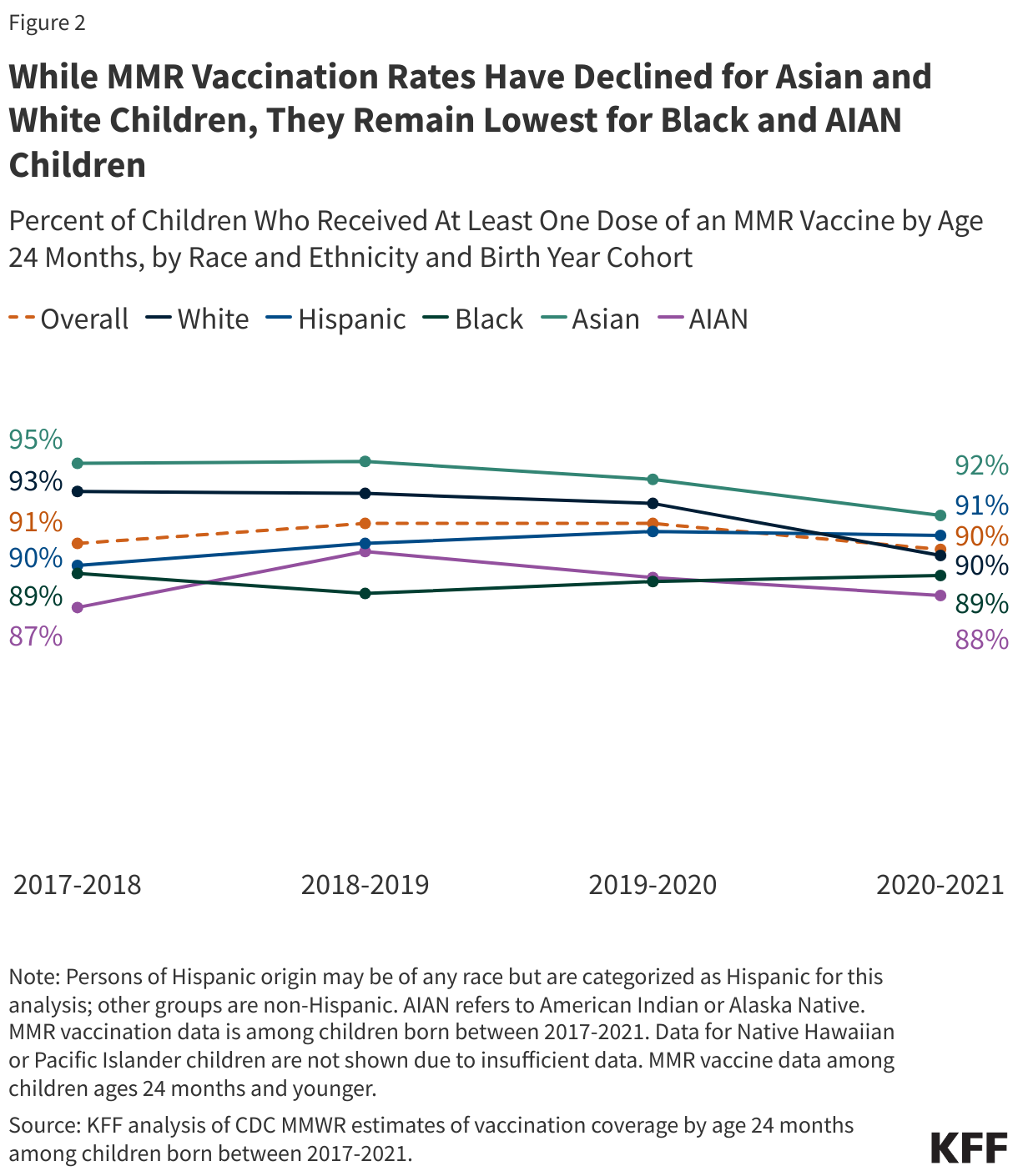
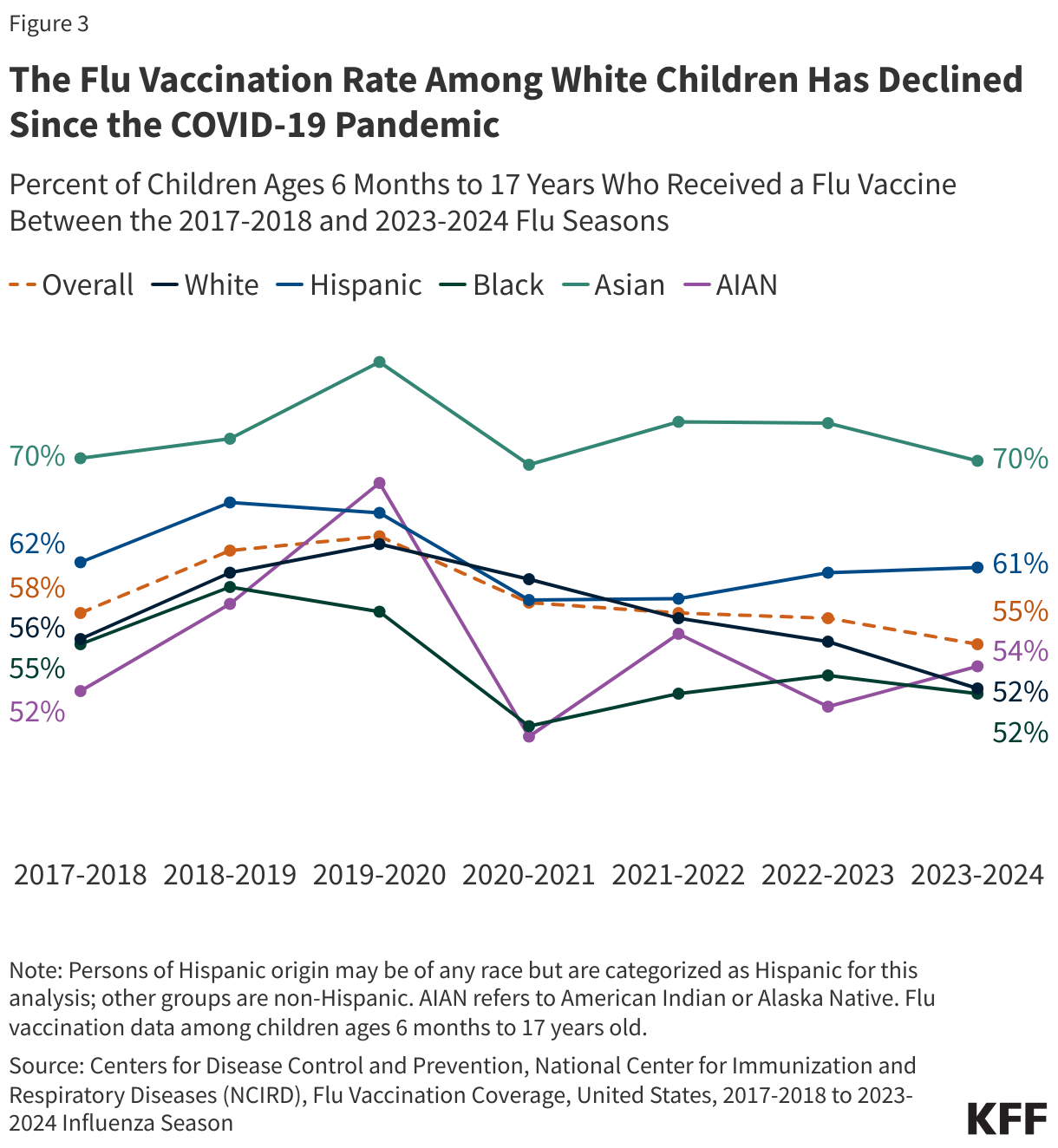
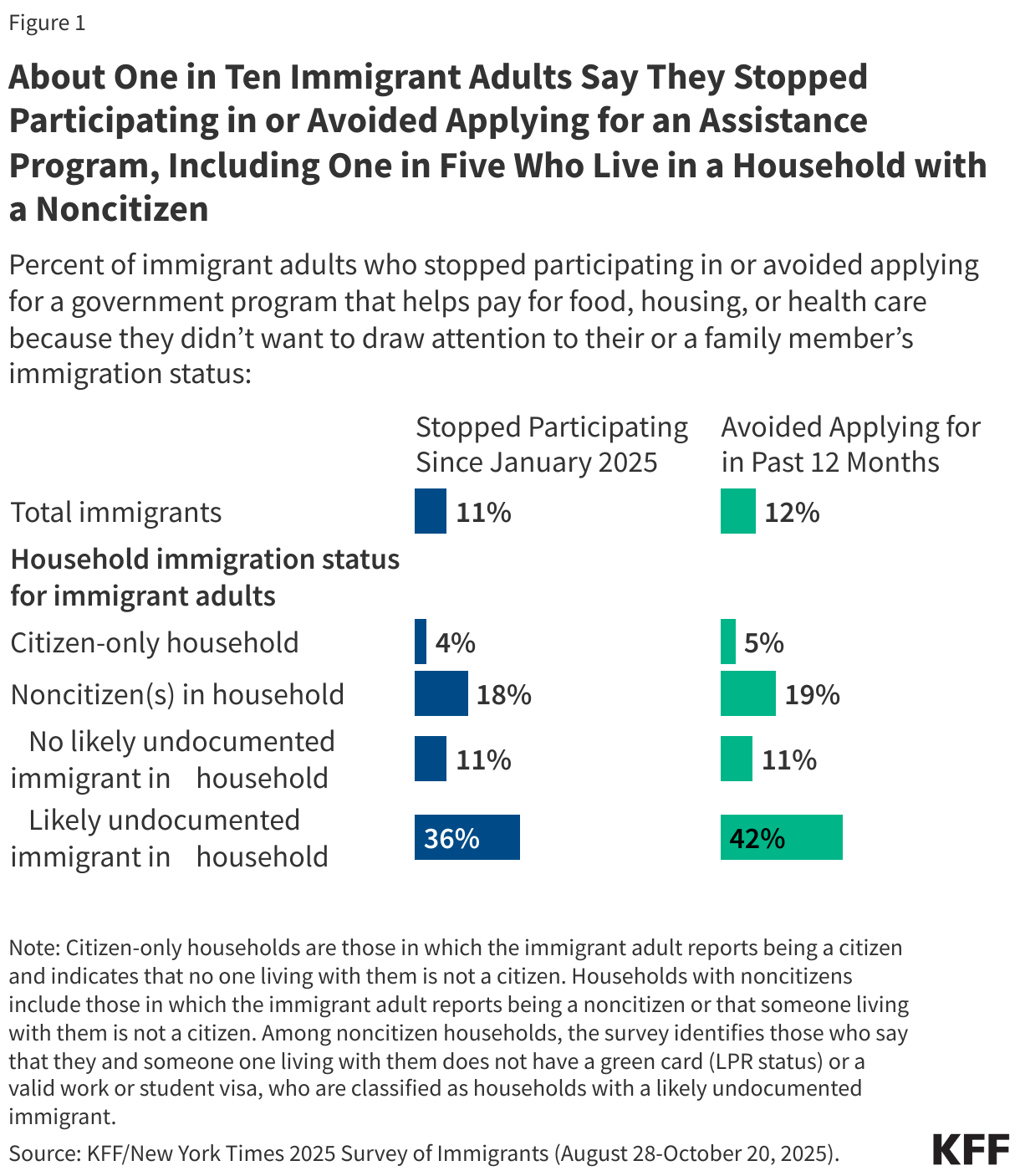
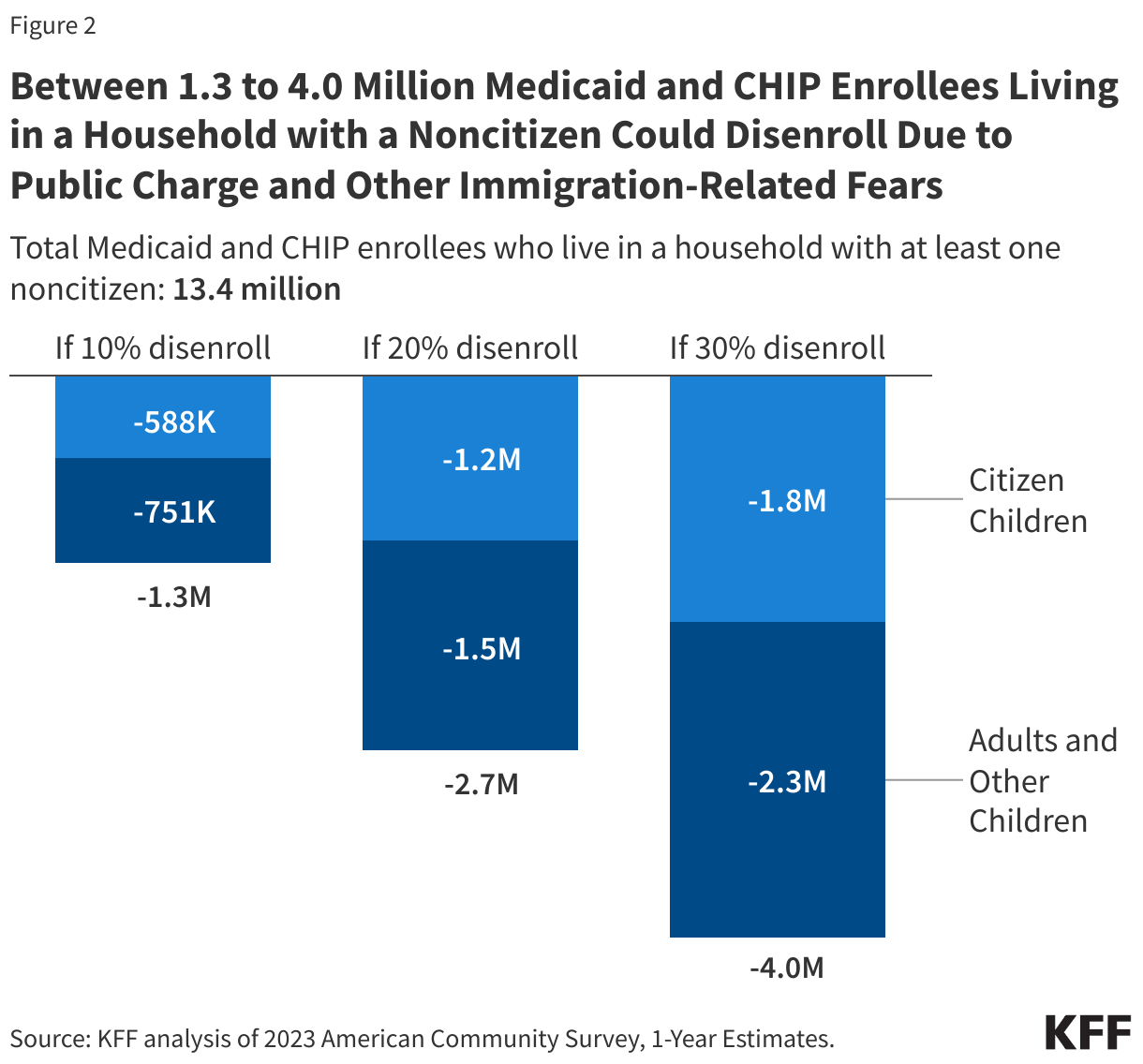
Litigation Challenging the 2025 Budget Reconciliation Law’s Provision Blocking Federal Medicaid Payments to Planned Parenthood
All ligation challenging Section 71113 has been voluntarily dismissed as of March 17, 2026:
- The Family Planning Association of Maine voluntarily dismissed their case on 12/29/25
- Planned Parenthood Federation of America voluntarily dismissed their case on 1/20/26
- State of California voluntarily dismissed their case on 3/17/26
This brief was updated on January 22, 2026, to reflect recent court decisions and developments.
Introduction
One of the immediate impacts of the 2025 Federal Budget Reconciliation Law is a provision, Section 71113, that blocks certain reproductive health care providers (who also provide abortions) from receiving federal Medicaid reimbursement for one year. The law impacts three organizations: (1) Planned Parenthood; (2) Maine Family Planning, a network of clinics in Maine, (3) and Health Imperatives, a network of specialized reproductive health clinics based in Massachusetts. Shortly after President Trump signed the law, Planned Parenthood, Maine Family Planning (legally known as the Family Planning Association of Maine), and 22 states and the District of Columbia filed separate legal challenges to prevent the implementation of Section 71113. One of the key issues in the litigation is whether Congress acted rationally to reach its policy goal of reducing the number of abortions across the country or whether they unlawfully targeted Planned Parenthood. This brief provides an overview of these legal challenges and summarizes the key positions of the plaintiffs and the defendants, Health and Human Services (HHS) and the Centers for Medicare and Medicaid Services (CMS).
Background
The 2025 Federal Budget Reconciliation Law is not the first instance of federal policy targeting funding for abortion and abortion providers. Blocking coverage of abortion under Medicaid and limiting abortion access has been a priority for abortion opponents since soon afterRoe v. Wade granted individuals the right to an abortion. Since 1977, Congress has included the Hyde Amendment every year as a rider to the federal appropriations budget prohibiting federal funds for abortion except in situations where the pregnancy is the result of rape or incest, or the pregnant person’s life is at risk. Federal Medicaid dollars may only reimburse abortion providers in these very limited situations. Some states make their own funds available to pay for abortion in other circumstances for their Medicaid enrollees. Yet, despite this longstanding provision, abortion opponents at the state and federal level have advocated going further in restricting federal funding. They argue that by allowing providers that also offer abortion services to remain in the Medicaid program, federal funds that go to these providers for other services indirectly subsidize abortion services with federal Medicaid dollars. While federal policy blocks funding for abortions under most circumstances, federal law mandates the coverage of family planning services, including contraception. Unique among covered benefits, federal Medicaid law classifies family planning services and supplies as a “mandatory” benefit category that states must cover and established a 90% federal matching rate (FMAP) for the costs of services categorized as family planning, a higher proportion than for other services. States pay the remaining 10% of costs. Many abortion providers also offer family planning services to their patients.
Over the years, anti-abortion policy makers and advocates have utilized many different strategies to block Planned Parenthood from participating in Medicaid. In 2017, Congress attempted to block Medicaid reimbursement to entities (including Planned Parenthood affiliates) that provide abortions outside of the Hyde Amendment exceptions as part of efforts to repeal and replace the Affordable Care Act. That year, the House of Representatives passed a Reconciliation bill that would have blocked federal Medicaid funds to Planned Parenthood; however, the bill failed in the Senate and did not become law. In addition to federal attempts to block Planned Parenthood from Medicaid reimbursement, in the past decade at least 14 states (AL, AR, AZ, FL, IA, ID, IN, KS, LA, MO, MS, OK, SC, TN, TX) have used state-level policies or sought federal permission to block the provider from participating in their state Medicaid programs. Though most of these provisions were blocked by court action, the Supreme Court’s June 2025 decision in a case challenging South Carolina’s exclusion of Planned Parenthood from their Medicaid program changed this precedent. In Medina v. Planned Parenthood South Atlantic the Court allowed states to exclude providers from their Medicaid programs if they provide abortions or other services the state does not condone. Since Medina, a few states (Indiana, Nebraska, Oklahoma) have taken new actions to exclude Planned Parenthood from their state Medicaid programs.
Box 1: Key Facts – Section 71113 of the 2025 Budget Reconciliation Law
Section 71113 of the 2025 Federal Budget Reconciliation Law prevents federal reimbursement to certain entities for services provided to Medicaid patients for one year from the date of enactment (July 4, 2025).
Entities including its affiliates, subsidiaries, successors, and clinics who are restricted from receiving Medicaid payments are those that as of October 1, 2025:
- Are 501(c)(3) non-profit organizations;
- Are considered essential community providers under the Affordable Care Act that are primarily engaged in family planning services or reproductive health services;
- Provide abortions outside of the Hyde Amendment exceptions (rape, incest, or life endangerment) and;
- Received more than $800,000 or more in Medicaid payments in 2023.
Who is Challenging Section 71113 of the 2025 Federal Budget Reconciliation Law?
Planned Parenthood, Maine Family Planning, and 22 states and the District of Columbia (CA, NY, CT, CO, DE, HI, IL ME, MD, MA, MI, MN, NV, NJ, NM, NC, OR, the Governor of PA, RI, VT, WA, WI and the District of Columbia) have filed separate legal challenges claiming that Section 71113 violates the United States Constitution. Health Imperatives has not filed a lawsuit challenging this provision, but Massachusetts is one of the states challenging the law.
The plaintiffs in these lawsuits say that restricting Medicaid funding to entities under Section 71113 would harm affected organizations and lead to increased costs to states. Further, they say this provision would decrease access to millions of Medicaid enrollees who receive reproductive health care including contraception, sexually transmitted infection testing and treatment, pregnancy testing, cancer screening, and other preventive services from affected providers. Section 71113 blocks federal Medicaid funding from certain entities for one year starting the date the law was signed, July 4, 2025; however, the provision states that entities are only designated as a “prohibited entity” if they meet certain criteria on October 1, 2025.
Status of Cases Challenging Section 71113
Section 71113 is currently in effect for all three entities, and they are blocked from receiving federal Medicaid reimbursement for services provided to patients; however, throughout the course of the ongoing litigation the enforceability of Section 71113 has varied. A district court initially granted a preliminary injunction for ten Planned Parenthood affiliates that do not individually meet the criteria of Section 71113 on July 21, 2025. On July 28, 2025, the district court extended the preliminary injunction to all Planned Parenthood affiliates. On September 11, 2025, the First Circuit Court of Appeals reversed the district court ruling by pausing the preliminary injunction, therefore blocking Planned Parenthood sites across the country from receiving federal Medicaid funds for any service they provide to Medicaid enrollees while the case is ongoing. On December 12, 2025, the First Circuit permanently blocked the district court’s preliminary injunction. The First Circuit held that Section 71113 does not impose punishment on Planned Parenthood and instead is a lawful exercise of Congress’ taxing and spending power. The district court is continuing to consider this case in accordance with the First Circuit’s ruling.
Maine Family Planning also requested a preliminary injunction to block the enforcement of Section 71113, but the district court denied their request, and Maine Family Planning appealed. However, on December 29, 2025, Maine Family Planning filed a motion to voluntarily dismiss their case.
On December 2, 2025, a district court granted a motion for a preliminary injunction blocking enforcement of Section 71113 for the 22 states and DC. However, the judge stayed her order for seven days, and HHS has appealed. The court held that text of Section 71113 does not provide clear notice to states of how a Medicaid provider is determined to be a “prohibited entity.” The court reasoned that Section 71113 does not sufficiently give states: (1) the criteria to determine whether an entity is “primarily engaged in family planning”; (2) guidance on calculating Medicaid expenditures (for the purpose of Section 71113’s $800,000 threshold) of entities in their state that are members of multistate organizations; and (3) how to identify “affiliates” when those organizations operate outside the state’s boarders. Further, the court asserted that the plaintiff states would be harmed by enforcement of Section 71113 including retroactive enforcement because the retroactive nature of the provision conflicts with states obligation to make payments on providers claim within 30 days of receipt.
After initially blocking the law from being implemented, the First Circuit subsequently granted the Government’s request on December 30, 2025, allowing the provision to be enforced in the 22 Plaintiff States and DC. The First Circuit held that the asserted ambiguities in Section 71113, including the definition of “affiliate” are unlikely to support the granting of a preliminary injunction. Currently states may reimburse Planned Parenthood and other affected providers with non-federal dollars, but only a handful of states have indicated their willingness to provide this support.
Box 2: Plaintiffs Claims Regarding the Impact of the Loss of Medicaid Funds from Section 71113
Planned Parenthood
- Planned Parenthood operates over 600 health centers nationwide, serving over 2 million patients per year, with more than half of those patients using Medicaid health coverage.
- In 2023, more than one-third of Planned Parenthood affiliates aggregate revenue was from federal reimbursement for services provided to Medicaid patients.
- 74% of Planned Parenthood Member health centers are in rural areas, Health Professional Shortage Area (HPSA), or Medically Underserved Areas. Without federal Medicaid funds, many Planned Parenthood health centers may be forced to close or face service reductions.
Maine Family Planning
- Maine Family Planning operates 18 clinics and a mobile health center that serves residents in 12 out of Maine’s 16 counties, providing primary care, family planning services, and abortion. In nearly all the counties where Maine Family Planning operates clinics, more than half of the population lives in a rural area, which can make health care difficult to access.
- Almost half of Maine Family Planning’s patients rely on Medicaid for their health insurance. Without the ability to bill for Medicaid services, a substantial portion of these patients will no longer be able to receive covered health care including primary care and sexual and reproductive health care.
- Clinics run by Maine Family Planning already face significant financial hardships, including two clinics only being able to operate one or two days per week. Without Medicaid funding, clinics will be forced to limit or eliminate certain services.
22 States and the District of Columbia
- The states argue that they will be the ones to enforce Section 71113, and the enforcement of this provision will cause states to financially harm themselves. The states argue that Section 71113 will lead to increased Medicaid expenses due to delays in care that will result from the closures of Planned Parenthood clinics, or force states to divert millions of dollars in state funds from other programs to keep Planned Parenthood as a Medicaid provider.
- Section 71113 will harm state health care infrastructure as Planned Parenthood clinics close or reduce hours with no alternatives available for patients, thus threatening state’s health care ecosystems.
On What Grounds are the Plaintiffs Suing the Federal Government?
The heart of the litigation is the constitutionality of Congress’ action of prohibiting payments to certain entities that provide services to Medicaid patients.
While all plaintiffs concede that Congress has the power to dictate spending guidelines for Medicaid funds, the constitutionality of Congress’ actions in enacting Section 71113, and the resulting burden on states is at the heart of this litigation. The Spending Clause in the United States Constitution grants Congress the power to, “lay and collect taxes…and provide for the common defense and general welfare of the United States,”— but this power is not unlimited. All exercises of Congress’ Spending Clause authority are subject to constitutional limitations. These limitations include the requirement that Congress must give clear notice of what actions are required in exchange for federal funds. Further, Congress cannot attach discriminatory conditions to federal funds that would violate other constitutional rights. At issue in this litigation is whether Section 71113 provides clear notice of state’s enforcement obligations, and whether the provision violates Maine Family Planning and Planned Parenthood’s constitutional rights.
First Amendment Claims
The First Amendment of the United States Constitution states that, “Congress shall make no law… abridging the freedom of speech…or the right of the people to assemble.” The First Amendment has been interpreted not only to guarantee freedom of speech, but also the right to associate with others. Planned Parenthood and the states contend that Section 71113 unconstitutionally burdens Planned Parenthood’s First Amendment right to freedom of association. The text of Section 71113 includes affiliates of an entity that meet the criteria of a prohibited entity. They argue that penalizing Planned Parenthood affiliates that do not independently meet the criteria of Section 71113 because they are associated with other Planned Parenthood affiliates that are prohibited entities, hinders the affiliates’ ability to freely associate.
Plaintiffs further allege that Section 71113 violates Planned Parenthood’s First Amendment rights because Section 71113 could be considered unconstitutional retaliation. The First Amendment prevents the government from restricting speech or retaliating against protected speech. Retaliation is an adverse action a plaintiff experiences where the motivating factor behind the action was the plaintiff’s protected speech. In their complaints, Plaintiffs, Planned Parenthood, and the states allege that the motivation for enacting Section 71113 is to stop Planned Parenthood’s advocacy for sexual and reproductive health care. The states and Planned Parenthood claim that due to the narrow criteria of abortion providers affected by Section 71113, the provision was specifically written to target Planned Parenthood and its affiliates.
Bill of Attainder Claims
In addition to the First Amendment claims, Planned Parenthood Federation of America and the states contend that Section 71113 is a violation of the US Constitution because it is an unlawful “bill of attainder.” A bill of attainder is a law that imposes a penalty without trial and circumvents due process of law. In their complaint, plaintiffs draw on the legislative history of Section 71113, and statements made by lawmakers to support their claim that Section 71113 was enacted specifically to exclude Planned Parenthood from receiving federal Medicaid funds. Planned Parenthood and the states also cite a 2017 attempt by Congress to block Medicaid payments from entities that provide abortions outside of the Hyde Amendment exceptions. During the 2017 attempt, the enacted provision would have solely excluded Planned Parenthood. Planned Parenthood argues that the text of the 2017 and 2025 provisions that block payments to prohibited entities are similar, demonstrating Congress’ intent to specifically penalize Planned Parenthood.
Fifth Amendment Claims
In all three cases, the plaintiffs allege some violation of the Fifth Amendment Equal Protection Principle. All Plaintiffs allege that Section 71113 violates the equal protection component of the Due Process Clause of the Fifth Amendment because it only applies to “prohibited entities.” The Fifth Amendment prevents arbitrary discrimination and generally requires that similarly situated entities are treated alike. If an entity believes they have been unfairly discriminated against by a government entity, they can file a claim alleging a violation of their Fifth Amendment rights. The courts will then analyze the government’s purpose for enacting the law, and whether the government has violated the entities’ guaranteed rights such as freedom of speech and equal protection under the law. Planned Parenthood maintains they are being treated differently than other abortion providers who provide abortions outside of the Hyde Amendment exceptions. They argue that there is not a legitimate government interest in drawing this distinction, and thus the provision violates the Fifth Amendment.
Planned Parenthood also asserts a separate Fifth Amendment claim, maintaining that Section 71113 is invalid because it is vague. The Fifth Amendment Due Process Clause states that a person cannot be deprived of their life, liberty, or property without due process of law. This has been interpreted to mean that statutes that lack specificity or definiteness can be invalidated because they do not give clear warning of a prohibited action (void for vagueness). Planned Parenthood alleges that Section 71113 does not define essential terms in the statutory text, thus making it void. Planned Parenthood maintains that the words “affiliates, subsidiaries, successors, and clinics,” in the statute are undefined leaving it unclear whether Section 71113 applies to Planned Parenthood affiliates who do not independently meet the provisions criteria. In a November 21, 2025, letter to State Medicaid Directors CMS provided guidance defining the statutory term “affiliate “as “a corporation that is related to another corporation by shareholdings or other means of control; a subsidiary, parent, or sibling corporation.” In the guidance CMS defines “control” as: “the direct or indirect power to govern the management and policies of a person or entity, whether through ownership of voting securities, by contract, or otherwise; the power or authority to manage, direct or oversee.” Planned Parenthood filed a response to this guidance in the district court contending that CMS’ definition of affiliate still leaves ambiguity on who is considered an “affiliate,” because of the overbroad terms used in the CMS guidance. Therefore, Planned Parenthood continues to allege that Section 71113 is “void for vagueness.”
The Trump Administration Contends Section 71113 is Constitutional
The Trump administration maintains similar legal arguments in all the lawsuits challenging the constitutionality of Section 71113. The government contends that Section 71113 is a component of duly enacted legislation, and plaintiffs’ attempts to block implementation of this provision seek to override the intentions of Congress. The Trump administration also argues that Congress has broad discretion to tax and spend for the general welfare, which includes the ability to alter federal Medicaid expenditures. The government also states that the parameters for designating entities to be removed from Medicaid reimbursement are not arbitrary and serve a legitimate government purpose. Further, the government rejects plaintiffs’ argument that Section 71113 was enacted due to animus toward plaintiffs. Instead, the government offers the alternative explanation that Section 71113 was enacted to reduce the number of abortions performed in the United States, a legitimate government interest.
What are the Next Steps?
Currently, Section 71113 in effect, and Planned Parenthood affiliates, Maine Family Planning, and Health Imperatives are blocked from receiving federal reimbursement for services provided to Medicaid patients. Litigation challenging Section 71113 has the potential to directly impact the over 2.1 million people who receive health care from these organizations. To continue to qualify for federal Medicaid funding it was reported that Planned Parenthood of Wisconsin responded by first stopping abortion care before the October 1, 2025, deadline and then when they resumed abortion care, they relinquished their Essential Community Provider status, to avoid meeting the criteria set forth in Section 71113. However, it is unclear whether this will be enough for them and other affiliates which do not meet the criteria of Section 71113 on their own to not land on HHS’s prohibited entity list based on CMS’ guidance released on November 21, 2025. If Section 71113 remains in effect, Planned Parenthood and Maine Family Planning may close clinics, reduce services, or be unable to serve Medicaid patients. While some states have announced plans to fill in gaps created by the loss of Medicaid funding, this is not likely enough to make up for lost federal funds. Planned Parenthood Mar Monte, Planned Parenthood North Central, Planned Parenthood Northern New England, Planned Parenthood of Greater New York, Planned Parenthood of Michigan, Planned Parenthood of Western Pennsylvania, and Planned Parenthood Greater Northwest, have already experienced clinic closures or service interruptions in the face of financial uncertainty, and Maine Family Planning has announced that they will no longer offer primary care services to Medicaid enrollees at three of their 18 sites.
Even if the Plaintiffs ultimately prevail on their challenges to Section 71113, the Supreme Court’s decision in Medina v. Planned Parenthood South Atlantic, opens the door for states to block providers from their Medicaid program. Without a federal law preventing Planned Parenthood from being blocked from Medicaid, more states may choose to exclude Planned Parenthood from their state’s Medicaid program. Ultimately, the constitutionality of Section 71113 may be decided by the Supreme Court; however, because Section 71113 only prohibits funding from July 4, 2025, to July 3, 2026, the year may be up before there is a final decision. Some states, such as Colorado and California, have provided temporary financial support to Planned Parenthood and other affected providers to fill the gap from the loss in federal funding. Yet, many anticipate that this provision could be inserted into a similar reconciliation bill in future years. It is not clear whether states will be able to continue to provide this support if there is a future reconciliation bill blocking federal Medicaid payments to these providers, especially in the face of future fiscal challenges and competing programmatic demands.