The Last Major Phase of the COVID-19 Vaccination Roll-out: Children Under 5
On June 17, the Food and Drug Administration authorized emergency use of both Moderna and Pfizer’s COVID-19 vaccines for children between the ages of 6 months and 5 years and on June 18, the Director of the Centers for Disease Control and Prevention (CDC) recommended their use for this population, noting their safety as well as their effectiveness in preventing severe COVID-19 outcomes. This marks the first time COVID-19 vaccines are available to virtually all people in the United States. The CDC has already provided guidance to jurisdictions to help in their planning, including instructing them to pre-order doses in anticipation of a recommendation. Pre-orders began on June 3 and shipments as soon as the emergency use authorizations (EUA) were granted on June 17; vaccination can begin now that the CDC has provided its recommendation.
We previously explored policy considerations for vaccinating 5-11 year-olds, who became eligible in November of last year, and have continued to track vaccine progress and demand among this group, both of which may be instructive for this next phase. Among 5-11 year-olds, vaccination rose quickly after first recommended but then dropped just two weeks in; as of June 16, just 29.5% have been fully vaccinated compared to 75% of those ages 12 and above. While a similar pattern may be expected for younger children – our most recent COVID-19 Vaccine Monitor survey, fielded before vaccines were recommended for them, found that only 18% of parents said they would get their young child vaccinated right away – there are also unique issues to consider for younger children that may present additional barriers and issues for policymakers, public health practitioners, and parents and caregivers. This brief provides an overview of the characteristics of children under the age of 5 nationally and by state and discusses some of the particular issues to consider in rolling out vaccination to this age group.
What are Characteristics of Children Under Age 5?
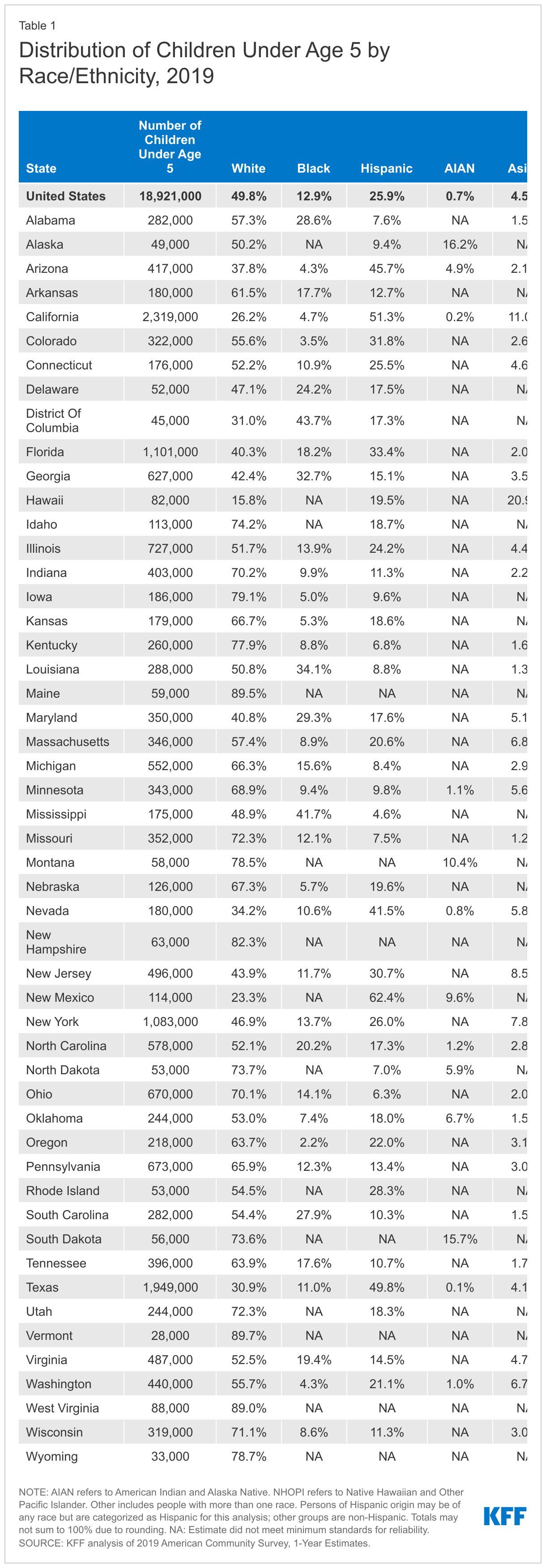
There are approximately 19 million children under the age of 5 in the United States. They account for 6% of the U.S. population. The share of the population represented by young children varies by state, ranging from a low of 4.6% in Maine to a high of 7.7% in Utah. Data about the size and composition of children under age 5 across the country come from the 2019 American Community Survey. Due to data on age only being collected in years, we include all children under the age of 5, including children less than 6 months old who would not be eligible to receive the vaccine under this authorization.
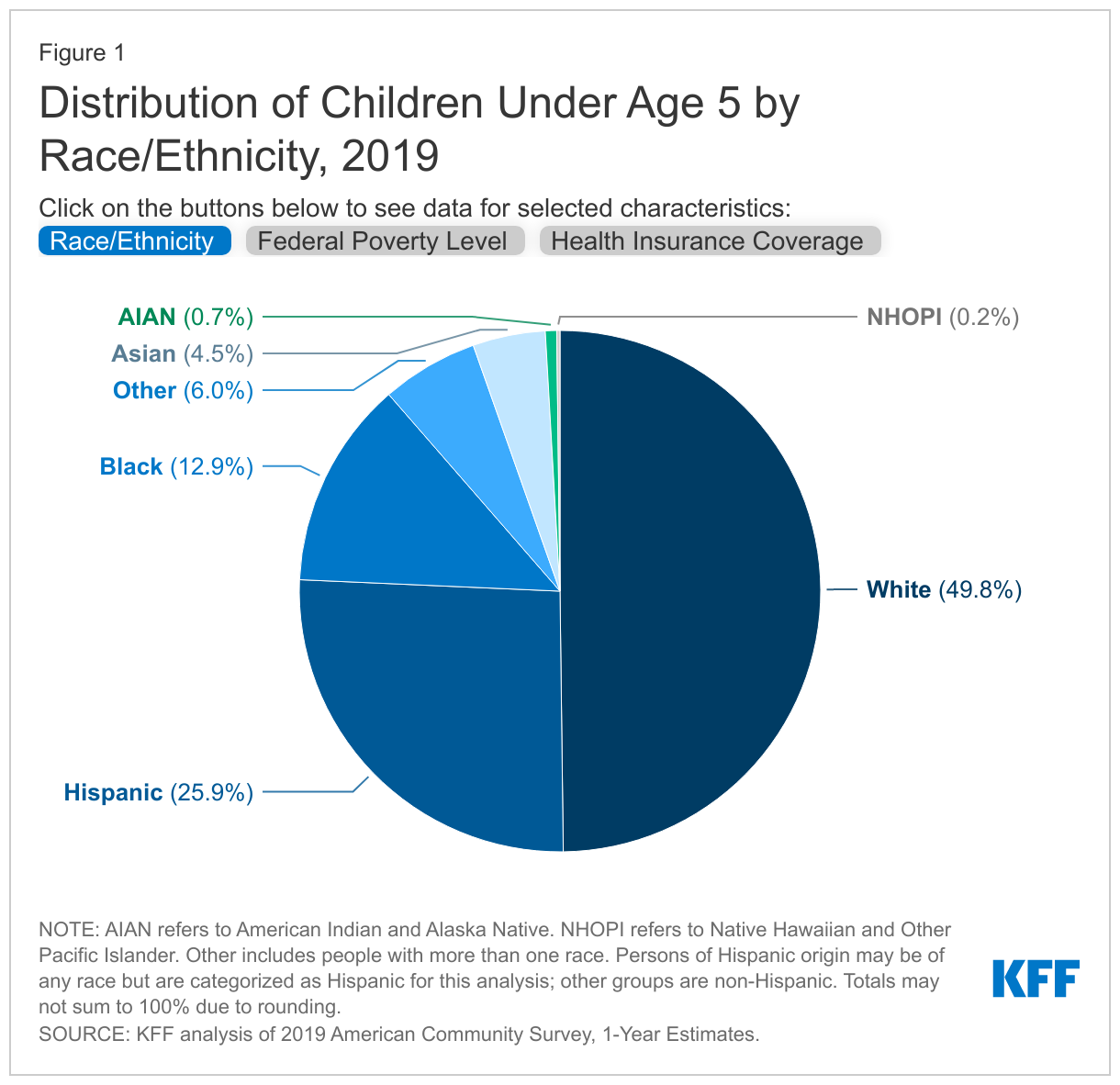
Half of children under the age of 5 are children of color, including more than a quarter who are Hispanic (25.9%), 12.9% who are Black, and 4.5% who are Asian (Figure 1). Smaller shares are American Indian or Alaska Native or Native Hawaiian or Other Pacific Islander (<1 % each). The distribution varies across the country (Table 1). For example, in four states, a third or more of younger children are Black – Georgia (32.7%), Louisiana (34.1%), Mississippi (41.7%) and DC (43.7%). States in the West and South include higher shares of Hispanic children; at least half of younger children are Hispanic in New Mexico (62.4%), California (51.3%), and Texas (49.8%).
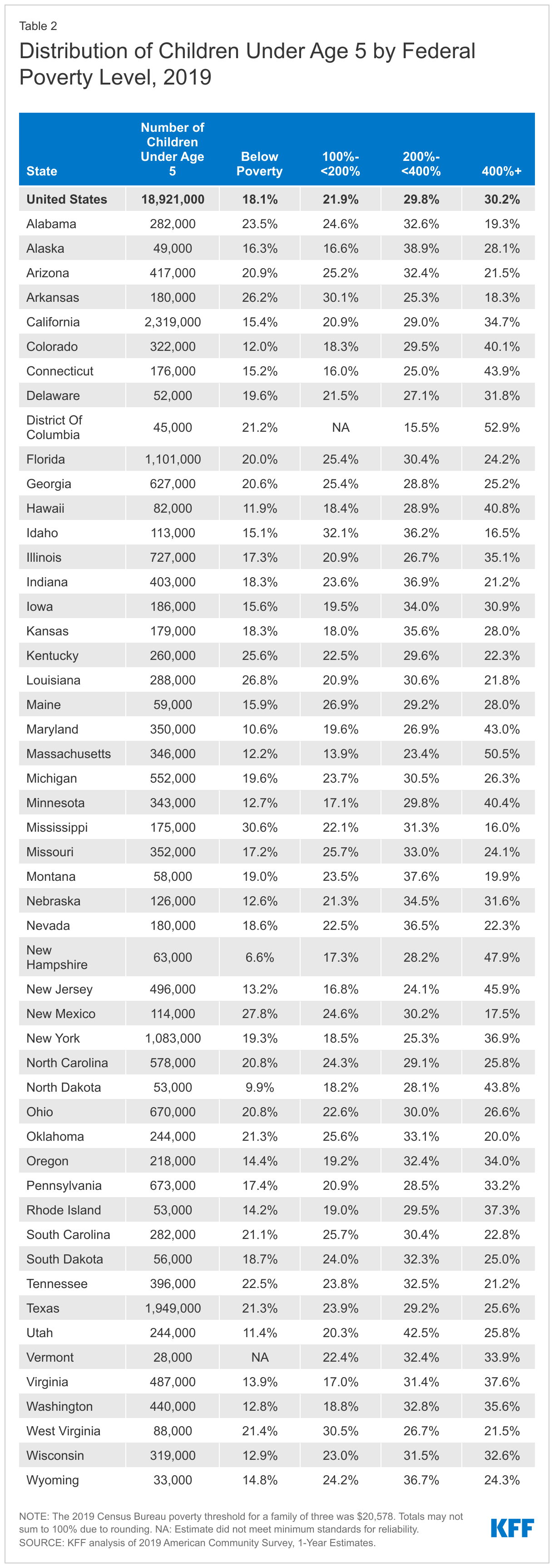
Four in ten children under the age of 5 live in a family with income below 200% of the Federal Poverty Level (FPL), including 18.1% below poverty and 21.9% between 100-200% FPL (Figure 1). An additional 29.8% live between 200-400% FPL and 30.2% are above 400% FPL. This income distribution varies significantly by state (Table 2). For example, the share living in a low-income family (below 200% FPL) ranges from 23.9% in New Hampshire to 56.3% in Arkansas. In four states, the share living below 200% FPL is greater than 50%; In 5 states, more than a quarter of younger children live below the poverty level.
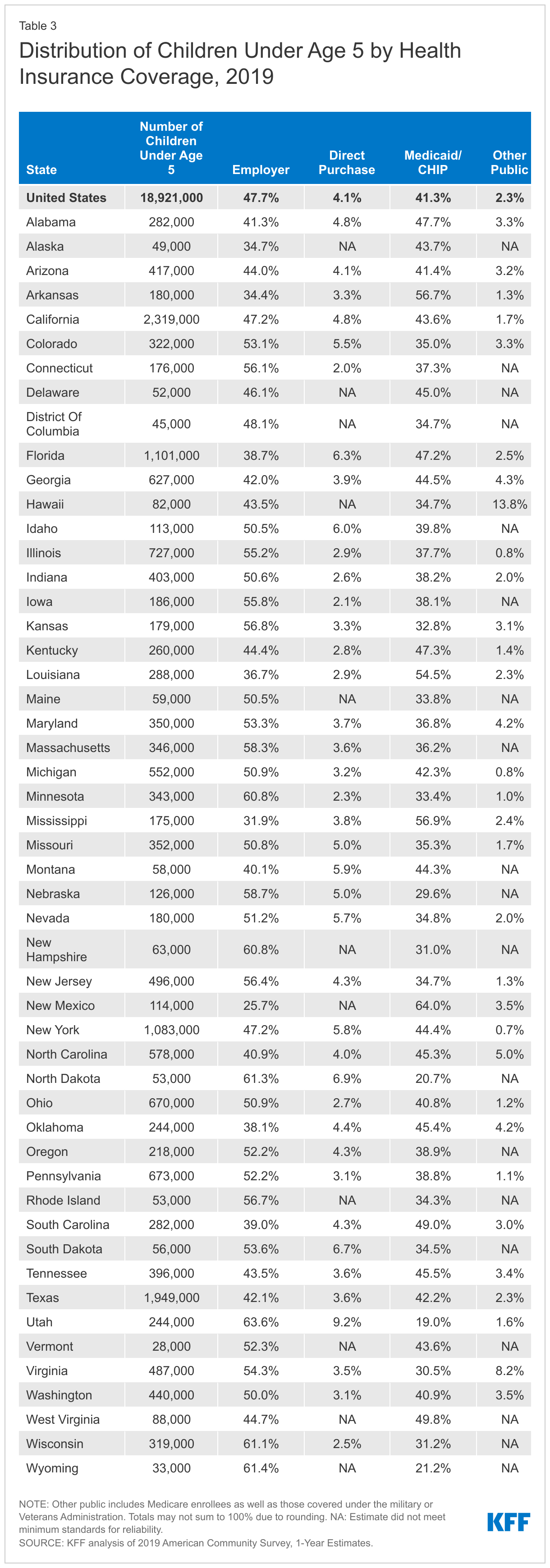
Just over half of children under the age of 5 have private insurance coverage while 41.3% are covered by Medicaid/CHIP, and 4.5% are uninsured. This too, varies by state (Table 3). For example, the share of young children who are privately insured ranges from 35.7% in Mississippi to 72.8% in Utah, while the share covered by Medicaid/CHIP ranges from just 19% in Utah to 64% in New Mexico. The share of young children who are uninsured ranges from 1.3% in Massachusetts to 9.8% in Texas.
How will vaccine roll-out to children under 5 be similar or different to 5-11 year-olds?
Many of the issues that have affected vaccine roll-out to 5-11 year-olds will likely apply in the case of younger children as well, including access challenges in some places. As with 5-11 year-olds, access will likely vary across the country, depending on jurisdictional decisions and implementation plans, the number and location of pediatric vaccinators and sites, the adequacy of provider networks, and communication and outreach plans. Unlike with adults, for example, where vaccines have been widely available at multiple locations to reach the close to 260 million who are eligible, the small population size of children has meant fewer locations with vaccines, an issue that has created access barriers in some rural areas. There are also other factors in play that will likely result in even fewer vaccination sites being available for those under age 5, as discussed below.
COVID-19 vaccination for children under age 5 will require yet another formulation and new doses and supplies to be shipped, and there are differences between the two newly authorized vaccines that may present new challenges for parents in their decision-making. One issue that arose during the roll-out of vaccination to 5-11 year-olds was that their vaccine dosage was lower than that for adults and required new vials to be shipped out to states and pharmacies. This delayed access in the beginning days of eligibility and has meant that vaccinators have had to specifically order and stock pediatric vaccines. For those under 5, there will again be different product configurations, compared to those for 5-11 year-olds, requiring new shipments to jurisdictions. This may once again hold up vaccination opportunities in the early days of authorization.
Moreover, unlike for those ages 5-11 where only one vaccine, Pfizer’s, was authorized for them last November (Moderna’s vaccine was only authorized for 5-11 year-olds on June 17), two vaccines have been authorized for those under 5 at the same time, each with a different number of doses, dosing schedules, and age ranges (see Table 4). Moderna’s vaccine requires two doses, with 28 days between them, while Pfizer’s requires three doses over a much longer time period. As a result, full protection will be reached several weeks sooner with Moderna compared to Pfizer. In addition, while both were found to be safe and effective, their trial results cannot be directly compared due to differences in trial designs. At the same time, parents and vaccine providers are likely more familiar with Pfizer’s vaccine, which has been more widely available to all ages for months. Finally, there are slight differences in the age range authorized for each. These unique factors may present new challenges for parents in their decision-making. It may also be the case that vaccine providers stock only one of the vaccines.
Pharmacies and schools, while important components of the COVID-19 vaccination effort for older children, will be less likely to reach those under 5. Many states limit the age at which pharmacists are allowed to vaccinate children. To address this issue in the context of COVID-19, the federal government amended the PREP Act under the COVID-19 public health emergency to allow pharmacists and pharmacist technicians to vaccinate children as young as 3 for routine immunizations as well as against COVID-19 (upon authorization). However, the Act has not been amended to allow for children under 3 (who account for almost 60% of all children under 5) to be vaccinated at a pharmacy, and according to data from the National Alliance of State Pharmacy Associations (NAPS) and American Pharmacists Association (Alpha), most states do not permit this. In addition, while schools have been important sites for providing access as well as information to help expand vaccination take-up among children as young as 5, and have been encouraged by the federal government to do so, most children under 5 are not yet enrolled in school, limiting this option for younger kids. Lastly, many of the larger vaccine clinics organized by jurisdictions, which were able to expand to new age groups when eligible, are no longer operating.
Pediatricians will likely be more important for vaccinating young children than even their slightly older counterparts. Pediatricians, who are already cited by parents as a highly trusted source of information about COVID-19 for children, will likely play an even more important role in vaccinating children under 5. This is in part due to the more limited access to vaccinations at pharmacies and schools, but also because parents are even more accustomed to getting their routine immunizations for younger children at their doctor’s offices. CDC reports, for example, that in the 2020-21 flu season, 80% of children ages 6 months to 4 years old received their flu vaccine at a doctor’s office, while only 4% of children 2–4 years old and <1% of those under age 2 received their vaccine at a pharmacy. Still, pediatricians face unique challenges with pediatric COVID-19 vaccinations for children, relative to other vaccinations. In addition to cold storage requirements (Pfizer’s vaccine requires an ultra-low temperature for long-term storage), a COVID-19 pediatric vaccine vial, for either vaccine, contains 10 doses which must be used within 12 hours after opening. This means that pediatricians, or any other pediatric vaccinator, would need to be able to vaccinate 10 children within this time period or risk wasting doses. Importantly, the CDC reports that as of early May 2022, more than two-thirds of Vaccines for Children (VFC) program providers were enrolled as COVID-19 vaccine providers, which could help to facilitate access, and encourages jurisdictions to enroll more VFC providers. But it also reports that while 73% of VFC providers intend to offer COVID-19 vaccines children under 5, only 52% intend to do so for children who are not regular patients.
Medicaid will be an especially important avenue for reaching younger children, as will community health centers. Among all children under age 5, over four in ten (41.3%) are covered by Medicaid, and almost three-quarters (74.1%) of children under age 5 with incomes below 200% of FPL are covered by Medicaid. We previously identified how state Medicaid programs and Medicaid managed care plans can facilitate access to vaccines for young, low-income children. In addition to these strategies to increase vaccine uptake, CMS released guidance requiring states to cover COVID-19 vaccine counseling visits for children under Medicaid’s Early and Periodic Screening, Diagnostic, and Treatment (EPSDT) benefit. Community health centers also offer vaccine access points for families with young children. A national network of safety net primary care providers, they are a primary source of care for many low-income populations (91% have incomes below 200% FPL) and communities of color (62%), and they have already been mobilized to provide vaccinations to their clients through the federal Health Center COVID-19 Vaccine Program. Community health centers serve between 5 and 6 million children under the age of 12, including about 2 million under the age of 5.
As with 5-11 year-olds, parents and caregivers will determine how quickly and how many younger children get vaccinated. Our latest COVID Vaccine Monitor report found that about one in five (18%) of parents of children under 5 say they’ll get their child vaccinated against COVID-19 right away once a vaccine is authorized, 38% say they will wait and see, and almost 4 in 10 say they won’t get their younger child vaccinated at all or only will if required. If the experience of vaccinating 5-11 year-olds is a guide for what might happen with younger children, vaccination coverage will likely be quite slow. While the share of parents of 5-11 year-olds who say they have or will get their child vaccinated has increased over time, more than 4 in 10 say they will not do so, or will only do so if required. Our analysis of vaccine coverage among 5-11 year-olds found that while vaccination rose sharply for the two-week period after eligibility, it then dropped steeply and daily rates of administration have remained low. As of June 16, just 29.5% of children ages 5-11 have been fully vaccinated, compared to 75% of those ages 12 and above.
Prioritizing equity is particularly important as vaccination efforts extend to the youngest group of children. Of the estimated 19 million children in the U.S. under age 5, half are children of color (compared to 40% of the U.S. population overall), including more than a quarter who are Hispanic (25.9%) and 12.9% who are Black. Over the course of the vaccination rollout, Black and Hispanic people have been less likely than their White counterparts to receive a vaccine, but these disparities have narrowed over time and been reversed for Hispanic people. Disparities in children’s take-up of the vaccine could reverse that trend. Another KFF COVID Vaccine Monitor report found Hispanic parents, Black parents, and parents with lower incomes were more likely to say they might have to miss work to get their child vaccinated, that they won’t have a trusted place to go, or that they’ll have difficulty traveling to a vaccination location compared to other parents. To mitigate similar disparities in vaccination rates among children, it will be important to address potential access barriers, ensure vaccinations are available through trusted sites, and address parent/caregiver concerns and questions through trusted individuals in the community. CDC guidance provided to jurisdictions asks them to take equity into consideration when selecting vaccine sites, particularly those initially selected in the early days of roll-out. Data on vaccination rates for children by race and ethnicity is important for being able to identify disparities and to direct resources to address them. However, the federal government is not currently reporting vaccinations among children by race and ethnicity, and only a handful of states report these data.
Implications
Many of the issues that presented challenges to vaccinating 5-11 year-olds will likely be at play in the effort to vaccinate younger children; however, there are additional factors to consider. These include the likelihood that there will be fewer places to get vaccinated for this age group, due to more limited opportunities at pharmacies and schools, and reduction in larger scale vaccination clinics formerly operated by jurisdictions across the country. Parents may be more likely to seek out pediatricians, who themselves may face new challenges to vaccinating younger children against COVID-19, or to encounter an offer for COVID-19 vaccination only when they bring their child to a routine check-up, a time period which may or may not correspond to a COVID-19 surge. In addition, there will be new communication challenges to reaching parents of children in this age group, given that two vaccines are being made available at the same time, each of which requires a different number of doses and time period to achieve full protection. Finally, particular attention to equity will be important given that large shares of children under 5 are children of color and live in low-income households.