Will 1999 Be The Year For Mifepristone (RU-486) And, An Update on Women’s Other Options for Very Early Abortion – Fact Sheet
Abortion in the U.S.
- Most abortions in the U.S. today are performed surgically, typically using vacuum aspiration (VA).
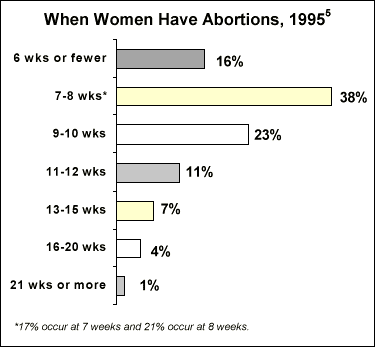
- 9 in 10 abortions occur within the first 12 weeks of pregnancy.5
- Surgical abortions can be performed as early as pregnancy is first detected, using electric VA or manual vacuum aspiration (MVA).21
- Medical abortions are procedures using medications to induce abortion, such as mifepristone (also called RU-486) or methotrexate in combination with misoprostol.
Unintended Pregnancy And Surgical Abortion
- Almost half (49%) of the 6.3 million pregnancies each year are unintended; 54% of unintended pregnancies end in abortion.
- In 1994, there were about 1.43 million abortions in the United States; the abortion rate was 24.2 per 1,000 women aged 15-44.10
- The U.S. abortion rate and the percent of pregnancies ending in abortion have declined slightly but steadily since 1980. Since 1990, all measures of abortion, including the annual incidence, have declined.6
- About 15,000 women have abortions each year because they became pregnant after rape or incest.6
- 48% of women 15*44 have at least one unplanned pregnancy sometime in their lives; 28% have one or more unplanned births, 30% have one or more abortions and 11% have both.9
- An estimated 43% of women will have an abortion by the time they are 45, given current abortion rates.9
Who Has Abortions
- 55% of women having abortions are younger than 25; 33% are aged 20-24 and 22% are teenagers.8
- Six in 10 abortions occur to white women; however, their abortion rate is below that of minority women. Black women are nearly 3 times as likely as white women to have an abortion and Hispanic women are about twice as likely.8
- Two-thirds of all abortions are to never-married women.8
Who Provides Surgical Abortions
- 93% of abortions take place in clinics or doctors’ offices.6
- The number of known current abortion providers (hospitals, abortion clinics, family planning clinics that offer surgical services, and physicians’ offices) declined by 8% between 1988 and 1992 (from 2,582 to 2,380).6
- In 1992, just 12% of all ob/gyn residency programs provided routine first-trimester abortion training; three in 10 programs offered no first-trimester training.12
- According to a national survey of ob/gyns in 1997, 36% of ob/gyns say they “ever perform” surgical abortions.18
Barriers & Access To Surgical Abortion Services
- 84% of all U.S. counties lacked an abortion provider in 1992.6V52% of all abortion facilities provide services only through the 12th week of pregnancy.7
- In 1992, 4 in 10 clinics and physicians’ practices would not perform surgical abortions before 6 weeks since the beginning of a woman’s last menstrual period (LMP). Another quarter of facilities do not perform abortions until even later in pregnancy.7
- One-quarter of women who have non-hospital abortions travel at least 50 miles from their home to the abortion facility.7
- From 1996 to 1997, the number of arsons doubled and bombings tripled against abortion providers.13
Very Early Abortion Methods3,14,21
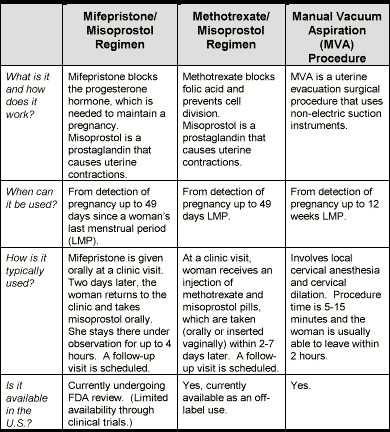
Medical Abortion
- Among women participating in the U.S. clinical trials who underwent an abortion within 49 days (7 weeks) LMP, the use of mifepristone (with misoprostol) was 92% effective in terminating pregnancy.16
- Two percent of women who had a mifepristone abortion 49 days LMP in clinical trials required hospitalization, surgical intervention, and/or intravenous-fluid administration.16
- Several published studies on the use of methotrexate (with misoprostol) for abortion have found the drug to be up to 96% effective in terminating pregnancy within 63 days LMP.1,2,4
- Of 178 women who underwent pregnancy termination with methotrexate within 63 days LMP in one study, 7 required surgical intervention.1
- A review of 12 international published studies conducted between 1979 and 1993 of patient attitudes and reactions to early first-trimester medical abortion found that 60-70% of women chose medical abortion when offered a choice between surgical and medical abortion.19
- Over 2,000 U.S. women, who had a medical abortion with mifepristone and misoprostol during the U.S. clinical trials (1994-1995), found the method highly acceptable: 96% would recommend it to others, 91% would choose it again and 88% found it very or moderately satisfactory.20
Who Pays For Surgical Abortions
- In 1993, the average amount paid for a non-hospital abortion at 10 weeks was $296; however, the cost ranged from $140 to more than $1,700.7
- About 13% of all abortions are paid for with public funds, virtually all of which are state funds. Currently, 16 states pay for all or some abortions for poor women.15
- In the private sector, over two-thirds of typical fee-for-service and managed care plans routinely cover abortions.17
Provider & Public Opinion On Medical Abortion
- As of 1997, most providers said they were at least somewhat familiar with mifepristone; fewer were familiar with methotrexate.18
- About half of all ob/gyns (54%) said they would offer mifepristone abortions if approved in the U.S., including 35% of ob/gyns who do not perform surgical abortions.18
- Among providers familiar with mifepristone, most said they considered it safe (96% of ob/gyns) and effective (94% of ob/gyns).18
- As of 1997, 43% of women and 51% of men had heard of either mifepristone or methotrexate; among women who had heard of the methods, 72% knew they could be used to end a pregnancy.18
References
1. Creinin M, Darney P. “Methotrexate and Misoprostol for Early Abortion.” Contraception. 1993; 48:339-348.
2. Creinin M. “Methotrexate for Abortion at 42 Days Gestation.” Contraception. 1993; 48:519-525.
3. Expanding Options For Early Abortions Packet. Overview and Fact Sheets. Reproductive Health Technologies Project, Washington D.C.: 1998.
4. Hausknecht R. “Methotrexate and Misoprostol to Terminate Early Pregnancy.” The New England Journal of Medicine. 1995;333:537-540.
5. Henshaw S. “Abortion Services in the United States, 1995-1996.” Family Planning Perspectives. (Forthcoming, Nov/Dec 1998).
6. Henshaw S, Van Vort J. “Abortion Services in the United States, 1991 and 1992.” Family Planning Perspectives. 1994; 26:100-106, 112.
7. Henshaw S. “Factors Hindering Access to Abortion Services.” Family Planning Perspectives. 1995; 27:54-59, 87.
8. Henshaw S, Kost K. “Abortion Patients in 1994-1995: Characteristics and Contraceptive Use.” Family Planning Perspectives. 1996; 28:140-147, 158.
9. Henshaw S. “Unintended Pregnancy in the United States.” Family Planning Perspectives. 1998; 30:24-29.
10. “Induced Abortion.” Facts in Brief. Jan. 1997, New York, N.Y. The Alan Guttmacher Institute.
11. Forrest JD. “Timing of Reproductive Life Stages.” Family Planning Perspectives. 1993; 82.
12. MacKay H, Trent MacKay A. “Abortion Training in Obstetrics and Gynecology Residency Programs in the United States, 1991-1992.” Family Planning Perspectives. 1995; 27:112-115.
13. National Abortion Federation Website (http://www.prochoice.org)
14. Schaff E, Eisinger S, Franks P, Kim S. “Methotrexate and Misoprostol for Early Abortion.” Fam Med. 1996;28:198-203.
15. Sollom T, Gold R, Saul R. “Public Funding for Contraceptive, Sterilization and Abortion Services, 1994.” Family Planning Perspectives. 1996; 28:166-173.v
16. Spitz I, Benton L, Bardin W, Robbins A. “The Safety and Efficacy of Early Pregnancy Termination with Mifepristone and Misoprostol: Results from the First Multicenter U.S. Trial.” The New England Journal of Medicine. 1998; 338:1241-1247.
17. Uneven & Unequal: Insurance Coverage and Reproductive Health Services. 1994, New York, N.Y. The Alan Guttmacher Institute.
18. Views of Americans and Health Care Providers on Medical Abortion: What They Know, What They Think, and What They Want.. 1998, Menlo Park, CA. Kaiser Family Foundation.
19. Winikoff B. “Acceptability of Medical Abortion in Early Pregnancy.” Family Planning Perspectives. 1995;27:142-148 & 185.
20. Winikoff B, Ellertson C, Elul B, Sivin I. “Acceptability and Feasibility of Early Pregnancy Termination by Mifepristone-Misoprostol.” Archives of Family Medicine. 1998; 7:360-366.
21. Winkler J, Blumenthal P, Greenslade F. “Early Abortion Services: New Choices for Providers and Women.” Advances in Abortion Care. 1996; 5. Carrboro, NC. IPAS.