Preventive Services Covered by Private Health Plans under the Affordable Care Act
Note: This content was updated on February 28, 2024 to incorporate new FAQs from CMS. Tables 1 and 2 were also updated to include updated recommendations.
It has been more than ten years since the Affordable Care Act (ACA) required private insurance plans to cover recommended preventive services without any patient cost-sharing. Research has shown that evidence-based preventive services can save lives and improve health by identifying illnesses earlier, managing them more effectively, and treating them before they develop into more complicated, debilitating conditions, and that some services are also cost-effective. Since the preventive services coverage policy went into effect, there have been numerous additions, changes, and updates to the policy as well as specific recommendations. There have also been legal challenges over elements of the preventive services requirement, including in the pending case, Braidwood Management Inc. v. Becerra. This fact sheet summarizes the federal requirements for coverage for preventive services in private plans, major updates to the requirement, and recent policy activities on this front.
ACA Requirements for Coverage of Preventive Services
Under Section 2713 of the ACA, private health plans must provide coverage for a range of recommended preventive services and may not impose cost-sharing (such as copayments, deductibles, or co-insurance) on patients receiving these services.1 These requirements apply to all private plans—fully insured and self-insured plans in the individual, small group, and large group markets, except those that maintain “grandfathered” status. In 2019, 13% of workers covered in employer sponsored plans were still in grandfathered plans. The requirements also apply to the Medicaid expansion eligibility pathway.
The required preventive services come from recommendations issued by four expert medical and scientific bodies—the U.S. Preventive Services Task Force (USPSTF), the Advisory Committee on Immunization Practices (ACIP), the Health Resources and Services Administration’s (HRSA’s) Bright Futures Project, and the HRSA-sponsored Women’s Preventive Services Initiative (WPSI). Individual and small group plans in the health insurance marketplaces are also required to cover an essential health benefit (EHB) package—that includes the full range of preventive requirements described in this fact sheet.
Clinical Preventive Services for Adults and Children
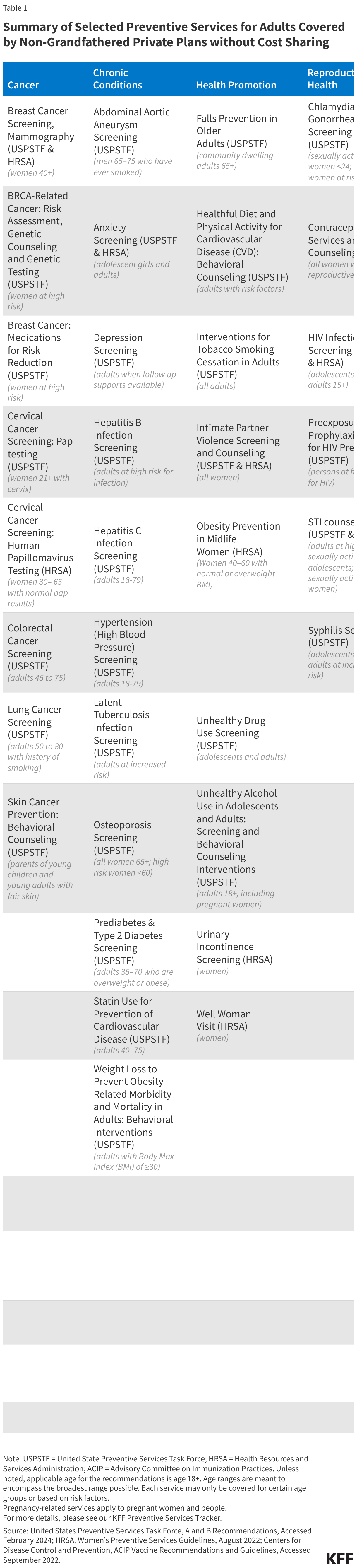
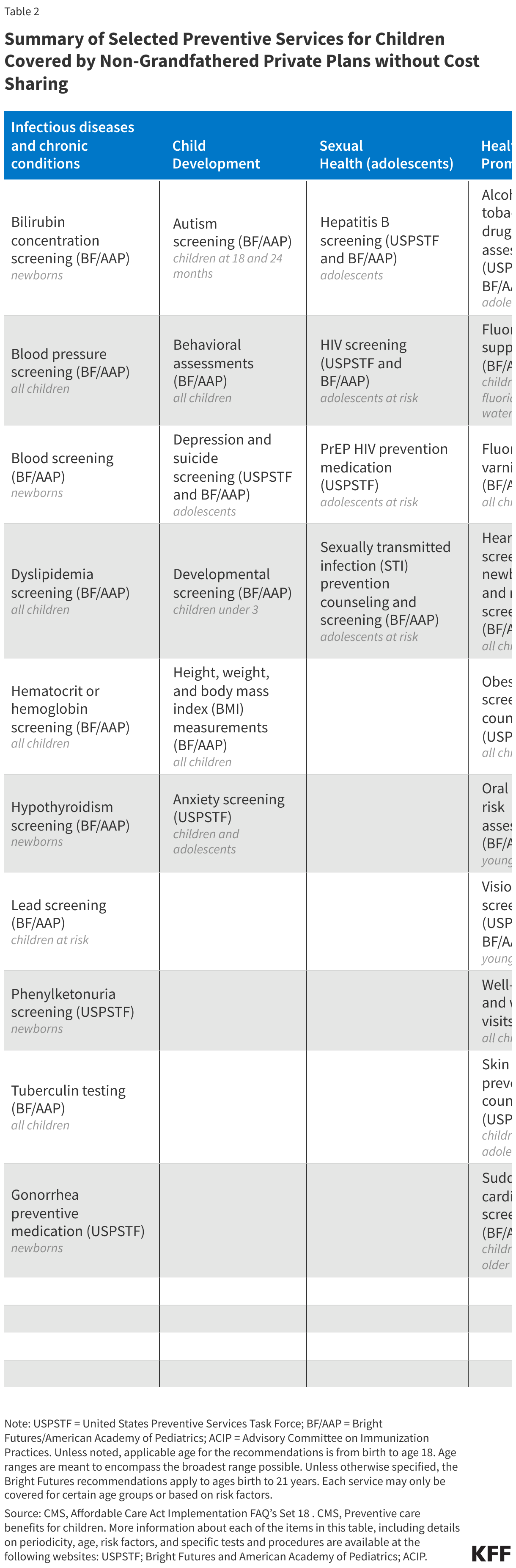
The ACA requires private plans to cover the following four broad categories of services for adults and children (summarized in Tables 1 and 2):
I. Evidence-Based Screenings and Counseling
Insurers must cover evidence-based services for adults that have a rating of “A” or “B” in the current recommendations of USPSTF, an independent panel of clinicians and scientists commissioned by the federal Agency for Healthcare Research and Quality. An “A” or “B” letter grade indicates that the panel finds there is high certainty that the services have a substantial or moderate net health benefit. The services required to be covered without cost-sharing include screenings for depression, diabetes, obesity, various cancers, and sexually transmitted infections (STIs), prenatal tests, medications that can help prevent HIV, breast cancer, and heart disease, as well as counseling for drug and tobacco use, healthy eating, and other common health concerns. The effective date for a new recommendation from USPSTF is considered to be the last day of the month in which it is published or otherwise released.
II. Routine Immunizations
Health plans must also provide coverage without cost-sharing for immunizations that are recommended and determined to be for routine use by the ACIP, a federal committee comprised of immunization experts that is convened by the Centers for Disease Control and Prevention (CDC). A new ACIP recommendation is considered to be issued on the date that it is adopted by the Director of the CDC. The preventive services guidelines require coverage for adults and children and include immunizations such as influenza, meningitis, tetanus, HPV, hepatitis A and B, measles, mumps, rubella, varicella, and COVID-19. With regard to the COVID-19 vaccine, Congress waived the typical one year delay in implementation and required private insurance plans to begin full coverage 15 days after ACIP recommendation. Going forward, any COVID-19 vaccine recommended by ACIP, including updated boosters, will continue to be fully covered for people enrolled in non-grandfathered plans starting 15 days after the vaccine is recommended by ACIP, irrespective of whether the vaccine is under an emergency use authorization or fully approved by the FDA.
III. Preventive Services for Women
In addition to the recommendations issued by USPSTF and ACIP, the ACA authorized HRSA to make coverage requirements for women for services not addressed by the other recommending bodies. HRSA turns to evidence-based recommendations issued by the Women’s Preventive Services Initiative (WPSI), to identify gaps in recommendations for women and review the evidence regarding the effectiveness of the recommendations. Current recommendations include well-woman visits, all FDA-approved, -granted, or -cleared contraceptives and related services, breastfeeding support and supplies, broader screening and counseling for a range of conditions, including intimate partner violence, urinary incontinence, anxiety, STIs and HIV. Some of the HRSA recommendations for women are similar to recommendations from USPSTF, but with slight variations in the population that is addressed.
Table 1 summarizes the full slate of adult preventive services subject to the preventive services coverage requirements.
IV. Preventive Services for Children and Youth
In addition to services for adults, the ACA requires that private plans cover without cost-sharing the preventive services recommended by the HRSA’s Bright Futures Project, which provides evidence-informed recommendations to improve the health and wellbeing of infants, children, and adolescents. The preventive services covered for children and adolescents include well child visits, immunization and screening services, behavioral and developmental assessments, fluoride supplements, and screening for autism, vision impairment, lipid disorders, tuberculosis, and certain genetic diseases. immunization and screening services, behavioral and developmental assessments, fluoride supplements, and screening for autism, vision impairment, lipid disorders, tuberculosis, and certain genetic diseases.
Table 2 summarizes the full slate of preventive services for children and adolescents.
Coverage Rules and Clarifications
The recommending bodies periodically issue new recommendations and update existing ones based on advances in research. Plans are required to provide full coverage for new and updated recommendations one year after the latest issue date, beginning in the next plan year.2 If a recommendation is changed during a plan year or a new recommendation is issued, an issuer is not required to make changes in the middle of the plan year, unless one of the recommending bodies determines that a service is discouraged because it is harmful or poses a significant safety concern.3 In these circumstances, federal guidance will be issued. There are limited circumstances under which insurers may charge copayments and use other forms of cost-sharing for preventive services:
- If the primary reason for the visit is not the preventive service, patients may have to pay for the office visit. For example, if an adult man sees a clinician for ongoing management of a chronic condition such as diabetes and also receives a COVID vaccine at that appointment, the plan may charge a co-payment for the office visit but may not charge for the vaccine, which is a recommended preventive service.
- If the preventive service is performed by an out-of-network provider when an in-network provider is available to perform the service, insurers may charge patients for the office visit and the preventive service. However, if an out-of-network provider is used because there is no in-network provider able to provide the service then cost-sharing cannot be charged.
- If a treatment is given as the result of a recommended preventive service, but is not the recommended preventive service itself, cost-sharing may be charged in some cases. For example, the USPSTF recommends a CT scan for some adults to screen for lung cancer. If cancer is detected during the scan, treatments such as surgery or medication may be prescribed. While plans must cover the screening test services in full, they may charge for the treatments.
The Public Health Service Act (PHSA) and federal regulations also allow plans to use “reasonable medical management” techniques to determine the frequency, method, treatment, or setting for a preventive item or service to the extent it is not specified in a recommendation or guideline. While there is no formal regulatory definition or parameters for reasonable medical management, medical management techniques are typically used by plans to control cost and utilization of care or comparable drug use. For example, plans can impose limits on number of visits or tests if unspecified by a recommendation, cover only generics or selected brands of pharmaceuticals, or require prior authorization to acquire a preferred brand drug. If a plan makes any material modifications that would affect the content of the plan’s Summary of Benefits and Coverage (SBC) during the plan year, the plan must notify enrollees of the change at least 60 days before it takes effect.
Since the policy took effect, a number of questions have arisen about how plans should implement the preventive services policy and the extent to which plans can use medical management practices to limit the frequency, range of covered services, and the types of providers that are subject to the policy. Over the years, the Departments of Health and Human Services, Labor, and Treasury have jointly issued a number of clarifications as” about different aspects of coverage of preventive services.
Notable highlights from clarifying documents include:
- Colon cancer screening – USPSTF recommends screening for colorectal cancer in adults ages 45-75 using either stool-based testing or procedural screening, such as sigmoidoscopy or colonoscopy. There have been some cases of insured asymptomatic patients being charged unexpected cost-sharing for anesthesia and polyp removal during screening colonoscopies. The federal government has clarified multiple times that insurers must cover the full cost of medically necessary anesthesia services, polyp removal and related pathology performed in connection with a preventive colonoscopy in asymptomatic individuals, and follow up colonoscopies in the event of positive findings on stool-based tests, CT, or sigmoidoscopy.
- Well-woman visits – The HRSA clinical preventive services for women include coverage for at least one well-woman preventive care visit for adult women. WPSI has clarified that a series of well-woman visits may be required to fulfill all necessary preventive services and should be provided without cost-sharing as needed, determined by clinical expertise. Furthermore, the most recent recommendation states that prenatal visits are considered well woman visits, as are pre-pregnancy, postpartum, and interpartum visits WPSI has also published recommendations for services to be provided as part of well woman care.
- Testing and medications for the risk reduction of breast cancer – Federal guidance reinforces the USPSTF recommendation that women with family history of breast, ovarian, or peritoneal cancer should be screened for BRCA-related cancer, and those with positive results should receive genetic counseling and testing without cost-sharing when the services are medically appropriate and recommended by her provider. USPSTF also recommends the provision of chemo-preventive medications, such as tamoxifen and raloxifene, for women who are at increased risk for breast cancer and at low risk for adverse effects.
- Special populations – Some of the recommendations subject to the preventive services requirement apply to a certain population, such as “high risk” individuals. The government has clarified that it is up to the health care provider to determine whether a patient belongs to the population in consideration and that plans must cover services accordingly. An individual’s sex assigned at birth or gender identity also cannot limit them from a recommended preventive service that is medically appropriate for that individual; for example, a transgender man who has breast tissue or an intact cervix and meets other requirements for mammography or cervical cancer screening must receive those services without cost sharing regardless of sex at birth.
- Contraceptive coverage – Contraceptive services and supplies for women is one of the recommendations from HRSA, and since it was first issued there have been numerous federal clarifications. Plans must cover without cost sharing at least one product within each FDA-approved, granted, or cleared contraceptive method for women as prescribed. In addition to covering the cost of the contraceptive supplies, plans must cover related counseling, insertion, removal, and follow up services. While insurers may use reasonable medical management to limit full coverage to generic drugs within a method category, federal clarifications also state that plans must cover any contraceptive if deemed “medically necessary” by a health care provider. This means that plans must cover the following: brand name drugs if a generic is not available, a clinician-recommended brand name product, and contraceptive products that are not specifically identified by HRSA, such as new contraceptive products approved by the FDA. Some plans may choose to cover only one product within a category of contraceptives that has other therapeutic equivalent products. If this is the case, the plan must have a process in place to make exceptions for an individual who wants to access a therapeutic equivalent product if it is determined to be medically necessary by the individual’s clinician. Any "exceptions process" must be accessible and timely for patients and providers to request coverage for a medically necessary contraceptive.
- Houses of worship have always been exempted from the contraceptive requirement, and religiously affiliated nonprofit employers have had an accommodation if they have a religious objection to contraceptives. Some employers have challenged this regulation, claiming the accommodation offered by the government (where the method is covered by their plan but they are not required to pay towards its coverage as part of the premium) makes them complicit in the provision of contraception, a service they object to on religious or moral grounds. The federal policy regarding contraceptive coverage requirements for employer plans has undergone multiple changes in federal regulations and been contested in numerous legal cases, including three that reached the Supreme Court. The current regulations were issued during the Trump Administration and exempt nearly any employer that claims to have a religious or moral objection from providing contraceptive coverage.
- Coverage for HIV Preexposure Prophylaxis (PrEP) – In June 2019, PrEP, medications which can help prevent HIV, received an “A”’ grade recommendation from the USPSTF as “effective antiretroviral therapy to persons who are at high risk of HIV acquisition.” Plans or policy years beginning on or after June 30, 2020, must cover PrEP (consistent with the USPSTF recommendation) without cost sharing. Federal guidance clarified that plans and insurers must also cover ancillary and support services for PrEP, such as adherence counseling and risk-reduction strategies, without cost sharing, and cannot use reasonable medical management techniques to restrict access to these services.
Impact of the Preventive Services Rules
The federal HHS Assistant Secretary for Planning and Evaluation (ASPE) estimates that in 2020, approximately 151.6 million people (58 million women, 57 million men, and 37 million children) currently are enrolled in non-grandfathered private health insurance plans that cover preventive services with no-cost sharing. Research has documented the impact of the policy on access to care in some areas, including utilization of cancer screening and contraceptives.
The evidence on cancer screening utilization after the elimination of cost-sharing is mixed and varies by cancer type. Some studies have shown that while screening rates for colorectal cancer among privately insured individuals increased since the passage of the ACA, rates for Pap testing decreased. However, it is difficult to assess the impact of the coverage provision since the recommendations for cervical cancer screening have been revised since the policy went into effect. Screening rates for breast cancer remained stable, though one study found that mammography screening among African American women significantly increased after ACA implementation. Likewise, the elimination of cost-sharing is associated with increases in BRCA genetic testing which helps identify women who are at elevated risk for breast and ovarian cancer. Studies have also indicated that increased access to and affordability of preventive services has helped cancer survivors obtain necessary care.
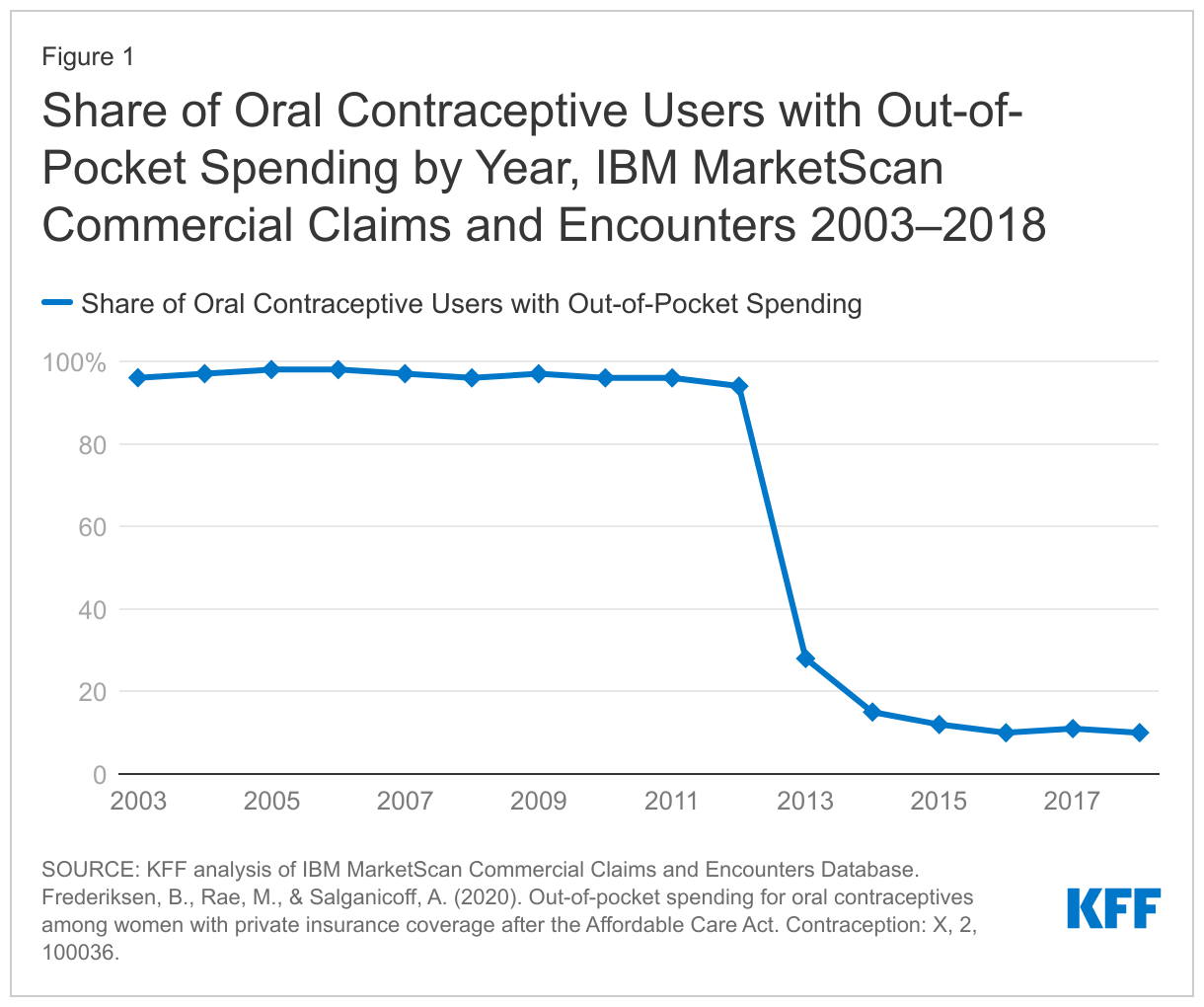
Several studies found that the contraceptive coverage requirement under the ACA has dramatically reduced OOP spending for contraceptives, including OOP spending for oral contraceptives (Figure 2). Multiple studies have shown increases in utilization for short-term birth control methods such as birth control pills, patches, and diaphragms. Studies have found that utilization of long-acting reversible contraceptives (LARCs), such as intrauterine devices (IUDs) and implants, increased after ACA implementation. Additional research also shows that OOP costs for LARCs—some of the most effective forms of pregnancy prevention—were also reduced under the ACA. These findings suggest that the lowered OOP costs from the contraceptive coverage requirement has improved contraception use and adherence.
The preventive services coverage policy has become an established part of health coverage for most people in the United States. Yet, the policy is currently facing legal challenges, notably in the case Braidwood Management Inc v. Becerra. The outcome of the latest legal challenge could affect whether people will continue to have full no-cost coverage for recommended preventive services in the future.
- Note that the rules described in this fact sheet apply to private insurers, self-insured employer plans, and are separate from preventive requirements for public programs like Medicare or Medicaid. ↩︎
- The final issue date for new or updated recommendations varies by recommending body. Recommendations are considered to be issued on the last day of the month on which the USPSTF publishes or releases the recommendation; recommendations from ACIP are considered issued on the date it is adopted by the Director of the CDC; and a recommendation or guideline supported by HRSA is considered to be issued on the date on which it is accepted by the Administrator of HRSA or, if applicable, adopted by the Secretary of HHS. Federal Register, Vol. 80, NO. 134, July 14, 2015. ↩︎
- These circumstances include downgrade of a USPSTF service from a rating of “A” or “B” to “D” (which means that USPTF has determined that there is strong evidence that there is no net benefit, or that the harms outweigh the benefits, and therefore discourages the use of this service), or a service is the subject of a safety recall or otherwise determined to pose a significant safety concern by a federal agency authorized to regulate that item or service. ↩︎