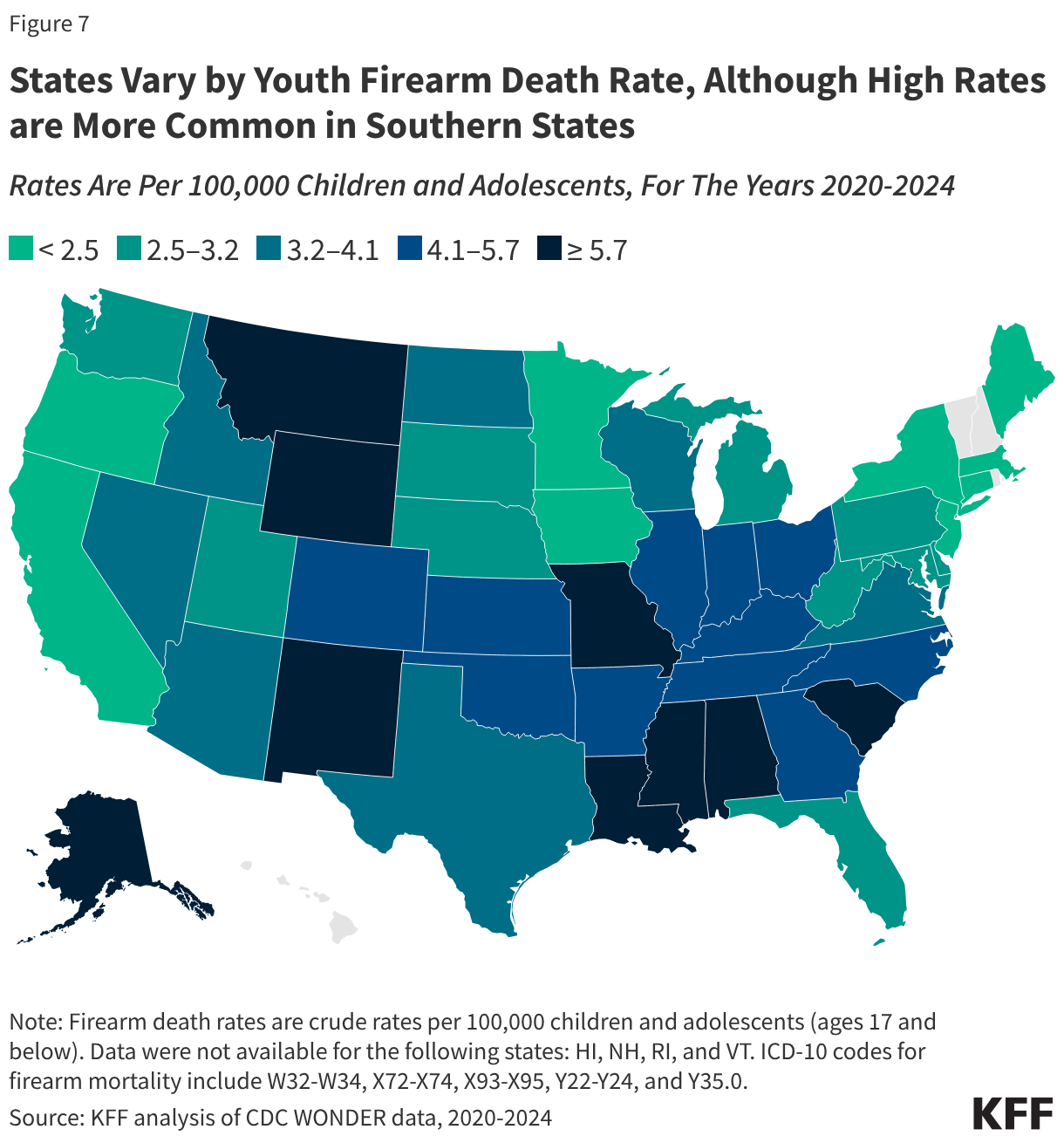
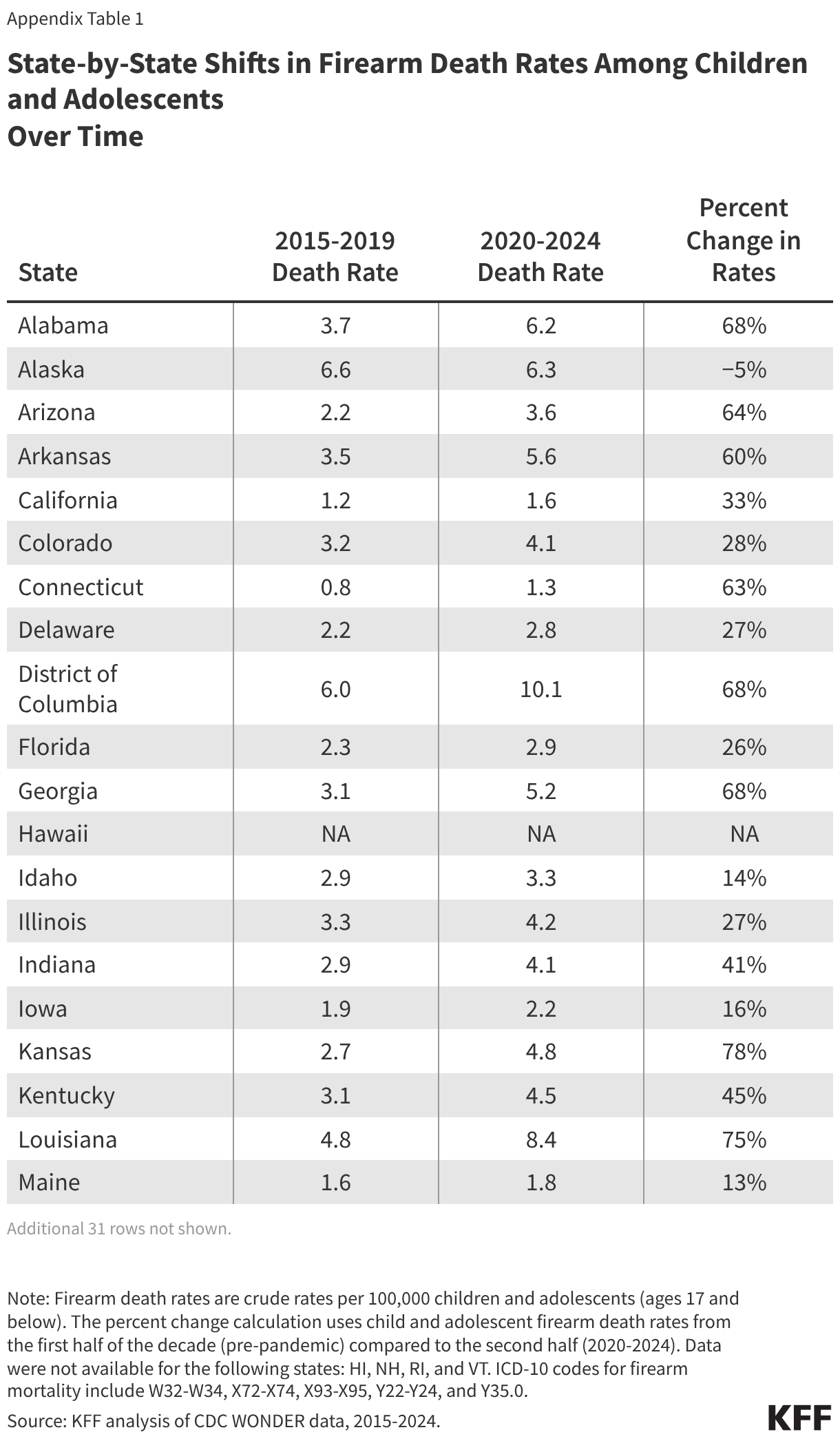
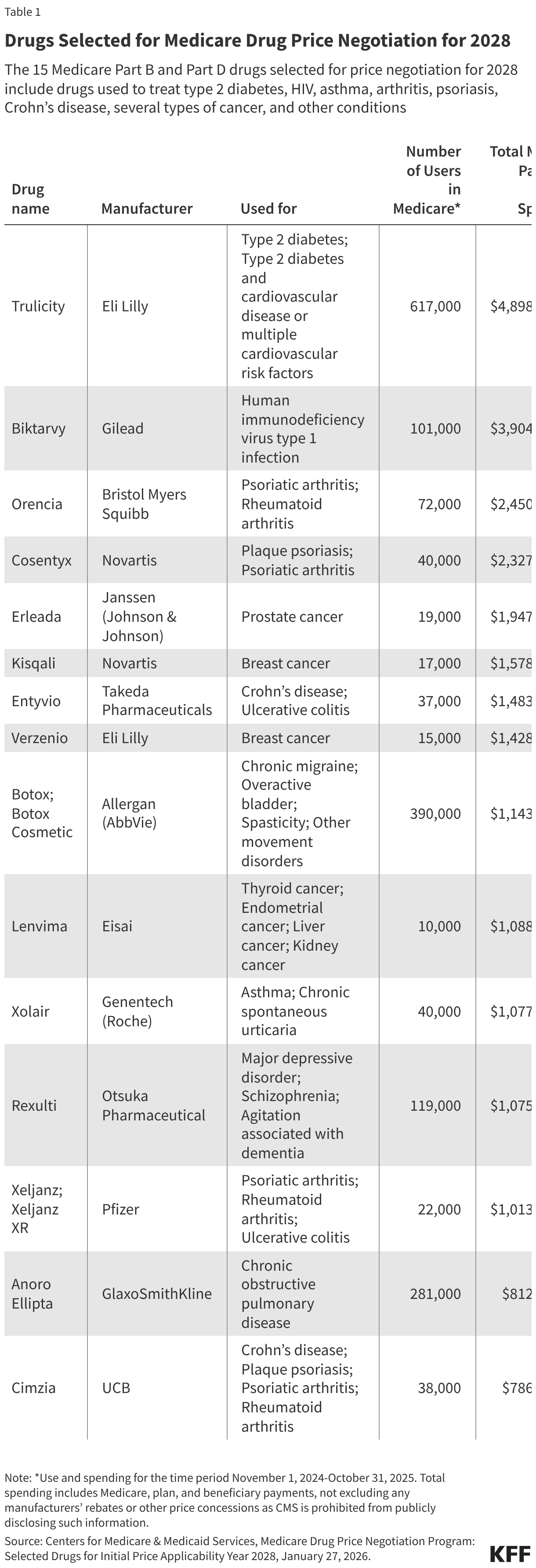
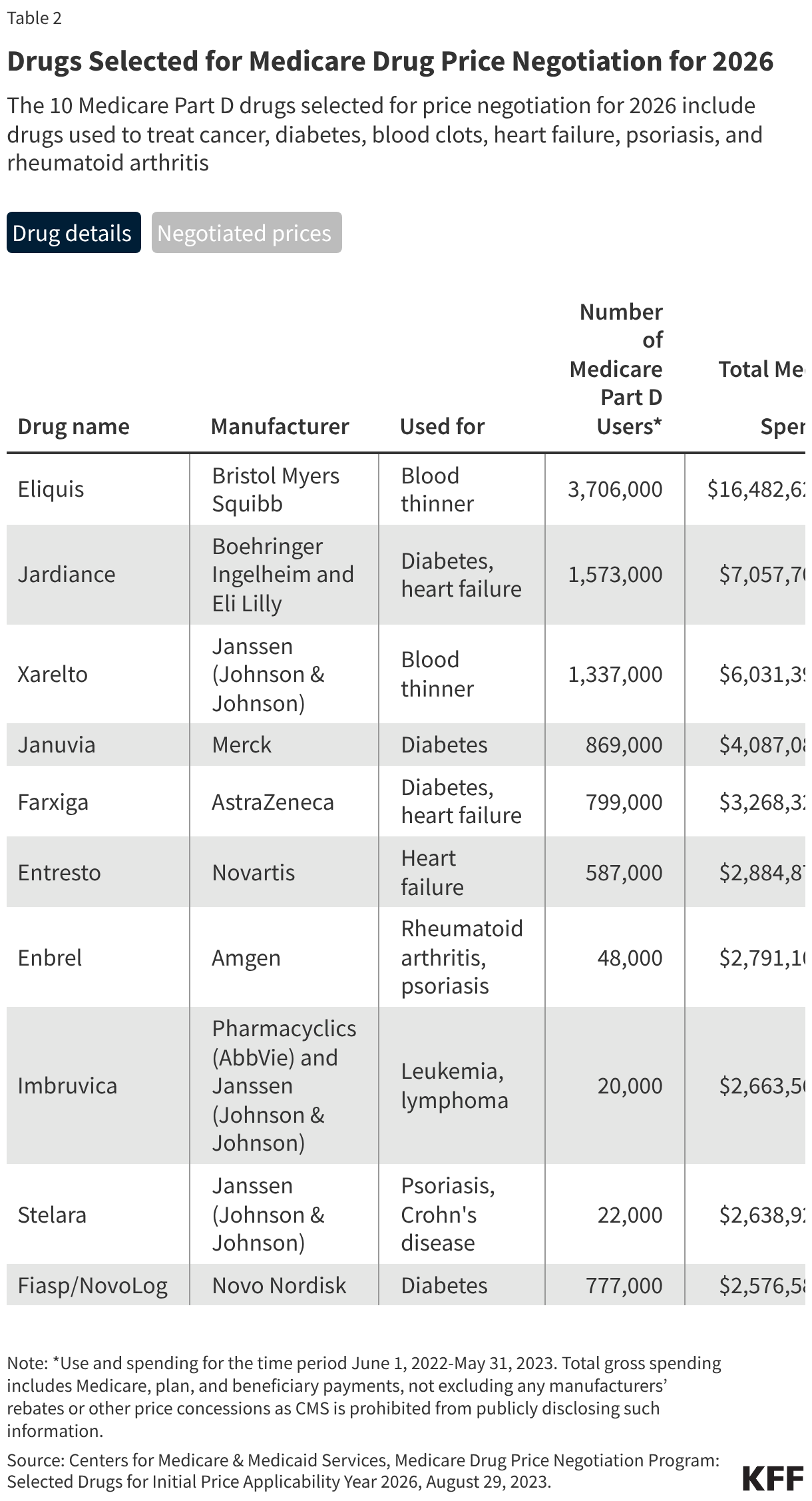
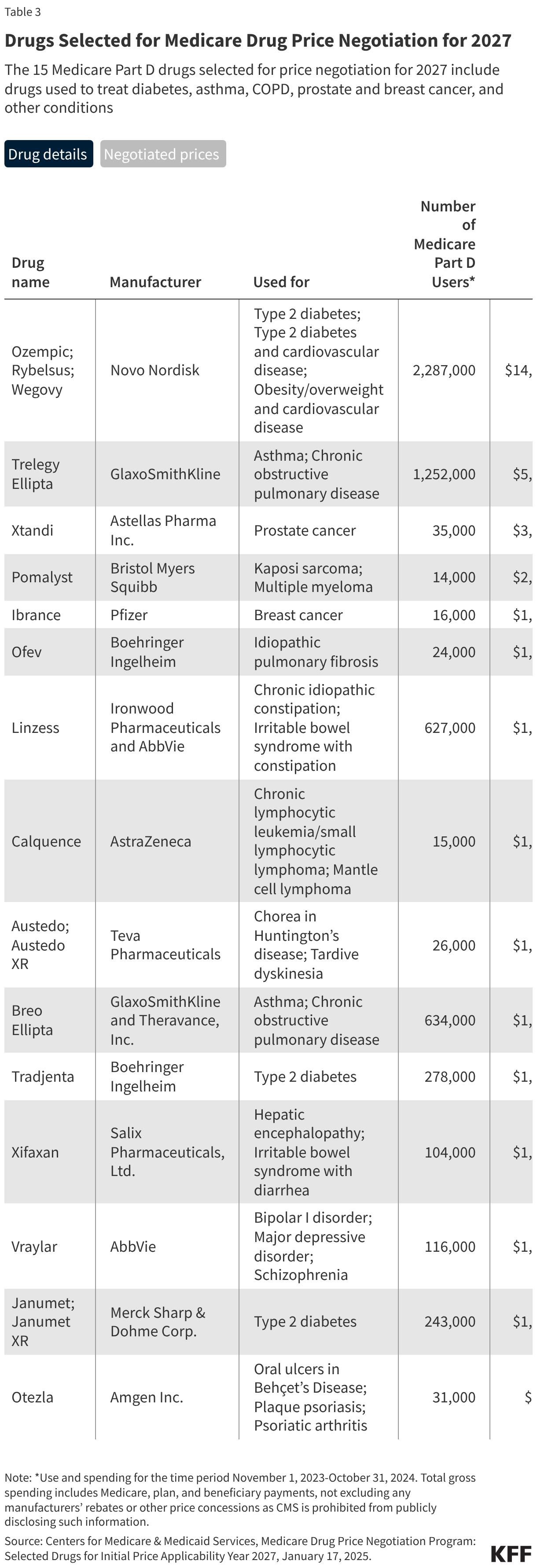
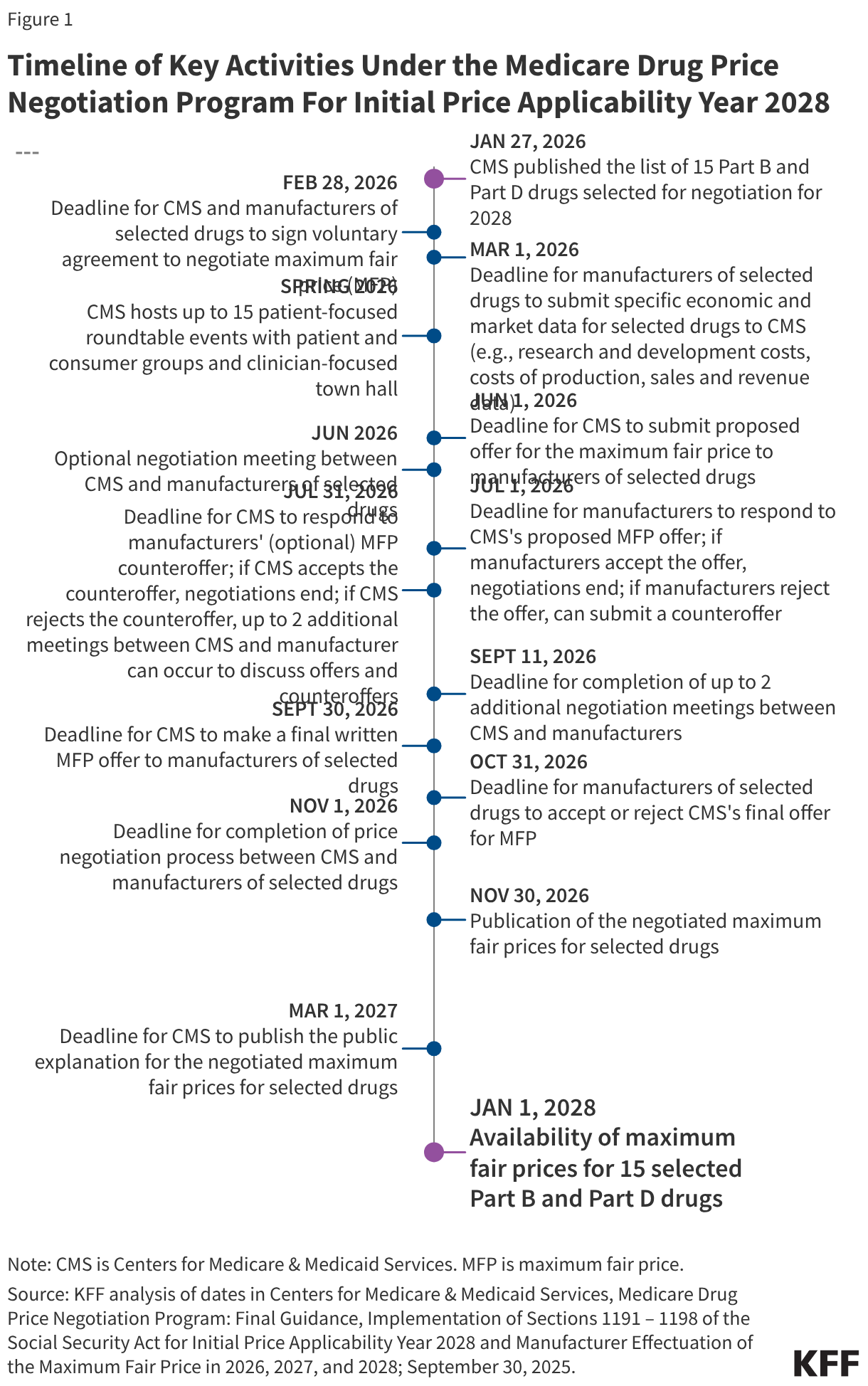
Fifteen drugs covered under Medicare Part B, which covers physician-administered drugs, or Medicare Part D, Medicare’s outpatient prescription drug benefit program, were selected for price negotiation in 2026, with Medicare’s negotiated prices for these drugs taking effect on January 1, 2028 (Table 1). These 15 drugs include treatments for type 2 diabetes, HIV, asthma, arthritis, psoriasis, Crohn’s disease, several types of cancer, and other conditions (See Table 1). Total gross Medicare spending on these 15 drugs between November 2024 and October 2025 was $27 billion, with 1.8 million Medicare beneficiaries using these medications during that time. Starting in 2027 and in each subsequent year, up to 20 additional drugs covered under Part B or Part D will be selected for negotiation. The number of drugs with negotiated prices available will accumulate over time.
Public Views on Prescription Drug Costs: Regulation, Affordability and TrumpRx
Findings
Key Takeaways
- In recent weeks, the Trump administration has renewed focus on lowering the cost of prescription drugs in the U.S., including the launch of TrumpRx. The latest polling from KFF shows that about four in 10 U.S. adults (41%) say it is likely the Trump administration’s policies will lower prescription drug costs for people like them, but views are largely influenced by partisanship. Only the president’s base remains positive, with 79% of Republicans and 88% of Make America Great Again (MAGA) supporters saying it is likely the administration will lower prescription drug costs, while much fewer independents (35%) and Democrats (11%) say it is likely. But there is broad, bipartisan agreement that the government should be playing a bigger role when it comes to regulating prescription drug costs, with at least two-thirds of Republicans, Democrats, and independents saying there is not as much government regulation as there should be in this area.
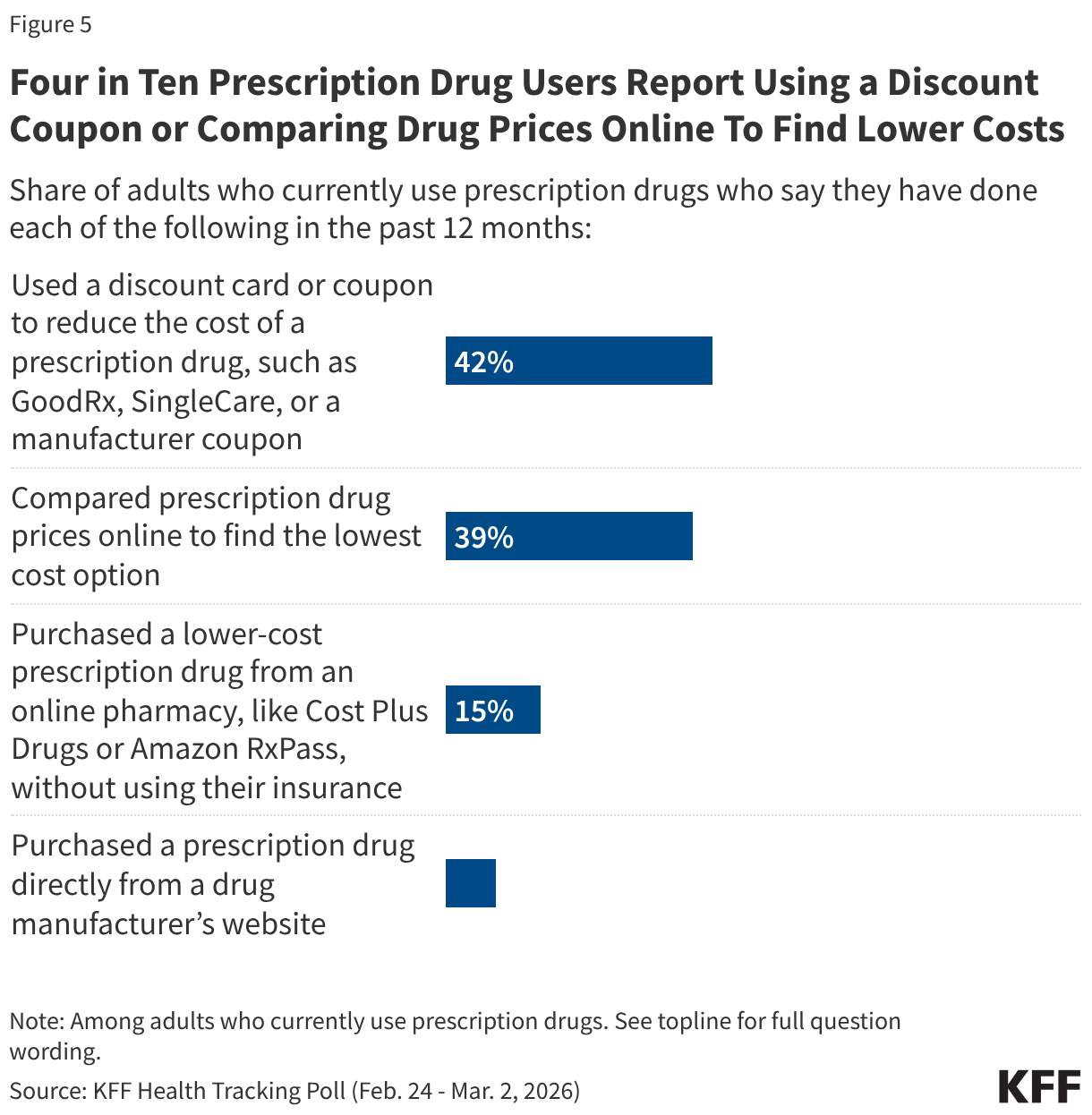
- TrumpRx, the new federal government-run website where people can get discounts to buy prescription drugs directly from some drug manufacturers or pharmacies, has gained some attention among those who currently take prescription medication, as one-third (35%) report having heard “a lot” or “some” about it. Seven percent of adults who currently take prescription medication say they have visited the TrumpRx website to compare prescription drug prices, rising to one in six (16%) of those who currently or have ever taken a GLP-1 medication, one of the classes of drugs consumers can get discounted rates through the new website. Beyond TrumpRx, drug discounts have long been available through third-party platforms, such as GoodRx, and directly from drug manufacturers. About four in 10 report that they have used a discount coupon to reduce the cost of a drug (42%) or gone online to compare prescription drug prices to find the lowest cost option (39%).
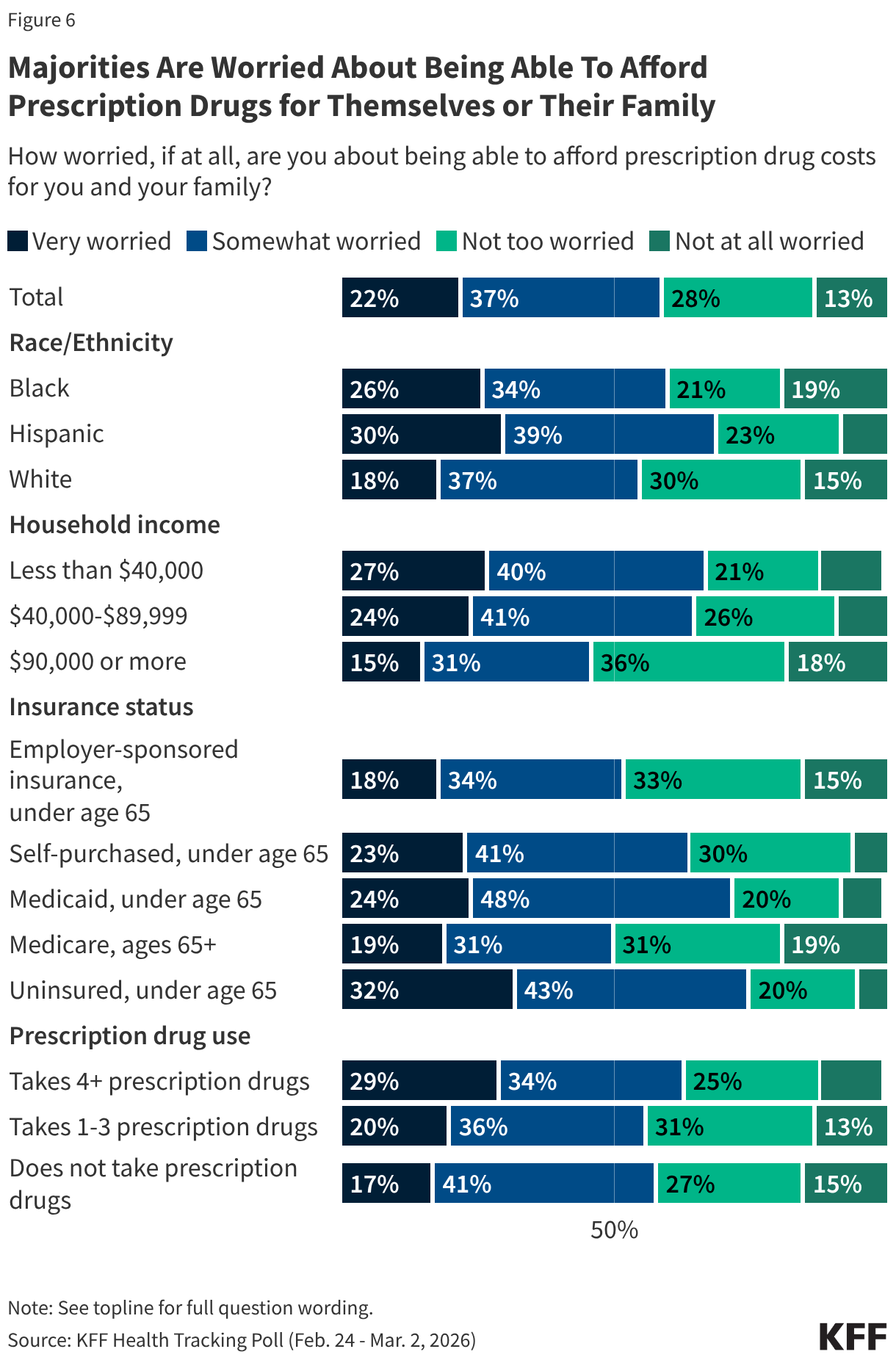
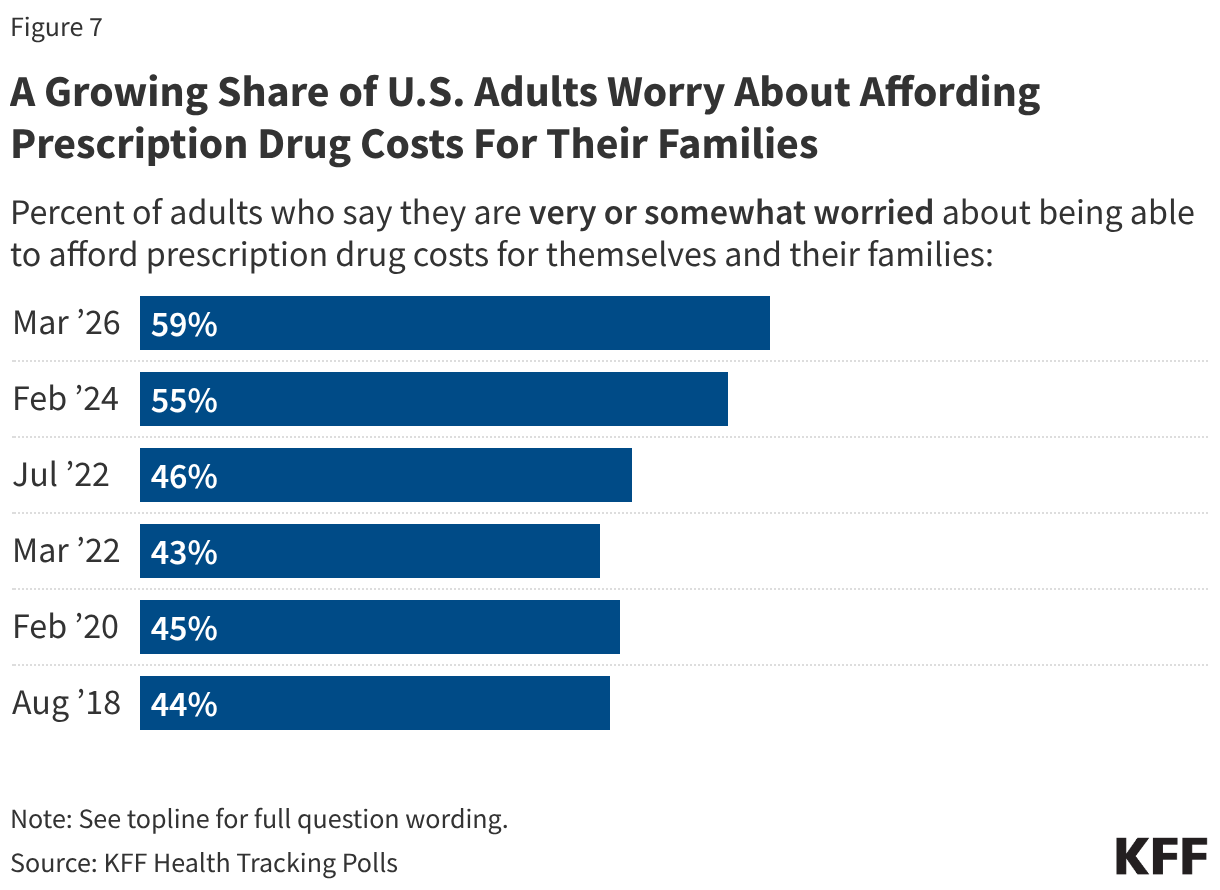
- The latest KFF poll shows that despite the Trump administration’s recent actions on prescription drug costs, a majority of the public (59%) is worried about affording prescription drugs for themselves and their families, the largest share since KFF first polled this question in 2018. The shares of adults worried about their prescription costs are larger among adults in households with annual incomes less than $40,000 (67%) and those who take at least four prescription medications (64%).
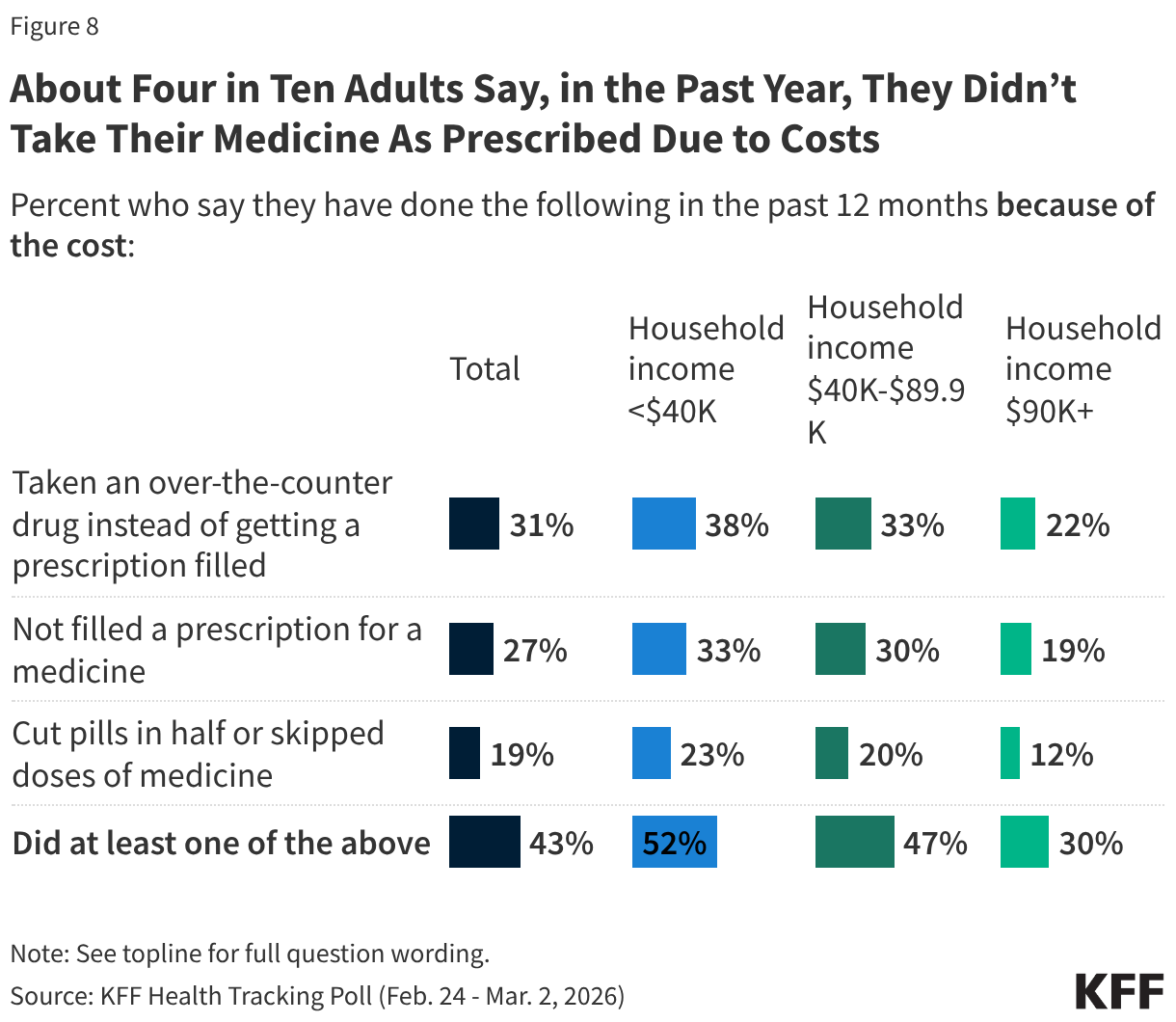
- About four in 10 (43%) U.S. adults say they have not taken their medication as prescribed in the past year due to costs. This includes three in 10 who say they have taken an over-the-counter drug instead of getting a prescription filled (31%), a quarter (27%) who have not filled a prescription, and one in five (19%) who have cut pills in half or skipped doses of medicine because of the cost. Larger shares of lower-income, uninsured, Black, and Hispanic adults report taking these measures.
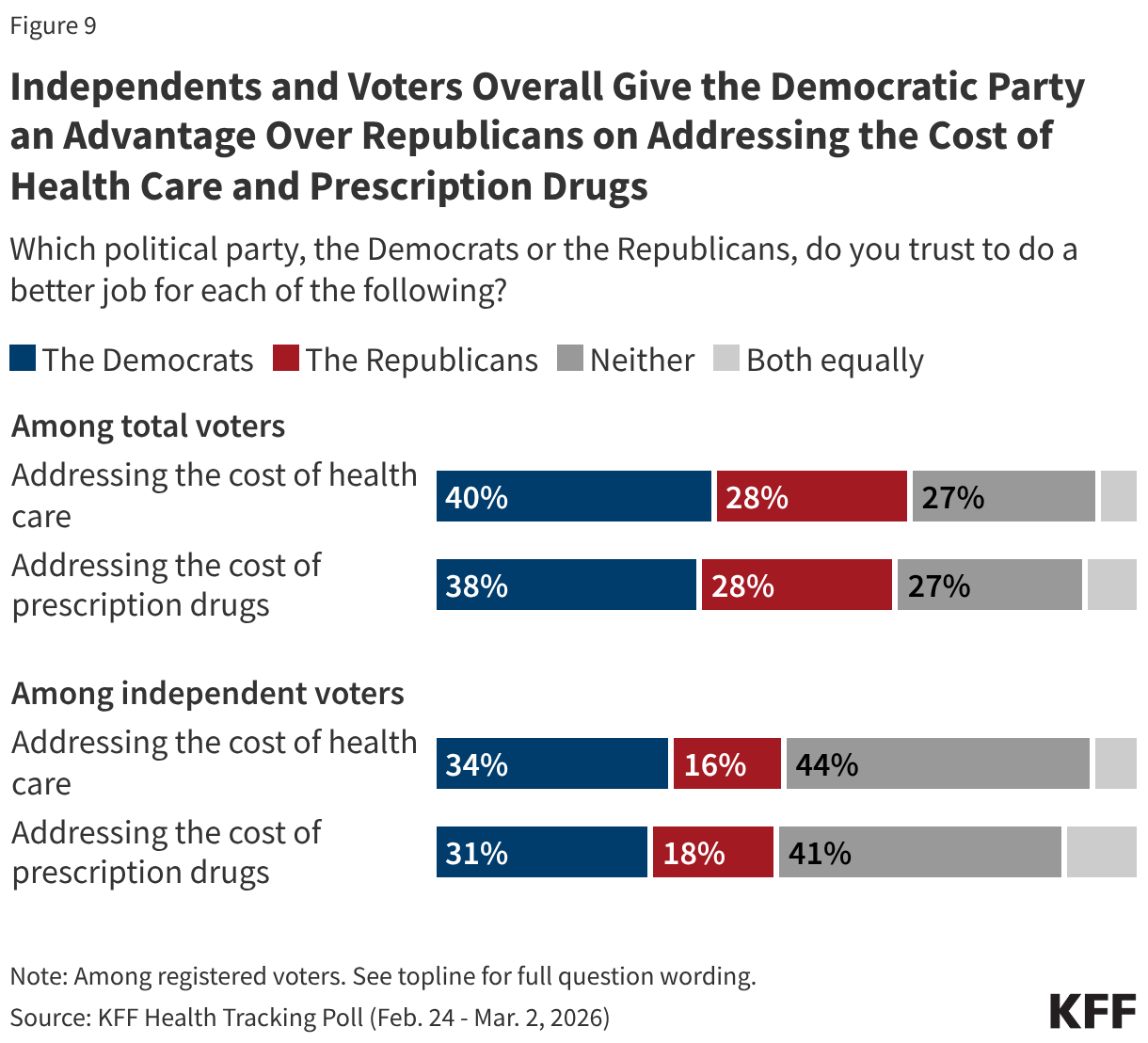
- Looking ahead to the 2026 midterm elections, the Democratic Party currently holds the advantage when it comes to who voters trust to address the cost of health care, including prescription drugs. Nearly four in 10 voters say they trust the Democratic Party to do a better job addressing the cost of prescription drugs (38%), 10 percentage points larger than the share who say they trust the Republican Party more (28%). However, reflecting frustrations with lawmakers over the rising costs, about one in four (27%) voters say they trust “neither party” to handle the issue.
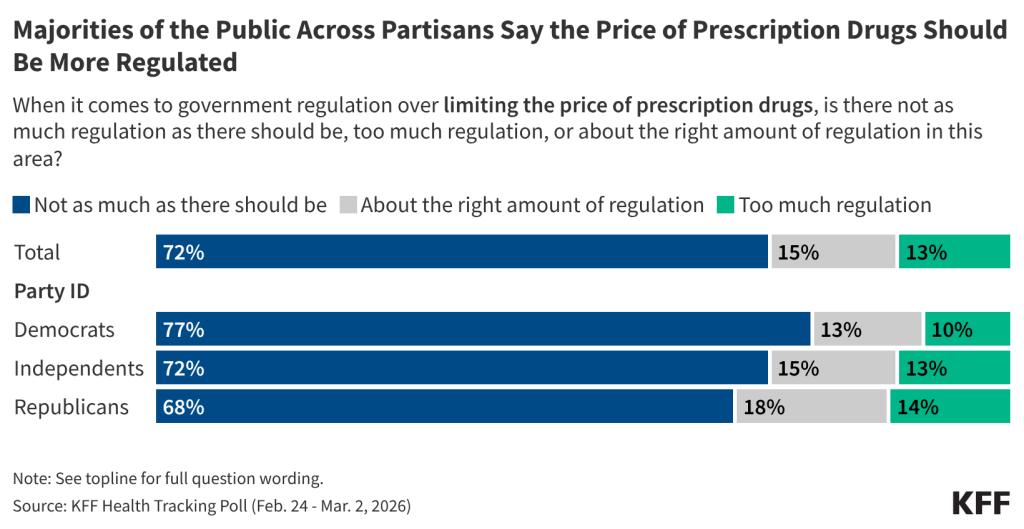
Most U.S. Adults Want More Regulation of Prescription Drug Pricing
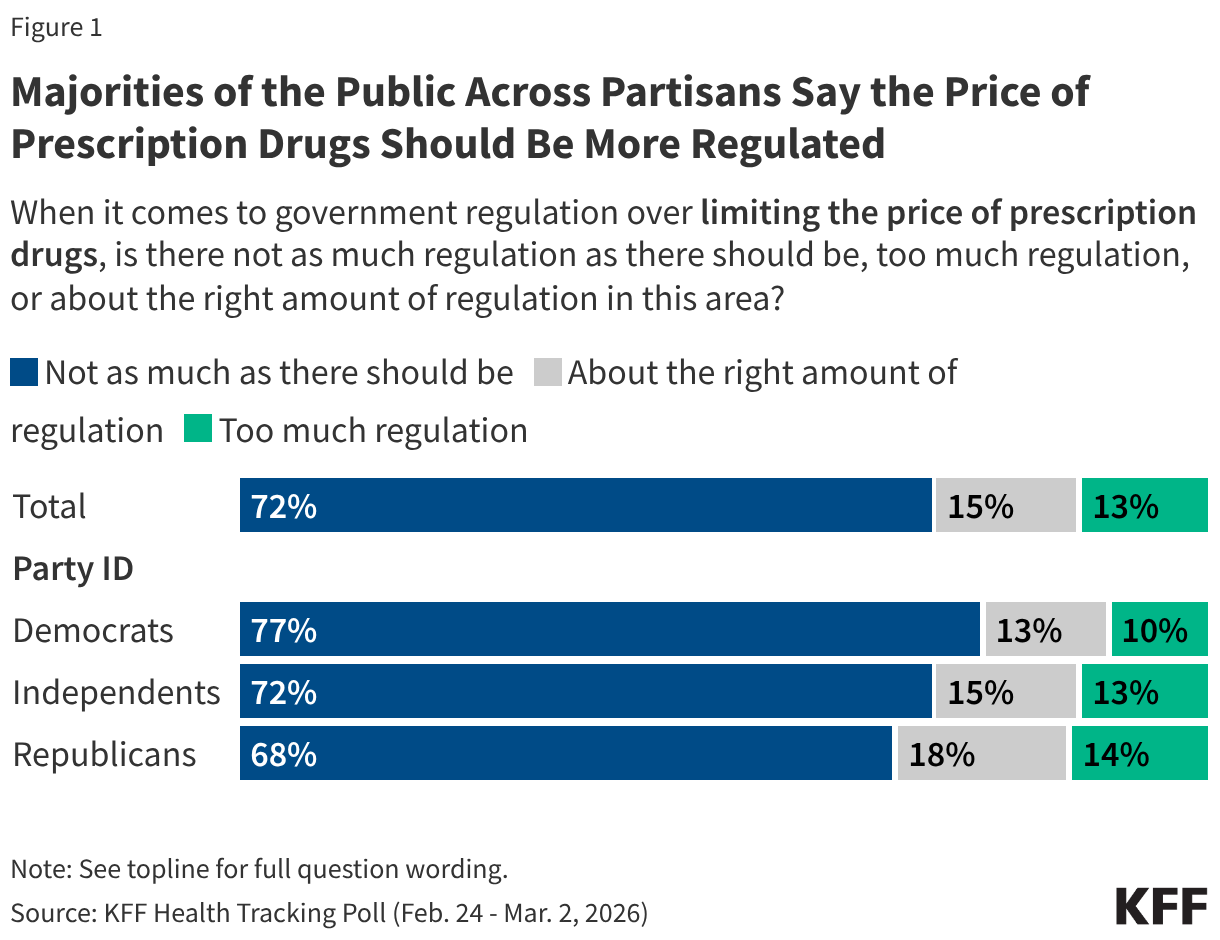
There is broad, bipartisan agreement that there should be more government regulation when it comes to prescription drug costs. About seven in 10 (72%) adults say there is not enough government regulation when it comes to limiting the price of prescription drugs, while 15% say there is “about the right amount,” and 13% say there is “too much” regulation in this area. At least two-thirds of Democrats (77%), Republicans (68%), and independents (72%) say there is not enough government regulation when it comes to limiting prescription drug prices.
Among adults who currently take prescription medications, more than three in four say there is not enough government regulation of prescription drug prices (77%), including similar majorities of those who take one to three medications (78%) and four or more (75%). Among those who do not currently take prescription medications, a smaller majority agrees (62%).
Some Adults Who Take Prescription Medication Report Visiting TrumpRx, But Most Doubt the Administration’s Policies Will Lower Costs
In early February, the Trump administration officially launched TrumpRx, the federal government-run website where people can get discounts to buy prescription drugs directly from some manufacturers or pharmacies, without using their health insurance. Few U.S. adults have heard much about the website in the weeks following its launch, and most remain skeptical that relief is coming.
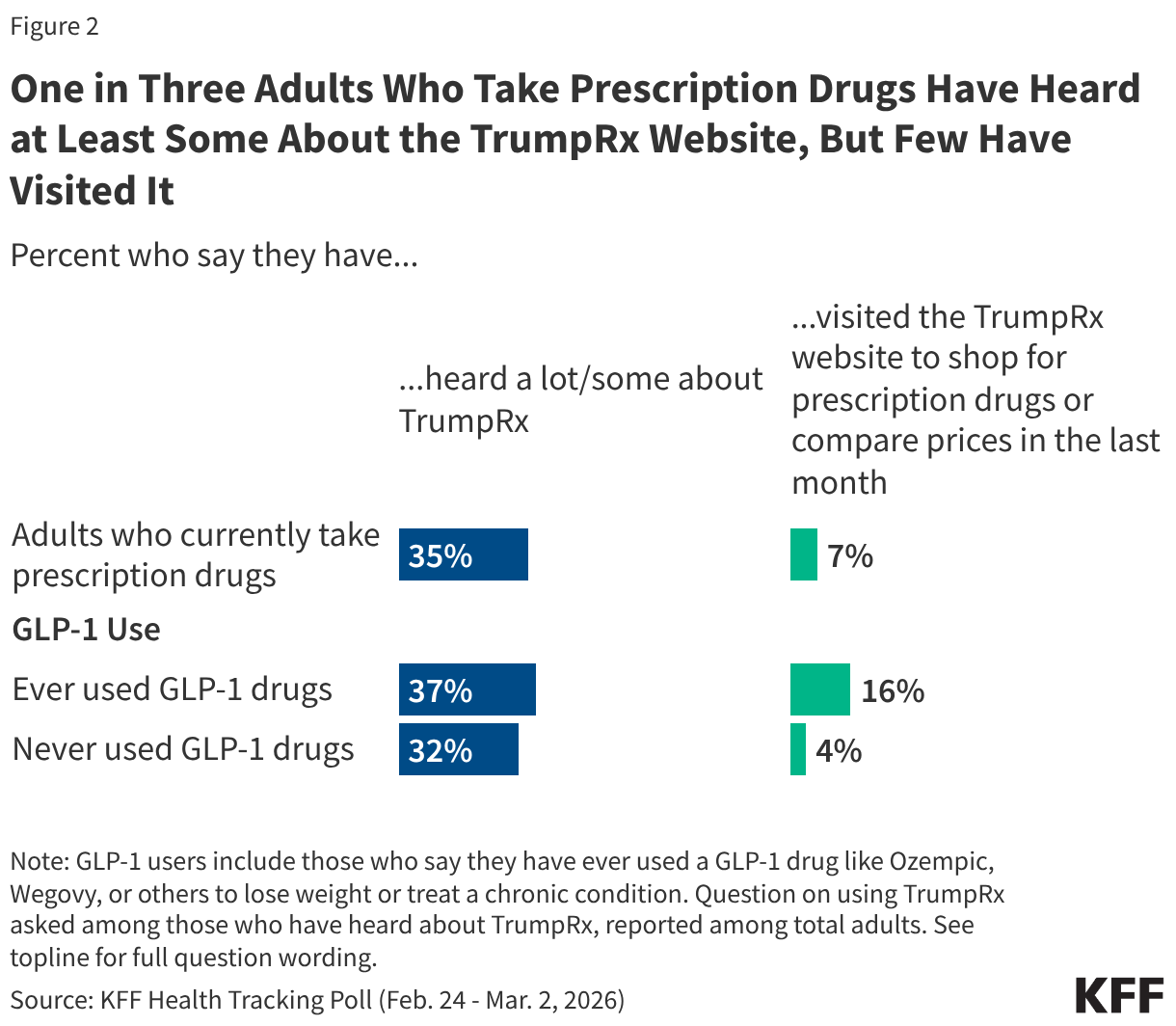
One-third (35%) of adults who currently take prescription medication (66% of all adults) say they have heard “a lot” (6%) or “some” (29%) about TrumpRx, up from about one in five (18%) who had heard about plans for the site in November 2025, leaving a large majority of prescription drug users still unaware of the new program. However, awareness has grown slightly since the site’s launch as about one-third (32%) of adults who take prescription drugs now say they have heard “nothing at all” about TrumpRx, compared to six in 10 (61%) in the months preceding the launch.
With GLP-1 agonist prescriptions on the rise, KFF polling finds nearly one in five adults (18%) have ever taken a GLP-1 medication, including 12% who report currently taking one. While few prescription drug users (7%) say they have visited the TrumpRx site to shop for or compare prescription prices in the past month, this rises to about one in six (16%) among those who currently take or have ever taken a GLP-1 medication for weight loss or certain chronic conditions. The TrumpRx website features at least four major GLP-1 medications among its initial 43 listed drugs.1
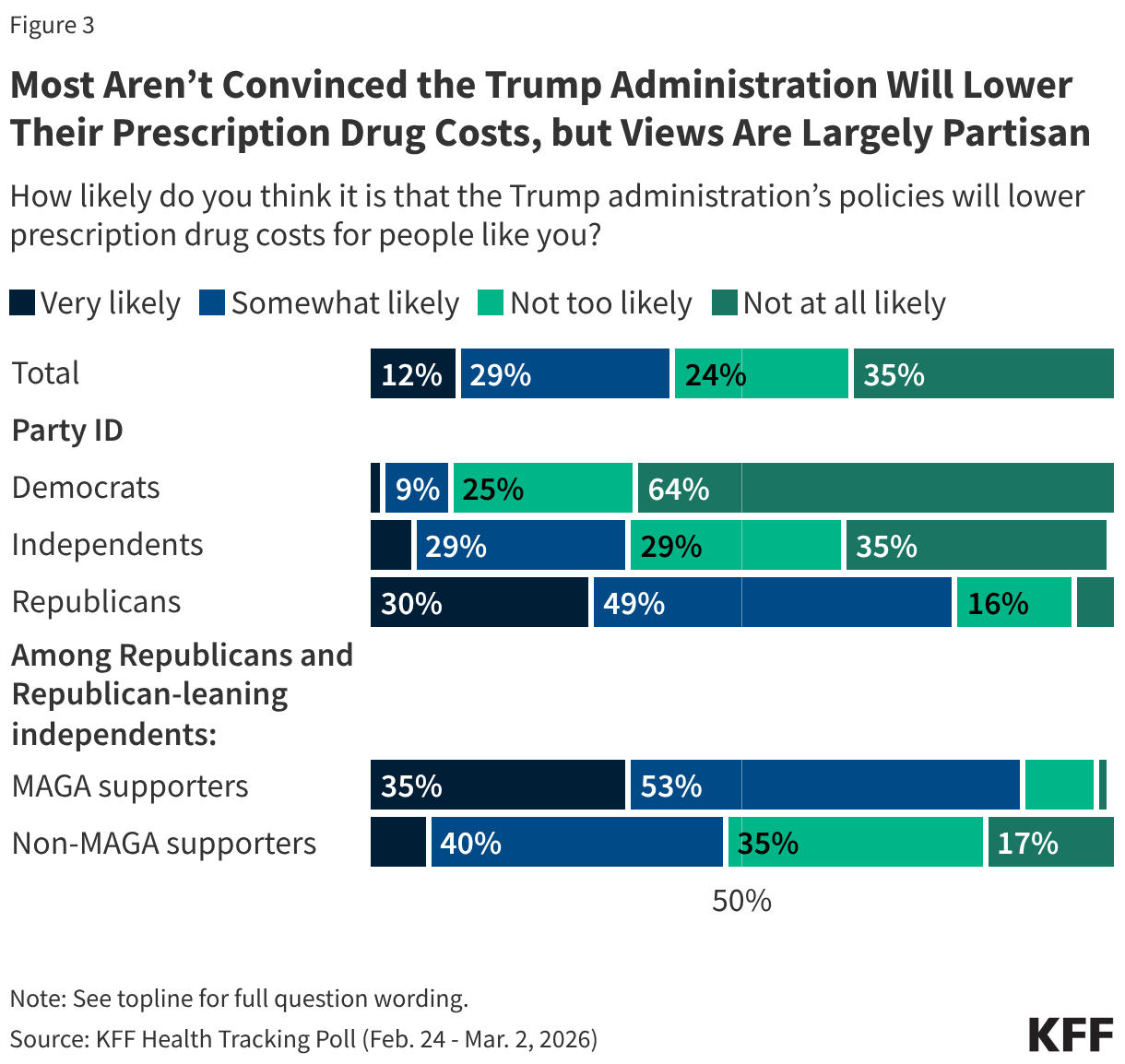
The public remains skeptical that the Trump administration’s policies will lower prescription drug costs for people like them. About six in 10 (59%) adults say it is “not too likely” or “not at all likely” that the policies will lower drug costs, compared to about four in 10 (41%) who say it is “very likely” or “somewhat likely.”
These expectations largely mirror overall partisan views of actions by the Trump administration, with large majorities of Republicans (79%) and MAGA-supporting Republican and Republican leaning independents (88%) saying it is likely the administration will lower drug costs for people like them. Much smaller shares of independents (35%) and Democrats (11%) say they think the administration will lower their prescription drug costs.
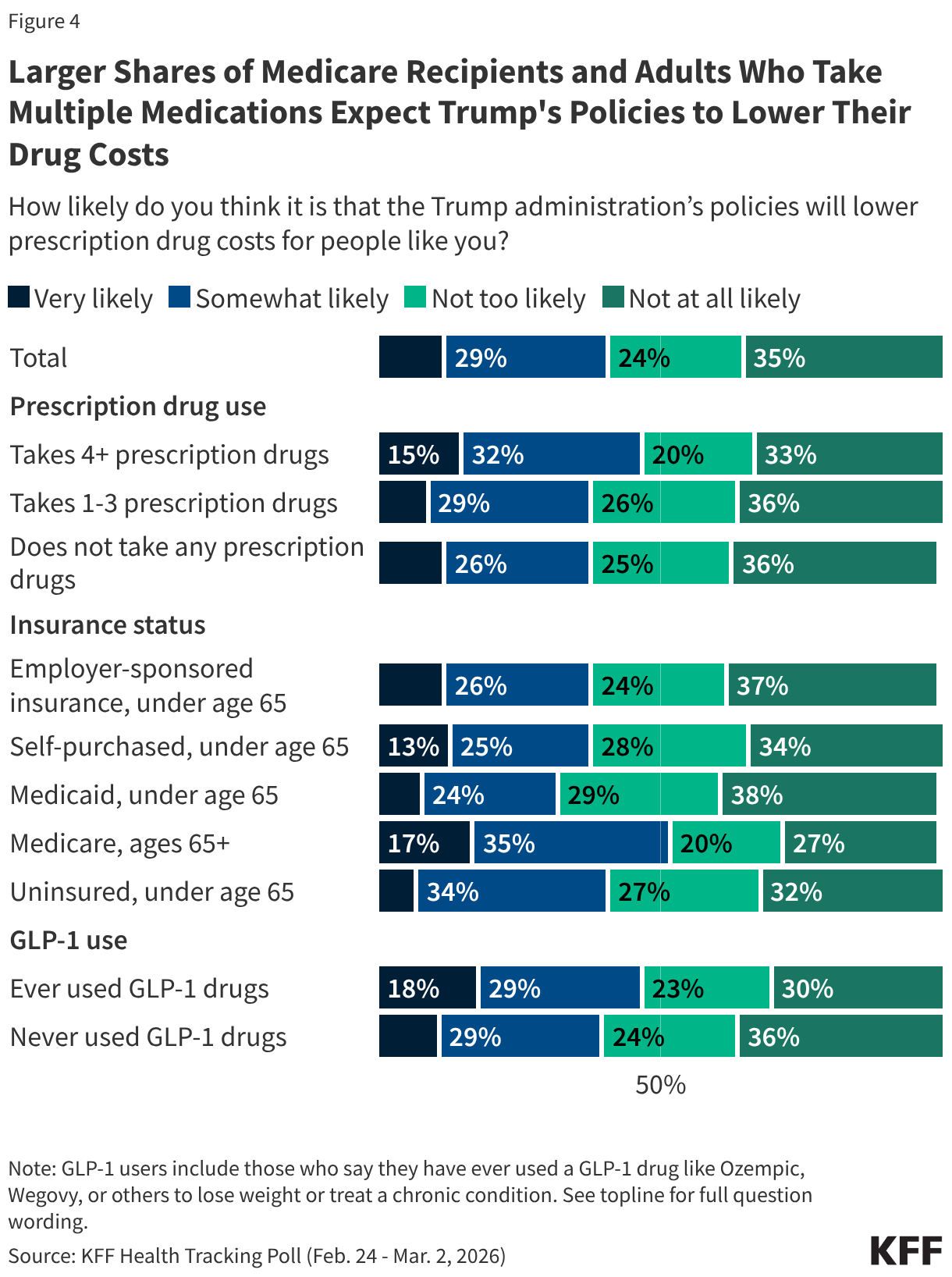
Adults ages 65 and older with Medicare coverage are split on whether the administration’s policies will lower prescription drug costs for people like them (53% say it is likely, 47% say it is unlikely). Similarly, nearly half of adults who take four or more prescription medications say the administration’s policies will lower their costs (47%), while half say it is unlikely (53%). Nearly half (46%) of adults who take or have taken GLP-1 medications say the Trump administration’s policies are likely to lower their costs, while 54% say it is unlikely.
Prior to TrumpRx, drug discounts have long been available through third-party platforms such as GoodRx and directly from drug manufacturers. About four in 10 adults who currently take prescription medication say, in the past year, they have used a discount card or coupon to reduce their prescription drug costs, such as GoodRx, SingleCare, or a manufacturer coupon (42%), or compared prescription drug prices online to find the lowest cost option (39%). Fewer say they have purchased a lower-cost drug from an online pharmacy without their insurance (15%) or directly from a drug manufacturer’s website (8%).
A Growing Majority of U.S. Adults Worry About Affording Their Prescription Medication Costs
Prescription drug costs are a widespread concern for U.S. adults. Two-thirds (66%) currently take prescription medication, including three in 10 (31%) who report taking four or more. Most U.S. adults are worried about affording prescription drugs for themselves and their families, and four in 10 say they have not taken their medication as prescribed in the past year due to cost.
Overall, about six in 10 U.S. adults say they are worried about being able to afford prescription drug costs for themselves or their families (59%), including about one in five (22%) who are “very worried.” Substantial shares of uninsured adults under age 65 (32%), Hispanic adults (30%), Black adults (26%), adults in households with annual incomes less than $40,000 (27%) say they are “very worried” about affording their prescription drug costs.
Among adults who take four or more prescription medications, about two-thirds (64%) report worrying about affording their medications, including about three in 10 (29%) who are “very worried.”
Notably, this KFF poll finds the largest share of U.S. adults saying they are “very” or “somewhat” worried about affording prescription drug costs for themselves or their families since KFF first polled on this question in 2018.
Among Medicare beneficiaries ages 65 and older, the share who report being worried about affording their prescription drug costs has remained unchanged from August 2018. The vast majority of Medicare beneficiaries are enrolled in Medicare Part D plans, giving them prescription drug coverage that has improved with recent policies in the Inflation Reduction Act of 2022.
About four in 10 (43%) U.S. adults say, in the past year, they have not taken their medication as prescribed due to the cost. This includes about three in 10 adults who say they have taken an over-the-counter drug instead of getting a prescription filled because of the cost (31%), one in four who say they have not filled a prescription for a medicine due to the cost (27%), and about one in five (19%) who say they have cut pills in half or skipped doses of medicine because of the cost in the past year.
Notably, larger shares of adults report not taking medication as directed due to the cost than KFF polls found three years ago, when about three in 10 (31%) reported taking at least one of these cost-saving measures.
Larger shares of adults in households with lower and middle incomes report resorting to these cost-saving prescription medication solutions compared to those with higher incomes. About half of adults in households with annual incomes under $40,000 (52%) or between $40,000 and $90,000 (47%) say they have not taken their medication as prescribed due to the cost in the last year, compared to three in 10 adults in households with incomes of $90,000 or more.
Democratic Party Holds the Advantage on Addressing Health Care, Prescription Drug Costs
Looking ahead to the 2026 midterm elections, the Democratic Party holds the advantage over the Republican Party on who voters trust to address health costs, including prescription drugs. Nearly four in 10 (38%) voters say they trust the Democratic Party to do a better job addressing the cost of prescription drugs, while about three in 10 (28%) say they trust the Republican Party. However, reflecting a general frustration over the cost of prescription drugs in the U.S., about one in four (27%) say they trust “neither party” to handle the issue.
The Democratic advantage on drug costs mirrors the party’s advantage on addressing health care costs overall. Four in 10 voters say they trust the Democratic Party to do a better job addressing the cost of health care, compared to about three in 10 (28%) who trust the Republican Party. Again, about one in four voters (27%) say they do not trust either party to handle the issue.
While partisans largely trust their own party to address the cost of prescription drugs and health care generally, independent voters are more likely to say they trust the Democratic Party over the Republican Party to address the cost of health care generally (34% vs. 16%) and prescription drugs (31% vs. 18%). However, more than four in 10 independent voters say they do not trust either party to do a better job handling either of these areas of affordability (44% and 41% respectively), suggesting that a substantial share of independents remain unconvinced that either party will deliver on these issues.
- Medicaid covers prescription drugs comprehensively, with enrollees paying little to nothing out of pocket. However, due to narrow exceptions, most states do not cover GLP-1 medications for weight loss. ↩︎
Methodology
This KFF Health Tracking Poll was designed and analyzed by public opinion researchers at KFF. The survey was conducted February 24 – March 2, 2026, online and by telephone among a nationally representative sample of 1,343 U.S. adults in English (n=1,268) and in Spanish (n=75). The sample includes 1,019 adults (n=62 in Spanish) reached through the SSRS Opinion Panel either online (n=995) or over the phone (n=24). The SSRS Opinion Panel is a nationally representative probability-based panel where panel members are recruited randomly in one of two ways: (a) Through invitations mailed to respondents randomly sampled from an Address-Based Sample (ABS) provided by Marketing Systems Groups (MSG) through the U.S. Postal Service’s Computerized Delivery Sequence (CDS); (b) from a dual-frame random digit dial (RDD) sample provided by MSG. For the online panel component, invitations were sent to panel members by email followed by up to three reminder emails.
Another 324 (n=13 in Spanish) adults were reached through random digit dial telephone sample of prepaid cell phone numbers obtained through MSG. Phone numbers used for the prepaid cell phone component were randomly generated from a cell phone sampling frame with disproportionate stratification aimed at reaching Hispanic and non-Hispanic Black respondents. Stratification was based on incidence of the race/ethnicity groups within each frame. Among this prepaid cell phone component, 142 were interviewed by phone and 182 were invited to the web survey via short message service (SMS).
Respondents in the prepaid cell phone sample who were interviewed by phone received a $15 incentive via a check received by mail or an electronic gift card incentive. Respondents in the prepaid cell phone sample reached via SMS received a $10 electronic gift card incentive. SSRS Opinion Panel respondents received a $5 electronic gift card incentive (some harder-to-reach groups received a $10 electronic gift card). In order to ensure data quality, cases were removed if they failed two or more quality checks: (1) attention check questions in the online version of the questionnaire, (2) had over 30% item non-response, or (3) had a length less than one quarter of the mean length by mode. Based on this criterion, 1 case was removed.
The combined cell phone and panel samples were weighted to match the sample’s demographics to the national U.S. adult population using data from the Census Bureau’s 2024 Current Population Survey (CPS), September 2023 Volunteering and Civic Life Supplement data from the CPS, and the 2025 KFF Benchmarking Survey with ABS and prepaid cell phone samples. The demographic variables included in weighting for the general population sample are gender, age, education, race/ethnicity, region, civic engagement, frequency of internet use and political party identification. The weights account for differences in the probability of selection for each sample type (prepaid cell phone and panel). This includes adjustment for the sample design and geographic stratification of the cell phone sample, within household probability of selection, and the design of the panel-recruitment procedure.
The margin of sampling error, including the design effect for the full sample, is plus or minus 3 percentage points. Numbers of respondents and margins of sampling error for key subgroups are shown in the table below. For results based on other subgroups, the margin of sampling error may be higher. Sample sizes and margins of sampling error for other subgroups are available on request. Sampling error is only one of many potential sources of error and there may be other unmeasured error in this or any other public opinion poll. KFF public opinion and survey research is a charter member of the Transparency Initiative of the American Association for Public Opinion Research.
| Group | N (unweighted) | M.O.S.E. |
|---|---|---|
| Total | 1,343 | ± 3 percentage points |
| Party ID | ||
| Democrats | 449 | ± 6 percentage points |
| Independents | 449 | ± 6 percentage points |
| Republicans | 373 | ± 6 percentage points |
| MAGA Republicans/Republican leaning independents | 334 | ± 6 percentage points |