What Could New Anti-Obesity Drugs Mean for Medicare?
This post has been updated to reflect the introduction of legislation in the 118th Congress in July 2023 that would authorize Medicare coverage of weight-loss medications.
A relatively new class of medications is generating excitement in the medical community and among people with obesity and others who struggle to lose weight. Initially approved to treat type 2 diabetes, these drugs, known as GLP-1 (glucagon-like peptide-1) agonists, including Novo Nordisk’s Ozempic and Wegovy (semaglutide) and Eli Lilly’s Mounjaro (tirzepatide), are also highly effective weight loss agents. But they are also expensive, and not covered by Medicare when used for weight loss. In this piece, we discuss Medicare coverage of obesity treatments, the potential cost implications if Medicare covers anti-obesity drugs, and how the Inflation Reduction Act could potentially address these cost concerns.
Medicare coverage of obesity services and treatments currently includes obesity screening, behavioral counseling, and bariatric surgery, but not drugs that are prescribed for weight loss. The 2003 law that established the Medicare Part D prescription drug benefit explicitly prohibits Part D plans from covering drugs used for weight loss, along with some other types of drugs, including agents used for cosmetic purposes or hair growth, fertility drugs, and drugs prescribed to treat sexual or erectile dysfunction.
With evidence that GLP-1s lead to significant weight loss and may offer additional health benefits to people with medical conditions exacerbated by excess weight, manufacturers of these drugs and other stakeholders are pushing for a change in law to allow coverage under Medicare. This push for expanded coverage under Medicare and other insurance plans is taking place despite some uncertainty about the risks associated with their use. A bipartisan group of lawmakers has introduced legislation, the Treat and Reduce Obesity Act, that would authorize Part D coverage of medications when used for the treatment of obesity or weight loss management in overweight individuals with related comorbidities.
Lifting the current law prohibition on coverage of weight-loss drugs would come at a cost to Medicare, given the high price and expected demand. Wegovy, for example, has an annual estimated net price of $13,600. According to a recent study, if 10% of Medicare beneficiaries with obesity use Wegovy, the annual cost to Medicare could be $13.6 billion (based on a 19% obesity rate from traditional Medicare diagnoses in 2021) to $26.8 billion (based on a 41.5% obesity rate from survey data for adults ages 60 and older). Higher take-up rates would mean higher Medicare spending. For context, total annual Part D spending in 2021 was $98 billion. Of note, these estimates do not account for potential reductions in Medicare spending that could occur if weight loss drugs reduce medical spending associated with other diseases, such as heart disease.
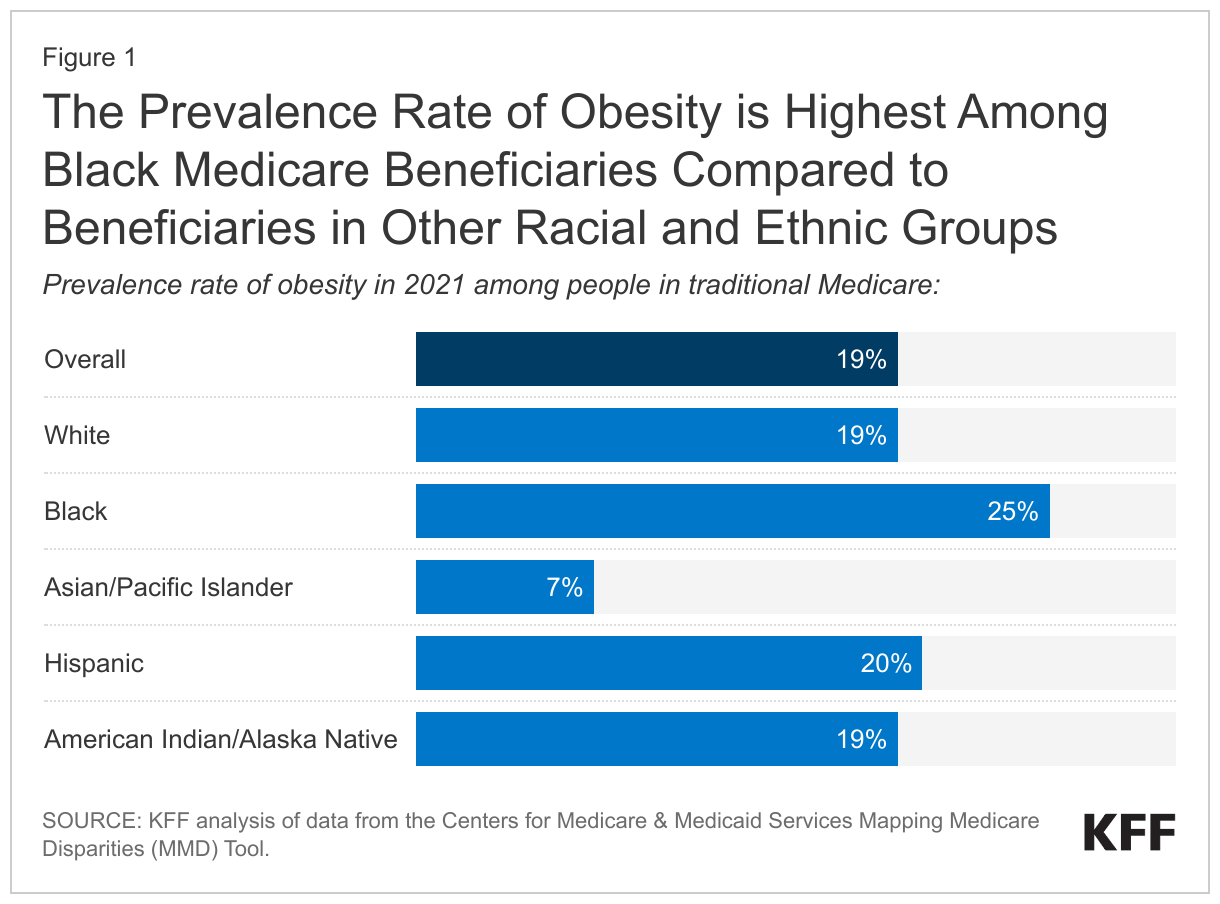
Maintaining the status quo, however, could raise both access and equity concerns, given the current price. The annual cost of Wegovy is unaffordable for many Medicare beneficiaries. Moreover, the rate of obesity is highest among Black Medicare beneficiaries compared to beneficiaries in other racial and ethnic groups (Figure 1). Without Medicare coverage, Black beneficiaries may be least able to afford these new drugs, given their substantially lower incomes and assets than White beneficiaries.
The Inflation Reduction Act of 2022 could help lessen the cost impact of weight loss drugs on Medicare and Part D enrollee out-of-pocket spending. If covered by Medicare, weight loss drugs could be among the limited number of drugs that will be subject to Medicare’s new drug price negotiation authority, but not for several years. At the earliest, a negotiated price for semaglutide, for example, would not be available before 2027 (based on FDA approval in late 2017) and not before 2031 for tirzepatide (based on FDA approval in 2022). Another provision would subject weight-loss drugs to the new law’s inflation rebate that aims to limit annual increases in drug prices. A third provision of the law will cap out-of-pocket Medicare Part D spending for covered drugs at $2,000 in 2025, which would certainly help to make these drugs more affordable. But even paying $2,000 out of pocket would still be beyond the reach of many people with Medicare who live on modest incomes.
To limit the potential cost impact to Medicare, lawmakers could consider shortening the period between FDA approval and the year negotiated prices take effect, as proposed in the Biden Administration’s FY2024 budget, but adopting this change to the newly-launched negotiation program seems unlikely in the current political climate. If the underlying Part D law is not changed, adding Medicare coverage for obesity drugs could possibly be achieved through other pathways, such as a demonstration program through the Centers for Medicare & Medicaid Innovation or Section 402 authority.
The availability of effective weight-loss drugs has the potential to be transformative for people who struggle with obesity and obesity-related medical conditions, but without insurance coverage, access to these relatively high-priced drugs will be limited to those who can afford them. While competition among GLP-1 medications could have a moderating effect on launch prices, the combination of intense demand and high prices for these treatments is likely to place tremendous pressure on Medicare spending if coverage is authorized, even in the wake of the prescription drug provisions in the Inflation Reduction Act. A decision to cover weight-loss drugs under Medicare could have ripple effects for employers and other payers if they follow Medicare’s lead.
This work was supported in part by Arnold Ventures. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.