Update on Children’s COVID-19 and Routine Vaccination Trends Heading into Winter and as Respiratory Viruses Surge
For updated data on child vaccination trends, including MMR vaccination coverage and vaccine exemption rates, read our latest analysis: Headed Back to School in 2024: An Update on Children’s Routine Vaccination Trends
In recent weeks, cases of respiratory syncytial virus (RSV) in younger children have surged, and the Centers for Disease Control and Prevention (CDC) has reported seeing early increases in seasonal flu cases. The CDC notes that the past two years have seen low flu activity, likely meaning reduced population immunity headed into this years’ flu season, especially among young children who have never been exposed to or vaccinated against the flu. Winter could also bring an increase in COVID-19 cases, as the weather gets colder and activities move indoors. While children’s cases of COVID-19 are usually mild, this is not always the case, and children are particularly vulnerable to flu and RSV. The combination of all three could have more serious ramifications for children and also has the potential to overwhelm hospitals. Vaccination against COVID-19 and flu can provide protection; however, COVID-19 vaccination rates have stalled and remain low for younger children and other routine vaccination rates may have been impacted by the pandemic. This policy watch describes recent trends in children’s COVID-19 and routine vaccinations and explores strategies to increase vaccination rates among children as we head into the winter season.
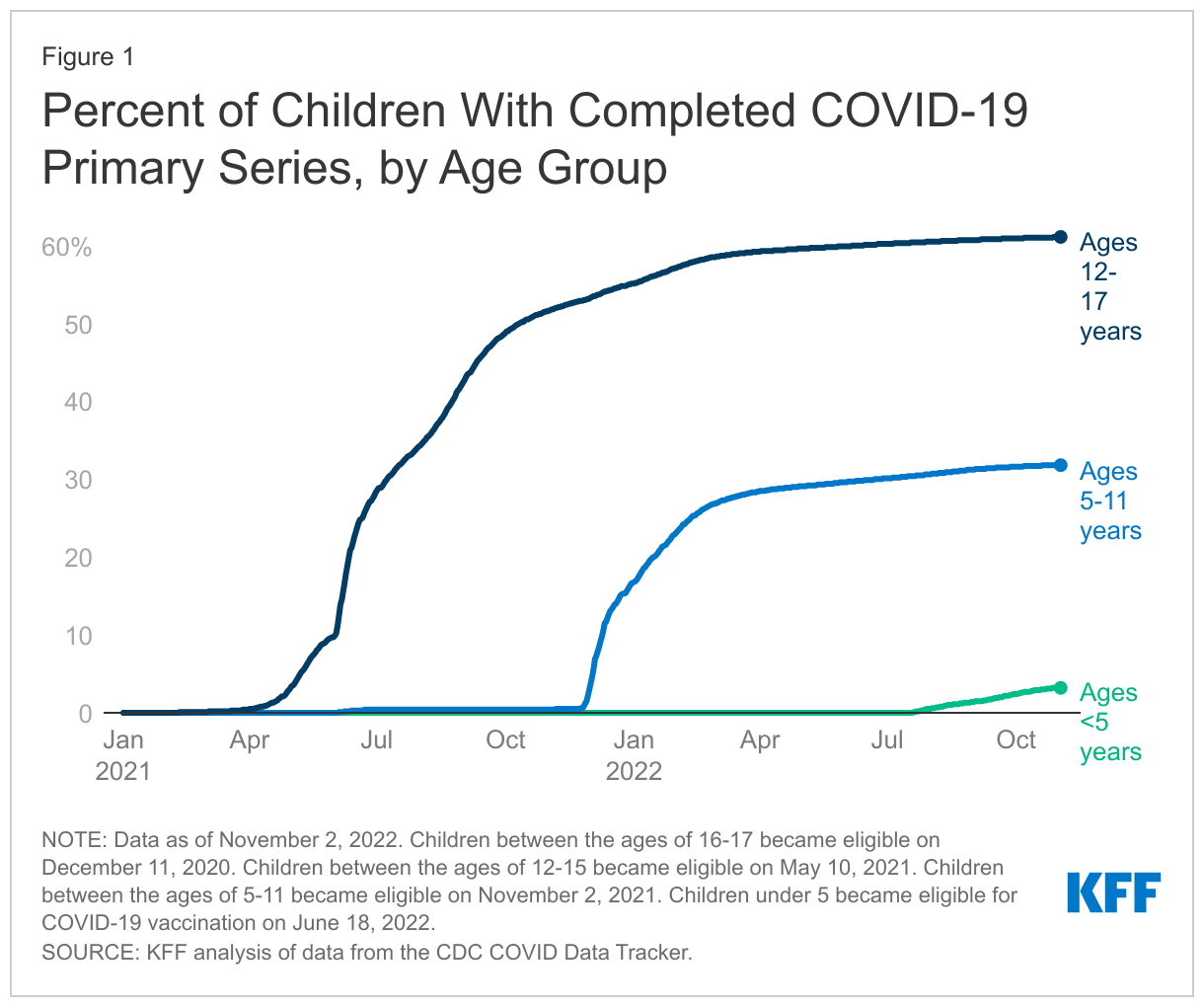
COVID-19 vaccine uptake among children has stalled and vaccination rates remain low for young children. As of November 2, 2022, 3.2% of children under age five and 31.8% of children ages 5-11 had completed their primary series, which is the initial doses of a COVID-19 vaccine, most commonly two shots of a mRNA vaccine but can vary by age, immune status, and vaccine product (Figure 1). Uptake is higher among those ages 12-17, at 61.1%. Some of this variation in uptake reflects the amount of time since COVID-19 vaccines were authorized by the FDA for different age groups. The vaccine was first authorized for 16 and 17 year-olds in December 2020 and for 12-15 year-olds in May 2021. It was authorized for 5-11 year-olds in November of 2021 and finally, for children under age five in June of 2022. However, some of it reflects parental views and concerns. KFF’s COVID-19 Vaccine Monitor from September 2022 reported that over half (53%) of parents of children under five and over a third (35%) of parents of children ages 5-11 said they will “definitely not” get their child vaccinated. KFF surveys from July 2022 found parents of young children were concerned about the newness of the vaccine and not enough testing or research, side effects, and worries over the overall safety of the vaccines.
Bivalent boosters were recently authorized for children ages five and older, but it is unclear how many children will get the new booster. Children’s vaccination rates for the first booster (no longer authorized) were low. As of November 2nd, 5.3% of children ages 5-11 and 18.2% of children ages 12-17 had received their first booster dose. Completion of the primary series at least two months earlier is required to receive the new bivalent booster; thus, low primary series rates, especially among children ages 5-11, means fewer children are eligible for the new booster. As of November 2nd, 0.5% of children ages 5-11 and 2.6% of children ages 12-17 have received an updated (bivalent) booster dose. While children usually have more mild COVID-19 cases, some children do develop severe illness and some have shown symptoms of long COVID following diagnosis. Boosters combat waning immunity and can help reduce the risk of infection and onward transmission.
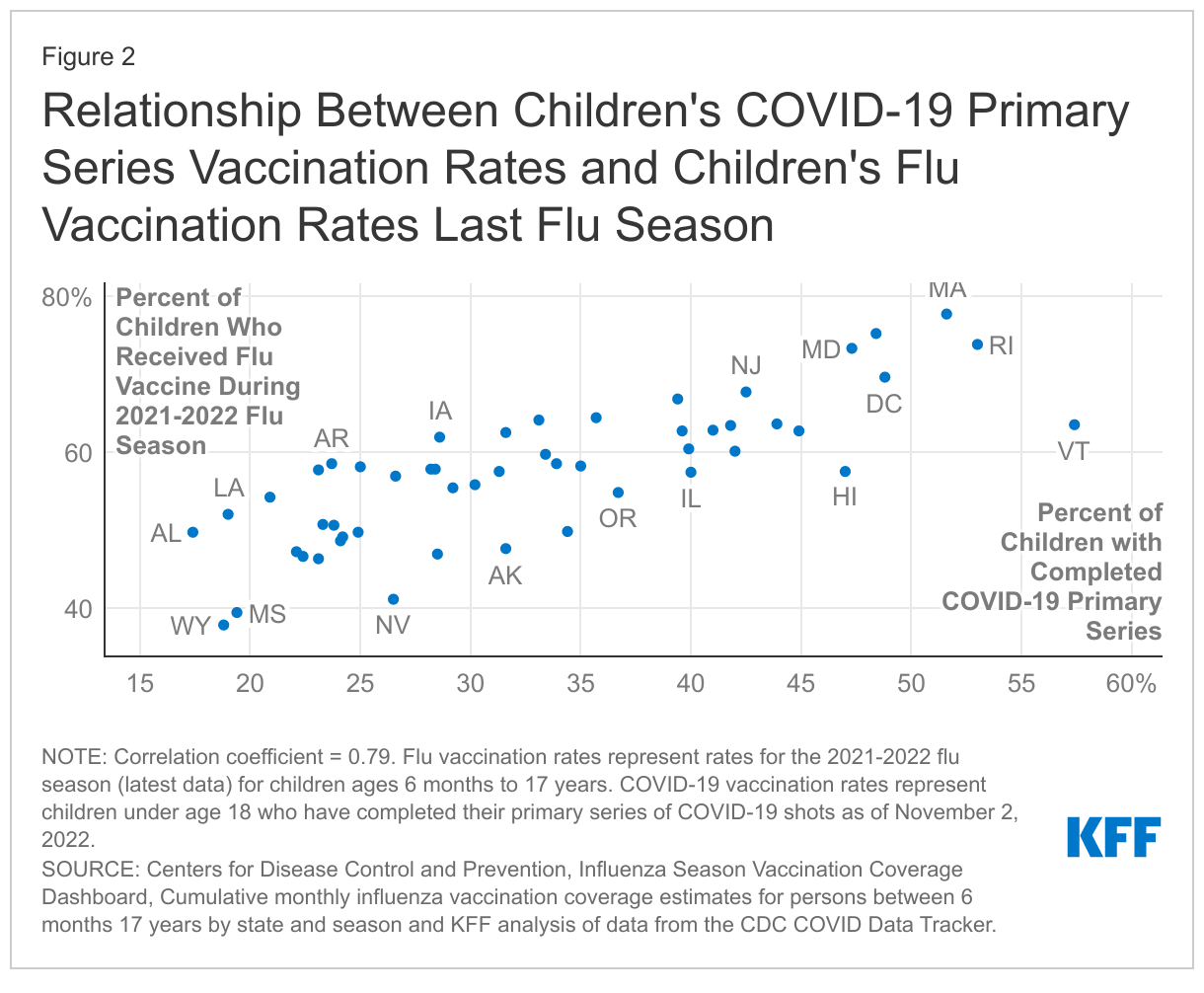
The pandemic has also led to declines in children’s flu vaccinations, though the impact of the pandemic on other routine vaccinations remains uncertain. While children’s flu vaccination rates from last year’s flu season (2021-2022) were similar to the previous year (2020-2021), they were almost 6 percentage points lower than in 2019-2020, right before the pandemic began. Flu vaccination rates among children vary widely by state and appear to be correlated with COVID-19 vaccination, meaning states with higher uptake of flu vaccination last season also have higher uptake of COVID-19 vaccination, and vice versa (Figure 2). Some public health leaders have expressed concern that COVID-19 vaccine hesitancy may be spilling over to routine child immunizations, and one study found factors impacting COVID-19 vaccine uptake could be impacting flu vaccine uptake. In addition, vaccination rates for other routine childhood vaccines also declined early in the pandemic, and the CDC reports vaccination coverage of all state-required vaccines for children in kindergarten (MMR, DTaP, and varicella) declined slightly by 1% in the 2020-2021 school year (the first full-pandemic school year) compared to the previous school year. Some states and local areas have more recently reported seeing reduced childhood immunization rates. However, CDC data for the most recent two school years are not yet available, and the overall impact on childhood routine vaccination rates remains unclear at this time.
There are specific policy considerations for vaccinating young children, and some states have developed successful strategies for increasing vaccination rates among children. States with the highest COVID-19 vaccination rates among children ages 5-11 used incentives, school-based vaccination clinics, parent-friendly websites, and the media to encourage parents to vaccinate their young child. School vaccine mandates can also be used as a tool to increase COVID-19 vaccine uptake among children, but at this time, only a few states have COVID-19 vaccine mandates for school staff or students. Preventive care appointments are also an important component when addressing routine vaccination rates, as some parents may not encounter an offer of a vaccine until they go in for a routine visit to a pediatrician and pediatricians are considered highly trusted sources of information by parents. Recently, the CDC’s Advisory Committee on Immunization Practices (ACIP) voted to add the COVID-19 vaccines to the recommended pediatric immunization schedule that includes the other routine vaccines for children depending on age. This is part of a regular, annual process in which ACIP meets to vote on adding newly recommended vaccines to the child and adult immunization schedules.
Because Medicaid covers four in ten children in the U.S., the program can play an important role in facilitating access to COVID-19 and other routine vaccines for children, especially those who are low-income. To increase COVID-19 vaccine uptake, state Medicaid programs and Medicaid managed care plans have undertaken various initiatives, including financial incentives for managed care plans that meet vaccination targets. Other recent federal actions can help increase routine vaccination rates more broadly. Data for children enrolled in Medicaid and CHIP showed a 9% decline in all routine vaccinations when comparing the COVID-19 public health emergency (PHE) period (March 2020 – April 2022) to a pre-PHE period (January 2018 – February 2020), with the largest declines for HPV, hepatitis A, and flu vaccines. Provisions included in recent legislation to bolster Medicaid’s Early and Periodic Screening, Diagnostic and Treatment (EPSDT) benefit can help more Medicaid-covered children receive all recommended screenings and services, which includes routine vaccinations. Further, the Child Core Set measures, designed to improve the quality of care for children in Medicaid and CHIP, will become mandatory to report in 2024 and track state-level immunization rates for children and adolescents.
Children will still be able to access needed COVID-19 vaccines for free following the end of the COVID-19 PHE and even when federal supplies of vaccines run out. While the end of the PHE as well as the depletion of federally-purchased supply could curtail access to some COVID-19 countermeasures like tests and treatments, COVID-19 vaccines, including boosters, will continue to be available for free to all children even when there is no longer any federally-purchased supply remaining or PHE protections in place. Once the supply of government-purchased vaccines runs out, the Vaccines for Children program (VFC) will provide access to COVID-19 vaccines for children who have Medicaid, who are uninsured or underinsured, or identify as American Indian or Alaska Native. While vaccines are free through the VFC program, participating health care providers can charge an administrative fee. Children who are uninsured may be eligible for free or reduced cost off vaccine administration through a community health center.
As we head into the winter season, RSV and other respiratory viral infections are surging in young children, flu infections are higher than usual for the time of year, and COVID-19 cases are also expected to rise. At same time, children’s COVID-19 vaccination rates have stalled and remain low, and other routine children’s vaccinations may have also declined since the pandemic began. While most children have mild COVID-19 cases, vaccines can offer protection for children and families as travelling and gatherings ramp up for the holiday season. Various tools, including incentives, outreach, and the media, can be used to increase children’s COVID-19 vaccination rates as well as routine vaccinations more broadly.