Editorial Note: Originally published in June 2001, this resource is updated as needed to reflect the latest developments.
Key Facts
- HIV, the virus that causes AIDS (acquired immunodeficiency syndrome), is one of the world’s most serious health and development challenges. Approximately 40.8 million people are currently living with HIV, and tens of millions of people have died of AIDS-related causes since the beginning of the epidemic.
- Many people living with HIV or at risk for HIV infection do not have access to prevention, treatment, and care, and there is still no cure.
- In recent decades, major global efforts, PEPFAR (the President’s Emergency Plan for AIDS Relief, the U.S. government’s global HIV initiative), and the Global Fund to Fight AIDS, Tuberculosis and Malaria (Global Fund) have been mounted to address the epidemic, and despite challenges, significant progress has been made in addressing HIV. Current global health goals are to end AIDS as a public health threat by 2030.
- PEPFAR, in particular, has helped to change the trajectory of the HIV epidemic, and the U.S. is the single largest donor to international HIV efforts in the world, including the largest donor to the Global Fund. PEPFAR has directed over $130 billion toward HIV prevention, care, and treatment efforts since launched in 2003.
- Since the beginning of the second Trump administration, the U.S. global health response has undergone significant change, fundamentally altering the global health landscape and U.S. global HIV efforts, including through PEPFAR.
Global Response
HIV, the virus that causes AIDS (see box), has become one of the world’s most serious health and development challenges since the first cases were reported in 1981. Approximately 91.4 million people have become infected with HIV since the start of the epidemic.1 Today, there are approximately 40.8 million people currently living with HIV, and tens of millions of people have died of AIDS-related causes since the beginning of the epidemic.2
HIV: A virus that is transmitted through certain body fluids and weakens the immune system by destroying cells that fight disease and infection, specifically CD4 cells (often called T cells). Left untreated, HIV reduces the number of CD4 cells in the body, making it more difficult for the immune system to fight off infections and other diseases. HIV can lead to the development of AIDS, “acquired immunodeficiency syndrome,” also known as Advanced HIV Disease.3
AIDS: Advanced HIV Disease (AIDS), used to be seen as an issue of late diagnosis and treatment of HIV, and while that remains a concern, AIDS is now most common in people who have received treatment (antiretroviral therapy) but have stopped.4
Over the past two decades in particular, major global efforts have been mounted to address the epidemic, and significant progress has been made. The number of people newly infected with HIV, especially children, and the number of AIDS-related deaths have declined over the years, and the number of people with HIV receiving treatment increased to 31.6 million in 2024.5
Still, remaining challenges continue to complicate HIV control efforts. Many people living with HIV or at risk for HIV infection do not have access to prevention, treatment, and care, and there is still no cure. HIV primarily affects those in their most productive years, and it not only affects the health of individuals, but also impacts households, communities, and the development and economic growth of nations. Many of the countries hardest hit by HIV also face serious challenges due to other infectious diseases, food insecurity, and additional global health and development problems.
Latest Estimates6
- Global prevalence among adults (the percent of people ages 15-49 who are infected) has leveled since 2001 and was 0.7% in 2024, though prevalence was higher for certain groups of people, including key populations (i.e., men who have sex with men, sex workers, people who inject drugs, transgender people, and people in prisons).
- There were 40.8 million people living with HIV in 2024, up from 32 million in 2010, the result of continuing new infections and people living longer with HIV. Of the people living with HIV in 2024, 39.4 million were adults and 1.4 million were children under age 15.
- Although HIV testing capacity has increased over time, enabling more people to learn their HIV status, about one in eight people with HIV (13%) are still unaware they are infected.
- While there have been significant declines in new infections since the mid-1990s, there were still about 1.3 million new infections in 2024, or about 3,500 new infections per day. The pace of decline varies by age group, sex, race, and region, and progress is unequal within and between countries.7
- HIV remains a leading cause of death worldwide and the leading cause of death globally among women of reproductive age.8 However, AIDS-related deaths have declined, due in part to antiretroviral treatment (ART) scale-up. 630,000 people died of AIDS in 2024, a 55% decrease from 1.4 million in 2010 and a 70% decrease from the peak of 2.1 million in 2004. Among women and girls, mortality has declined by 58% since 2010.
- Sub-Saharan Africa,9 home to approximately two-thirds of all people living with HIV globally, is the hardest hit region in the world, followed by Asia and the Pacific. Latin America, Western and Central Europe and North America, as well as Eastern Europe and Central Asia are also heavily affected.
Affected/Vulnerable Populations
- Most HIV infections are transmitted heterosexually, although risk factors vary. In some countries, men who have sex with men, people who inject drugs, sex workers, transgender people, and prisoners are disproportionally affected by HIV.
- Women and girls represent over half (53%) of all people living with HIV worldwide, and HIV (along with complications related to pregnancy) is the leading cause of death among women of reproductive age.10 Gender inequalities, differential access to service, and sexual violence increase women’s vulnerability to HIV, and women, especially younger women, are biologically more susceptible to HIV. In many countries in sub-Saharan Africa, HIV incidence among adolescent girls and young women ages 15-24 is more than three times that among adolescent boys and young men.
- Young people in particular face barriers to accessing HIV and sexual and reproductive health services, including age-appropriate comprehensive sexuality education.
- Globally, in 2024, children accounted for 1.4 million people living with HIV; among children, there were 75,000 AIDS-related deaths and 120,000 new infections, the lowest number of new infections in children since the 1980s. Since 2010, new HIV infections among children have declined by 62%, though progress has stalled in recent years.
HIV & TB
HIV has led to a resurgence of tuberculosis (TB), particularly in Africa, and TB is a leading cause of death for people with HIV worldwide.11 In 2024, approximately 6% of new TB cases occurred in people living with HIV.12 However, between 2010 and 2024, TB deaths in people living with HIV declined substantially, largely due to the scale-up of joint HIV/TB services.13 (See the KFF fact sheet on TB.)
Prevention and Treatment14
Numerous prevention interventions exist to combat HIV, and new tools such as vaccines, are currently being researched.15
- Effective prevention strategies include behavior change programs, condoms, HIV testing, blood supply safety, harm reduction efforts for injecting drug users, and male circumcision.
- Additionally, recent research has shown that engagement in HIV treatment not only improves individual health outcomes but also significantly reduces the risk of transmission (referred to as “treatment as prevention” or TasP). Those with undetectable viral loads (known as being virally suppressed) have effectively no risk of transmitting HIV sexually.16
- Pre-exposure prophylaxis (PrEP) has also been shown to be an effective HIV prevention strategy in individuals at high risk for HIV infection. In 2015, the World Health Organization (WHO) recommended PrEP as a form of prevention for high-risk individuals in combination with other prevention methods.17 Further, in 2016, the U.N. Political Declaration on HIV/AIDS stated PrEP research and development should be accelerated, and in 2022, WHO released guidelines for the use of long-acting PrEP.18 Most recently, WHO released new guidelines recommending the use of a twice-a-year, long-acting injectable PrEP.19 These products signal an expansion and diversification of HIV prevention options.
- Experts recommend that prevention be based on “knowing your epidemic” (tailoring prevention to the local context and epidemiology), using a combination of prevention strategies, bringing programs to scale, and sustaining efforts over time. Access to prevention, however, remains unequal, and there have been renewed calls for the strengthening of prevention efforts, particularly as funding cuts from donors threaten progress on prevention.20
HIV treatment includes the use of combination antiretroviral therapy (ART) to attack the virus itself, and medications to prevent and treat the many opportunistic infections that can occur when the immune system is compromised by HIV. In light of research findings, WHO released a guideline in 2015 recommending starting HIV treatment earlier in the course of illness.21 Further, research on long-acting ART is currently underway.22
- Combination ART, first introduced in 1996, has led to dramatic reductions in morbidity and mortality, and access has increased in recent years, rising to 31.6 million people (77% of people living with HIV) in 2024.
- The percentage of pregnant and breastfeeding women receiving ART for the prevention of mother-to-child transmission of HIV increased to 84% in 2024, up from 49% in 2010.
- While access to ART among children has increased, treatment gaps still remain, and children are less likely than adults to receive ART; treatment coverage in children was 55% compared to 77% among adults in 2024.
- Approximately 73% of all people living with HIV are virally suppressed, which means they are likely healthier and less likely to transmit the virus. Viral suppression varies greatly by region, key population, age, and sex.
Global Goals
International efforts to combat HIV began in the first decade of the epidemic with the creation of the WHO’s Global Programme on AIDS in 1987. Over time, new initiatives and financing mechanisms have helped increase attention to HIV and contributed to efforts to achieve global goals; these include:
- the Joint United Nations Programme on HIV/AIDS (UNAIDS), which was formed in 1996 to serve as the U.N. system’s coordinating body and to help galvanize worldwide attention to HIV/AIDS; and
- the Global Fund to Fight AIDS, Tuberculosis and Malaria (Global Fund), which was established in 2001 by a U.N. General Assembly Special Session (UNGASS) on HIV/AIDS as an independent, international financing institution that provides grants to countries to address HIV, TB, and malaria (see the KFF fact sheet on the Global Fund).
The contributions of affected country governments and civil society have also been critical to the response. These and other efforts work toward achieving major global HIV/AIDS goals that have been set through:
- the Sustainable Development Goals (SDGs). Adopted in 2015, the SDGs aim to “end the AIDS epidemic,” or end AIDS as a public health threat,23 by 2030 under SDG Goal 3, which is to “ensure healthy lives and promote well-being for all at all ages.”24
- UNAIDS targets to end the epidemic by 2030. On World AIDS Day 2014, UNAIDS set targets aimed at ending the AIDS epidemic by 2030. To achieve this, countries are working toward reaching the interim “95-95-95” targets—95% of people living with HIV knowing their HIV status; 95% of people who know their HIV positive status on treatment; and 95% of people on treatment with suppressed viral loads—by 2025.25 These targets are successors to the earlier 90-90-90 targets for 2020, which were missed.26 Based on the 2024 data and trends (the latest data available),27 87% of people living with HIV knew their status; among those who knew their status, 89% were accessing treatment; and among those accessing treatment, 94% were virally suppressed.28 Additional interim “95-95-95” targets have also been set for 2025, which place a greater emphasis on social services and reducing stigma and discrimination to address inequalities that hinder the HIV response.29
Over the past decade, world leaders reaffirmed commitments to end AIDS by 203030 and adopted a Political Declaration with global commitments and targets for 2025 to address inequalities that impede the AIDS response.31 The next Global AIDS Strategy for the period 2026-2031 is currently under development.32
Global Resources
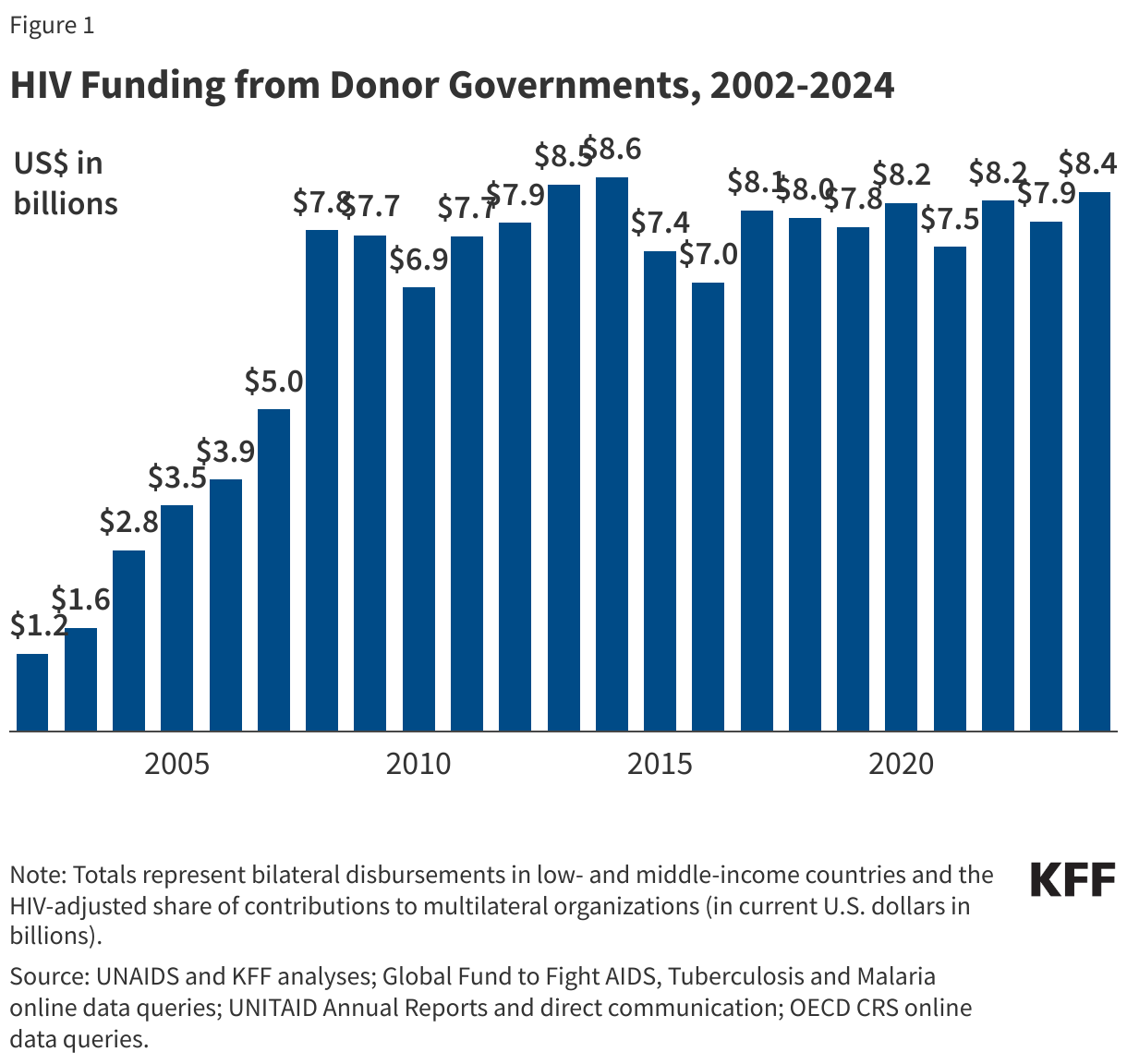
UNAIDS estimates that $18.7 billion was available from all sources (domestic resources, donor governments, multilaterals, and foundations) to address HIV in low- and middle-income countries in 2024. Of this, donor governments provided $8.4 billion (or 44% of total available resources) (see Figure 1).33 Other governments and organizations that contribute substantially to funding the global response include:
- hard-hit countries, which have also provided resources to address their epidemics;
- the Global Fund, which has approved over $29 billion for HIV efforts in more than 100 countries to date;34 and
- the private sector, including foundations and corporations, which also plays a major role (the Gates Foundation, for one, has committed more than $3 billion in HIV grants to organizations addressing the epidemic, as well as provided additional funding to the Global Fund).35
Looking ahead, UNAIDS estimates at least $21.9 billion annually will be needed to meet global targets to end AIDS as a global public health threat by 2030.36
U.S. Government Efforts
The U.S. has been involved in HIV efforts since the 1980s and is the single largest donor to international HIV efforts in the world, including the largest donor to the Global Fund.37 The U.S. first provided funding to address the global HIV epidemic in 1986. U.S. efforts and funding increased slowly over time through targeted initiatives to address HIV in certain countries in Africa, South Asia, and the Caribbean, but they intensified with the 2003 launch of the President’s Emergency Plan for AIDS Relief (PEPFAR), which brought significant new attention and funding to address the global HIV epidemic, as well as TB and malaria.38 Since the beginning of the second Trump administration, however, the U.S. global health response has undergone significant shifts, disruption, and retraction, fundamentally altering the global health landscape and U.S. global HIV efforts through PEPFAR in particular.
PEPFAR
Created in 2003, PEPFAR is the U.S. government’s global effort to combat HIV. PEPFAR has historically involved multiple U.S. departments, agencies, and programs, particularly USAID and CDC, although that has changed (see below for more details). The program had also been carried out in close coordination with host country governments and other organizations, including multilateral organizations such as the Global Fund and UNAIDS and non-governmental organizations, including civil society.39 U.S. bilateral HIV activities spanned more than 50 countries in Asia, West Africa, and the Western Hemisphere, with U.S. support for multilateral efforts reaching even more countries.40 (For more information, see the KFF fact sheet on PEPFAR.)
Since its creation, PEPFAR, which includes all bilateral funding for HIV as well as U.S. contributions to the Global Fund and UNAIDS, has totaled over $130 billion.41 For FY 2026, Congress appropriated $6 billion in total funding for PEPFAR, including $4.7 billion for bilateral HIV programs, $45 million for UNAIDS, and $1.25 billion for the Global Fund, matching funding levels for FY 2025.42 (For more details on historical appropriations for U.S. global HIV/AIDS efforts, see the KFF fact sheets on the U.S. Global Health Budget: Global HIV, Including PEPFAR and the U.S. Global Health Budget: The Global Fund, as well as the KFF budget tracker.)
Currently, PEPFAR faces significant change, brought on by a re-evaluation of U.S. foreign assistance, the dissolution of USAID (the main PEPFAR implementing agency), and the cancellation of most PEPFAR awards. While U.S. policymakers had been increasingly looking at when and how to transition PEPFAR services and financing to country governments, the Trump administration has sought to narrow PEPFAR’s scope and significantly accelerate this timeline. Per a new U.S. strategy, the America First Global Health Strategy, the administration is developing bilateral agreements with countries to integrate PEPFAR programming with other global health areas and is planning to scale down funding over the next few years, with country governments required to increasingly co-finance these activities. (See the KFF fact sheet on the status of PEPFAR for more information.)
Endnotes