Latest News
-
Key Data About LGBT+ Women’s Experiences and Their Access to Care
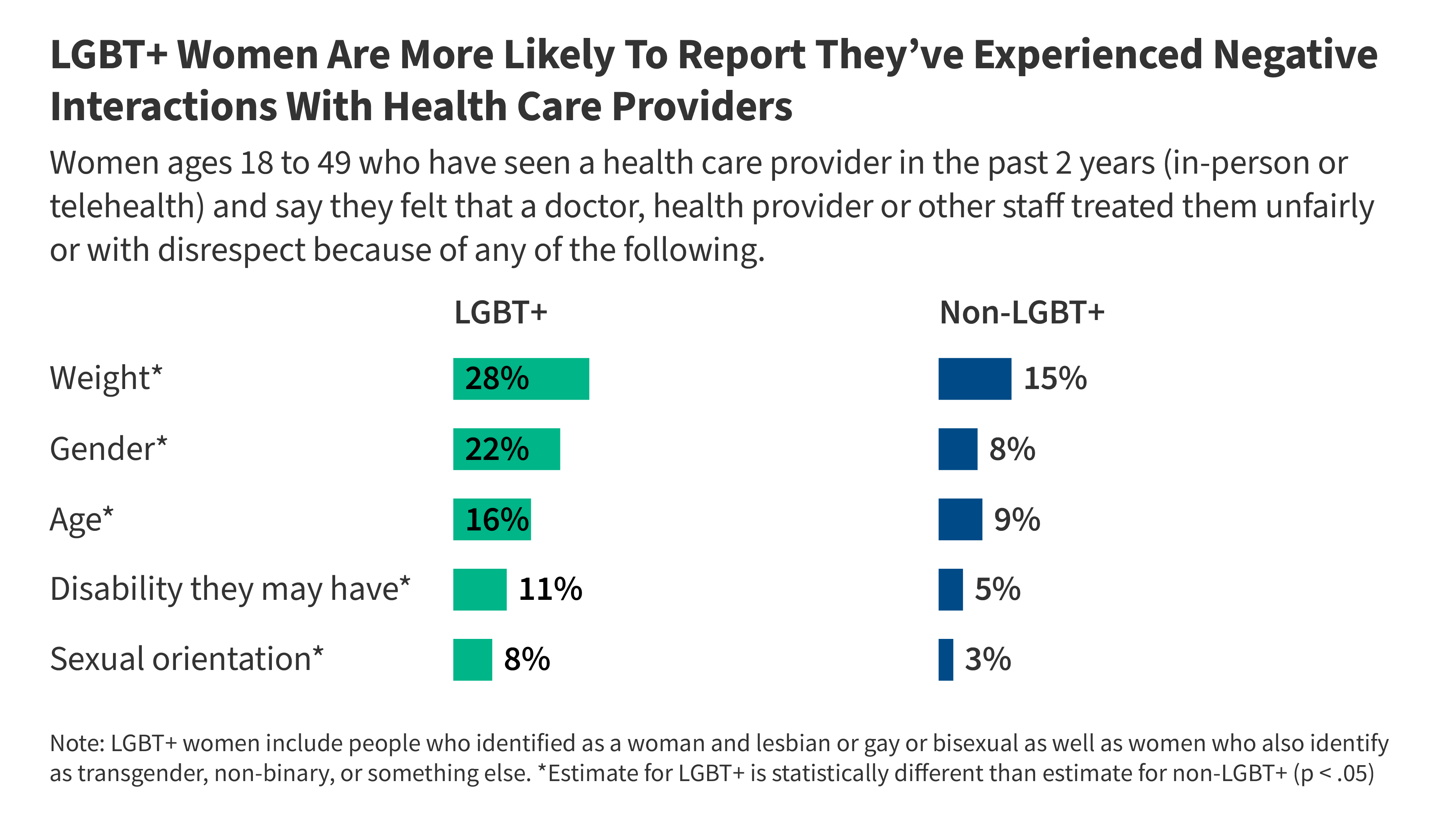
This brief presents key data points on the health care experiences of LGBT+ women from the KFF Women’s Health Survey, a nationally representative survey of women in the United States conducted from May 13 – June 18, 2024.
-
Fraud in Marketplace Enrollment, Eligibility: Five Things to Know

This brief evaluates what is currently known about fraud and abuse in the Affordable Care Act Marketplace, including how the final Marketplace Integrity and Affordability Rule and proposed budget reconciliation legislation would change existing Marketplace enrollment and eligibility standards.
-
Part B Costs for 7M on Medicare Could Rise Under Reconciliation Bill
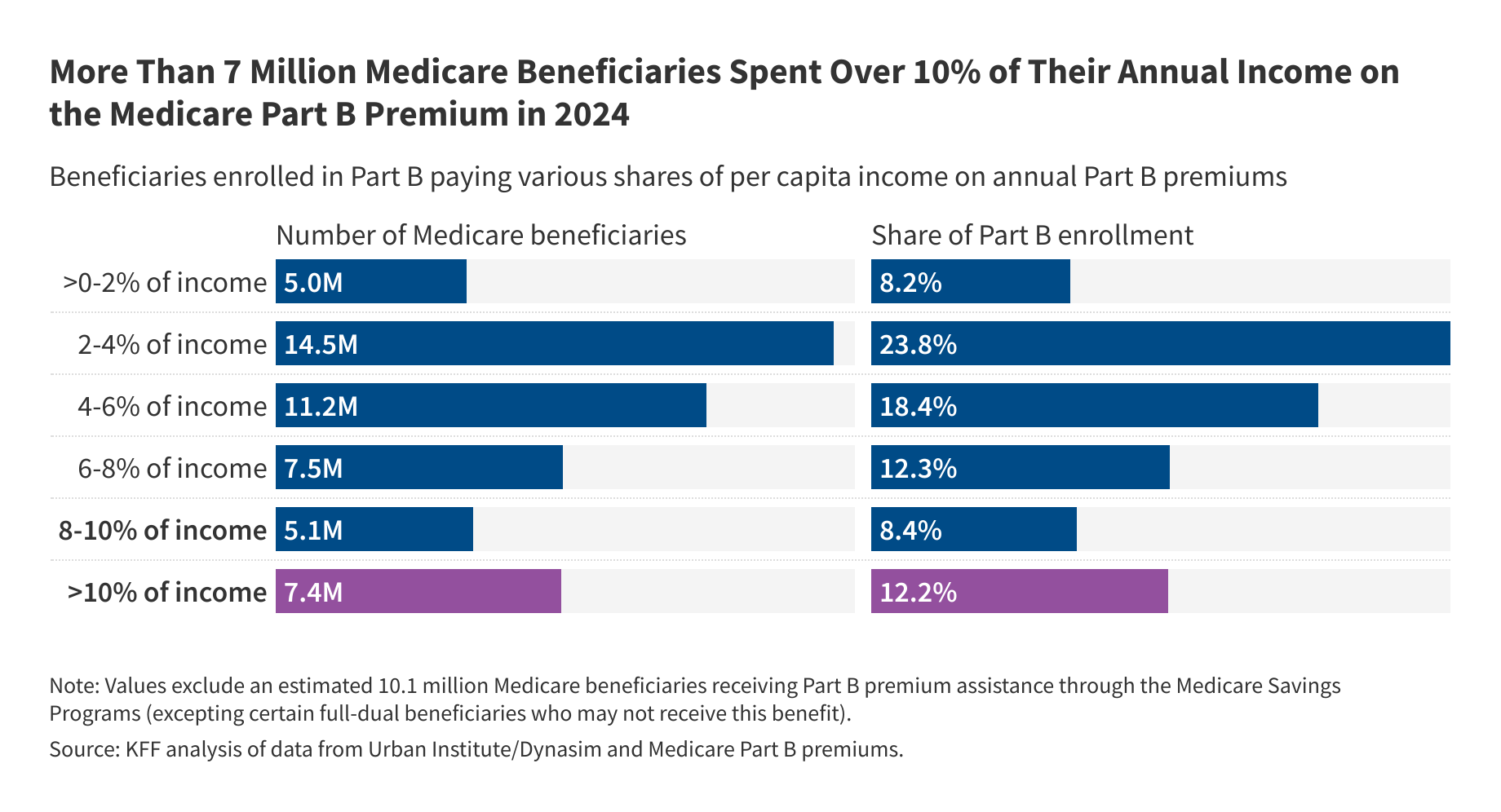
People with low incomes and limited financial resources can qualify for the Medicare Savings Programs, through which state Medicaid programs provide financial assistance with Medicare premiums and cost sharing. However, provisions in the GOP’s budget reconciliation bill would make it harder for people to enroll in these programs.
-
Beyond the Data, a Column by Drew Altman

In his regular Beyond the Data columns, CEO Drew Altman discusses what the data, polls, and journalism produced by KFF mean for policy and for people.