The Availability and Use of Medication Abortion
This factsheet was updated on March 10, 2025 to reflect policy updates and new data.
On June 13, 2024, the Supreme Court of the United States ruled in Alliance for Hippocratic Medicine (AHM) v. FDA that the AHM does not have standing to sue the FDA for injury. The Court had been asked to ascertain whether or not the FDA had violated federal law when it modified its guidelines for prescribing and dispensing mifepristone in 2016 and 2021. However, three state Attorneys’ Generals have intervened in this case in district court, and it is unclear how this action will shape the case when it goes back to the 5th Circuit Court of Appeals and then back to the originating federal district court.
On June 24th, 2022, the Supreme Court ruled on the Dobbs v. Jackson Women’s Health Organization, overturning Roe v. Wade. States can now set their own policies protecting or banning abortion without any federal standard protecting access to abortion. This has created a new focus on medication abortion as an option for expanding access to people facing barriers to abortion care.
Medication abortion, also known as medical abortion or abortion with pills, is a pregnancy termination protocol that involves taking two different drugs, Mifepristone and misoprostol, that can be safely used up to the first 70 days (10 weeks) of pregnancy according to the U.S. Food and Drug Administration (FDA). The World Health Organization has authorized use up to 12 weeks of pregnancy. Since the FDA first approved the drug in 2000, its use in the United States has quickly grown. In 2023, 63% of abortions in the US were medication abortions. The medication abortion drug regimen approved by the FDA is available in many states across the nation, however, dispensing these pills for the purpose of terminating a pregnancy is now banned in some states. This factsheet provides an overview of medication abortion, how it is used and regulated, the role of the drug in self-managed abortions, and an analysis of the intersection of federal and state regulations pertaining to its provision and coverage.
What is a Medication Abortion?
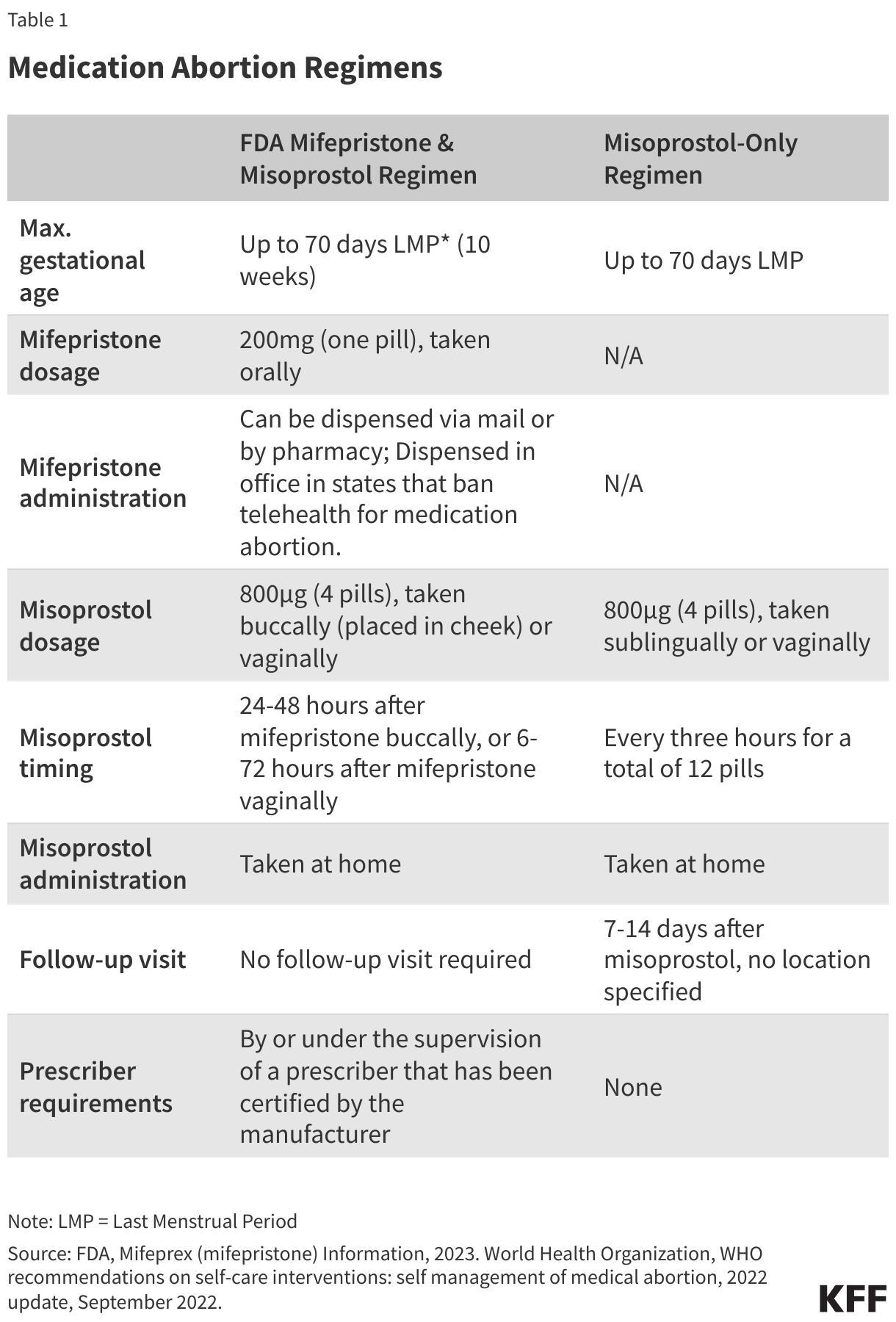
There are two medication abortion regimens that have a long safety and efficacy record: mifepristone with misoprostol and misoprostol alone. Both involve taking oral medications to terminate a pregnancy.
Mifepristone and Misoprostol Regimen
The most common medication abortion regimen in the United States involves the use of two different medications: mifepristone and misoprostol. Mifepristone, also known as the abortion pill, or RU-486 is sold under the brand name Mifeprex and through a generic manufactured by GenBioPro in the United States. Mifepristone works by blocking progesterone, a hormone essential to the development of a pregnancy, and thereby preventing an existing pregnancy from progressing. Misoprostol, taken 24–48 hours after mifepristone, works to empty the uterus by causing cramping and bleeding, similar to an early miscarriage. A follow-up visit can be scheduled a week or two later, to confirm that the pregnancy was terminated via ultrasound or blood test. The FDA has found that medication abortion is a safe and highly effective method of pregnancy termination. When taken, medication abortion successfully terminates the pregnancy 99.6% of the time, with a 0.4% risk of major complications, and an associated mortality rate of less than 0.001 percent (0.00064%).
The FDA first approved Mifeprex in 2000. In 2016, the FDA updated and approved a new evidence-based regimen and drug label. This regimen approves use of medical abortions for up to 70 days (10 weeks) of pregnancy (Table 1). Until 2019, mifepristone was only sold under the brand name Mifeprex, manufactured by Danco Laboratories. In 2019, the FDA approved GenBioPro, Inc.’s application for generic mifepristone. In 2021, the FDA announced they would not enforce the in-person dispensing requirement that had been in effect since the approval of mifepristone. With the new REMS in 2023, the in-person dispensing requirement was formally removed.
Misoprostol-Only Regimen
While the combined regimen of mifepristone and misoprostol for medication abortion is recommended, there is a second medication abortion protocol using misoprostol–only that is more commonly used internationally and currently not approved by the FDA. The regimen is also recommended for up to 70 days (10 weeks) of pregnancy. It involves taking 800 µg (4 pills) of misoprostol sublingually or vaginally every three hours for a total of 12 pills. Research has shown the misoprostol-only regimen to be a safe and highly effective method of pregnancy termination, however it may result in a higher incidence of side effects, particularly diarrhea, fever and chills. When taken, the misoprostol-only regimen successfully terminates the pregnancy approximately 80-100% of the time, with a complication rate of less than 1%. Some U.S. telehealth organizations have been providing the misoprostol-only regimen as an option for medication abortion for a number of years.
Risk Evaluation and Mitigation Strategies (REMS)
In 2011, the FDA added a Risk Evaluation and Mitigation Strategy (REMS) to the dispensing requirements for mifepristone permitting only medical providers who had obtained certification from the manufacturer to prescribe and directly dispense the drug. This requirement has had the effect of limiting the number of clinicians able to prescribe medication abortions, but also necessitated an in-person visit to a health care setting and meant patients could not obtain the medication from a retail pharmacy or by mail.
On December 16, 2021, the FDA removed the in-person dispensing requirement for mifepristone and expanded the distribution to include certified pharmacies in addition to certified clinicians. This change removed the requirement to dispense the medication in person and expanded the opportunity for telehealth in states that have not banned abortion. Despite the change to the in-person requirement, prescribers are still required to be certified by the manufacturers. On January 3, 2023, the FDA approved a protocol for pharmacies, allowing those that have been certified by the manufacturers to dispense mifepristone directly to patients. In March 2024, two major pharmacy chains in the U.S. announced they had become certified to dispense mifepristone and would start dispensing the medication to patients in certain states later that month. Table 2 shows the change in REMS from 2011 to 2023.
State Regulations and Availability
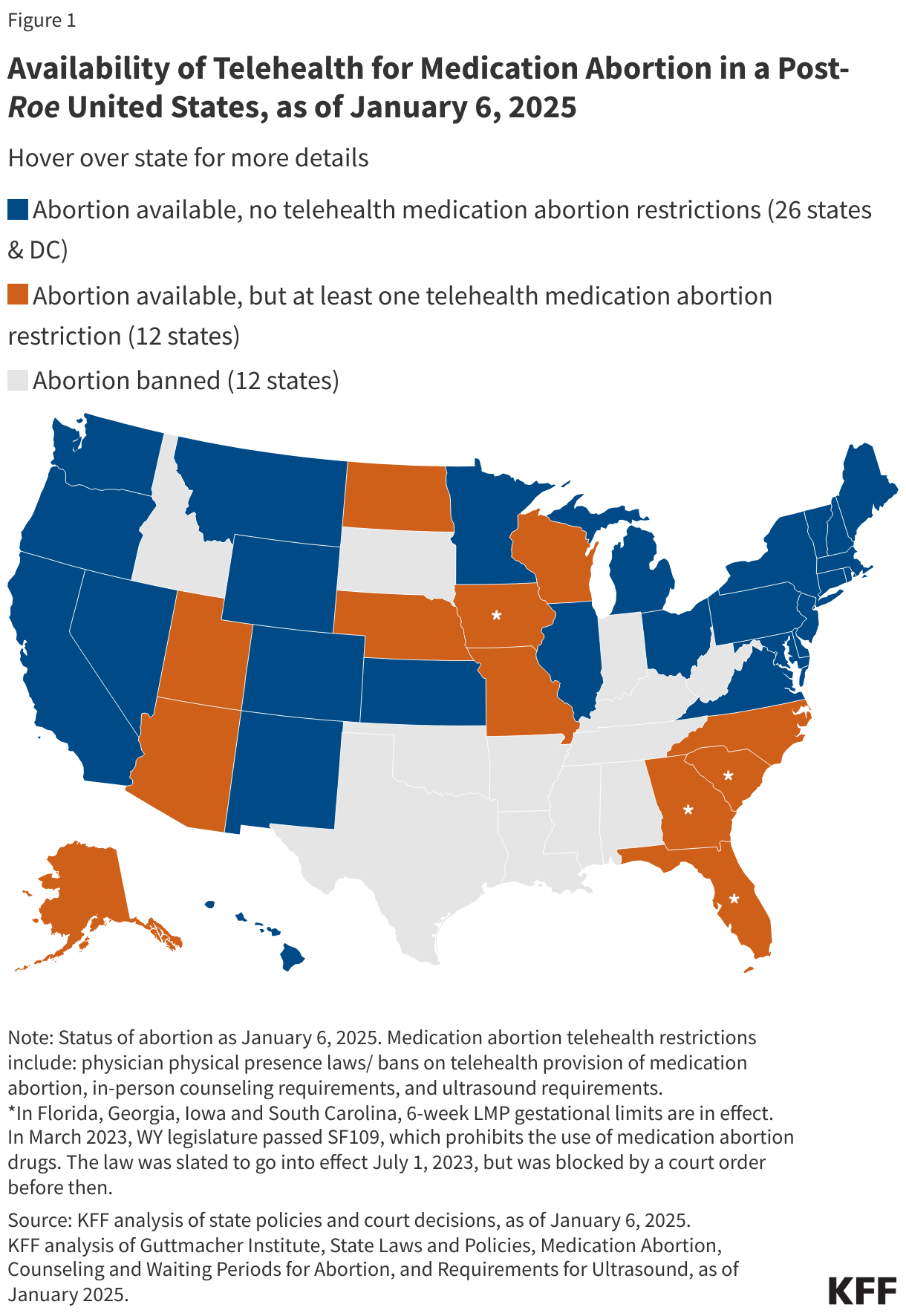
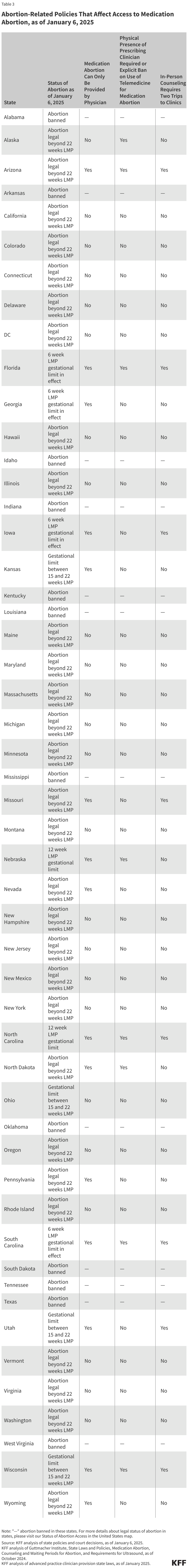
State laws that ban or restrict abortion apply to medication abortion just as they apply to abortion procedures. There are currently large swaths of the country, mostly in the South and Midwest that ban abortion. Even though the federal FDA has approved mifepristone as safe and effective, following the Dobbs decision, the availability of medication abortion today depends on state laws. Even before the Dobbs decision, however, some states restricted access to medication abortion either by blocking the use of telehealth abortions by mandating in-person visits for abortions, imposing requirements for in-person dispensing, or limiting the kinds of clinicians who could dispense the pills (only permitting MDs to dispense). In many states these laws are now superseded by state laws that ban abortion.
Box 1: Conflict Between Federal and State Regulations
There are two challenges in federal court to abortion prohibitions and restrictions on federal preemption grounds. The maker of a generic mifepristone medication, GenBioPro, Inc., is challenging West Virginia’s total abortion ban, and an ob-gyn, Dr. Bryant, is challenging the abortion restrictions in North Carolina, which include requirements that Mifepristone be dispensed in person by a physician after state-mandated counseling session and a 72-hour waiting period. In both cases, plaintiffs argue that the FDA’s authorization and regulation of Mifepristone preempt state law banning the use of the medication or regulating its use more strictly, and given this, enforcement of the state laws should be blocked.
There are other ways that state laws also affect use of and access to medication abortion. Some states require that patients be counseled about unsubstantiated claims about the ability to reverse an abortion after mifepristone is ingested. For example, Nebraska, a state that hasn’t banned abortion, requires patients to be counselled that medication abortion may be reversed if given a high dose of progesterone after taking mifepristone—despite a lack of scientific evidence to support this claim. Similarly, Utah requires counseling that mifepristone alone is not always effective in ending a pregnancy and that patients may still have a viable pregnancy after taking mifepristone despite its record of effectiveness. Prior to banning abortion Arkansas, Idaho, Kentucky, Oklahoma, South Dakota had similar requirements. Research demonstrates that APCs, such as nurse practitioners, physician assistants, or nurse-midwives, can provide medication abortion as safely as physicians can, but they are only permitted to do so in 24 states and DC out of the 38 states where abortion is not currently banned.
Telehealth
Telehealth can be used to expand access to health services in areas where the number of clinicians who provide abortion care is limited. Many patients, particularly those who live in rural communities, must travel long distances to obtain abortion services even in states where abortion is still permitted, which has raised interest in the potential of telehealth to expand access medication abortion. Because the updated FDA label now allows for telehealth, mifepristone has emerged as an option for patients who are either unable to travel to clinic or for other reasons wish to have an abortion in the privacy of their own home, if permitted by state law.
As part of efforts to limit abortion access, some states have taken action to block the use of telehealth for abortion. Among the states that have not banned abortion, thirteen states have at least one restriction that requires at least one trip to the clinic, and effectively ban telehealth for medication abortion (Figure 1).
Cost
According to a recent study, the median self-pay price for medication abortion significantly increased from $495 in 2017 to $560 in 2020. Although Danco Laboratories does not make the cost of Mifeprex public, providers report that Mifeprex pills alone cost them around $90 a pill. GenBioPro, the manufacturer of the generic mifepristone drug also does not report the cost of their pill but has stated that they want to drive down costs for those who choose medication abortion. Private insurance coverage of abortion services is variable and depends on the type of insurance plan, the policy holder’s state of residence, and employer coverage decisions. Federal Medicaid funding only pays for abortions when the pregnancy is a result of rape or incest or a threat to the pregnant person’s life. Seventeen states have opted to use their own state funds to pay for abortions, including medication abortions, for Medicaid enrollees. For those who do not have abortion coverage, there are limited means of financial support, promoted on both Danco and GenBioPro’s website. The National Abortion Federation, as well as local abortion funds are sometimes able to cover some of the cost of an abortion (including travel) for a pregnant person. Outside of these funding sources or a sliding fee scale clinic, there are few options to help with abortion costs.
Use
Although the overall rate of abortion has declined over the past two decades, the use of medication abortion as a share of all abortions has greatly increased over the years. According to Danco Laboratories, by 2016, over 2.75 million women in the United States had used Mifeprex since its FDA approval in 2000. Data from the Centers for Disease Control and Prevention (CDC) show medication abortions have increased steadily over the past 15 years. A Guttmacher Institute report found that medication abortion accounted for 63% of all nonhospital abortions in 2023, although this is likely an undercount since it does not include self-managed abortions.
Self-Managed Abortion
Self-managed abortion, sometimes referenced as “self-induced” or “at-home” abortion, is when a person ends a pregnancy outside the medical care setting, typically by ordering abortion pills online. Patients may seek to manage their own abortion for many reasons, including state bans, clinic access barriers, cost, transportation, time limitations, and privacy. There are different medication protocols that can be used for a self-managed abortion. As detailed in this JAMA review, an individual can take the FDA-approved medication abortion regimen of mifepristone and misoprostol pills or misoprostol pills alone.
There are a number of companies that offer self-managed abortion services using different approaches. Some companies have a clinician that reviews a customer’s medical information and may have a telehealth visit. Other companies, such as AidAccess based outside the United States, will mail abortion medications to an individual without requiring a clinician visit. Plan C Pills operates a website that provides a list of online retailers in every state and includes information about clinician involvement, price, ship time, product quality, as well as information about how to take the drugs and resources for financing assistance. Prices differ between companies, but typically costs are lower than average charges for a clinic abortion as there is typically no involvement with a brick and mortar clinic. One 2017 study found the median cost of mifepristone-misoprostol products ordered online is approximately $205.
Self-managed abortions have occurred around the world for years, particularly in areas where abortion access in the medical care system has been limited. Before the FDA removed the REMS requirement for in person dispensing of medication abortion, access to telemedicine abortion care (either through the medical system or self-managed) was extremely limited in the United States. Interest in self-managed abortion has grown in recent years in the United States, particularly since the Dobbs decision. For example, research has documented a sharp increase in requests for abortion pills to the company AidAccess since the Dobbs ruling, rising from an average of 82.6 requests daily before the ruling to 213.7/day after it was issued, with the largest increases in states that have banned abortion.
None of the current state abortion bans or restrictions criminalize pregnant people for obtaining self-managed abortions, yet there have been many documented cases of people facing criminal charges for self-managing an abortion. Some states impose criminal penalties on clinicians or others who help an individual obtain abortion services. These policies can create a climate of fear related to self-managed abortion for both patients and clinicians, for instance if a patient were to present to a clinician for a complication or follow up care after a self-managed abortion. Some major medical and public health groups such as ACOG, AMA, and APHA oppose any criminalization of patients that seek self-managed abortion.
Conclusion
The use of medication abortion has grown significantly since its approval by the FDA in 2000. The FDA update of the REMS in 2023 has expanded the availability of medication abortion and broadened the use of telehealth dispensing. However, state abortion bans, specific bans on telehealth for medication abortion, and state-level requirements for in-person dispensation of mifepristone and for in-person counseling visits and ultrasounds that are not medically recommended will continue to restrict access in many states.