Assessing the Role of Treaties, Conventions, Institutions, and Other International Agreements in the Global COVID-19 Response: Implications for the Future
Key Findings
The COVID-19 pandemic has tested global health governance and international law in unprecedented ways, revealing weaknesses and gaps in the existing global health security architecture and fueling debate over how to strengthen global governance of disease. In December of 2021, the World Health Assembly (WHA) agreed to launch a process to develop a new international agreement on pandemic prevention, preparedness and response, with at least an initial outcome expected in 2024. To date, there has been no detailed examination of existing global health treaties, commitments, partnerships, organizations, and other global health agreements (hereafter referred to as “agreements”) to identify gaps and potential areas of collaboration moving forward. To help inform these discussions, we reviewed existing agreements to assess their potential role in pandemic preparedness and response efforts, and whether they were used to respond to COVID-19. Key findings are as follows:
- 71 agreements were identified as having a role in global health. These agreements, some of which are more than 100 years old and others that were established only recently, address a wide range of areas including diseases (HIV, malaria, tuberculosis, etc.), vulnerable populations (children, women, refugees, etc.), the environment, and trade/intellectual property, among others.
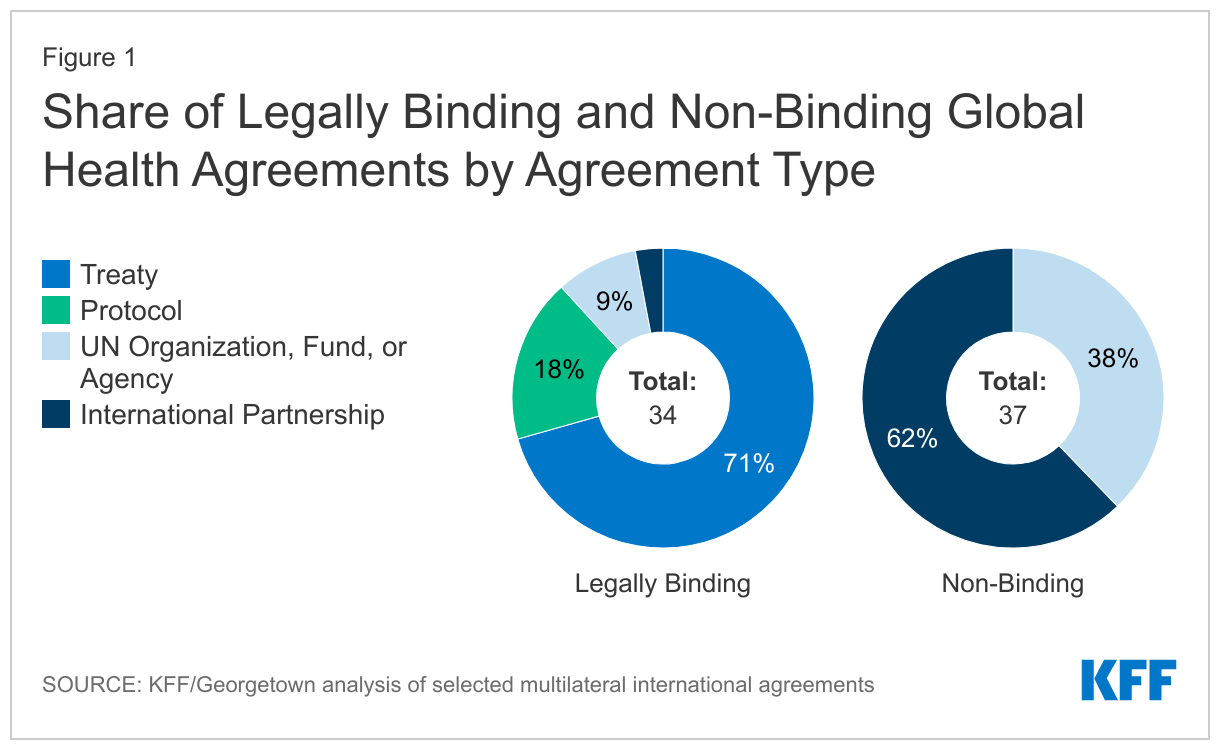
- Nearly half (34) of these agreements are binding under international law. Legally binding agreements are those intended to create enforceable legal obligations in the international arena on their parties.1 The 34 binding agreements include treaties, protocols (which are associated with existing treaties), and the terms of memberships in some United Nations (UN) entities such as the World Health Organization (WHO). The remaining 37 agreements, while not legally binding, may confer obligations and commitments upon participating countries.
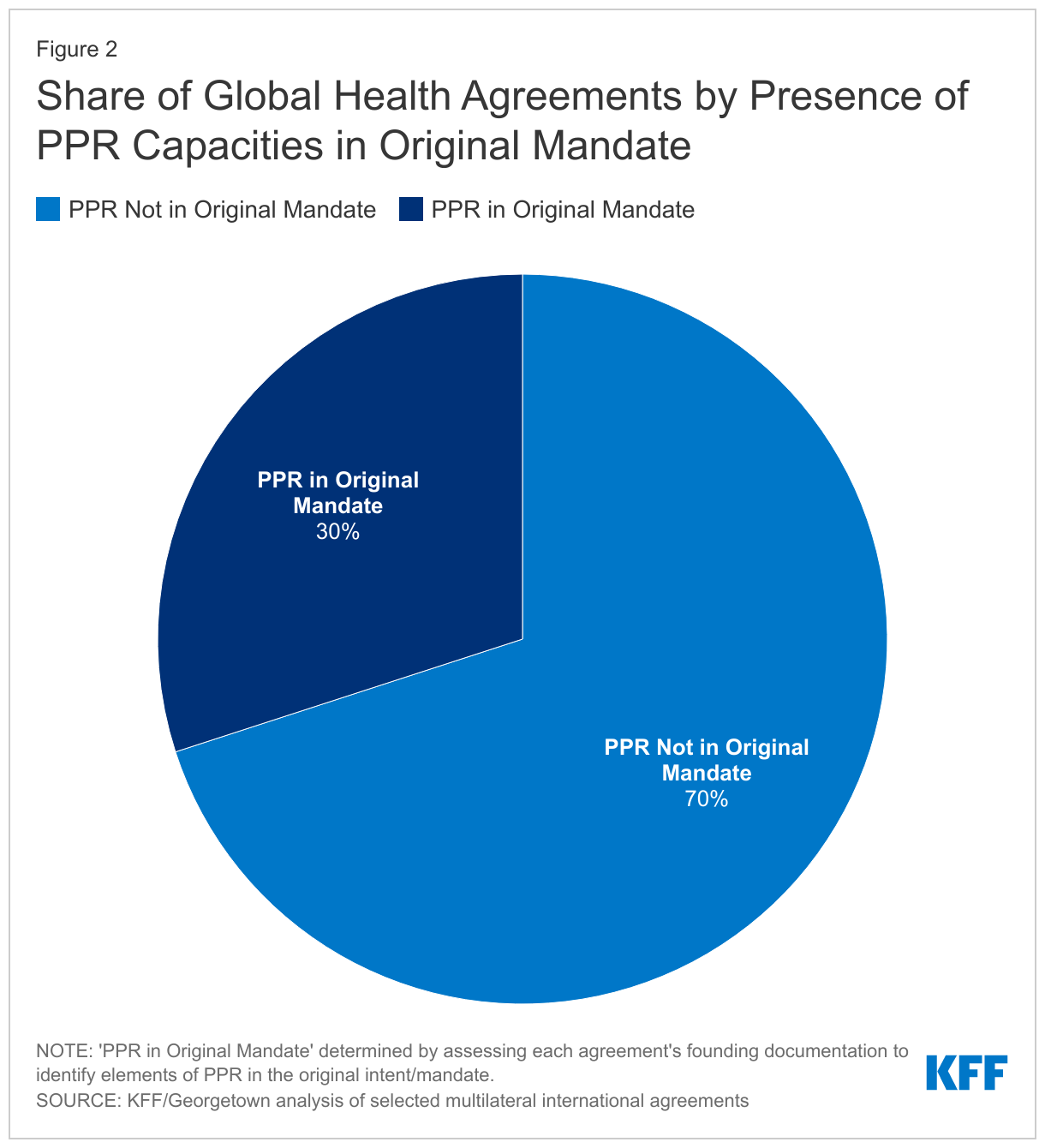
- Less than one-third (21 or 30%) of the agreements have pandemic preparedness and response (PPR) as part of their original mandate. Of these 21 agreements, 9 are legally binding under international law.
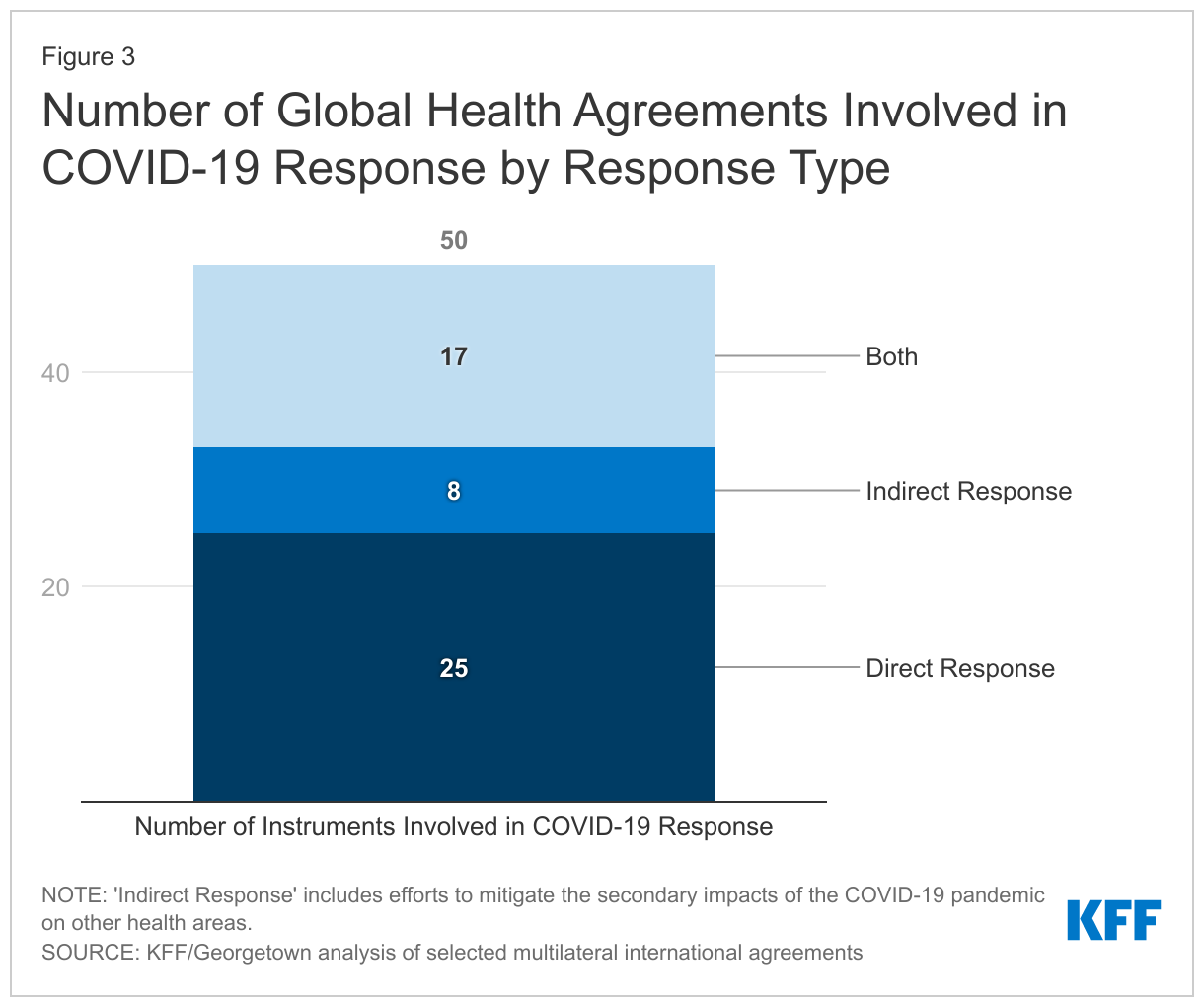
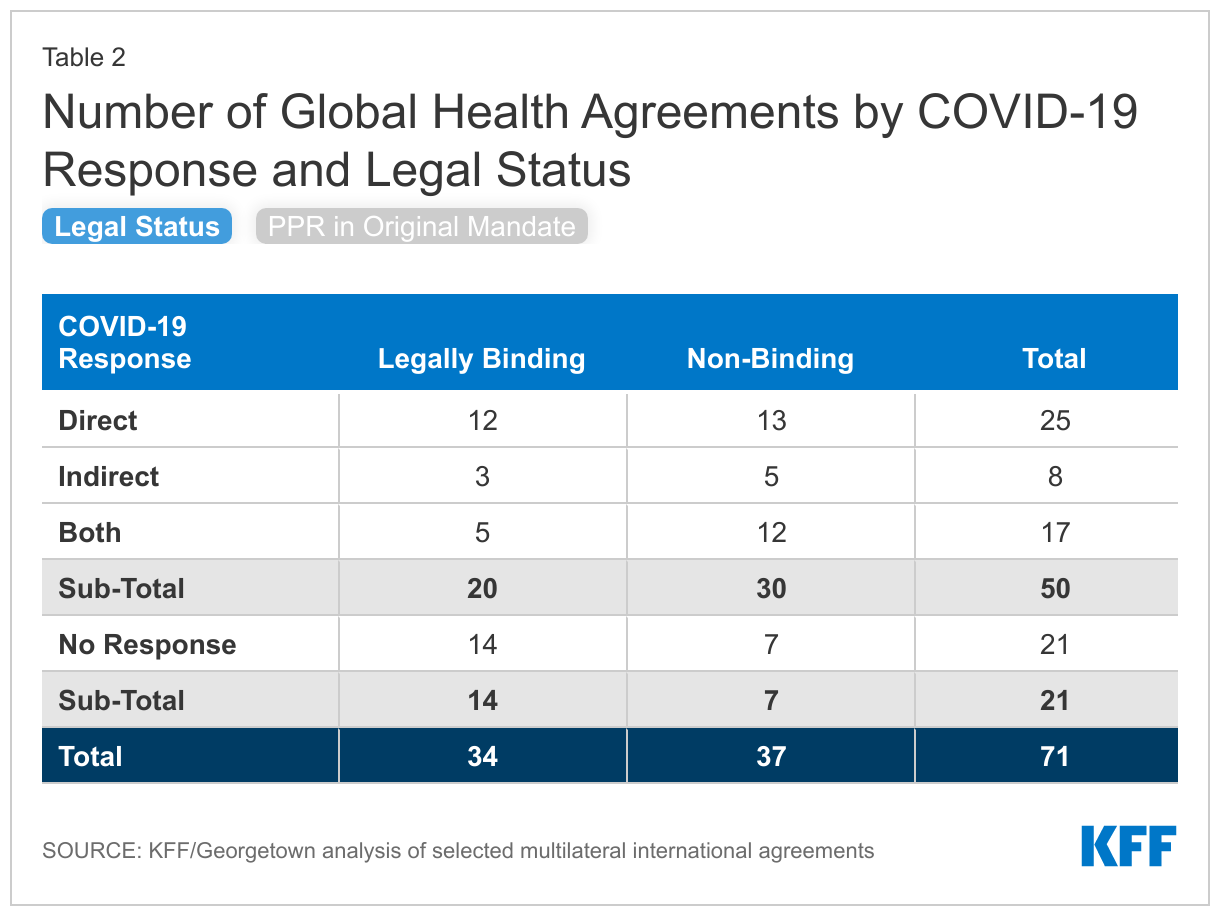
- Still, most (50 or 70%) of these agreements participated in the COVID-19 response. They were either involved directly (25), via activities specifically aimed at addressing the pandemic, indirectly (8), through activities that addressed the impacts of the pandemic on other health areas, or some combination of both (17). Thirty-six did not have PPR in their original mandate but either adapted existing efforts or started new activities to address the pandemic.
- The 71 agreements identified in this review could play a key role in the next phase of pandemic preparedness and response, including many that did not have PPR in their original mandate. As the global community considers a new international instrument to help prepare for and respond to future pandemics, our analysis finds that many of the existing agreements were mobilized to address COVID-19 and others have PPR as part of their original mandate. The extent to which these existing agreements could be part of future preparedness efforts remains to be seen but further research could help to highlight whether additional strengthening or further adaptation is needed, as well as highlight gaps that may persist.
Introduction
The COVID-19 pandemic tested global health governance and international law in unprecedented ways, revealing weaknesses and gaps in the existing global health security architecture and fueling debate over how to strengthen global governance of disease. Starting in late 2020, nations and expert groups began discussing the potential need for a new international agreement addressing pandemic preparedness and response (PPR). In December 2021, the World Health Assembly (WHA) agreed to launch a process to develop such an agreement. As part of this process, the WHA has established an Intergovernmental Negotiating Body (INB) “to draft and negotiate a WHO convention, agreement or other international agreement on pandemic prevention, preparedness and response” with an outcome for consideration expected in 2024. At its meeting held in July 2022, INB members agreed that at least some elements of the new international pandemic agreement should be legally-binding, although this decision is not yet final. The INB recently released a draft of the agreement which lays out a proposed governance structure for the new agreement as well as the PPR activities parties may be expected to undertake, such as improving supply chains and logistics networks, supporting information sharing and technology transfer, increasing research and development capabilities, and strengthening health systems and workforce. However, final text is not expected until 2024.
To help inform the current conversation, we reviewed existing international global health agreements (e.g. International Health Regulations), organizational charters (e.g., World Health Organization; Global Fund to Fight AIDS, Tuberculosis and Malaria; etc.), and institutions that have issued declarations addressing global health issues (e.g., World Bank, G7, G20, etc.) to assess their role in pandemic preparedness and response efforts, including whether they have played a role in the COVID-19 response, to help identify existing gaps and potential areas of collaboration with other organizations in the PPR space. This analysis builds on work researchers at KFF and Georgetown University conducted more than a decade ago exploring U.S. involvement in international health treaties and other global health organizations. For this analysis, we confirmed whether the agreements included in our original analysis were still active and identified additional agreements that should be included (e.g., those that have taken on a greater role in health or were created since the original paper was published). We assessed the extent to which each agreement has been involved in the COVID-19 response (either directly or indirectly) and/or possessed pandemic preparedness and response capacities. We considered agreements to be “directly” involved in the COVID-19 response if they addressed the impacts of COVID itself (e.g., provided medical countermeasures or other COVID-specific support). Agreements considered to be “indirectly” involved were those that sought to address the impacts of COVID on other health areas (e.g., provided support to maintain or reduce the effect of COVID on the response to another communicable disease). We defined agreements broadly to include both those that were legally-binding as well as non-binding agreements, partnerships, and the charters establishing United Nations entities and formally established global institutions (see Methodology for more detail).
Findings
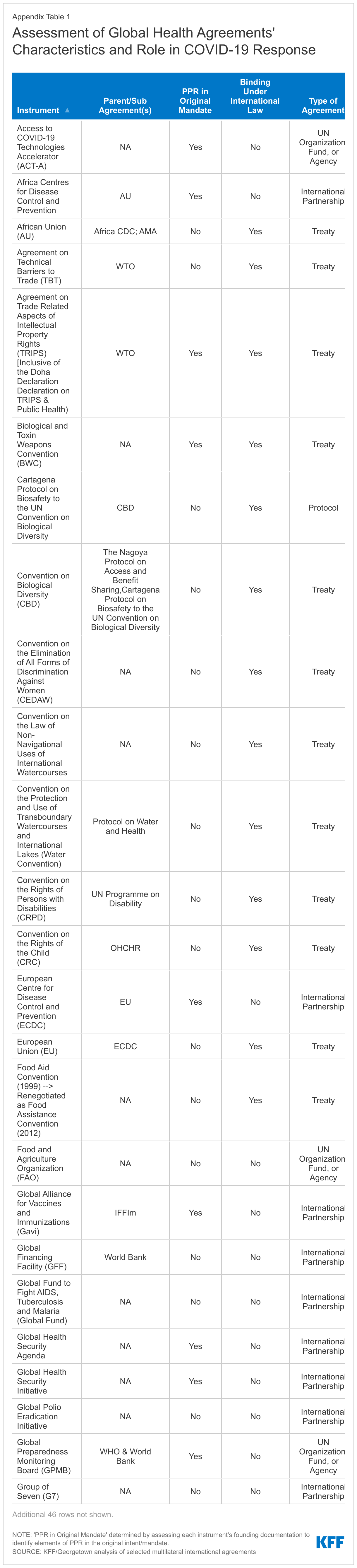
Overall, we identified 71 global health agreements, including 46 that were included in our original analysis.2 An additional 13 were already established but had not been originally included (they had taken on a greater role in health) and 12 were more recently created. The earliest agreement was established in 1902 (the Pan American Health Organization, originally the Pan-American Sanitary Bureau) and the most recent in 2021 (The Treaty of the African Medicines Agency). They span a range of focus areas, including diseases (HIV, malaria, tuberculosis, etc.), vulnerable populations (children, women, refugees, etc.), the environment, and trade/intellectual property, among others, and have varying levels of enforcement power and strength. Though a minority of the agreements reviewed had PPR in their original mandate, most were utilized in the COVID-19 response in some way, with many organizations taking on new roles to do so. Our specific findings are as follows:
Legal Status
- Nearly half (34) of the 71 agreements reviewed are legally-binding under international law (see Figure 1). These include 24 treaties, 6 protocols (which are associated with existing treaties), 3 United Nations organizations, funds, or specialized agencies (WHO, WTO, and WIPO), and one other international agreement (OECD) (see Table 1). Treaties include trade-related agreements under the World Trade Organization (WTO), which are binding under international law for WTO member countries, membership in international health organizations such as the World Health Organization (WHO) and the Pan American Health Organization (PAHO), and other legally binding agreements such as the International Health Regulations (2005)3 . Protocols include many of the agreements focused on environmental issues such as the Kyoto and Nagoya protocols.
- The remaining 37 agreements are not legally-binding under international law, but include agreements that confer obligations on members, including participation in organizations with global health mandates. Most of these (23) are “international partnerships”, which include initiatives such as the Sustainable Development Goals (SDGs) and Universal Health Coverage (UHC) 2030, as well as formal institutional arrangements of regional member states, such as the Africa Centres for Disease Control and Prevention (CDC), and broader alliances such as the Group of Seven (G7), Group of Twenty (G20), and North American Leaders’ Summit (NALS). “United Nations organizations, funds, or agencies” made up the remainder (14) and include entities such as the United Nations Environmental Programme (UNEP), United Nations Children’s Fund (UNICEF), and United Nations Population Fund (UNFPA).
PPR in Original Mandate
- Of the 71 agreements examined, 21 (30%) included PPR, in some capacity, as part of their original mandate (see Figure 2). For example, the International Health Regulations (IHR) (2005) require countries to possess certain capacities around PPR, including detecting, assessing, reporting, and responding to public health emergencies. The Africa CDC’s agreement obligates the body to work with member countries to build disease surveillance systems capable of identifying health threats and to provide response support during health emergencies. Similarly, the European Centre for Disease Control and Prevention’s founding documents outline a surveillance and response strategy for emerging health threats. The Agreement on Trade Related Aspects of Intellectual Property Rights (TRIPS) and the accompanying Doha Declaration on the TRIPS Agreement and Public Health recognize the gravity of public health emergencies and speak to the potential for intellectual property rights flexibilities in the context of these public health emergencies.
- Nine of these 21 are legally-binding. These are: The Agreement on the Application of Sanitary and Phytosanitary Measures (SPS Agreement), Biological and Toxin Weapons Convention (BWC); The IHR (2005); The Nagoya Protocol on Access and Benefit-Sharing; The Pan-American Sanitary Code; The Protocol on Water and Health; The TRIPS Agreement; the Treaty of the African Medicines Agency, and the World Health Organization.
COVID-19 Responses
- Most (50 or 70%) of the agreements reviewed were utilized to respond to the COVID-19 pandemic either directly (25), via activities specifically aimed at addressing the pandemic, indirectly (8), through activities that addressed the impacts of the pandemic on other health areas, or some combination of both (17) (see Figure 3). Twenty-one of these agreements are legally-binding (see Table 2).
- The 25 agreements that exclusively supported the direct impacts of COVID-19 undertook a range of activities, such as provisioning medical countermeasures, training health care workers, shoring up laboratories and other health systems, and developing new governance mechanisms to coordinate the response. For example, the Access to COVID-19 Tools Accelerator (ACT-A) supported a range of activities focused on addressing the COVID-19 pandemic, including supporting the procurement of diagnostic tools, therapeutics, vaccines, and personal protective equipment, the World Organisation for Animal Health created several expert groups to develop guidelines related to animal health and COVID-19, and the Global Polio Eradication Initiative shifted its efforts to assist in the COVID-19 response by training laboratory workers to detect the virus.
- Eight addressed only the indirect impacts of COVID-19 on other areas that aligned with their mandates. For example, the Stop TB Partnership issued guidance to help high TB burden countries integrate their TB and COVID-19 testing systems, and the Joint United Nations Programme on HIV/AIDS (UNAIDS) provided guidance on maintaining progress on HIV prevention in the context of the COVID-19 pandemic.
- Seventeen engaged in both direct and indirect COVID-19 responses. For example, the Global Fund to Fight AIDS, Tuberculosis and Malaria procured COVID-19 diagnostics, treatments, and oxygen for the countries in which it works and set up the COVID-19 Response Mechanism (C19RM) to fund countries’ efforts to mitigate the impacts of COVID-19 on HIV, TB, and malaria; the United Nations World Food Programme supported both the delivery of COVID-19 supplies, such as personal protective equipment (PPE), as well as food assistance to address food insecurity resulting from the pandemic; and the Pan American Health Organization (PAHO) assisted countries in the procurement and delivery of vaccines, as well as provided guidance on maintaining essential health services in the context of COVID-19.
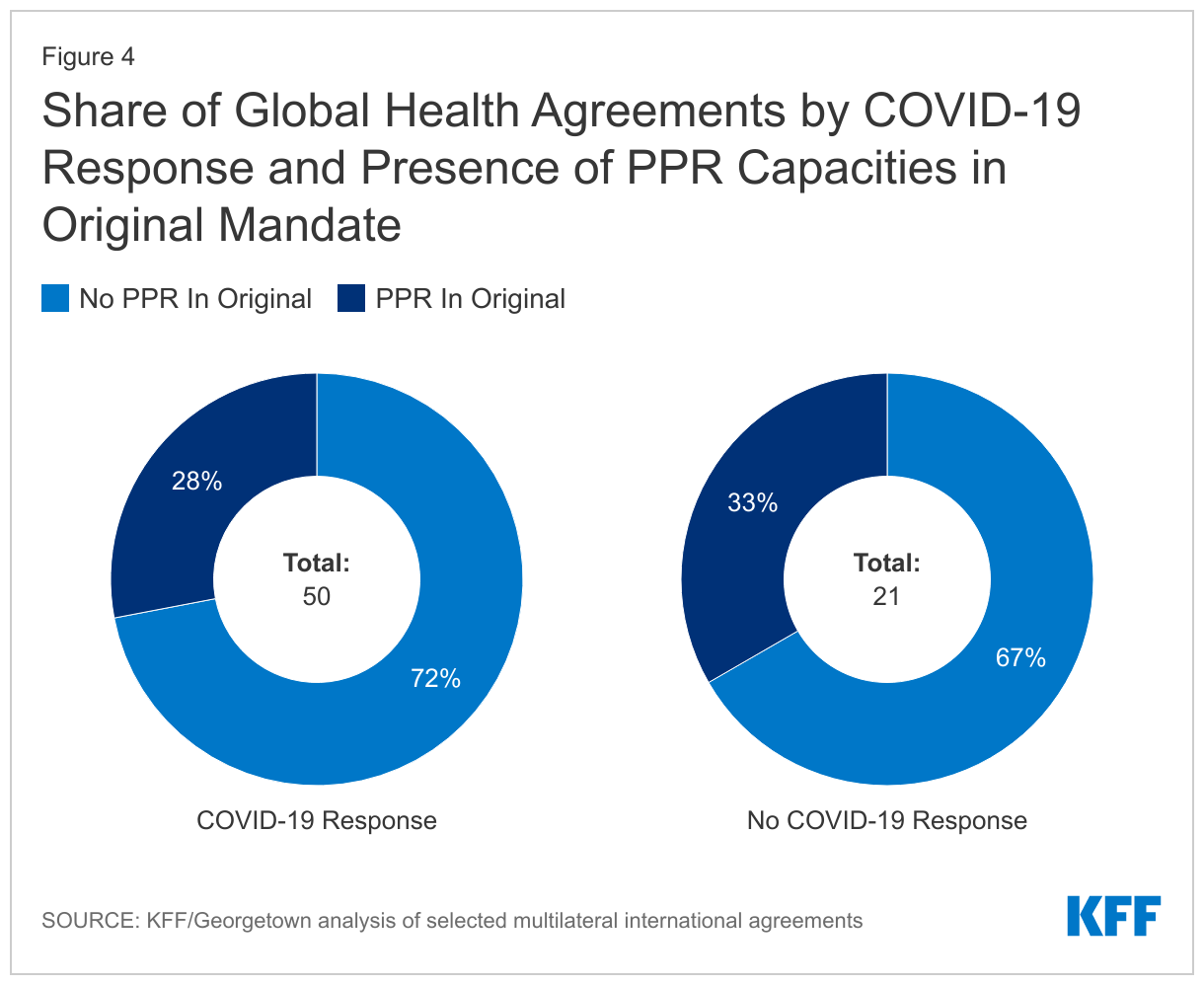
- Most of the agreements involved in responding to COVID-19 (36 of 50) did not have PPR in their original mandate but adapted to address the pandemic (see Figure 4). These include the Global Fund, which established the COVID-19 Response Mechanism (C19RM) to provide grants to low- and middle-income countries for the purchase of test, treatments, and personal protective equipment (PPEs), and the World Bank, which established the COVID-19 Fast Track Facility and provided support for vaccine rollout.
- A subset of agreements that were not involved in the COVID-19 pandemic response (7) contain PPR capacities in their original mandate. In some cases, this may be because COVID-19 fell outside their organizational scope (for example, water-borne diseases) or because these agreements were created very recently (for example, the Treaty of the African Medicines Agency).
Discussion
The COVID-19 pandemic has highlighted major gaps in global pandemic preparedness and response capabilities. This analysis outlines the international agreements already involved in pandemic preparedness and response to inform the development of a new pandemic agreement designed to address these gaps. Overall, we find that most of the agreements reviewed (50 of 71) have been involved in addressing the COVID-19 pandemic, either directly or indirectly. Most of these do not have PPR in their original mandate but, rather adapted to address the pandemic. In addition, half of the 71 agreements (34) are legally-binding, including 20 that responded to COVID. This suggests that there are already capacities to address PPR within existing agreements that could potentially be coordinated with a new framework. Importantly, though, this analysis does not attempt to assess the effectiveness of an agreement’s response to COVID-19 or its PPR capabilities more broadly, and future research could seek to do so. In addition, while most of the agreements responding to COVID-19 did so by adapting their work, we did not assess what was required to make such adaptations (e.g., whether new authorizations or mandates were needed). As global leaders continue to pursue the development of a new pandemic instrument, this review may help to identify agreements with existing PPR capabilities, including whether they need strengthening or further adaptation, as well as highlight gaps that may persist.
Methodology
We sought to identify multilateral international treaties, commitments, partnerships, organizations, and other agreements (“agreements”) that were health-specific or had a significant health component.4
We started with the list of agreements compiled and criteria used for our previous analysis, “U.S. Participation in International Health Treaties, Commitments, Partnerships, and Other Agreements”. Since the original report was completed over a decade ago, we revisited and reviewed the initial list to confirm whether each agreement was still active. We also revisited the sources (listed below) to determine if additional agreements warranted inclusion in this analysis (for example, if they had taken on a greater role in health or were created after the initial analysis). Each agreement was reviewed by multiple members of the team to ensure the accuracy of categorizations.
We analyzed both legally binding and nonbinding agreements and categorized each by type of agreement, and whether components of PPR were present in the agreement’s founding documents.5 Legally binding agreements were compiled based on a review of the Department of State’s required annual report to Congress on all Treaties in Force, the United Nations Treaty Collection, the Library of Congress reference collection on Treaties, the Congressional Research Service, and other reference documents. Nonbinding agreements were compiled based on a desktop review and include multilateral organizations, partnerships, consortia, and other arrangements. As such, the nonbinding list of agreements is comprehensive to the best of our knowledge but may not be exhaustive.
We then assessed each agreement to determine if it was involved in responding to COVID-19 either directly (through actions or the provision of services aimed at addressing the COVID-19 pandemic) or indirectly, to address the secondary effects of the pandemic (e.g., actions or the provision of services aimed at addressing other health-related activities, such as food aid, that have been directly affected by the pandemic). If an agreement was deemed to be “directly” involved in the COVID-19 response, it was automatically included under the PPR categorization. Agreements addressing the “secondary effects” of the COVID-19 response might not necessarily mean they would be involved in PPR activities. As such, there were several that were not involved in the COVID-19 response that were included under the PPR categorization. If an agreement was not deemed to be involved in the COVID-19 response or PPR, it was listed as “Other”. Several agreements resulted from or are directly related to previously established agreements (e.g., the Nagoya Protocol was established following the Convention on Biological Diversity) or are under the auspices of another entity (e.g., WHO manages the implementation of the International Health Regulations). In these instances, the parent agreement is classified as having the same role in the COVID-19 response as the sub-agreements.
| Table 3: Agreement Role(s) in Global COVID-19 Response: Definitions | |
| Category | Definition |
| COVID-19 Response | |
| Direct | Supports efforts aimed directly at responding to and mitigating the impacts of the COVID-19 pandemic. |
| Secondary Effects | Supports mitigating the potential effects on other health activities resulting from the COVID-19 pandemic. |
| Pandemic Preparedness & Response (PPR) | Manages, directs, or supports efforts to prepare and respond to potential disease outbreaks. If an agreement is categorized as contributing to the COVID-19 response (either direct or secondary effects), it is deemed to also support PPR, but not vice versa. |
| Other | Aims to prevent or reduce the likelihood of virus emerging that could result in a pandemic or aids in the strengthening of country health systems. |
Appendix
Anna Rouw, Adam Wexler and Jen Kates are with KFF.Kate Toole, Anjali Britto, and Rebecca Katz are with Georgetown University.
- KFF, “U.S. Participation in International Health Treaties, Commitments, Partnerships, and Other Agreements.” September 2010. ↩︎
- This total is inclusive of entities that have transformed or been renamed since the last publication (e.g. the International Health Partnership is now Universal Health Coverage 2030). ↩︎
- The International Health Regulations (2005) were created under the auspices of another agreement included in this analysis, the WHO Constitution. ↩︎
- The United Nations defines multilateral treaties as international agreements concluded between three or more parties, each possessing treatymaking capacity. Parties that have treaty-making capacity include sovereign States as well as international organizations with treaty-making capacity (e.g., the European Union). See: The United Nations Treaty Handbook. Bilateral agreements (country-to-country) were not included in this analysis. ↩︎
- See KFF, “U.S. Participation in International Health Treaties, Commitments, Partnerships, and Other Agreements” for categorical definitions on the type of agreement, focus area, and legal status (September, 2010). ↩︎