A Look at the Medicaid Payment Error Rate Measurement (PERM) Program and Upcoming Changes and Impacts
Introduction
Medicaid is a very complex program that involves millions of enrollees, hundreds of thousands of providers, and significant federal and state expenditures. Both the federal government and states are responsible for ensuring the proper management and functioning of the Medicaid program to ensure it is providing quality and efficient care while using funds–taxpayer dollars–appropriately with minimal waste. Program integrity efforts historically have worked to prevent and detect fraud, waste, and abuse, to increase program transparency and accountability, and to work on corrective action plans and recover improperly used funds. The Trump Administration has signaled reducing fraud, waste, and abuse across federal programs (including Medicaid) is a priority. However, broader Medicaid cuts from 2025 reconciliation law may impede states’ ability to invest in oversight and other administrative initiatives to reduce fraud. The reconciliation law also includes changes that directly address the Medicaid Payment Error Rate Measurement (“PERM”) program, which over time could have financial and administrative implications for states. The Congressional Budget Office (CBO) estimates that Medicaid payment reductions related to PERM could reduce federal spending in Medicaid by $7.6 billion over ten years.
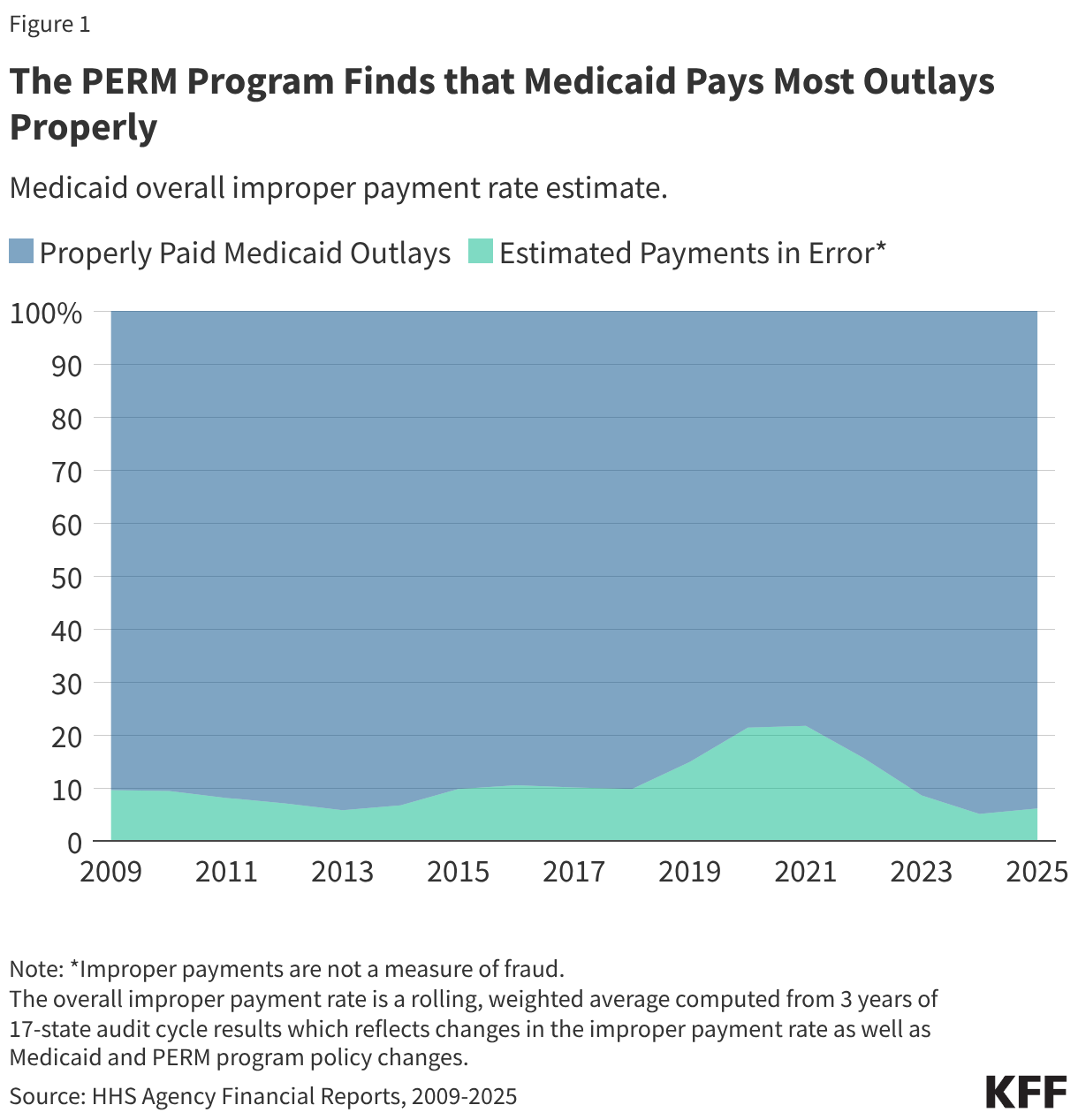
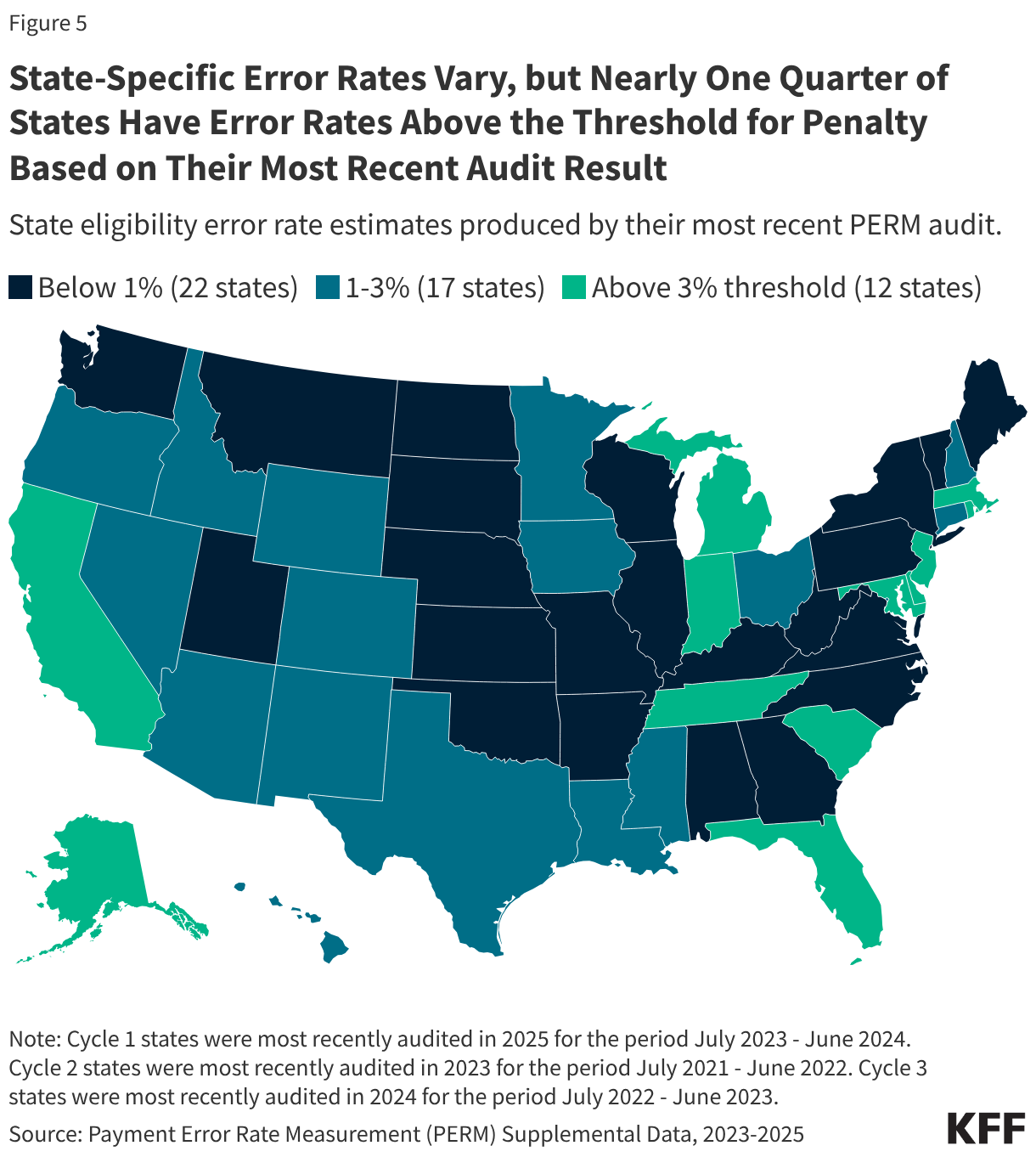
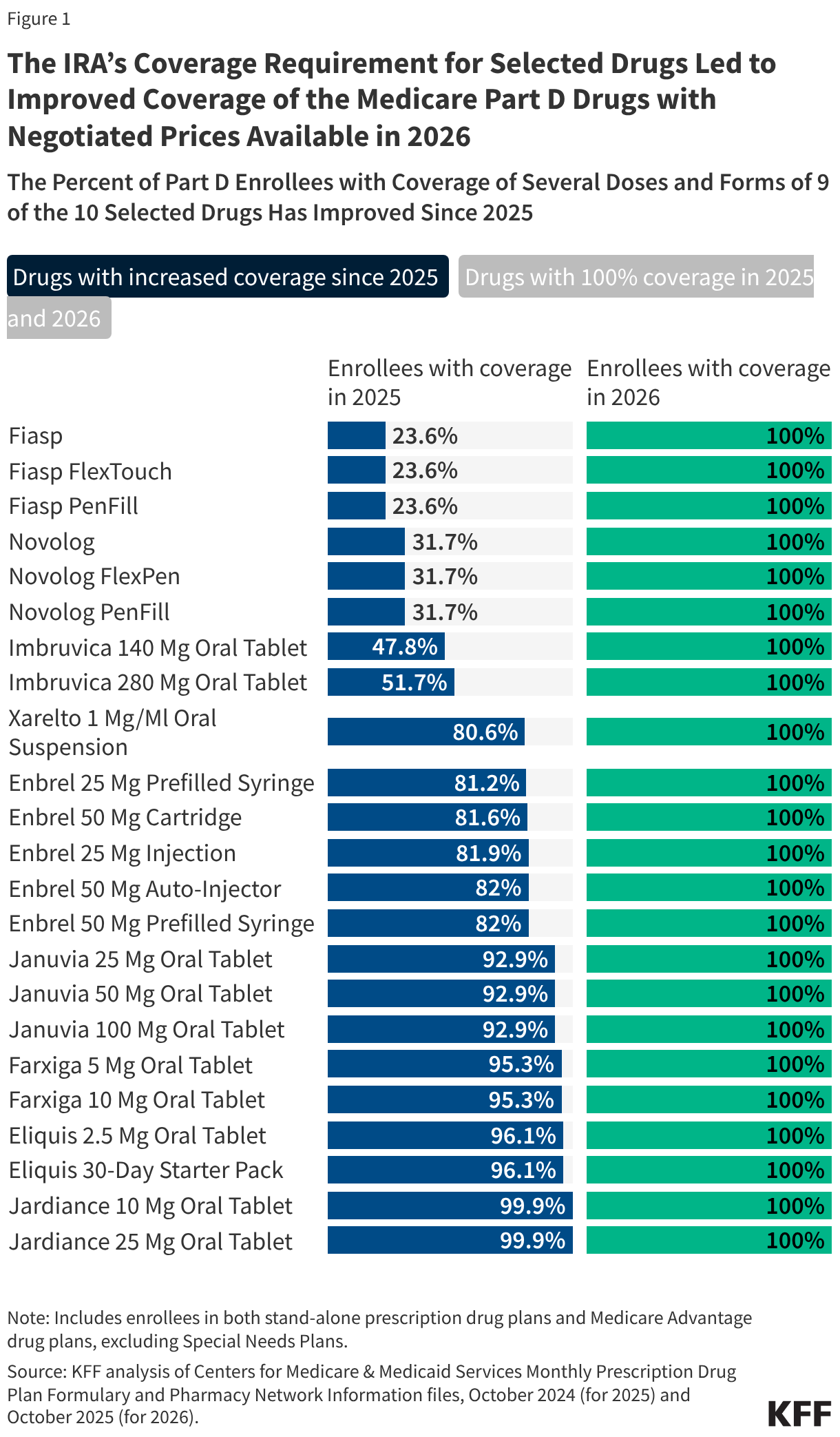
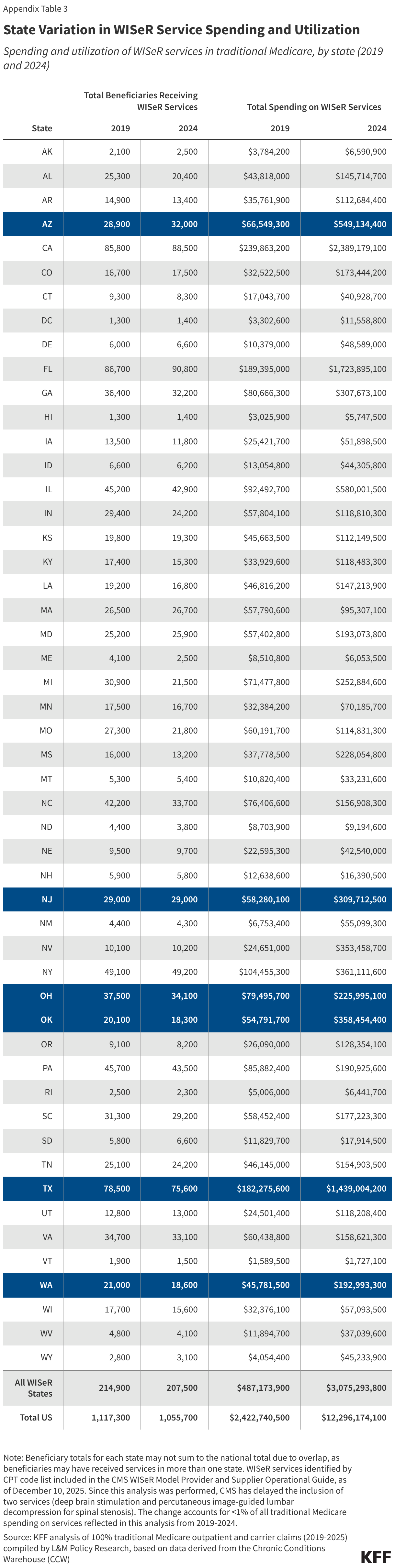
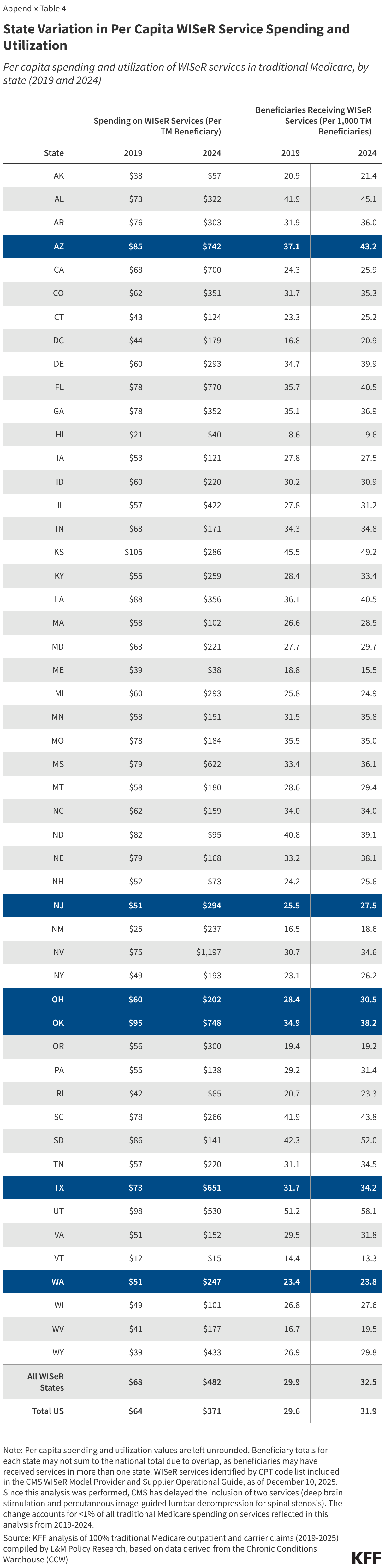
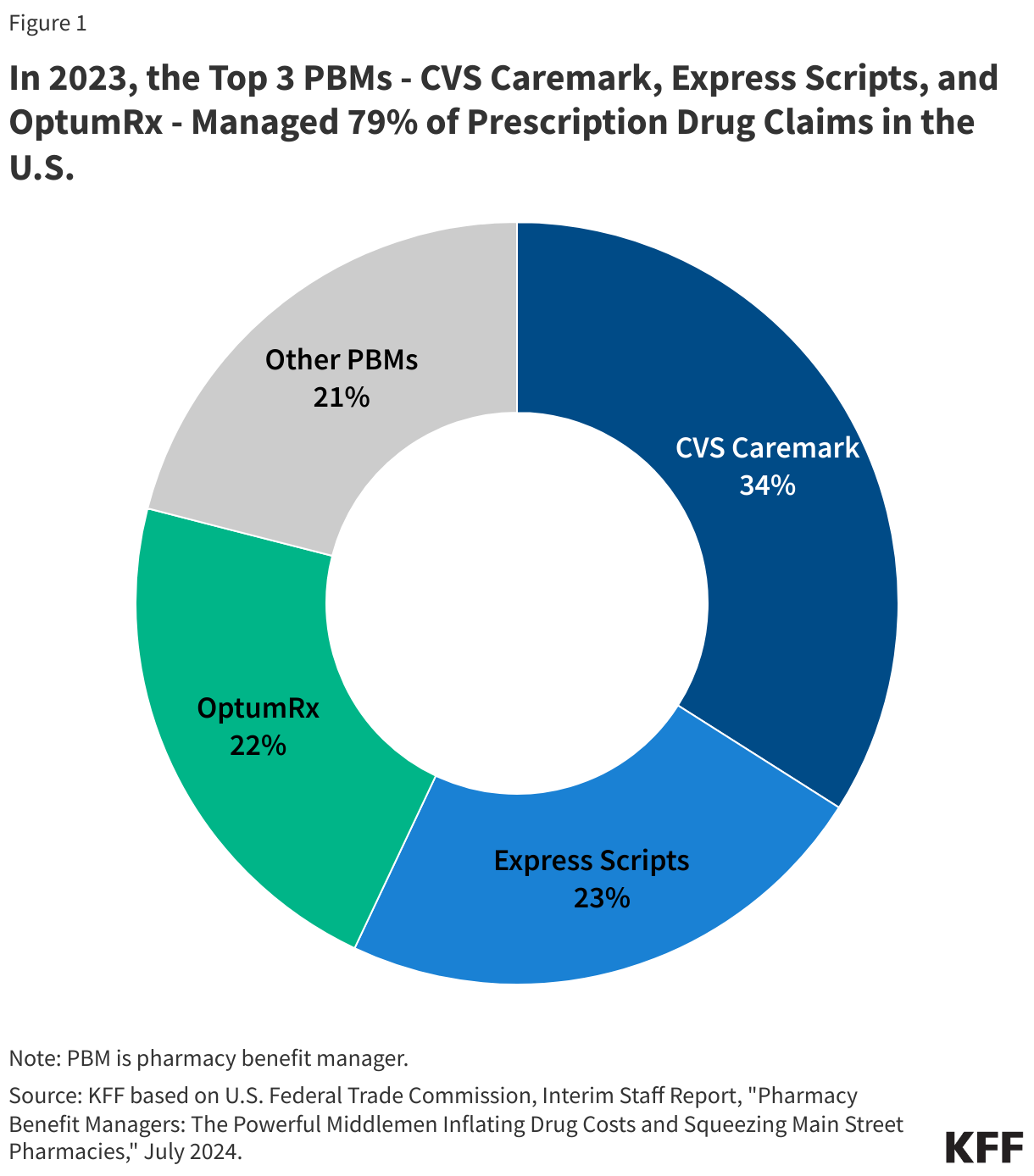
The PERM program measures “improper payments” in Medicaid to produce improper payment rates. Improper payments are payments that do not meet CMS program requirements, often the result of administrative or paperwork issues. PERM is designed to help states improve their operation of the program and reduce errors; it is not a measure of fraud against the program. Beginning October 1, 2029, the reconciliation law requires HHS to reduce federal Medicaid financial participation to states that exceed a three percent PERM eligibility error rate threshold. (SNAP policy changes will also require states to pay a portion of SNAP benefit costs, depending on the state’s payment error rate.) While Medicaid pays most outlays properly (93.9% in 2025) most improper payments are due to insufficient documentation (77.2% in 2025), including eligibility-related errors (68.1% due to insufficient documentation in 2025). State error rates vary, but nearly one quarter of states have eligibility error rates above the three percent PERM threshold based on their most recent audit result. This brief explains the PERM program and its three components as well as upcoming changes to PERM and possible state impacts.
What is PERM?
The PERM program operates according to requirements set in law and CMS rulemaking. The Improper Payments Information Act (IPIA) of 2002 (replaced by the Payment Integrity Information Act (PIIA) of 2019) requires the heads of federal agencies to annually review programs they administer and identify those that may be susceptible to significant improper payments, to estimate the amount of improper payments, to submit those estimates to Congress, and to submit a report on actions the agency is taking to reduce the improper payments. The federal government has designated Medicaid and other programs (including Medicare, SNAP, and TANF) as “risk-susceptible.” Medicaid receives this designation due to its size and complexity, as the program involves millions of enrollees, hundreds of thousands of providers, 51 state agencies (including DC), different delivery systems, complicated eligibility rules, and significant federal and state expenditures. GAO monitors high-risk programs and their improper payments, including Medicaid.To comply with IPIA requirements in Medicaid, CMS developed the PERM program.
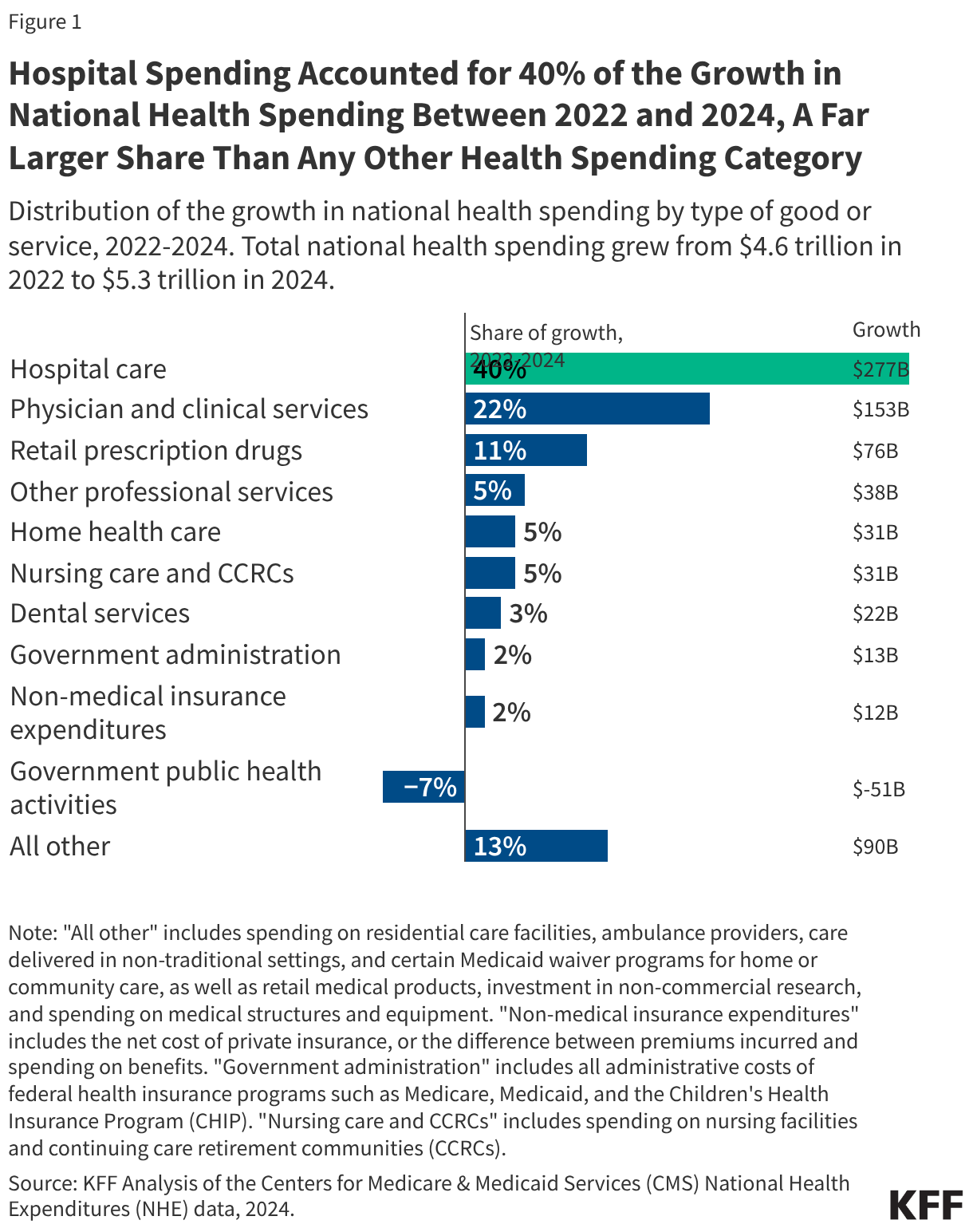
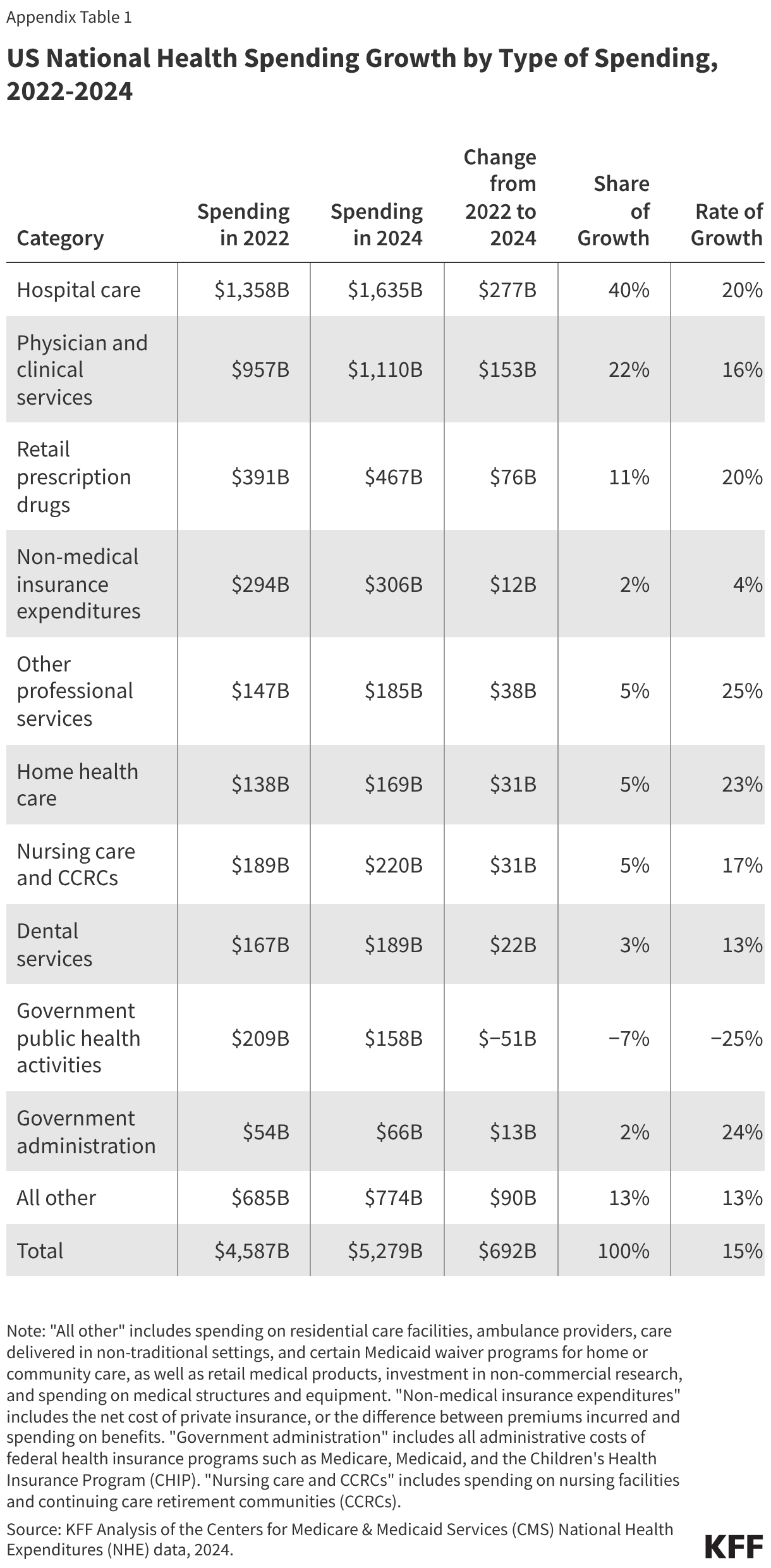
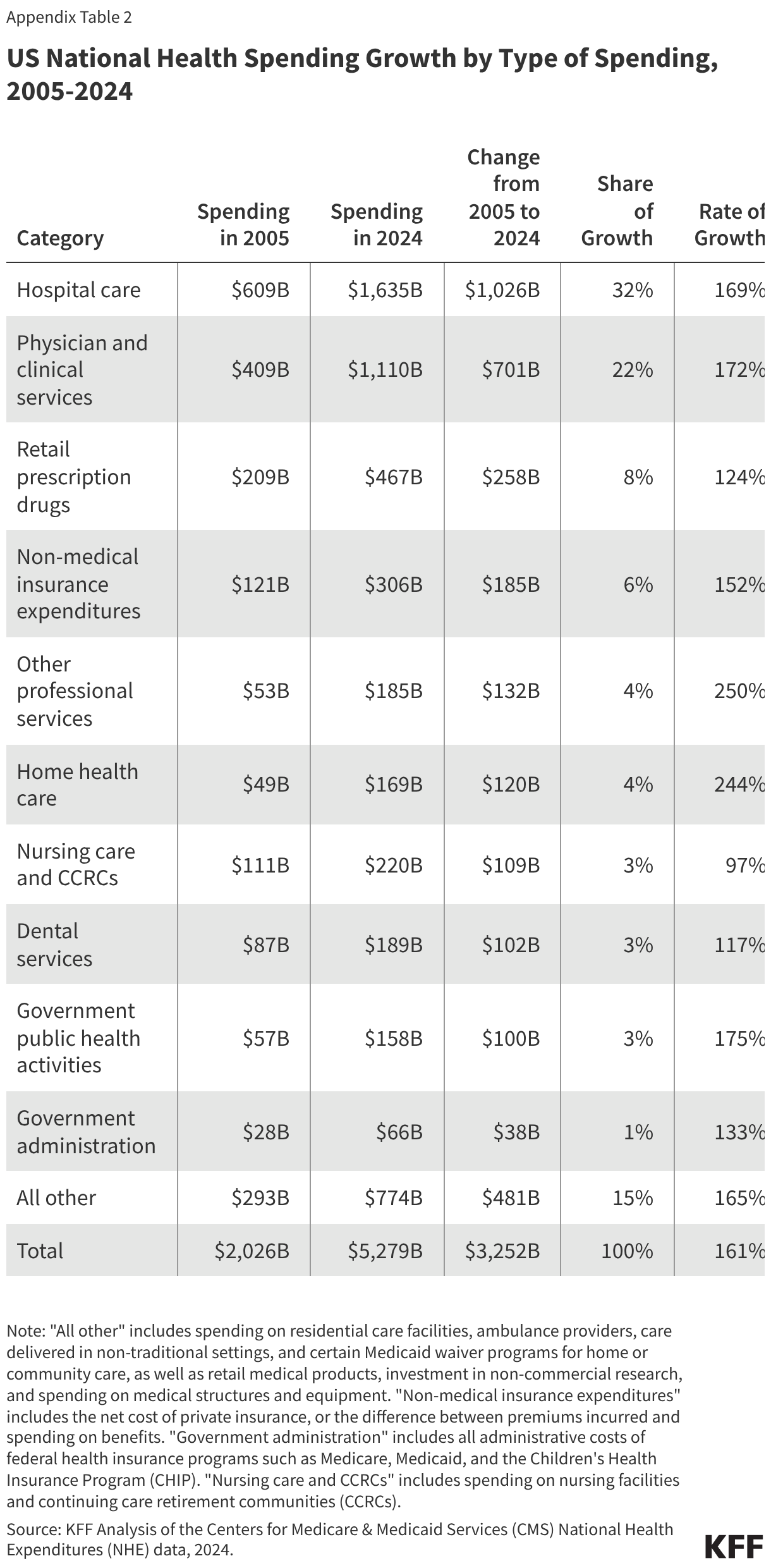
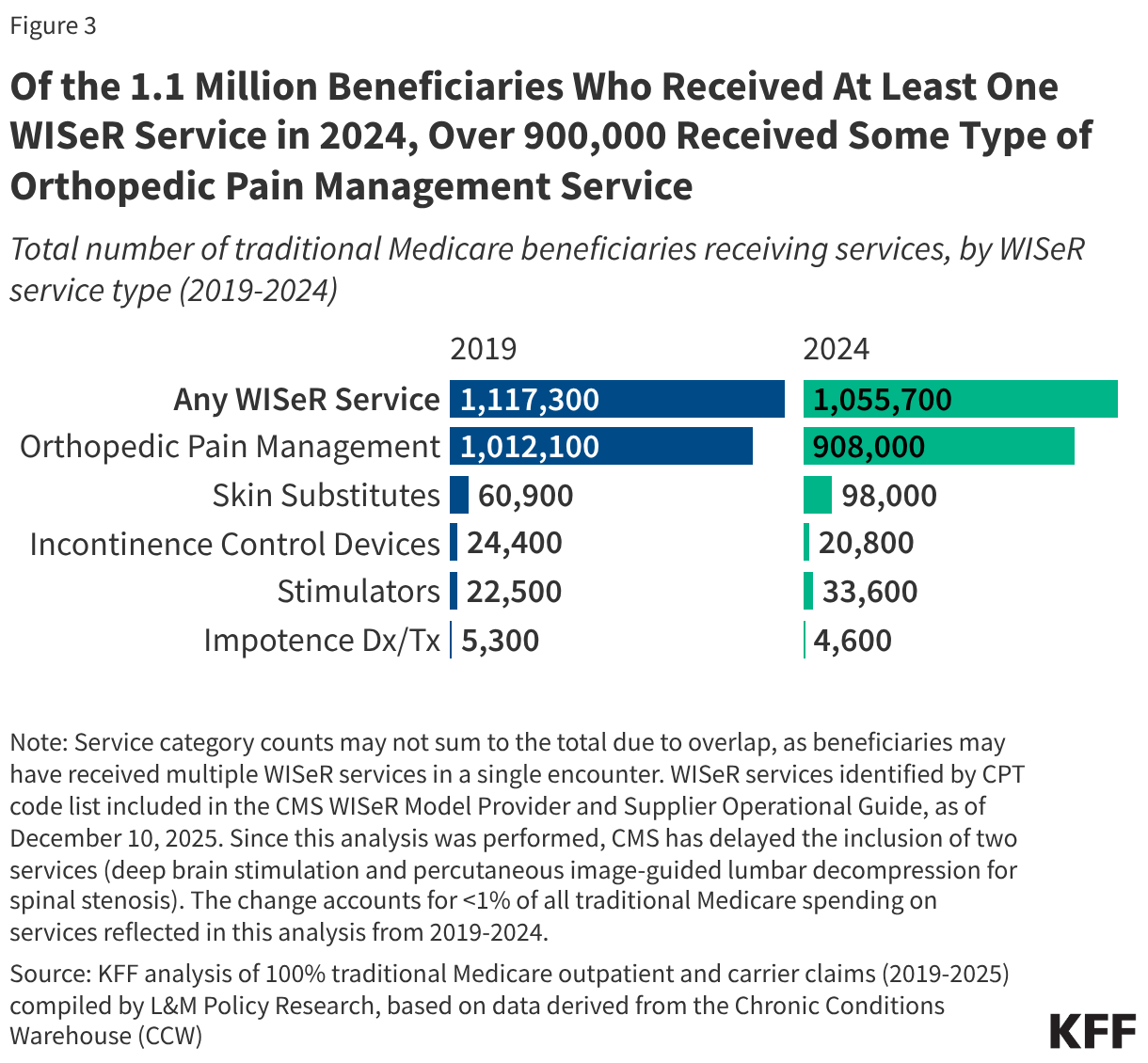
The PERM program measures improper payments in Medicaid by producing a national improper payment rate through cyclical audits of state Medicaid programs. Improper payments are payments that do not meet Medicaid program requirements. The improper payment rate is the estimated share of Medicaid claims that do not meet Medicaid program requirements. The error rate is not a “fraud” rate (or a waste or abuse rate), but a measurement of payments made that did not meet statutory, regulatory, or administrative requirements or are made in an incorrect amount including overpayments and underpayments. Each year (since 2009) HHS has reported an estimated national (or overall) Medicaid improper payment rate, which is based on reviews of three program components: fee-for-service (FFS) claims, managed care capitation payments, and eligibility determinations. In 2025, Medicaid paid an estimated 93.9% of outlays properly, representing $610.95 billion in proper federal payments (Figure 1). The overall estimated improper payment rate was 6.1%, or $37.39 billion in federal payments. (See Box 1 for discussion of improper payment rate trend over time.)
PERM audits a sample of state Medicaid FFS claims and managed care payments every three years. Federal contractors conduct standardized audits of states on a rolling three-year basis, meaning each PERM cycle measurement includes one-third of states (see Appendix). Annually, the most recent three cycles are combined to produce a national improper payment rate (weighted by state size). Audits include a random sample of FFS claims and managed care payments from a 12-month period. To create the 2025 improper payment rate, PERM program contractors reviewed about 48,000 claims and managed care payments.1 States receive results of their audits and use the information to improve compliance during the next audit cycle. States are often required to develop and implement corrective action plans to reduce mistakes and improve recordkeeping.
What types of errors does the improper payment rate capture?
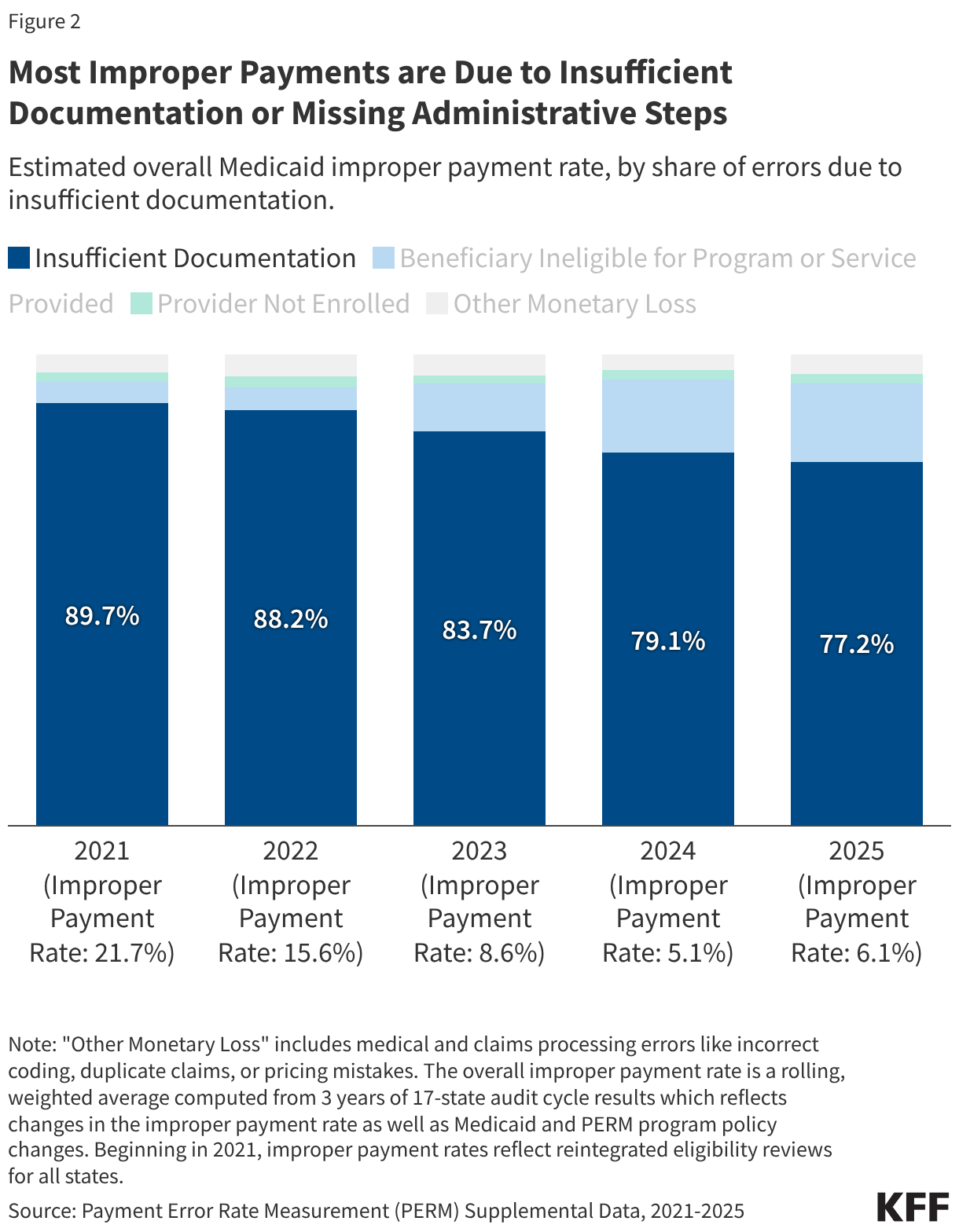
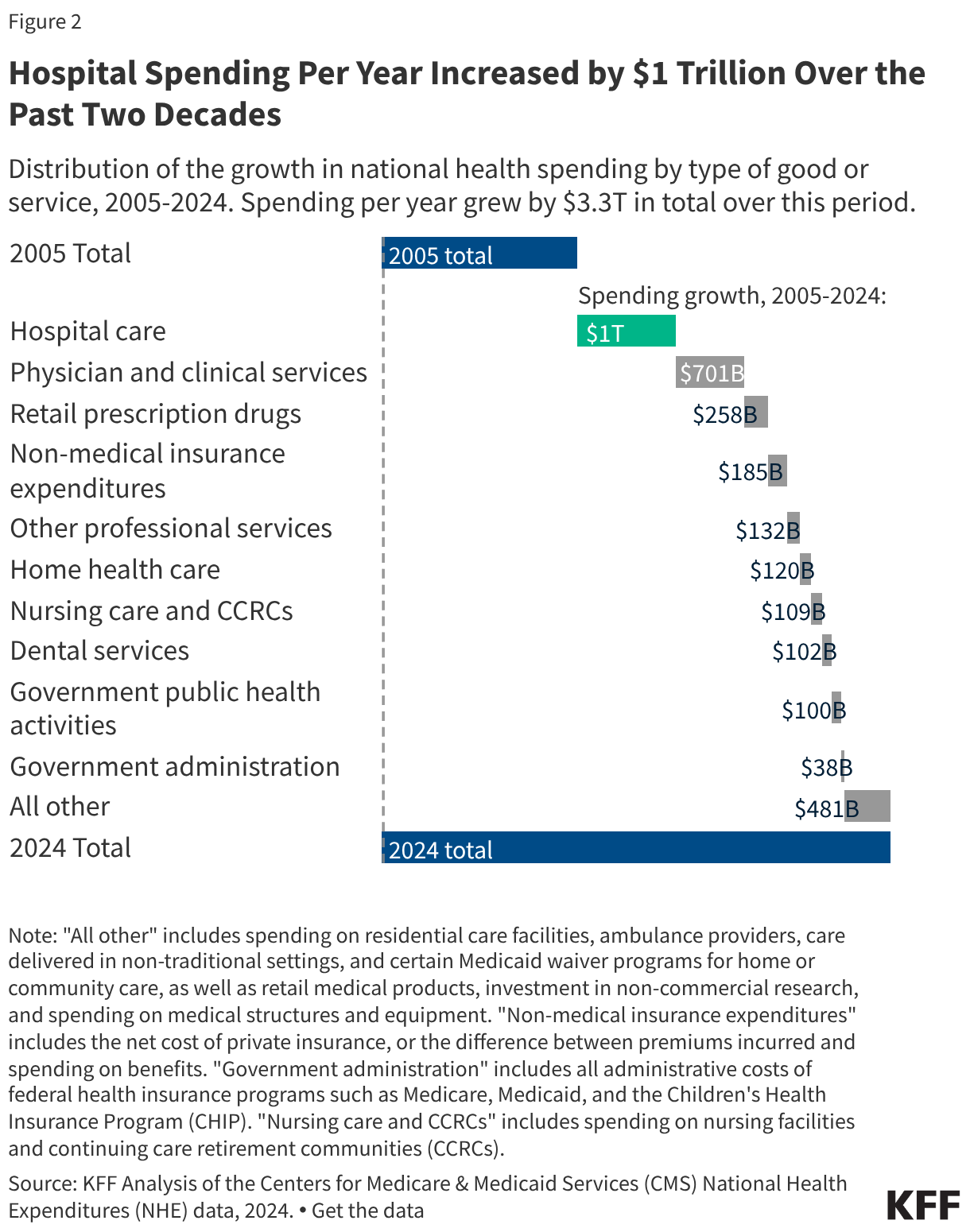
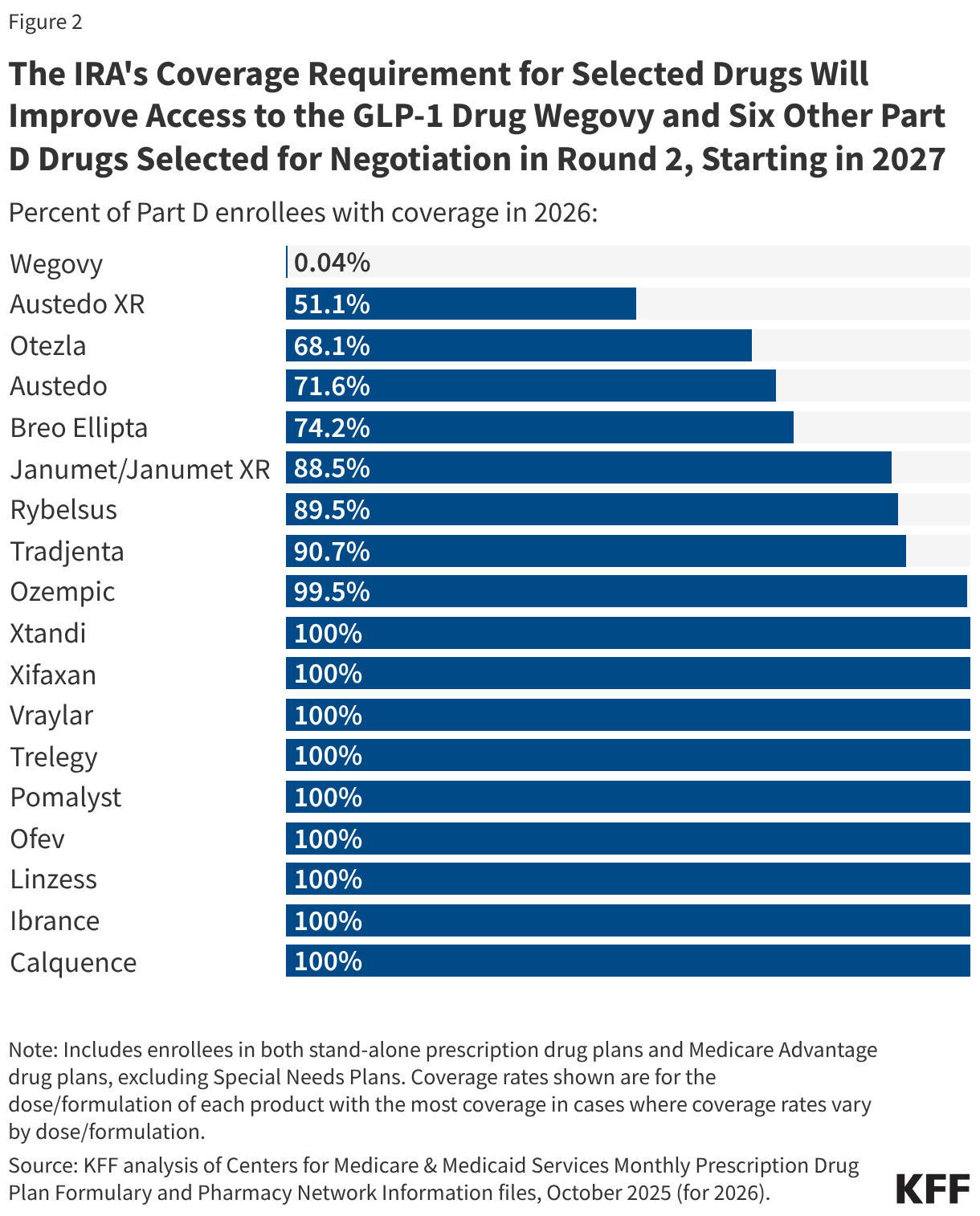
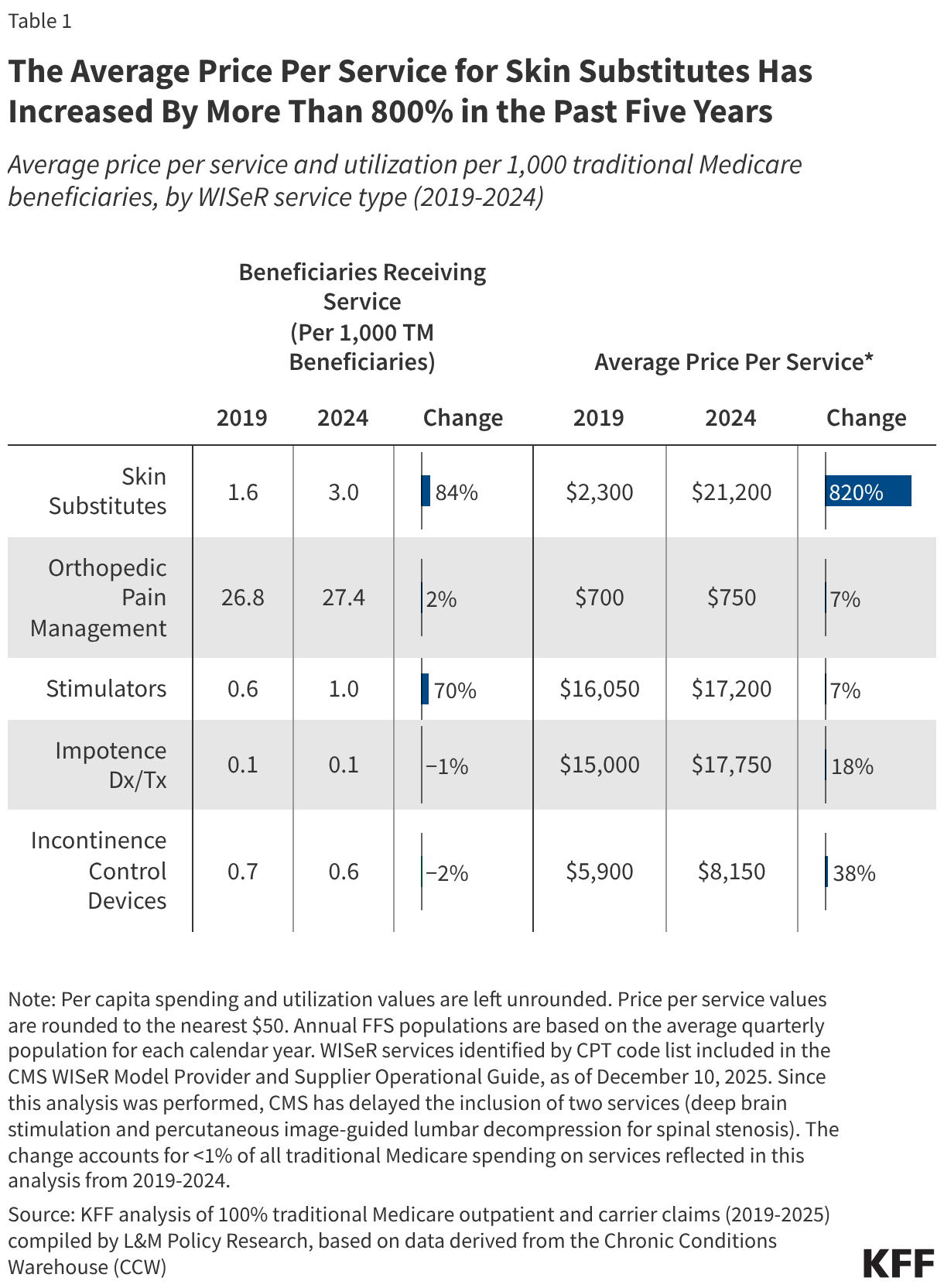
Most improper payments are due to insufficient documentation or missing administrative steps. HHS reports the different types of errors that contribute to the improper payment rate each year. In 2025, 77.2% of improper Medicaid payments were the result of insufficient documentation or missing administrative steps (Figure 2). These payments were not necessarily for ineligible enrollees, providers, or services since they may have been payable if the missing information had been on the claim and/or the state had complied with requirements. Examples include state failure to retain documentation of enrollee eligibility or to appropriately screen enrolled providers, or medical records not submitted or missing required documentation to support the medical necessity of a claim. Errors that represent “monetary loss” made up the remaining one quarter (22.8%) of improper payments in 2025 (Figure 2). Monetary loss represents a subset of improper payments where CMS has sufficient information to conclude the payment should not have been made or was made in the incorrect amount. These errors include payments made for enrollees who are ineligible for the program or for services and providers not enrolled.
How do the different PERM components contribute to the improper payment rate?
The overall improper payment rate is calculated based on three component error rates: fee-for-service, managed care, and eligibility. The PERM program uses a random sample of Medicaid claims and managed care payments to calculate component error rates. Those component error rates are combined to produce an overall improper payment rate.
Fee-for-Service. Within a FFS delivery system, the state Medicaid agency pays providers or groups of providers directly. The FFS PERM component estimates the payment error rate for fee-for-service claims, where selected claims undergo a medical review (focused on documentation in the medical record) and a data processing review (checking if claims followed correct procedures –e.g., coding, billing and payment amounts).
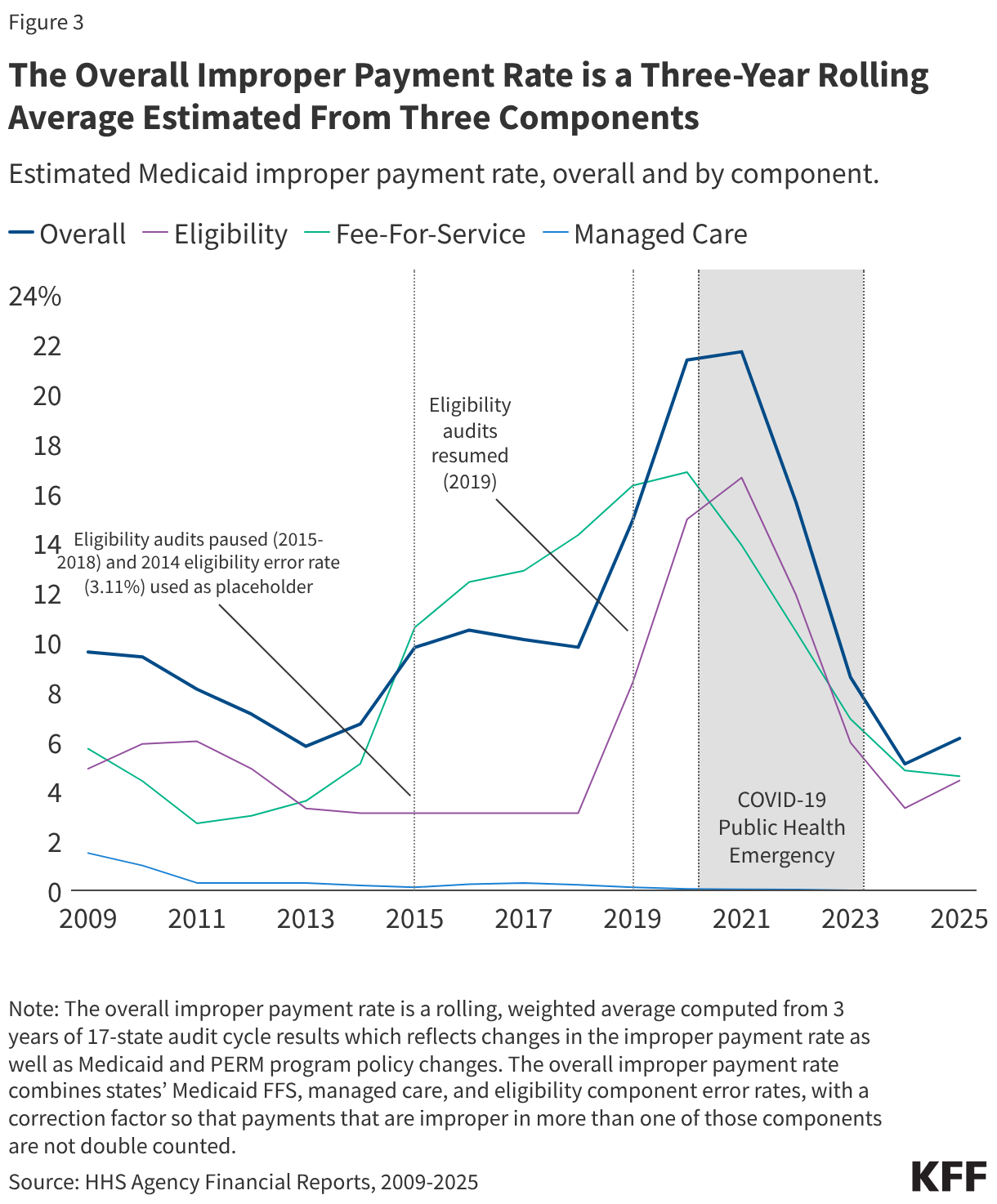
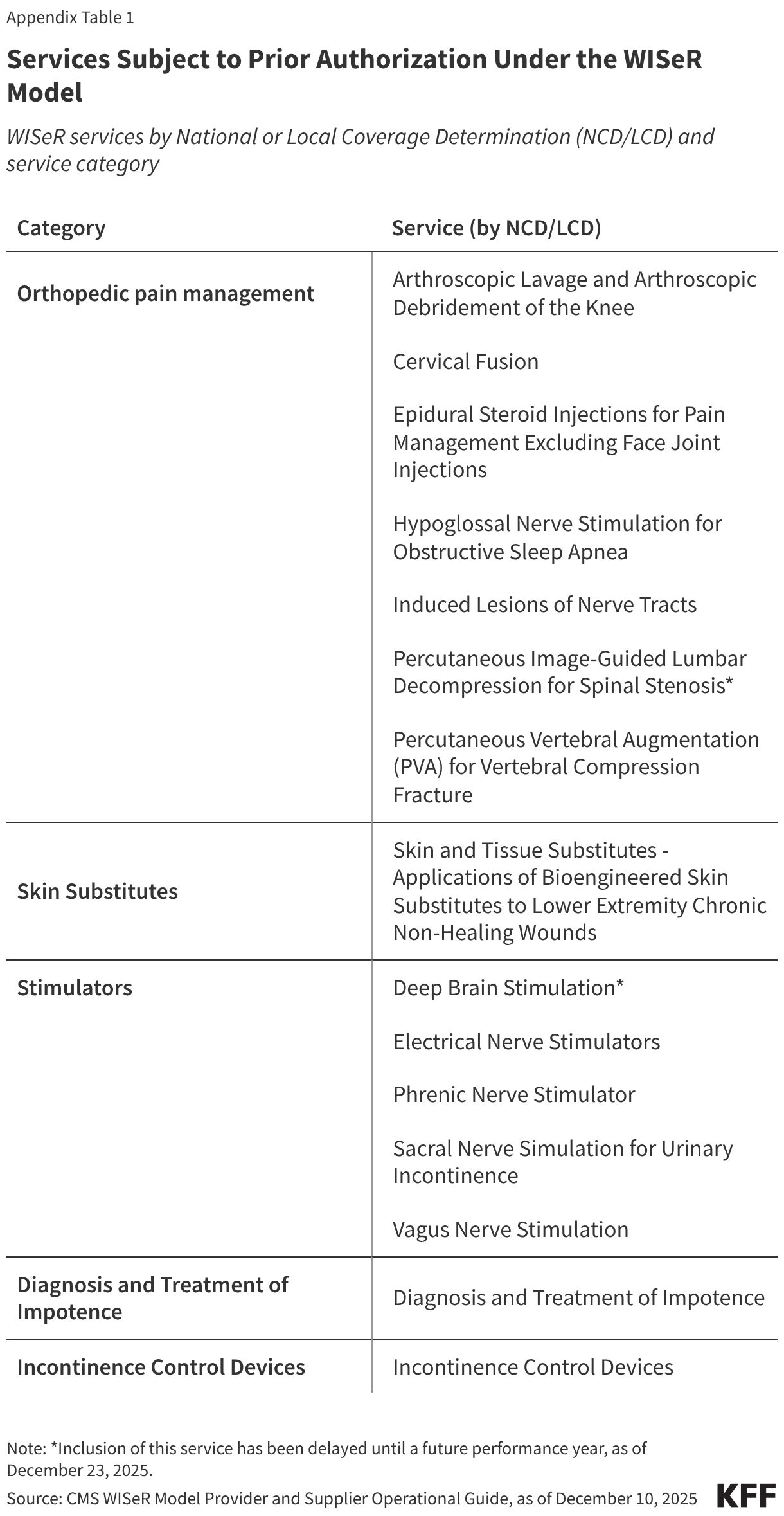
Managed Care. States that contract with Medicaid managed care plans pay a set per member per month (“capitation”) payment to the plan for the services covered by the contract. The managed care PERM component estimates Medicaid managed care capitation payments made by the state in error (plan payments to providers are not reviewed under PERM). The PERM program’s first report (2009) estimated a managed care component error rate of 1.5%, and since 2011, the estimated managed care error rate has remained below one-half of one percent (Figure 3).
Eligibility. The eligibility component assesses the state’s application of federal rules and documented state eligibility policies and procedures. States must provide records or documentation to support eligibility determinations. Non-compliance with eligibility requirements may include not performing a required element (e.g., income verification); not providing or retaining sufficient documentation for a required element; failing to conduct timely eligibility redeterminations; determining eligibility under the incorrect eligibility category; or enrolling an individual who is not eligible. (Note that PERM does not attempt to identify or measure instances where a state incorrectly determines an individual is ineligible for Medicaid – i.e., where eligibility rules were applied inaccurately.)
Box 1: The improper payment rate has changed over time, largely due to the ACA implementation and the COVID-19 public health emergency.
Pre-ACA: Between 2009 and 2013, the overall improper payment rate trended downward as information systems improved states’ ability to determine provider and enrollee eligibility . The PERM program’s eligibility component error rate decreased from 2011 to 2014, when it was estimated at 3.11 percent (Figure 3). The Patient Protection and Affordable Care Act (ACA) passed in 2010 and phased in several changes to the Medicaid program, including the 2014 Medicaid expansion.
ACA Implementation: During the 2013-2019 period, the overall improper payment rate trended upward, driven by errors in FFS claims related to state non-compliance with new provider screening, enrollment, and documentation requirements introduced in 2012 under the ACA. From 2015 to 2018, HHS suspended eligibility audits as states began to implement new eligibility standards and determination requirements under the ACA. During this period, the overall PERM rate was calculated using a proxy eligibility estimate of the 2014 eligibility component error rate, 3.11 percent to provide states with time to adjust to eligibility process changes in the Affordable Care Act.
Post ACA: Beginning in 2019, PERM reintroduced the eligibility component under updated rules, for the first time requiring states to work with an independent contractor using nationally standardized eligibility audit procedures2. Between 2019 and 2021 the overall improper payment rate rose again (Figure 3), driven by the new standardized PERM eligibility audits. Most of the eligibility error rate was attributed to errors due to insufficient documentation or administrative mistakes. The 2021 improper payment rate is the first estimate that includes reintegrated eligibility error rates (conducted under updated rules) for all three audit cycles (i.e., all states).
COVID-19 Public Health Emergency (PHE): Beginning in 2021, the overall improper payment rate decreased sharply. In 2024, the improper payment rate (5.1%) was less than one-fifth of the 2021 estimate (21.7%). The 2024 improper payment rate was the lowest rate since the COVID-19 pandemic began, which HHS reported was due in part to the exclusion of certain audit review elements as policies adopted during the PHE that paused eligibility renewals and disenrollments and reduced requirements for provider enrollment and revalidations were taken into consideration and due to improved state compliance with program rules. The COVID-19 PHE and related flexibilities were in place from early 2020 through early 2023.
Post-COVID-19 PHE: In early 2023 states began to phase-out the continuous enrollment provision that suspended Medicaid eligibility renewals and disenrollments during the pandemic and flexibilities that reduced provider enrollment requirements. Most states completed eligibility redeterminations for all Medicaid enrollees and resumed disenrollments by August 2024. Improper payment rates since 2021 include at least some audits conducted while COVID-19 PHE flexibilities were in place. The 2025 improper payment rate incorporated the first full audit conducted following the end of the PHE (July 2023 – June 2024). The 2025 improper payment rate (6.1%) increased relative to 2024 (5.1%). From early 2023 through late 2024, states resumed eligibility renewals and disenrollments and phased out provider screening flexibilities allowed during the PHE.
What types of errors are driving the eligibility component error rate?
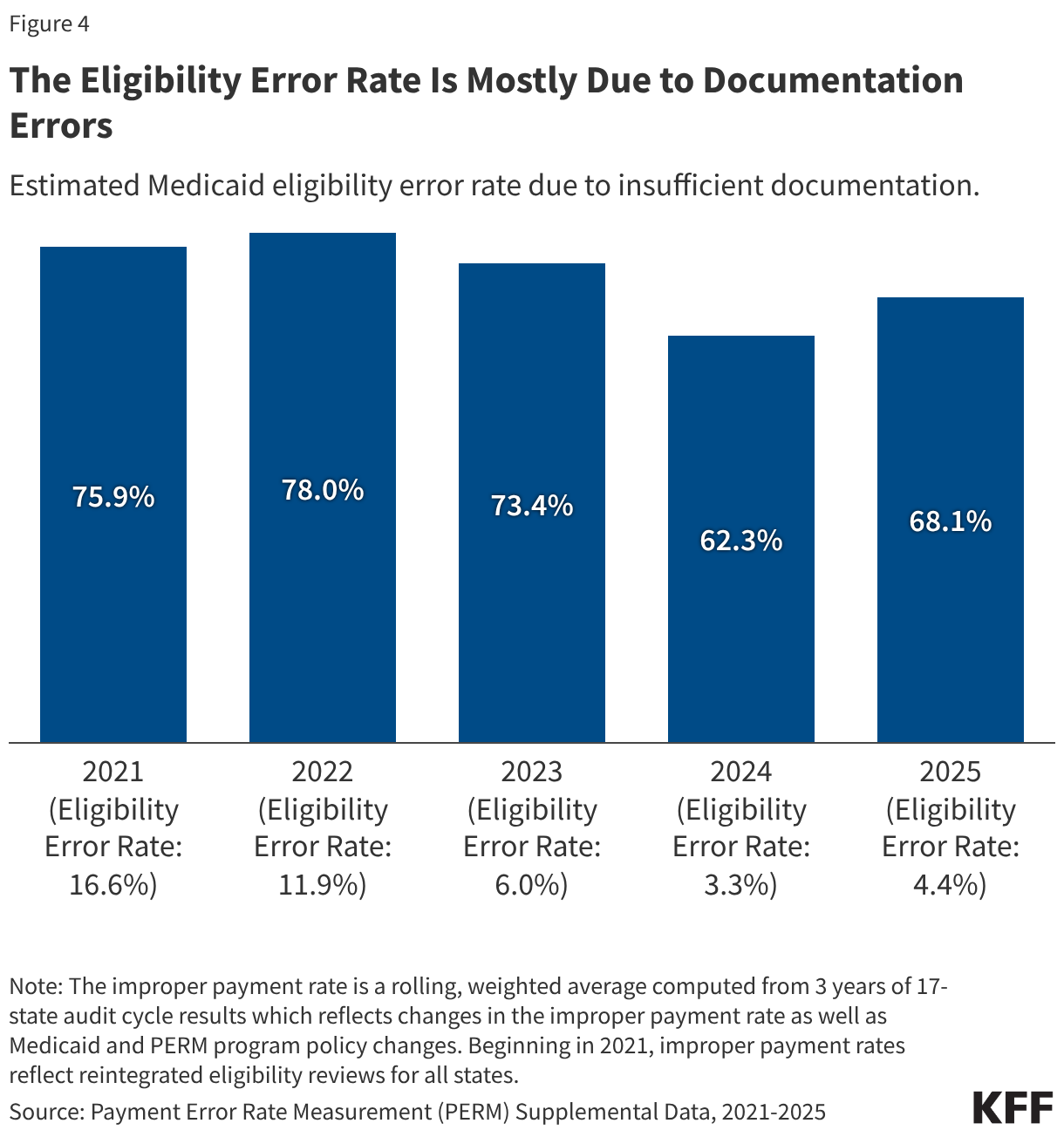
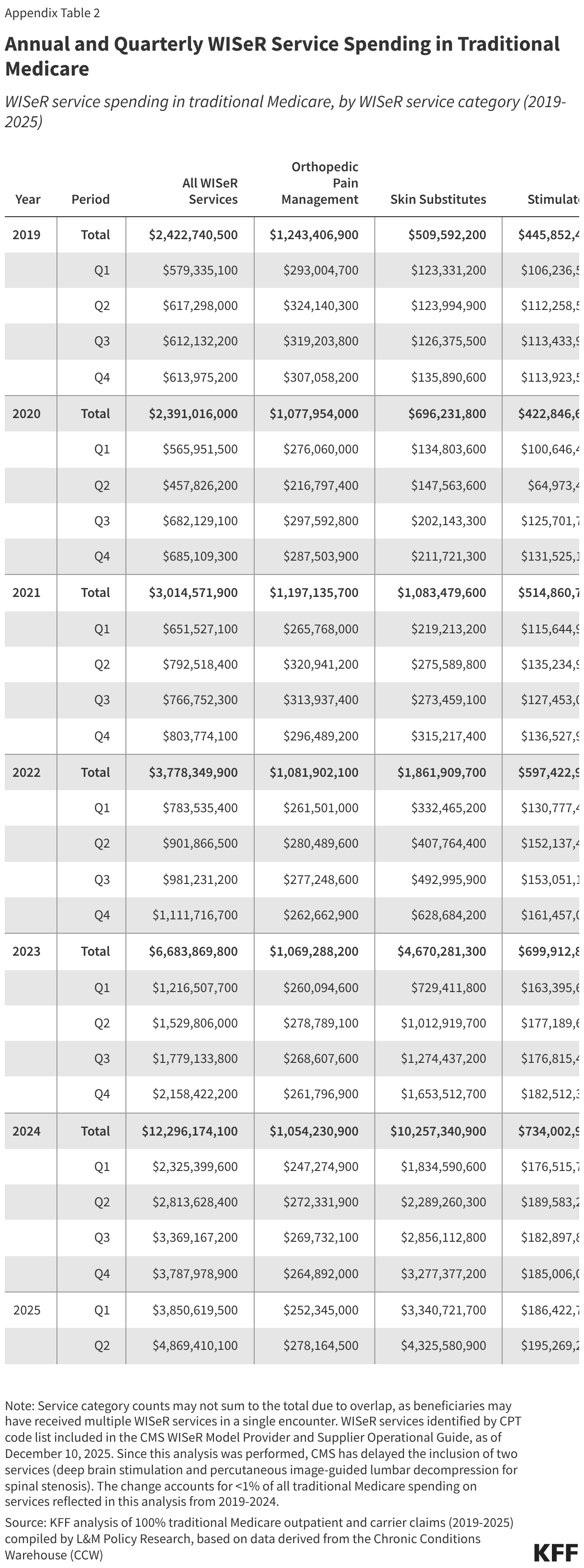
Insufficient documentation errors are the cause of most eligibility errors. While the national eligibility error rate decreased from 2021-2024, due in part to audits that accounted for the continuous enrollment provision that paused Medicaid eligibility renewals and disenrollments from March 2020 through March 2023 (e.g., excluding errors due to out-of-date redeterminations or changes in the enrollee’s circumstances), the eligibility error rate rose from 2024 to 2025 as states resumed normal eligibility operations (Figure 3 and Box 1). Insufficient documentation errors continue to be the cause of most eligibility errors (Figure 4). Eligibility determination errors caused by reasons other than insufficient documentation include enrollees receiving services for which they are financially or categorically ineligible (for example, an enrollee who is eligible for a certain eligibility group and receives services through a program for another eligibility group).
Recent federal rules increase recordkeeping requirements aimed at reducing insufficient documentation errors. Beginning in 2026, states must adhere to updated eligibility and payment recordkeeping standards required in the 2024 Eligibility and Enrollment Final Rule. This rule aimed to reduce “paperwork” errors that lead to the majority of eligibility-related improper payments by implementing new documentation and retention requirements and procedures. The Eligibility and Enrollment rule specifies that states must maintain records for all eligibility determinations in an electronic format, clarifies what information must be contained in the records, and requires that records be kept for a minimum of three years. States must comply with these requirements by June 2026.
How is the reconciliation law changing the PERM program and how could these changes impact states?
PERM program changes in the reconciliation law focus on the eligibility component of the error rate. Beginning in 2029, states may be subject to a financial penalty if PERM eligibility error rates exceed three percent. Even before passage of the reconciliation law, HHS had the authority to recoup federal funds from states with eligibility-related improper payment error rates greater than three percent. This statutory recoupment authority was first implemented by CMS through the Medicaid Eligibility Quality Control (MEQC) program (which complements the PERM program). In 2020, GAO reported no recoupments due to MEQC eligibility errors had occurred since 1992. In 2017, CMS introduced new procedures to begin recouping funds based on PERM eligibility errors beginning in FY 2022, but there are no public reports of any recoupments. Prior law also gave HHS authority to grant states “good faith” waivers if they demonstrated a “good faith effort” to improve eligibility error rates. However, the reconciliation law effectively eliminates “good faith” waivers, requiring CMS to impose the penalty for states that exceed the PERM eligibility error threshold. Beginning October 1, 2029, states with eligibility error rates greater than three percent will have to repay the federal portion of the improper payment amount above the three percent threshold. The reconciliation law also expands the types of eligibility errors that are used to calculate the financial penalty. Insufficient documentation errors will be included in the federal financial recoupment calculation. Implementing federal financial recoupment will likely depend on further federal guidance to states.
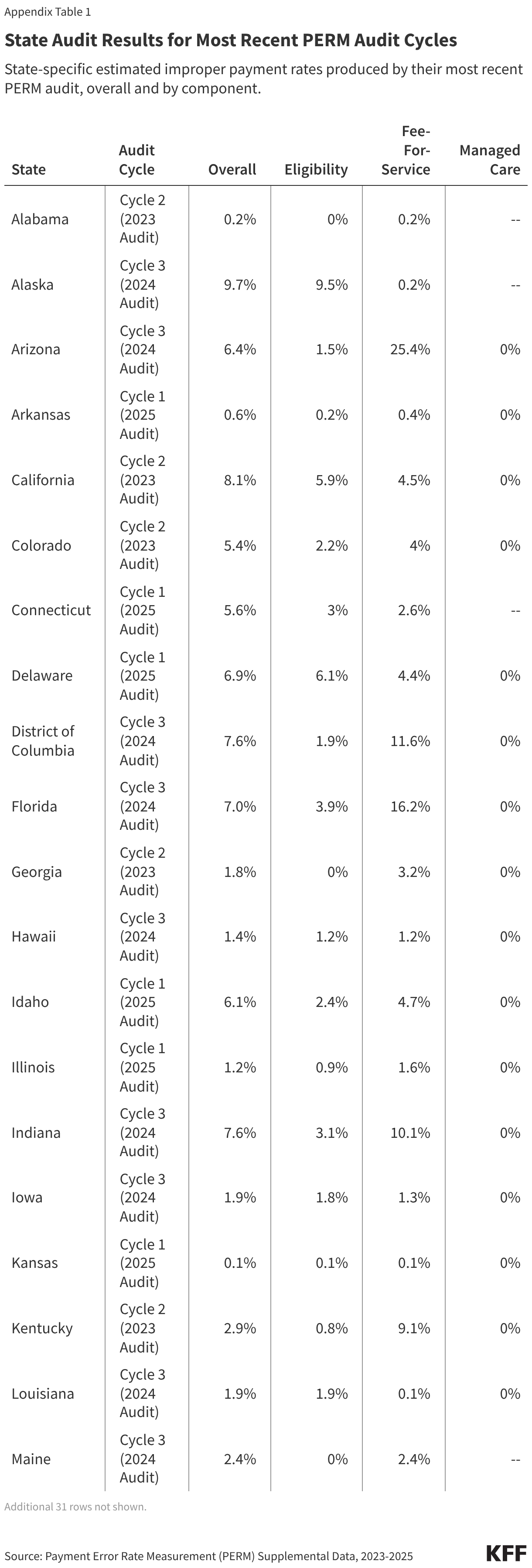
Based on the most recent PERM audits, nearly one quarter of states have eligibility error rates above the new threshold for penalty.3 Twelve states had eligibility error rates above three percent in their most recent PERM audit (Figure 5). The most recent three PERM audit cycles used Medicaid claims and payments from July 2021 – June 2024. During most of this period, eligibility redeterminations and disenrollments were paused under flexibilities related to the COVID-19 PHE, which likely decreased the rate of eligibility errors. As a result, the audit results may not indicate future state performance. The amount of federal funds recouped will vary across states due to differences in eligibility error rates and because of federal match rate (FMAP) differences across states. States with a higher traditional “FMAP” rate risk losing a greater share of any payments subject to recoupment because a larger share of their Medicaid payments come from federal funds.
States will need to prepare for PERM program changes and implement other complex eligibility and enrollment policy changes required by the reconciliation law at the same time. State readiness to comply with increased recordkeeping and electronic documentation requirements in the Eligibility and Enrollment rule will also vary. At a time of increasing state budget pressures, states may need to invest in upgraded technology, expanded audits or staffing, or other costly increases to program integrity capacity to maintain a low rate of improper payments. At the same time, states are also implementing other complex eligibility policy changes required by the 2025 reconciliation law, including several changes by January 1, 2027:
- The reconciliation law requires states to implement work requirements (at application and renewal) for Medicaid expansion adults. States will need to make significant changes to eligibility systems in a short timeframe to verify compliance as well as to identify individuals who qualify for exemptions or optional exceptions. Failure to document compliance with complex work-related federal and state eligibility determination policy could lead to increased PERM eligibility errors.
- The reconciliation law increases the frequency of required eligibility redeterminations from every 12 months to every 6 months for expansion adults. An increased number of eligibility determinations could result in additional eligibility errors.
- The reconciliation law also decreases the time period that states must provide retroactive Medicaid coverage (from three months to one month of retroactive coverage for expansion adults and two months for traditional enrollees). Narrowed claims payable under new retroactive coverage rules could result in additional payment errors.
Beginning in 2029, states risk losing federal funds if their PERM eligibility error rate is above three percent – a threshold nearly one quarter of states met prior to implementing complex eligibility changes included in the reconciliation law (12 states in 2025, Figure 5). However, because these audits occurred during the PHE and unwinding periods, the results may not predict future improper payment rates. CBO estimated $7.6 billion over 10 years in Medicaid payment reductions related to the reconciliation law’s changes to PERM. Updated electronic documentation and recordkeeping requirements may help reduce some errors due to insufficient documentation or administrative missteps. However, to limit the risk of financial penalty due to eligibility errors, states may add administrative steps or impose more onerous paperwork requirements on enrollees or applicants.
Appendix
The overall improper payment estimate for each state combines the state’s fee-for-service, managed care, and eligibility estimates, with a correction factor so that sampled payments that are improper in more than one of those components are not double counted.
- HHS selects a sample size for each PERM component for each state. The number of claims or payments sampled for a state audit ranged between 288 and 1,782 FFS claims, between 38 and 200 managed care payments, and between 97 and 644 eligibility determinations. ↩︎
- Under PERM program procedures prior to a 2017 Final Rule (implemented in 2019), the PERM program’s eligibility component error rate was state reported rather than measured by standardized methodology under an external auditor. The PERM FFS and managed care components have continuously been measured by an external auditor. ↩︎
- In the past, CMS has cautioned that comparing state PERM rates to one another is difficult due to variation in state systems and policies as well as due to external factors (that can make year to year comparisons difficult). ↩︎