Community Health Centers Are A Key Source of COVID-19 Rapid At-Home Self-Tests For Hard-To-Reach Groups
Across the country, many Americans are having difficulty accessing COVID-19 tests amid the ongoing surge in cases driven by the new Omicron variant. The Biden administration has taken a multi-pronged approach to increase access to COVID-19 tests that includes allowing every household to order up to four free at-home tests, increasing testing in schools, expanding the number of federal testing sites, and requiring private insurers to cover the costs of at-home tests. In addition, under the American Rescue Plan Act (ARPA), state Medicaid and Children’s Health Insurance Program (CHIP) programs are required to cover FDA-authorized at-home COVID-19 tests without cost-sharing. As part of an effort to promote equitable access to tests, the administration also launched a testing supply program in late December that has set aside 25 million rapid at-home self-test kits (or 50 million tests because there are two tests per kit) for distribution by the more than 1,300 community health centers.
Health centers are a national network of safety-net primary care providers that have played a significant role in the COVID-19 response, including facilitating equitable access to vaccinations. To date, health centers have administered nearly 19 million doses of the vaccine, of which more than two-thirds (68%) have gone to people of color. While health centers make up a relatively small proportion (4%) of total vaccinations administered nationally (roughly 530 million doses administered as of mid-January), health centers’ role has focused on vaccinating and providing primary care to some of the hardest-to-reach populations.
Like the vaccine program, the objective of the health center COVID-19 testing supply program is to provide COVID-19 rapid self-tests to communities that are currently facing difficulties accessing COVID-19 tests. Under the program, health centers will be distributing self-tests to patients and community members, with a focus on populations at greatest risk from adverse outcomes related to COVID-19. While the 25 million test kits set aside by the administration for the program is notably less than 28.6 million patients served by health centers in 2020, the federal government expects the number of test kits for the program to last through May 2022. The self-tests provided through the new federal program should supplement other testing services that health centers have been providing throughout the pandemic. Beyond the supply of free self-tests available through the new program, health center patients with Medicaid, CHIP, or other coverage may have other options to access these free tests through pharmacies or other sources and health centers may be able to seek reimbursement from public and private payers, including Medicaid/CHIP reimbursement, for self-tests ordered for patients covered by these programs depending on state rules.
Data collected through a biweekly survey conducted by the Health Resources and Services Administration (HRSA) show that health center patient demand for COVID-19 tests has increased recently (as it has across the US). In the two weeks ending January 14, 2022, health centers reported administering an average of 794 PCR and antigen tests per responding health center, more than double the number reported in the December 17, 2021 survey. Notably, roughly two-thirds (64%) of tests were provided to people of color. These data indicate that supplying health centers with self-test kits should further increase access to testing in underserved communities during the current period of high demand.
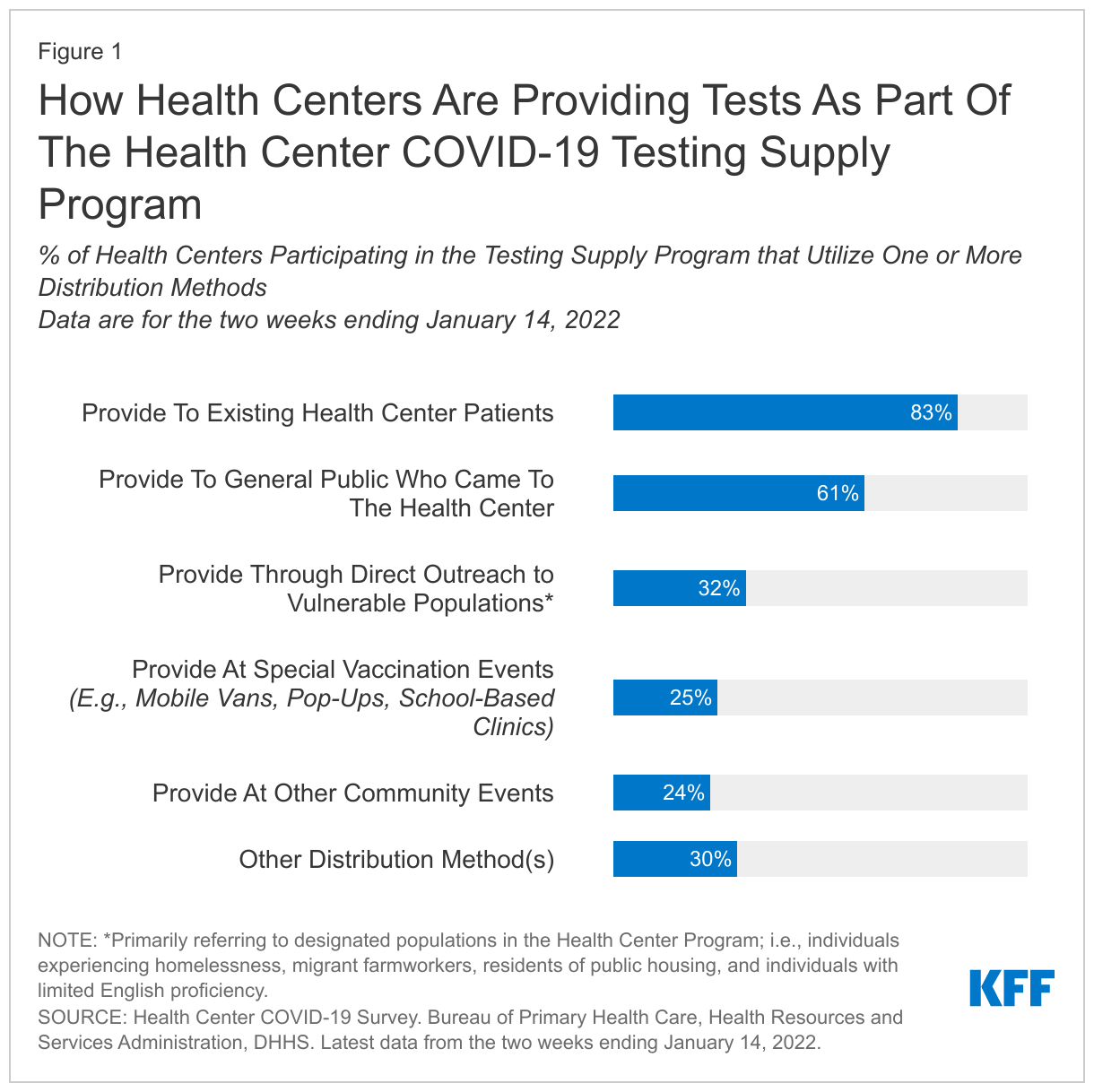
HSRA’s most recent biweekly survey also shows how health centers are distributing the self-test kits through the federal partnership program as of early January. In the two weeks ending January 14, 27% of responding health centers reported distributing 1.08 million kits to community members (or roughly 2 million tests because each kit includes two tests). That number is expected to increase as more health centers register for the program and begin placing orders. Health centers also reported that they distributed the self-tests in a variety of ways (Figure 1). Most reported passing out the test kits to existing health center patients (83%) or to others in the community who came to the health center (61%). Roughly one-third (32%) of health centers reported distributing tests through direct outreach to underserved populations, such as individuals experiencing homelessness, migrant farmworkers, and low-income populations.
Looking ahead, health centers are expected to participate in other efforts to slow the spread and lessen the severity of COVID-19 disease. HRSA has begun the initial phase for a separate program that supplies health centers with oral antiviral pills to treat patients who recently tested positive for COVID-19 to lower their risk of more serious illness. The initial phase of this program includes 200 health centers; HRSA will include more participants as supply of the antivirals increases. In the most recent biweekly survey of health centers (as of January 14), 59 health centers had begun distributing antiviral pills through the new program, a number that we expect will grow as the program ramps up. In addition to supplying self-tests and antiviral pills, the administration recently began distributing N95 masks through community health centers alongside retail pharmacies, another program that should be ramping up in coming weeks.
Health centers are growing their role in the nation’s pandemic response efforts (in addition to state and local efforts) and have been important players in coordinating response efforts quickly and more equitably. Taken together, health centers’ federal partnership programs to administer vaccinations, distribute self-test kits, provide free N95 masks (in the coming weeks), and eventually prescribe and distribute oral antivirals should strengthen health centers’ ability to help prevent, detect, and treat COVID-19 in underserved communities.