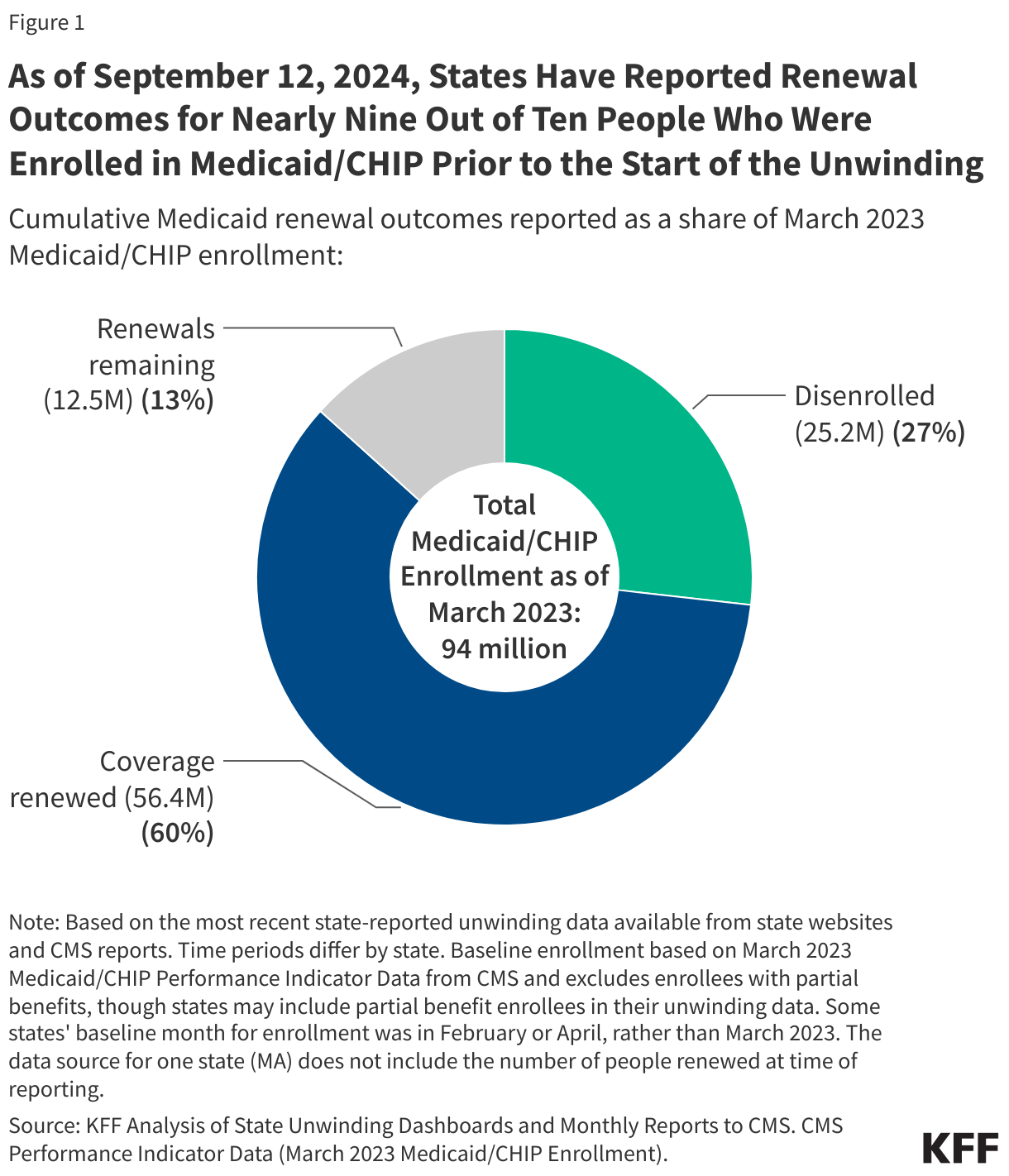
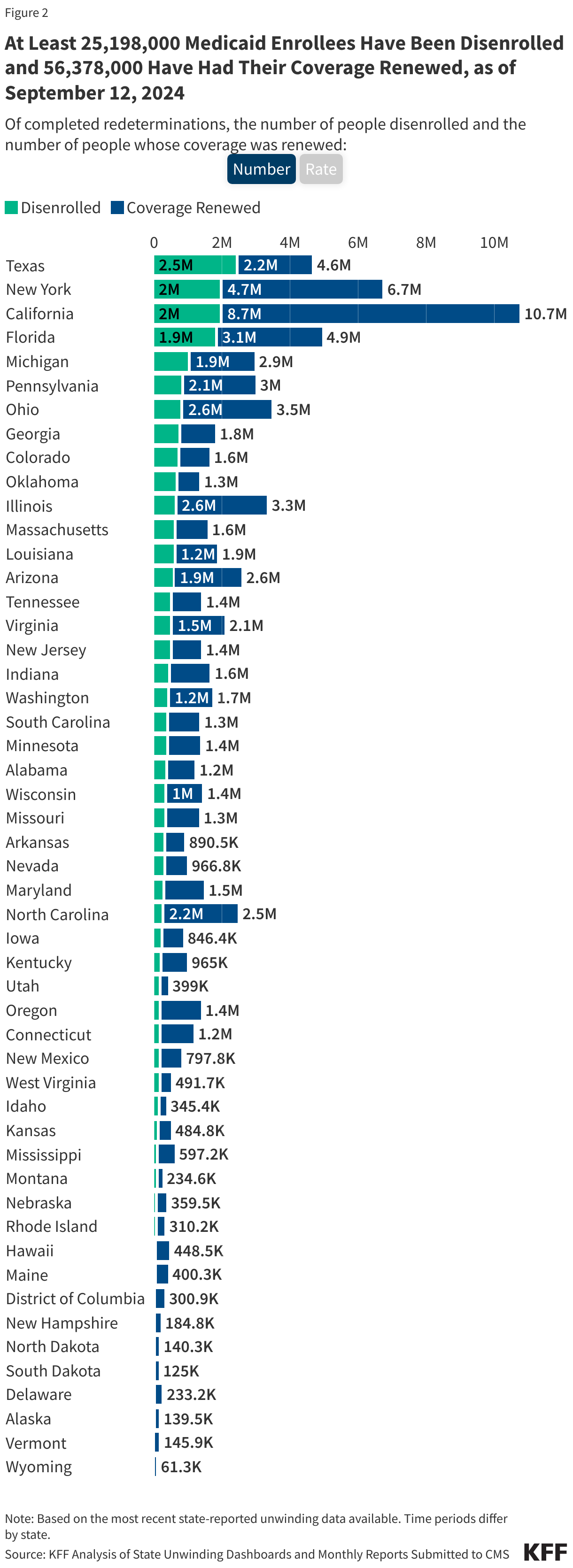
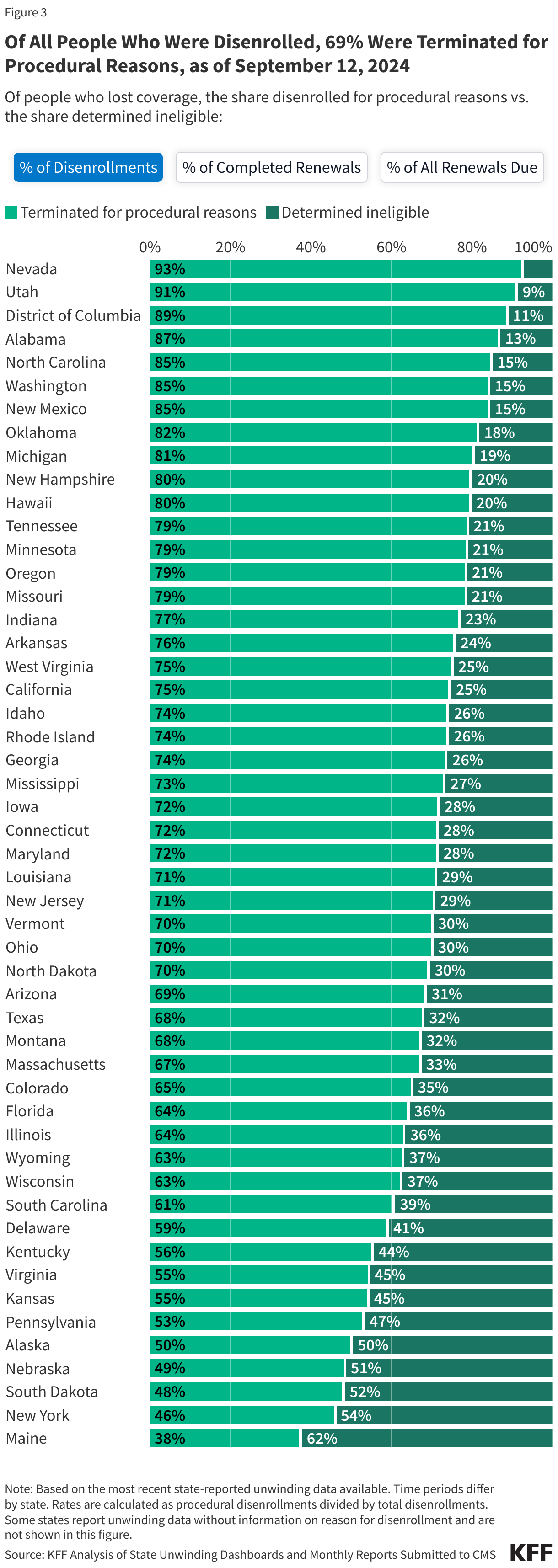
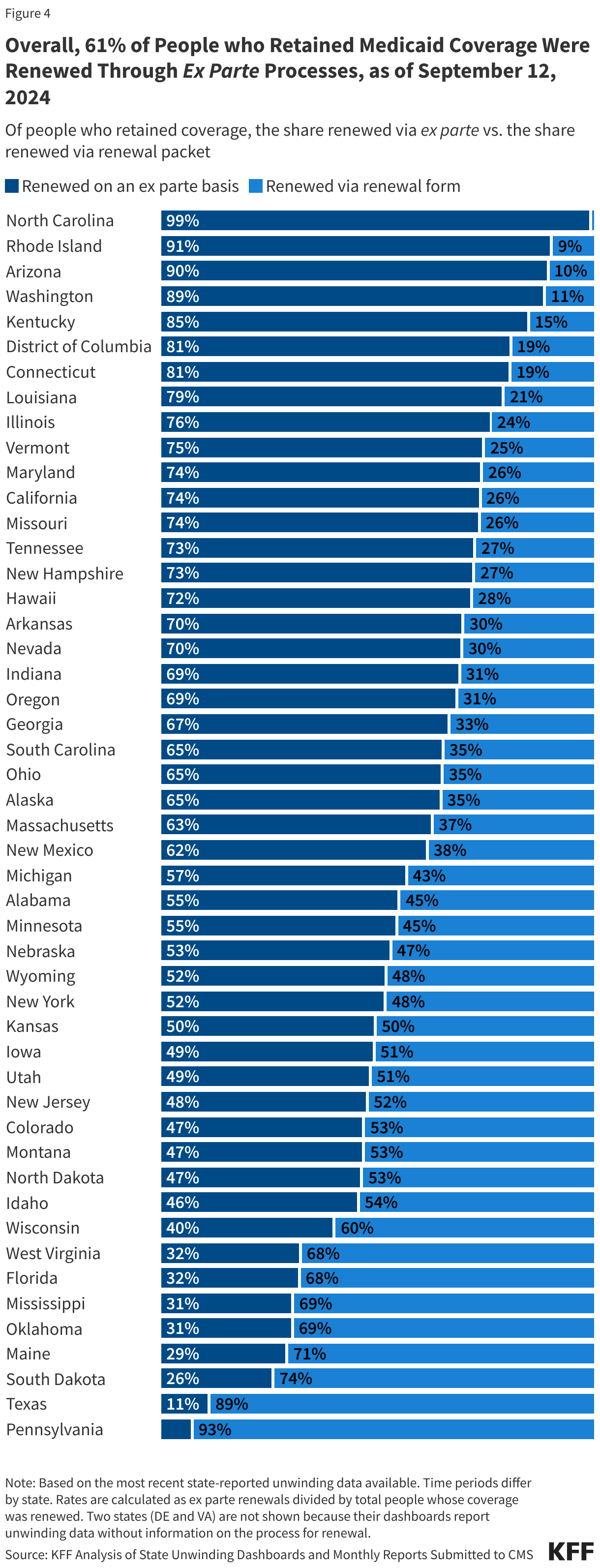
KFF Health Tracking Poll: The Public’s View of Immigration Enforcement Activities in Health Care Settings
Findings
Soon after taking office, President Trump reversed longstanding policy that had protected against immigration enforcement in “sensitive locations” including health care facilities, schools, and places of worship. Following this recission, as well as an overall increase in enforcement activity, there have been reports of Immigration and Customs Enforcement (ICE) agents showing up at hospitals and other health care facilities. ICE presence at and around health care facilities has led to growing concerns among health care providers about the impacts on the health and safety of the community, including adults and children going without care. These actions sit against a backdrop of broad increased enforcement activity and policies restricting access to health coverage and care for immigrant families. Research shows that this environment has negative impacts on the mental and physical health of immigrant families, including the millions of U.S. citizen children living in them, as well as broader economic effects on communities.
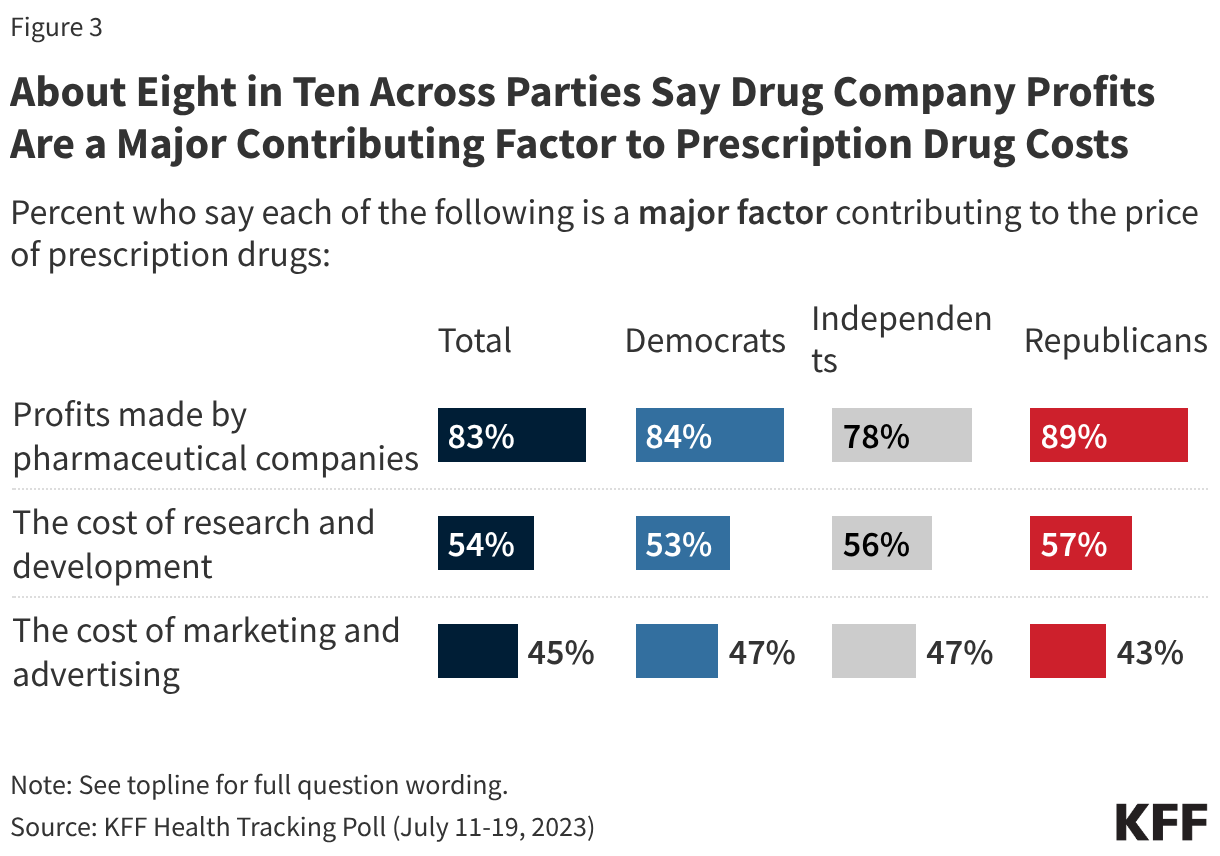
KFF has conducted multiple surveys examining immigrants’ health and experiences amid the policy environment as part of its Surveys of Immigrants conducted since 2023. New data from the KFF Health Tracking Poll provides insights into how the general public views the Trump administration’s immigration enforcement polices in health care settings. Overall, it shows the public is split along partisan lines on the administration’s approach to immigration enforcement in health care facilities. Democrats and independents express concern over the administration’s tactics, whereas Republicans are largely not concerned. Majorities of Democrats and independents say they do not think ICE or Customs and Border Control (CBP) should be allowed to arrest or detain people in and around health care facilities, whereas a majority of Republicans say they should be allowed. On each of these measures, Republicans and Republican-leaning independents who support President Trump’s MAGA movement are much more likely to support these tactics or say they are not concerned than non-MAGA supporters.
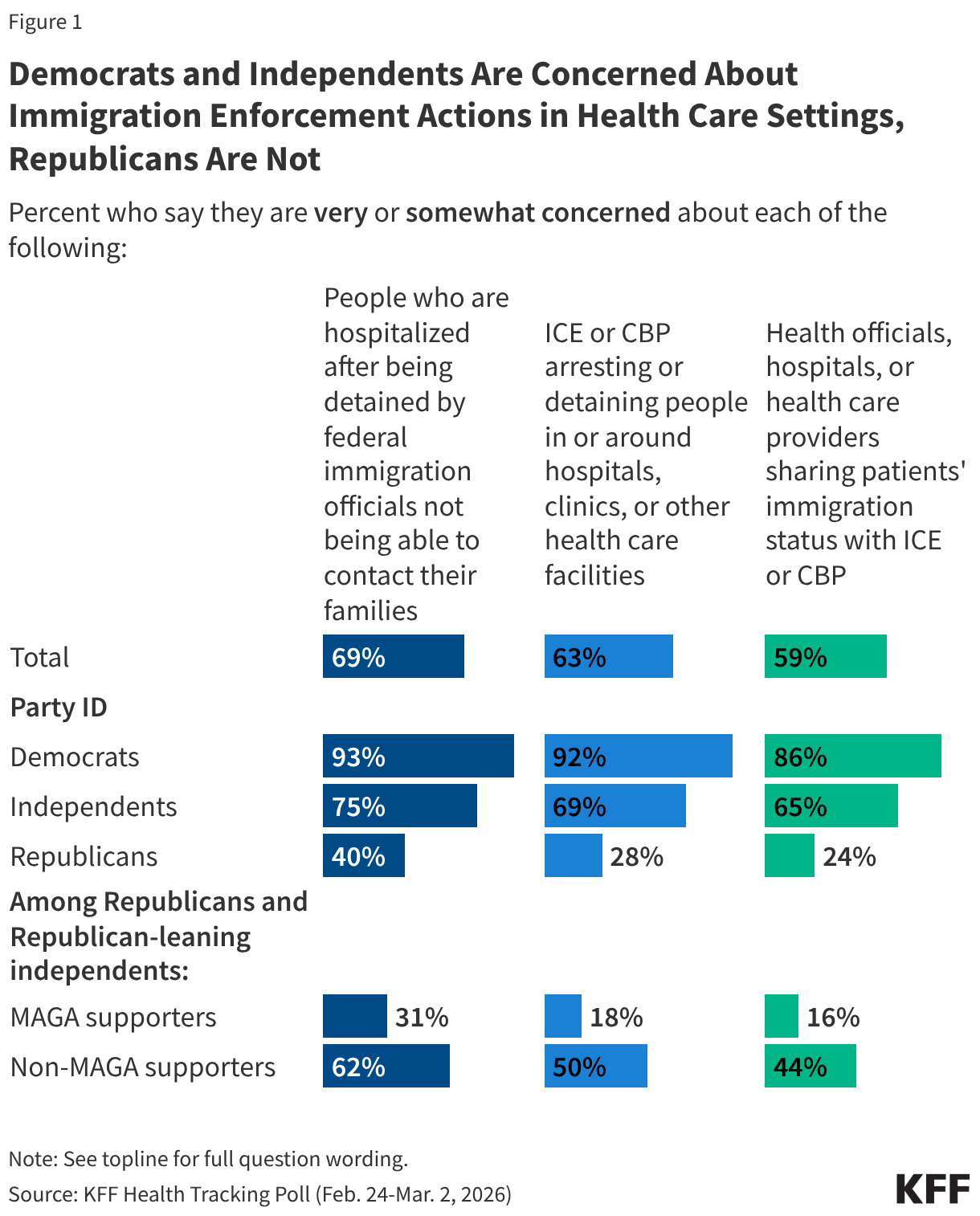
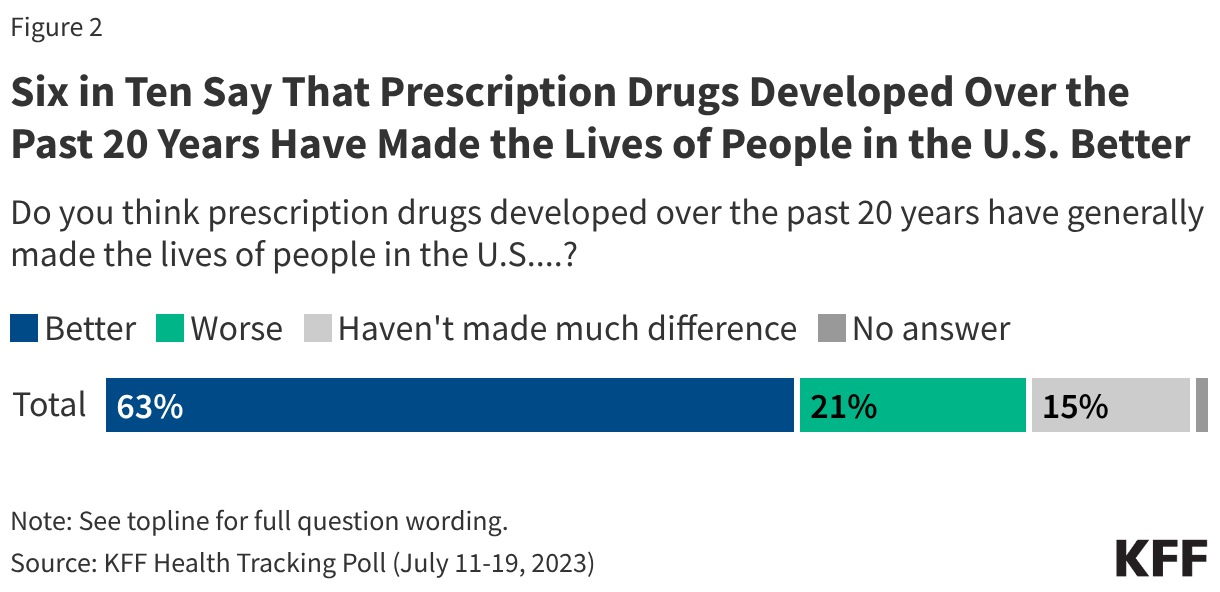
A majority of the public say they are concerned about the Trump administration’s tactics and immigration enforcement activities in or around health care settings. This includes majorities of the public who say they are “very” or “somewhat” concerned about people who have been hospitalized after being detained by federal immigration officials not being allowed to contact their families (69%), ICE or CBP arresting or detaining people in health care settings (63%), and health care providers and officials sharing patients’ immigration status with ICE or CBP (59%).
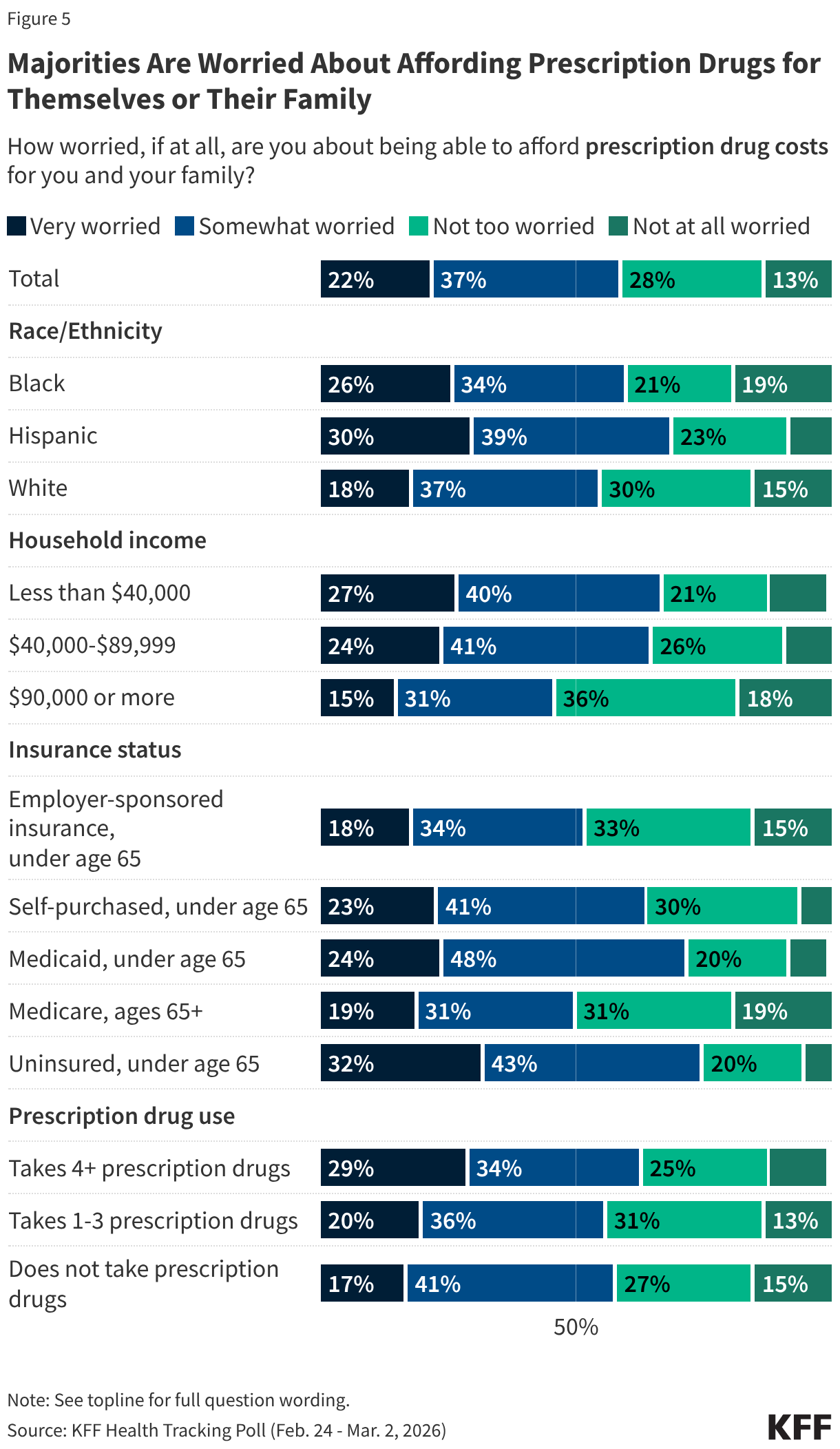
While majorities of the public are concerned about each of these tactics, there are big differences by partisanship, with Democrats and independents more likely than Republicans, especially MAGA Republicans, to say they are concerned about these. For example, about nine in ten Democrats and three-quarters of independents say they are concerned about people who have been hospitalized after detainment not being allowed to contact their families, whereas four in ten Republicans express concern. There are larger divides by partisanship on the two other tactics asked about, with Democrats about three times as likely as Republicans to say they are concerned about ICE or CBP detaining people in or around hospitals, clinics, or other health care facilities (92% vs. 28%) and health officials, hospitals, or health care providers sharing patients’ immigration status with ICE or CBP (86% vs. 24%). For each of these immigration enforcement tactics, two-thirds or more of independents say they are “very” or “somewhat” concerned about them. There are also differences among Republicans and Republican-leaning independents who support MAGA versus those who do not support the MAGA movement, with non-MAGA supporters at least twice as likely to say they are concerned about these tactics than MAGA supporters.
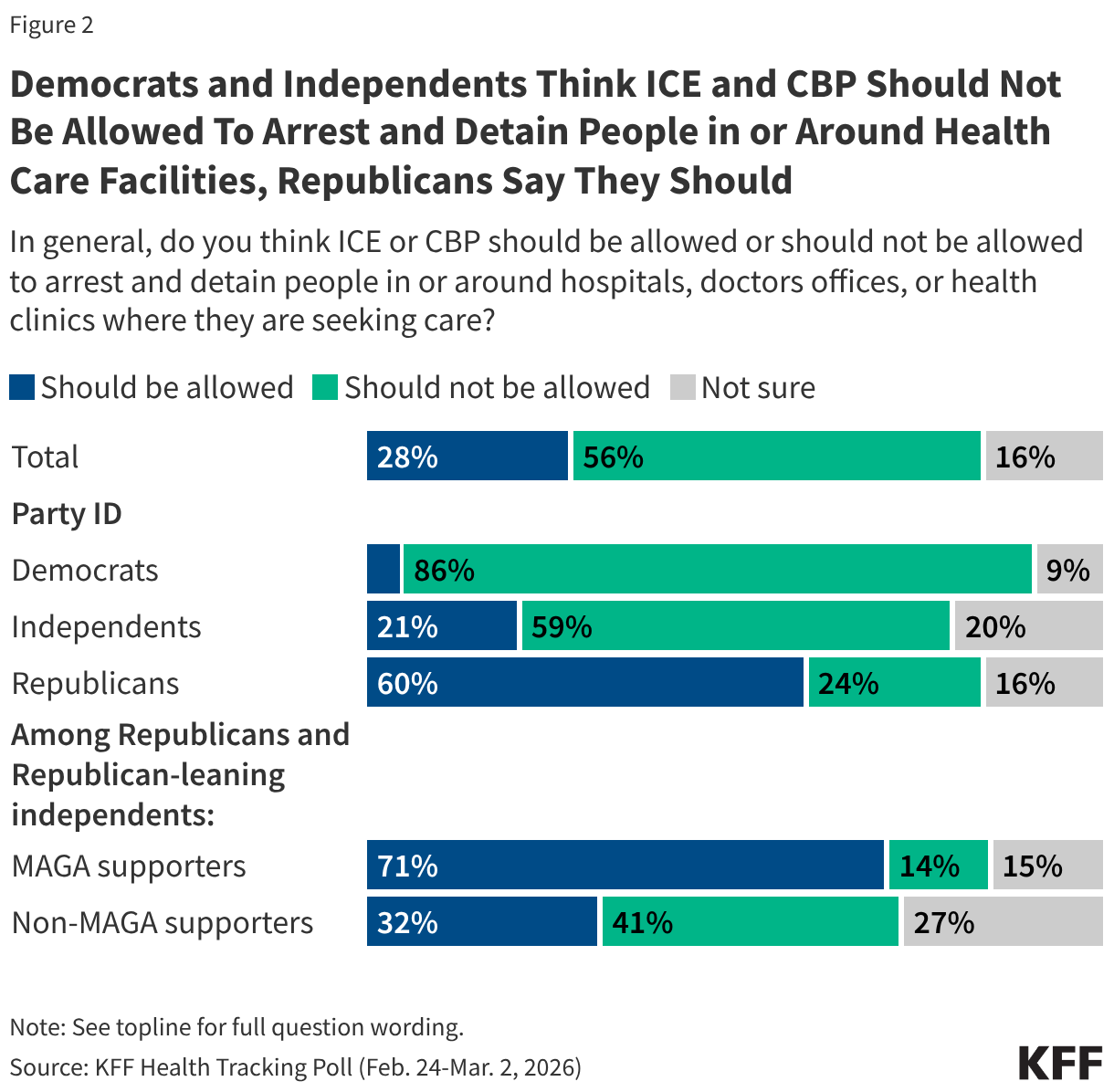
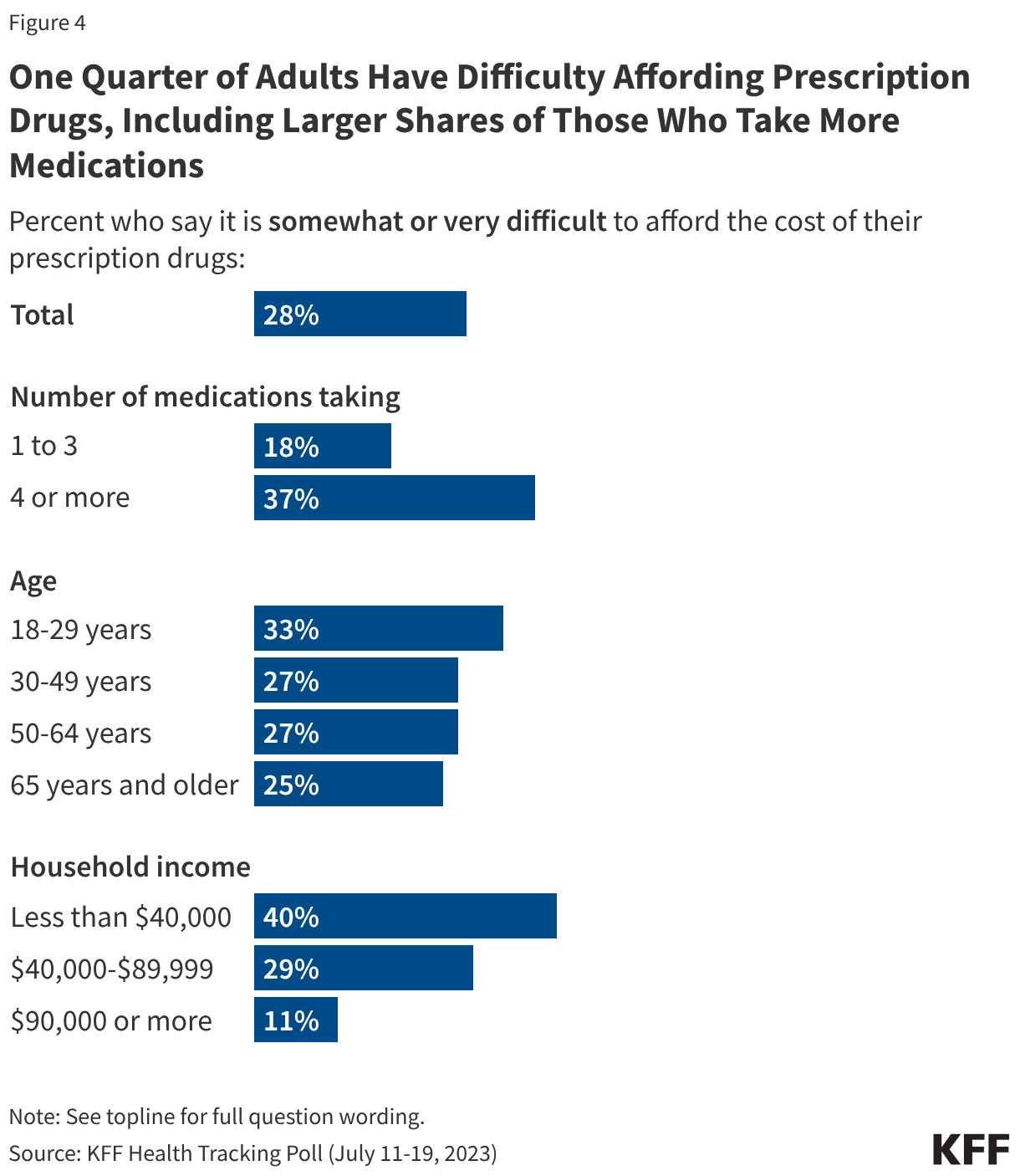
A majority of the public (56%) says ICE and CBP should not be allowed to arrest and detain people in or around hospitals, doctors’ offices or health clinics where they are seeking care, while about three in ten (28%) say they should be allowed, and one in six (16%) are “not sure.” There are stark divides by partisanship on this tactic. Nearly nine in ten (86%) Democrats and about six in ten (59%) independents say immigration enforcement activity should not be allowed in or around health care facilities, whereas six in ten Republicans say it should be allowed. But again among Republicans on this question, MAGA supporters are much more likely than non-MAGA supporters to say this tactic should be allowed (71% vs. 32%), and four in ten (41%) non-MAGA Republicans say it should not be allowed.
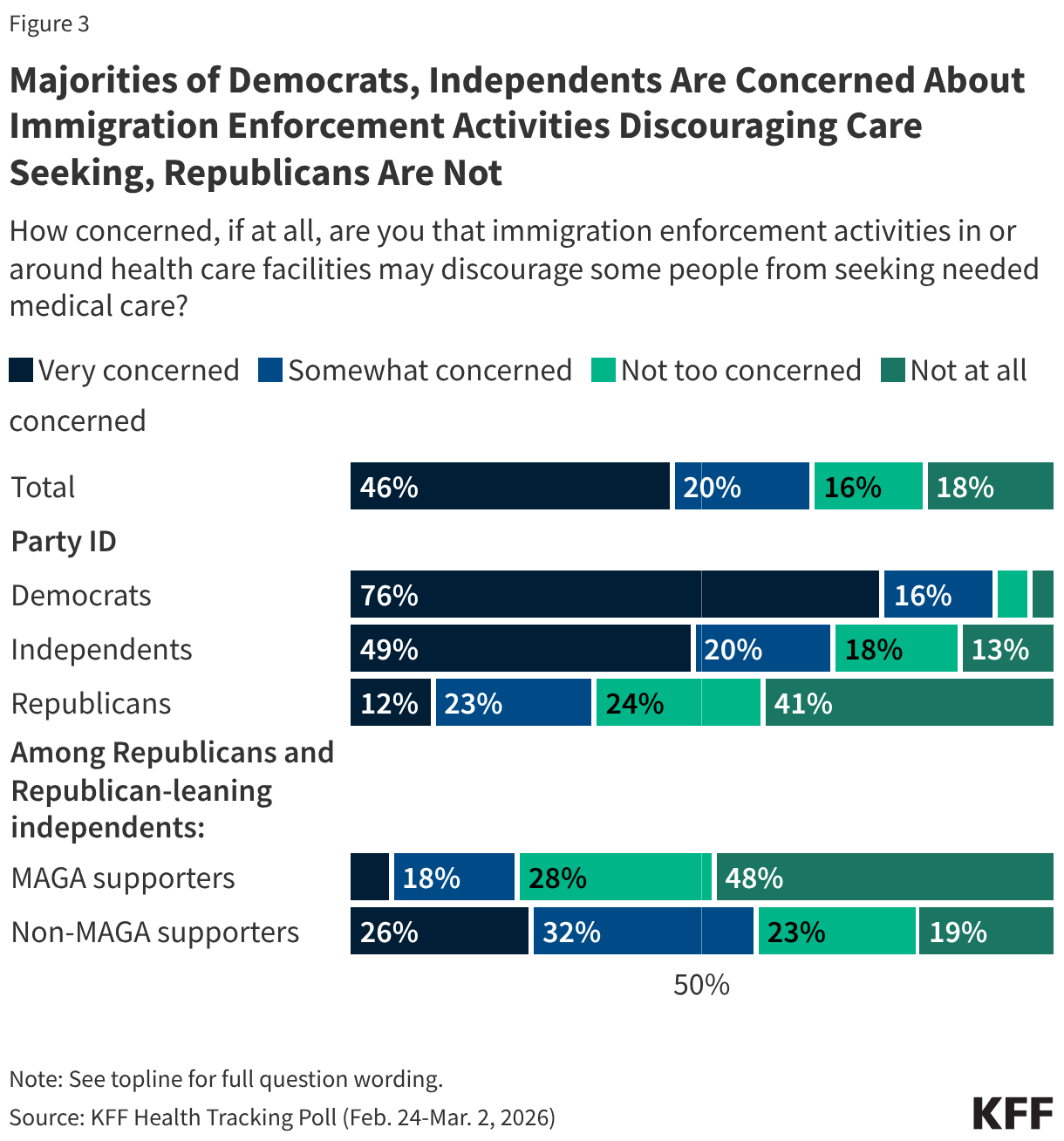
Two-thirds (65%) of adults say they are concerned that immigration enforcement activities in or around health care facilities may discourage some people from seeking needed medical care, while a third (34%) say they are not concerned. There are large differences by party identification on this topic, with about nine in ten Democrats (91%) and about seven in ten (69%) independents saying they are concerned about this, including three-quarters (76%) of Democrats who say they are “very concerned.” In contrast, most (65%) Republican say they are not concerned about this, including four in ten (41%) who say they are “not at all concerned.” However, there are stark divides by MAGA support among Republicans, with a majority of MAGA Republicans (76%) saying they are not concerned and a majority of non-MAGA Republicans (58%) saying they are concerned. Since the rescission of the “sensitive areas” policy and ramp up of ICE activity across the country, there have been reports of immigrants avoiding seeking medical care, which can lead to negative and costly health outcomes. Even prior to the recent uptick in public enforcement activity in areas such as Minneapolis and other parts of the country, the 2025 KFF/New York Times Survey of Immigrants found that many immigrants said they were avoiding seeking medical care due to concerns about drawing attention to someone’s immigration status. As of Fall 2025, 14% of immigrants overall said they have avoided seeking medical care since January 2025, rising to nearly half (48%) of likely undocumented immigrants.
Methodology
This KFF Health Tracking Poll/KFF Tracking Poll on Health Information and Trust was designed and analyzed by public opinion researchers at KFF. The survey was conducted February 24 – March 2, 2026, online and by telephone among a nationally representative sample of 1,343 U.S. adults in English (n=1,268) and in Spanish (n=75). The sample includes 1,019 adults (n=62 in Spanish) reached through the SSRS Opinion Panel either online (n=995) or over the phone (n=24). The SSRS Opinion Panel is a nationally representative probability-based panel where panel members are recruited randomly in one of two ways: (a) Through invitations mailed to respondents randomly sampled from an Address-Based Sample (ABS) provided by Marketing Systems Groups (MSG) through the U.S. Postal Service’s Computerized Delivery Sequence (CDS); (b) from a dual-frame random digit dial (RDD) sample provided by MSG. For the online panel component, invitations were sent to panel members by email followed by up to three reminder emails.
Another 324 (n=13 in Spanish) adults were reached through random digit dial telephone sample of prepaid cell phone numbers obtained through MSG. Phone numbers used for the prepaid cell phone component were randomly generated from a cell phone sampling frame with disproportionate stratification aimed at reaching Hispanic and non-Hispanic Black respondents. Stratification was based on incidence of the race/ethnicity groups within each frame. Among this prepaid cell phone component, 142 were interviewed by phone and 182 were invited to the web survey via short message service (SMS).
Respondents in the prepaid cell phone sample who were interviewed by phone received a $15 incentive via a check received by mail or an electronic gift card incentive. Respondents in the prepaid cell phone sample reached via SMS received a $10 electronic gift card incentive. SSRS Opinion Panel respondents received a $5 electronic gift card incentive (some harder-to-reach groups received a $10 electronic gift card). In order to ensure data quality, cases were removed if they failed two or more quality checks: (1) attention check questions in the online version of the questionnaire, (2) had over 30% item non-response, or (3) had a length less than one quarter of the mean length by mode. Based on this criterion, 1 case was removed.
The combined cell phone and panel samples were weighted to match the sample’s demographics to the national U.S. adult population using data from the Census Bureau’s 2024 Current Population Survey (CPS), September 2023 Volunteering and Civic Life Supplement data from the CPS, and the 2025 KFF Benchmarking Survey with ABS and prepaid cell phone samples. The demographic variables included in weighting for the general population sample are gender, age, education, race/ethnicity, region, civic engagement, frequency of internet use and political party identification. The weights account for differences in the probability of selection for each sample type (prepaid cell phone and panel). This includes adjustment for the sample design and geographic stratification of the cell phone sample, within household probability of selection, and the design of the panel-recruitment procedure.
The margin of sampling error including the design effect for the full sample is plus or minus 3 percentage points. Numbers of respondents and margins of sampling error for key subgroups are shown in the table below. For results based on other subgroups, the margin of sampling error may be higher. Sample sizes and margins of sampling error for other subgroups are available on request. Sampling error is only one of many potential sources of error and there may be other unmeasured error in this or any other public opinion poll. KFF public opinion and survey research is a charter member of the Transparency Initiative of the American Association for Public Opinion Research.
| Group | N (unweighted) | M.O.S.E. |
|---|---|---|
| Total | 1,343 | ± 3 percentage points |
| Party ID | ||
| Democrats | 449 | ± 6 percentage points |
| Independents | 449 | ± 6 percentage points |
| Republicans | 373 | ± 6 percentage points |
| MAGA Republicans/Republican leaning independents | 334 | ± 6 percentage points |
| Used AI for health information or advice in the past year | 458 | ± 6 percentage points |
| Used for physical health information | 407 | ± 6 percentage points |
| Used for mental health information | 234 | ± 8 percentage points |