for Ryan White clients, covering 39%, including those dually eligible for Medicare. Other coverage includes: private insurance (20%), Medicare only (10%), and other or multiple sources of insurance (12%).
- Clients are largely male (72%); 25% are female and 3% are transgender. Approximately half (46%) are between the ages 45 and 64, up from 22% in 2016. More than one-third (39%) are between 25-44. Smaller shares are under 25 (4%) or over 64 (12%). Differing from the U.S. population overall, most clients are people of color (72%), including 45% who are Black and 25% who are Hispanic. Just over one-quarter of clients (25%) are White. Half (52%) are gay and bisexual men, or men who have sex with men.
Structure & Funding
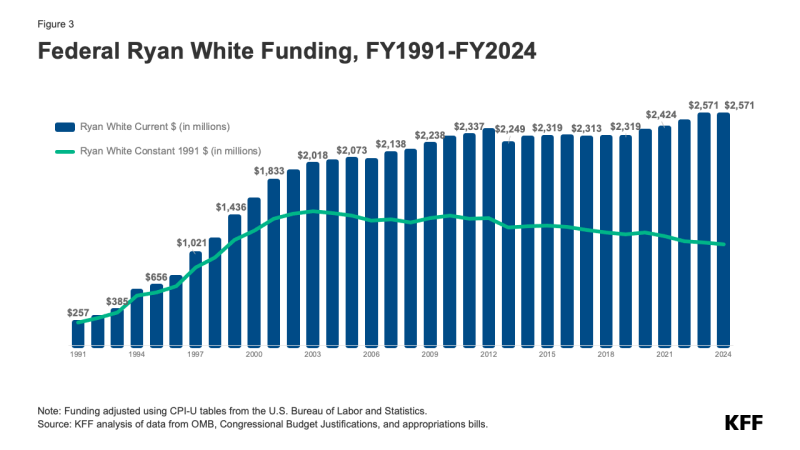
The Ryan White Program is the third largest source of federal funding for HIV care in the U.S., after Medicare and Medicaid. In FY24 funding for the program totaled $2.6 billion in FY 2024. Federal funding for the program, which is appropriated by Congress annually, began in FY1991 and increased significantly in the mid-1990s, primarily after the introduction of highly active antiretroviral therapy (HAART). For many years thereafter, funding continued to increase, first slowing down and then, eventually, flattening out. This trend began to shift modestly when new funding as part of the EHE Initiative marked the first significant increase to the program in many years. Since then, funding for EHE has risen substantially, increasing to $165 million in FY 2024. (See Figures 2 and 3)
However, funding for the Ryan White Program overall has not kept pace with inflation and does not necessarily meet the needs of a rising number of clients. (See Figure 3).
The Ryan White HIV/AIDS Program is composed of “Parts,” each with a different purpose and funded as a separate line item through annual Congressional appropriations. Funding is provided to states and territories (Part B) cities (Part A), and to providers, community-based organizations (CBOs), and other institutions (Parts C, D, and F), in the form of grants (described in detail in Table 1). In recognition of the varying nature of the HIV epidemic, grantees are given broad discretion to design key aspects of their programs, such as specifying client eligibility levels and service priorities. However, there are requirements, including that, unless granted a waiver, grantees must spend 75% or more of funds on “core medical services” under Parts A through C and that all state AIDS Drug Assistance Programs (ADAPs) must have a minimum formulary for medications
Ryan White HIV/AIDS Program & Care Outcomes
While many clients have gained coverage under the ACA, Ryan White continues to play a critical role as a safety net provider for those who remain uninsured or underinsured, helping to fill the gaps for clients with insurance, including assisting with insurance affordability and access to support services. Notably, Ryan White clients are significantly more likely to have sustained viral suppression compared to those without program support (68% v. 58%) and this pattern was observed across all coverage types. Viral suppression affords optimal health outcomes at the individual level and, because when an individual is virally suppressed they cannot transmit HIV, a significant public health benefit.
Key Issues
First enacted as an emergency measure, the Ryan White Program has grown to become a central component of HIV care in the U.S., playing a critical role in the lives of many low and moderate-income people with HIV. Looking ahead, there are several key issues facing the program that will be important to monitor, including:
- Future funding. As a federal grant program, funding is dependent on annual appropriations by Congress, and funding levels do not necessarily correspond to actual need (i.e. the number of people seeking services or the costs of services) and, as noted above, the program’s funding has not kept pace with inflation. As a result, not all states and communities have been able to meet the needs of people in their jurisdictions. Additionally, the Trump administration has proposed eliminating Part F of the program in its preliminary budget released in May 2025, and, in an earlier leaked budget document, proposed to eliminate the EHE. It has yet to be seen whether Congress will continue to appropriate funding to Part F or EHE and at what level.
- Structural changes and commitment to HIV. The Trump administration has also been actively scaling back the nation’s HIV response through staffing and funding cuts which have impacted HHS significantly, including HRSA/HAB. These changes could make program management, grant delivery, and data collection/analysis more challenging. This administration also plans to move HRSA to a new agency, the Administration for a Healthy America, but it is unclear how this will work and what impact it will have on the program.
- Major changes to the health policy landscape. The Trump administration and Congress are implementing or exploring significant, broader health policy changes that could impact the Ryan White Program. These include efforts to restrict access to gender affirming care, including in the Ryan White Program, as well as proposals to substantially change the Medicaid program which could lead to large coverage losses for individuals and/or cost-shifting to states and the Ryan White.


