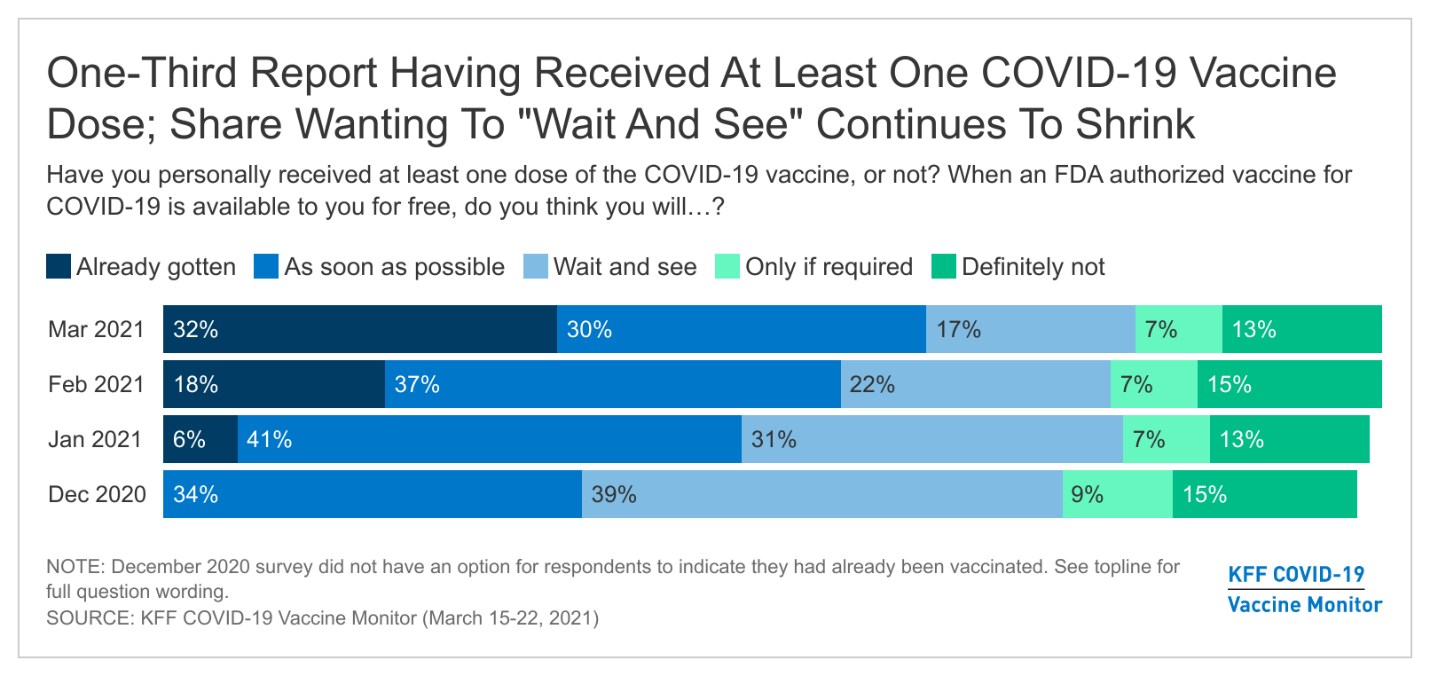
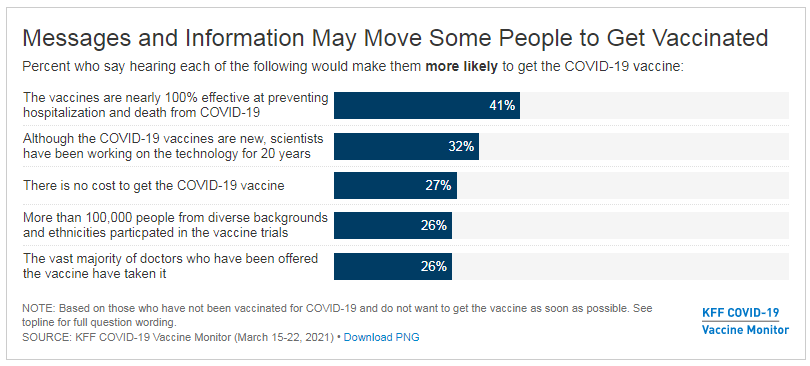
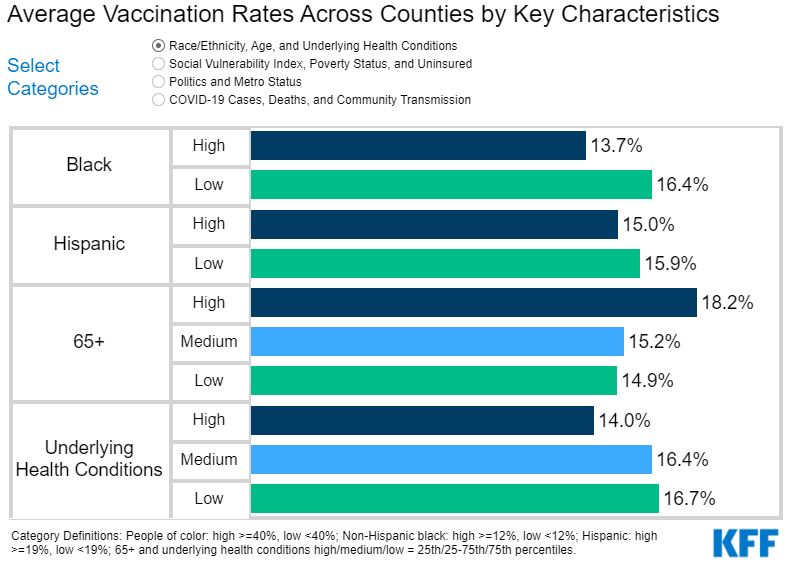
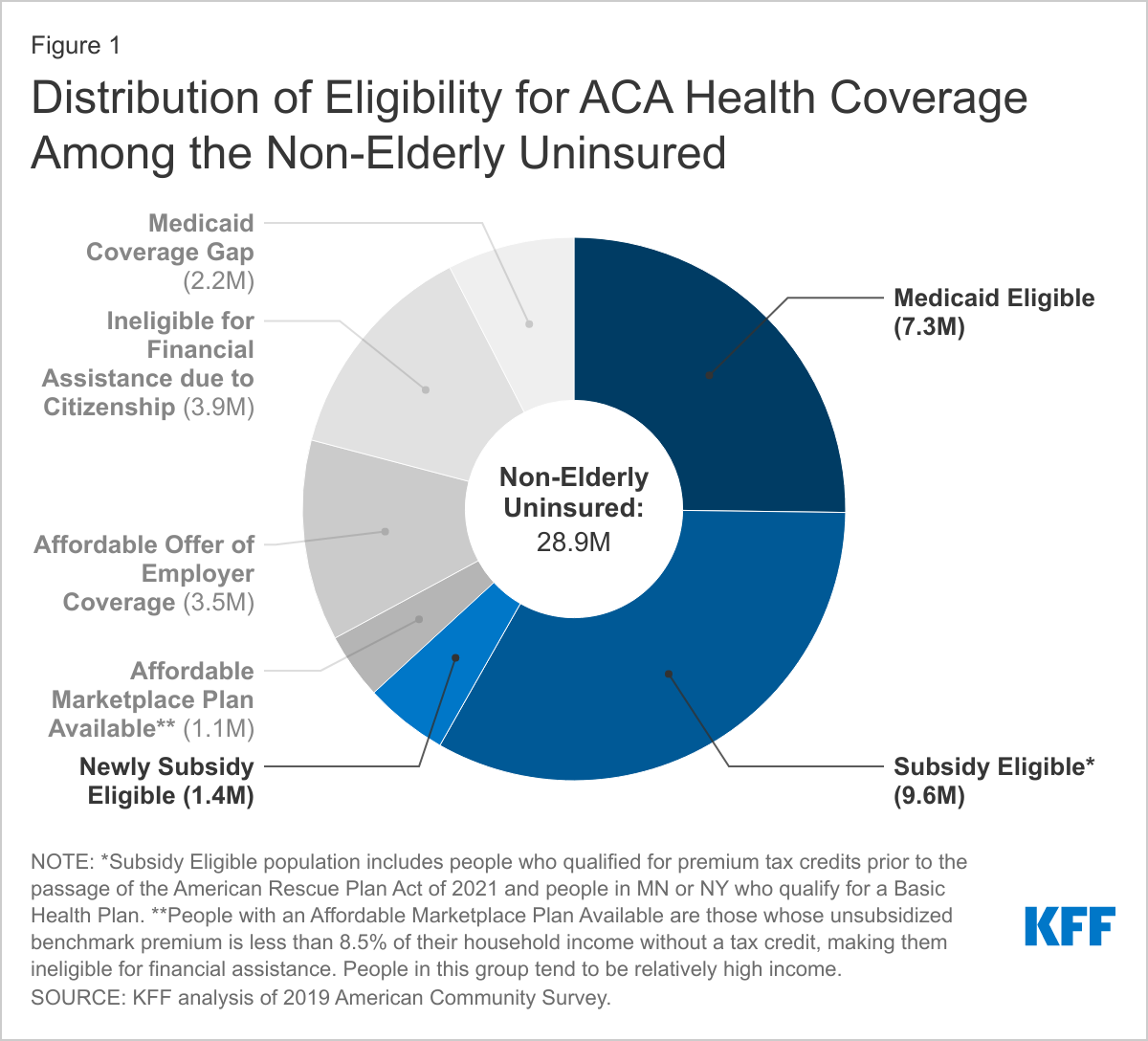
During the 2020 presidential campaign, President Biden supported several policies to lower prescription drug costs, including proposals to authorize the federal government to negotiate drug prices, cap out-of-pocket drug costs in Medicare Part D, and limit drug price increases to the rate of inflation. Whether or not the 117th Congress acts on these or other prescription drug proposals, the Administration has at its disposal a variety of administrative levers to address drug costs.
This brief focuses on two pathways through which the Biden Administration could use its executive authority to implement policy changes related to prescription drug costs. The first pathway would use the authority provided under the Affordable Care Act (ACA) through the Center for Medicare and Medicaid Innovation (CMMI, or Innovation Center) (established under Section 1115A of the Social Security Act). The Innovation Center is authorized to design, implement, and test new health care payment models to address concerns about rising costs, quality of care, and inefficient spending. The second pathway would use authority that was established soon after Medicare was enacted – known as Section 402 demonstration authority – to implement Medicare demonstrations to test new ways of delivering health care and paying health care providers.
We review examples of recent efforts to use these authorities to lower prescription drug costs, including the Trump Administration’s Most Favored Nation Model initiated through the Innovation Center and the Trump Administration’s proposed $200 prescription drug vouchers for Medicare Part D beneficiaries via Section 402 demonstration authority. We examine questions that have been raised regarding the use of these authorities and discuss how the Biden Administration might use them to implement prescription drug policies. In addition to these pathways, the Administration could employ other administrative tools to implement policy changes related to prescription drug costs, including through annual payment and policy rules, regulatory guidance, and waivers; those tools are not discussed further here.
Background
Since the early days of the Medicare program, the HHS Secretary has had the authority to initiate demonstrations that can lead to permanent changes in the Medicare program. The Secretary was first granted this authority under Section 402 of the Social Security Amendments of 1967, which was then modified by the Social Security Amendments of 1972. Section 402 provides broad authority to the Secretary to develop and implement demonstration projects to test new Medicare payment methodologies, and allows the Secretary to waive compliance with certain Medicare requirements relating to payment and reimbursement to conduct these demonstrations.1 In addition to demonstrations and models initiated by the Executive Branch, Congress has also mandated demonstration projects through legislation, either as part of more comprehensive legislation relating to the Medicare program or as part of appropriations legislation.
In the years leading up to the establishment of the Innovation Center, the Medicare Payment Advisory Commission (MedPAC) and other experts noted shortcomings with the demonstration process, including insufficient and declining funding, the long timeframe for research and evaluation of demonstration projects, administrative barriers including requirements under the Paperwork Reduction Act, implementation or expansion delays due to administrative and judicial review, and the condition of budget neutrality. Further, the process for scaling up successful models required Congressional action, which may delay or block their implementation. Additionally, the Centers for Medicare and Medicare Services (CMS) can be prevented from modifying or ending demonstration models based on early results (whether positive or negative) when models are specified in law.
Partly in response to these concerns, Congress established the CMS Innovation Center in the Affordable Care Act of 2010 (ACA). Congress specifically directed the Innovation Center to focus on models that could potentially lower health care spending for Medicare, Medicaid, and the Children’s Health Insurance Program (CHIP) while maintaining or enhancing the quality of care furnished under these programs. While the focus of the Innovation Center is on Medicare, Medicaid, and CHIP, Innovation Center models have also incorporated multi-payer and all-payer approaches that affect patients with private insurance to increase the potential impact of these models on the broader health system.
Through the Innovation Center, the ACA granted the HHS Secretary more tools to design, adapt, and test models that could produce savings and be more rapidly scaled. The Secretary has the ability to waive more regulations and has broader authority to expand and implement successful Innovation Center programs into Medicare, Medicaid, and CHIP, and terminate the models that fail. Further, the law allocated dedicated funding to the Innovation Center: $5 billion for fiscal year 2010, $10 billion for the years 2011 through 2019, and another $10 billion for each decade thereafter. These funds are not subject to annual appropriations and are designated for the operation of the Innovation Center, including direct support for individual models and delivery system reform initiatives, and operational expenses, including payroll expenses for full-time employees, contracts, and administrative expenses. Notably, Innovation Center models are not subject to the Paperwork Reduction Act, and there is no administrative or judicial review of the Secretary’s decisions for certain aspects of models (described in greater detail below). HHS also cannot require that an Innovation Center model initially be budget neutral.
With the passage of the ACA, the Innovation Center is the primary vehicle through which the Administration tests new models, even though Section 402 demonstration authority has existed much longer. More detailed discussion of these administrative pathways, plus examples of how they have been used to implement policy changes related to prescription drugs, is below.
Innovation Center Models
The ACA established the Innovation Center to test innovative payment and service delivery models that could lead to permanent program changes, with the goal of reducing program spending while preserving or enhancing the quality of care or improving quality of care without increasing spending. The law expanded the Secretary’s authority to waive Medicare and other program requirements in order to develop and conduct these models, and does not limit this authority to reimbursement or payment related changes. (This is in contrast to the Secretary’s waiver authority for Section 402 demonstrations, discussed more below.)
For the sole purpose of testing models, the Secretary may waive all requirements of Title XI of the Social Security Act (SSA) (General Provisions, Peer Review, and Administrative Simplification) and Title XVIII of the SSA (Medicare), three sections of Title XIX of the SSA relating to the Medicaid program, and nearly all sections of Title XIX of the SSA pertaining to the Program of All-Inclusive Care for the Elderly (PACE).2 The waiver authority for Title XI includes all sections, including fraud and abuse laws, such as the federal anti-kickback statute. The waiver authority for Title XVIII also includes all sections, including for example provisions related to Medicare eligibility, covered benefits, and payment rules, among others. However, provisions pertaining to Medicare that are not included in Title XVIII and not explicitly mentioned cannot be waived, such as the Medicare payroll tax of 1.45% on employers and employees, which is specified in the Internal Revenue Code.
Innovation Center Model Implementation and Expansion
Congress gave the Innovation Center more authority to initiate, terminate, modify, or expand models than what is allowed under Section 402 demonstration authority. As defined by statute, Innovation Center models are first implemented under Phase I, and if a model meets certain criteria, it can be expanded, including on a nationwide basis, under Phase II.
phase I
Phase I for Innovation Center models is the testing phase. The law directs the Secretary to select models for testing “where the Secretary determines that there is evidence that the model addresses a defined population for which there are deficits in care leading to poor clinical outcomes or potentially avoidable expenditures.” The law also mentions a list of possible models to be tested including but not limited to:
- Contracting directly with groups of providers of services and suppliers to promote innovative care delivery models, such as through risk-based comprehensive payment or salary-based payment.
- Promoting care coordination between providers of services and suppliers that transition health care providers away from fee-for-service based reimbursement and toward salary-based payment.
- Allowing States to test and evaluate systems of all-payer payment reform for the medical care of residents of the State, including dual eligible individuals.
The ACA prohibits the Secretary from requiring budget neutrality as a condition of testing during Phase 1 of the model. However, after testing of a model has begun in Phase I, the statute authorizes the Secretary to modify the design of a model or terminate a model unless it is expected to:
- improve quality of care without increasing spending under the applicable program (i.e., Medicare, Medicaid or CHIP);
- reduce program spending without reducing quality of care; or
- improve the quality of care and reduce program spending.
The statute also specifies that, following Phase I of the model, the Secretary shall conduct an evaluation of the model, which includes an analysis of the quality of care provided and changes in program spending attributed to the model.
phase ii
Phase II for Innovation Center models is the expansion phase. Under Phase II, taking into account the evaluation from Phase I, the Secretary may expand the duration and scope of a model, through rulemaking, including implementation on a nationwide basis. While not specifically required by statute, Phase I models that require mandatory participation have also typically been subject to the public rulemaking process.
In order for a model to be implemented nationwide, it must meet the following three criteria:
- The Secretary determines that such expansion of the model would reduce spending under the applicable program (i.e., Medicare, Medicaid or CHIP) without reducing the quality of care; or would improve the quality of care without increasing spending;
- The Chief Actuary of CMS certifies that such expansion would reduce (or would not result in any increase in) net program spending; and
- The Secretary determines that such expansion would not deny or limit the coverage or provision of benefits under the applicable program for beneficiaries.
To date, four Innovation Center models have met the statutory criteria to be certified by the Secretary for expansion. These four models are the Diabetes Prevention Program (DPP) model, the Home Health Value-Based Purchasing (HHVBP) Model, the Prior Authorization Model for Repetitive Scheduled Non-Emergent Ambulance Transport (RSNAT), and the Pioneer ACO model. These certifications enable the Secretary to expand the program to become a permanent part of Medicare, as the Secretary did for the Diabetes Prevention Program Model, which became a full preventive benefit in Medicare Part B for eligible beneficiaries (the “Medicare Diabetes Prevention Program”), effective April 2018.
In September 2020, CMS announced that the RSNAT model would be expanded nationwide, though the implementation of the expansion into new states is being delayed due to the COVID-19 pandemic. In January 2021, CMS announced its intent to expand the HHVBP model, and this expansion must be implemented through rulemaking to begin no earlier than January 1, 2022. The Secretary has not made the Pioneer ACO model a part of the full Medicare program.
In contrast to Section 402 demonstrations discussed below, the testing, evaluation and expansion of Innovation Center models are specifically exempted from the Paperwork Reduction Act, which can be time consuming and burdensome.3 Further, the statutory language notes that specific features of the model are not subject to administrative or judicial review:
- the selection of models for testing or expansion;
- the selection of organizations, sites, or participants to test those models selected;
- the elements, parameters, scope, and duration of such models for testing or dissemination;
- determinations regarding budget neutrality;
- the termination or modification of the design and implementation of a model;
- determinations about expansion of the duration and scope of a model.
Section 402 Demonstrations
Since the passage of the ACA, both the Obama and Trump Administrations relied more on the authority provided under the Innovation Center than on Section 402 demonstrations, with a few notable exceptions. Section 402 gives the Secretary authority to engage in demonstration projects to determine whether changes in methods of payment or reimbursement will have the effect of “increasing the efficiency and economy of health services” without affecting quality of care. Such demonstrations could include paying providers for services not otherwise covered by Medicare at the time of the demonstration or experimenting with changing the basis of provider payments.4 Section 402 allows the Secretary to waive requirements under Medicare and Medicaid, but only as long as they relate “to reimbursement or payment on the basis of reasonable cost, or (in the case of physicians) on the basis of reasonable charge.”
While budget neutrality is not specified in statute or regulation, according to CMS’ Medicare Waiver Demonstration Application, “Medicare-waiver-only demonstrations must be budget neutral. Budget neutrality means that the expected costs under the demonstration cannot be more than the expected costs were the demonstration not to occur.” Although most demonstrations have generally conformed to the administrative practice of budget neutrality, there have been exceptions, which are discussed in more detail below. Section 402 demonstrations also have typically been subject to certain types of review such as requirements under the Paperwork Reduction Act and administrative or judicial review.5 Under the Paperwork Reduction Act, agencies that plan to collect data on 10 or more individuals must submit information to the Office of Management and Budget (OMB) on proposed collection efforts as well as provide a 60-day period for public comment.6 Some Section 402 demonstrations, such as competitive bidding for clinical laboratory services and durable medical equipment, have also been subject to judicial review, a situation when the courts review various federal agencies’ actions, typically based on criteria included in the Administrative Procedure Act (APA).
Many of the demonstrations initiated by the Secretary under Section 402 and by Congress have ultimately led to permanent changes in Medicare law. For example, hospice demonstrations beginning in 1974 ultimately led to a hospice benefit becoming first authorized as part of Medicare in the Tax Equity and Fiscal Responsibility Act of 1982 (TEFRA). This benefit was originally scheduled to sunset in 1986 but then permanently added to Medicare benefits in the Consolidated Omnibus Budget Reconciliation Act of 1985. The inpatient prospective payment system (IPPS), which is how Medicare pays hospitals for acute inpatient stays, also had its origins in various demonstration projects. Starting in 1974, the Health Care Financing Administration (a precursor to CMS) engaged in various demonstrations that led to the design of a new Medicare prospective payment system, using a system of diagnosis-related groups (DRGs), which was enacted as part of the Social Security Amendments of 1983. Other demonstration projects that have been expanded program-wide include the skilled nursing facility and home health prospective payment systems; competitive bidding for durable medical equipment; and Medicare coverage for heart transplants.7
While the funding of some demonstrations has been Congressionally earmarked, CMS has a research, evaluations, and demonstrations (RDE) budget that is appropriated annually. In FY 2020, $20.1 million was appropriated for these activities, significantly less than the funding allocated to the Innovation Center, which averages $1 billion per year. Funding appropriated to the RDE generally fluctuates from year to year, but has been declining over time, particularly after the establishment of the Innovation Center, from $57.4 million in FY 2007 to $35.6 million in FY 2010 and $20.1 million in FY 2020. The RDE section of the budget notes that “CMS leverages other funding sources, such as ACA 3021 (Innovation Center) funding, to support RDE projects wherever possible.”
How Have Innovation Center Models and Section 402 Demonstration Authority Been Used to Address Prescription Drug Costs?
Innovation Center: The Trump Administration leveraged the flexibility and authority of the Innovation Center to address prescription drugs at least three times. Two of the models are ongoing: the Part D Senior Savings model, which limits out-of-pocket costs on insulin in participating Part D plans, and the Part D Payment Modernization Model, which tests the impact of a revised Part D payment structure that creates incentives for plans, patients, and providers to choose drugs with lower list prices in order to better manage spending in the catastrophic phase. This Part D Payment Modernization Model was updated by the Trump Administration for calendar year 2022 to waive the requirement that all drugs in five of six protected classes (all protected class drugs except antiretrovirals) be included in formularies, the requirement that plans cover a minimum of two drugs in other therapeutic classes, and the requirement that sponsors take on downside risk. However, the Biden Administration recently announced that they are not moving forward with these changes. Sponsors may not modify coverage of drugs in protected classes, they must continue to cover a minimum of two drugs in other therapeutic classes, and sponsors must continue to take on two-sided risk to participate in the model. To date, only two Part D plans are participating in this model.
The third model is the Most Favored Nation model, a mandatory nationwide model designed to lower payments for the top 50 Part B drugs that contribute to high Part B spending, by aligning payment for Part B drugs more closely to what other countries pay and removing incentives for doctors to prescribe higher-priced drugs. The model was set to go into effect January 2021, but stakeholders filed lawsuits to block the model from being implemented, with one of the primary complaints being that the Administration violated the APA by not following the appropriate rulemaking process. HHS has been temporarily blocked from implementing this rule under a preliminary injunction issued by the U.S. District Court for the Northern District of California, based on its ruling that the Trump Administration failed to follow the standard notice and comment rulemaking procedures. According to the Innovation Center, the Biden Administration will not implement this rule without further rulemaking.8
While the Obama Administration frequently used Innovation Center authority, such as for Accountable Care Organizations (ACO), bundled payment models, and in collaboration with states for all-payer ACO models such as in Vermont and Maryland, it did not do so as often relating to prescription drug costs.
In March 2016, the Obama Administration issued a proposed rule for a Part B Drug Payment Model to be implemented through the Innovation Center – although this model was ultimately withdrawn prior to implementation due to criticisms of the mandatory nature of the proposed model and opposition from patient and physician groups. The model offered alternative payment designs for Part B drugs to test whether these approaches would “strengthen the financial incentive for physicians to choose higher value drugs.” Under current law, for most Part B drugs, providers are reimbursed based on the average sales price (ASP) of a given drug plus a 6 percent add-on payment. The first phase of the model would have assigned certain providers an alternative payment approach of ASP plus 2.5 percent, plus a flat fee. The second phase of the model would have tested the application of value-based purchasing tools, in addition to the alternative ASP add-on payments. To eliminate selection bias and test whether these alternative payment approaches would change physician behavior and achieve savings, all providers and suppliers in selected geographic areas would have been required to participate.
While not solely a model pertaining to prescription drugs, the Obama Administration initiated the Medicare Advantage Value-Based Insurance Design (VBID) Model (though the model went into effect during the Trump Administration). Among the variety of payment and delivery approaches the model tests, the model provides Medicare Advantage plans with flexibilities, targeted to Medicare beneficiaries based on chronic conditions, including allowing reductions in cost-sharing for Medicare Part D covered drugs. For 2021, 19 sponsors are participating in this model.
The Obama Administration also initiated the Part D Enhanced Medication Therapy Management Model through the Innovation Center (though the model went into effect during the Trump Administration), which seeks to provide basic, stand-alone prescription drug plans with flexibility to implement innovative medication therapy management programs to optimize medication use. This model is ongoing.
Section 402: The Trump Administration proposed to use Section 402 demonstration authority relating to prescription drugs in order to send $200 prescription drug cards to Medicare Part D beneficiaries, though this proposal was not finalized. According to a draft document, the demonstration was designed to determine whether these $200 drug cards, which would help cover out-of-pocket costs for beneficiaries’ prescription drugs, would improve medication adherence, health outcomes, and improve the efficiency of Medicare payments. Questions pertaining to the design and cost of this proposed demonstration are discussed in more detail in the section below.
One of the Trump Administration’s stated rationales for using this demonstration authority for the proposed drug cards (rather than the Innovation Center) is that CMS has previously used Section 402 authority to test other changes to Part D, citing the Part D Low-Income Newly Eligible Transition Program and the Part D Reinsurance Payment Demonstration (Appendix Table 1). These demonstrations were initiated by the Bush Administration in the years after the enactment of the Medicare Modernization Act of 2003, which created the Part D program for prescription drugs, and before the Innovation Center was established. The Part D Low-Income Newly Eligible Transition Program adjusted weighting for regional benchmarks to ensure low-income Medicare beneficiaries had access to affordable plan choices and increase incentives for them to enroll in low-cost plans. The Part D Reinsurance Payment Demonstration was designed to encourage Part D sponsors to offer enhanced benefit packages by offering an alternative method for reinsurance financing, including paying organizations up-front capitated payments. While the Obama Administration did not use Section 402 demonstration authority relating to prescription drugs, it did use this authority to implement the Medicare Advantage Quality Bonus Payment demonstration, which is discussed below.
Questions Raised About the Use of Innovation Center Models and Section 402 Demonstration Authority
Over the years, the Government Accountability Office (GAO), the HHS Office of Inspector General (OIG), members of Congress, and other stakeholders have raised questions with respect to models initiated through the Innovation Center and demonstrations conducted under Section 402 authority.
Can models require mandatory participation?
Some stakeholders have questioned whether Innovation Center models are allowed to be mandatory, although the ACA does not specify that models must be voluntary. Part of the reason mandatory models have been proposed is to ensure that a sufficient number of providers participate in both the research and control groups, and minimize the risk of selection bias. In 2016, over 170 members of Congress stated in a letter to CMS that the Innovation Center was overstepping its authority by making models mandatory, mentioning the Comprehensive Care for Joint Replacement (CJR) model, and the proposed Part B Drug Payment Model discussed above, among others. The CJR model was initially mandatory for all hospitals in 67 designated areas of the country, but the Trump Administration pared back the mandatory hospital participation requirement, effective January 2018. However, in the final rule that made participation in the CJR model voluntary, CMS responded to commenters who contended that CMS lacks the authority to mandate participation in Innovation Center models, by affirming it does have this authority, stating that:
“We disagree that the Innovation Center lacks the authority to test mandatory models under section 1115A of the Act. Section 1115A of the Act authorizes the Secretary to test innovative payment and service delivery models to reduce program expenditures while preserving or enhancing the quality of care furnished to Medicare, Medicaid, and Children’s Health Insurance Program (CHIP) beneficiaries. Section 1115A of the Act does not specify that participation in models must be voluntary. Moreover, the Secretary has authority to establish regulations to carry out the administration of Medicare. Specifically, the Secretary has authority under both sections 1102 and 1871 of the Act to implement regulations as necessary to administer Medicare, including testing these Medicare payment and service delivery models.”
Are models subject to an adequate rulemaking process?
Some members of Congress have expressed concern with the rulemaking process for Innovation Center models. In a January 2019 letter to CMS, Ways and Means Chairman Richard Neal and Ranking Member Kevin Brady argued that the Innovation Center process has been historically opaque and does not use the traditional rulemaking cycle, particularly for models that are not mandatory. Further, in the 116th Congress, Representative Sewell sponsored H.R.5741, “The Strengthening Innovation in Medicare and Medicaid Act,” which seeks to address these and other concerns, such as requiring more advance public notice and opportunities for stakeholder input, limiting the scope of models to no more applicable individuals than necessary to obtain a statistically valid sample, and establishing judicial review of some aspects of models, including review of the elements, parameters, scope, and duration of such models for testing or dissemination as well as determinations about expansion of the duration and scope of a model.
Concerns over the rulemaking process were one of the main criticisms of the Trump Administration’s Most Favored Nation model, which was finalized in an interim final rule, meaning it can be effective immediately without public comment. This model was set to go into effect January 1, 2021, but stakeholders including PhRMA, BIO, and others, filed lawsuits to block the model from being implemented, with one of the primary complaints being that the Administration violated the APA by not following the appropriate rulemaking process. Generally, the APA requires agencies to publish notices of rulemaking in the Federal Register, providing at least 60 days for public comment before going into effect. The Trump Administration attempted to make a case for a “good cause” exception, which allows them to waive the notice and comment requirements under the APA, citing the “particularly acute need for affordable Medicare Part B drugs now, in the midst of the COVID-19 pandemic.” However, several U.S. district courts agreed with the reasoning in the complaints and have blocked implementation, including a preliminary injunction by the U.S. District Court for the Northern District of California based on its ruling that the Trump Administration failed to follow the standard notice and comment rulemaking procedures.9 According to the Innovation Center, this rule will not be implemented without further rulemaking.
Is the demonstration designed as an experiment, with a comparison group to test for significant outcomes?
One question that has been raised in relation to demonstrations is whether the demonstration has an adequate comparison group. This matter was recently highlighted with the Trump Administration’s proposed $200 prescription drug cards. According to the draft document, the demonstration would test the impact of lower out-of-pocket drug costs on medication adherence and discontinuation rates, as well as downstream effects on health care utilization and Medicare Part A and B spending. However, many experts have questioned the experimental design of the demonstration. Because every Medicare Part D beneficiary who does not receive the most generous level of low-income subsides would be eligible for the card, there would be no comparison group. Critics also argue that the “test” the demonstration seeks to carry out has already been well established, with many studies showing that lower cost sharing for drugs improves medication adherence.
Will the demonstration achieve savings or increase costs?
The high cost of demonstrations has also been a concern, particularly when they are not designed to achieve savings. While most demonstrations conducted under Section 402 authority are generally required to be budget neutral by OMB, this has not been true of all demonstrations. This issue was raised in regard to the Trump Administration’s proposed $200 prescription drug cards. The drug cards were intended to lower the cost of medications, which could improve medication adherence and reduce unnecessary health care utilization, ultimately lowering Medicare Part A and B spending over time. However, the draft document acknowledges that actually observing such reductions would be difficult given the short duration of the demonstration, and estimated that the cost of the demonstration would be $7.9 billion dollars without any estimate of potential savings or expectation of budget neutrality. Many stakeholders were critical of the cost of the plan, with members of Congress sending letters to HHS and GAO seeking to review the proposal’s legality, including a determination of whether the discount card would have “the effect of increasing the efficiency and economy of Medicare” – a condition of section 402 demonstrations.
Though it was not intended to address to prescription drug policy, questions about the cost a demonstration were also raised when the Obama Administration initiated the Medicare Advantage (MA) Quality Bonus Payment demonstration that was in effect from 2012 to 2014. The HHS Secretary initiated this demonstration under Section 402 authority to test an alternative method for calculating and awarding MA bonuses, rather than implementing the bonus structure set forth in the ACA. The Office of the Actuary (OACT) estimated the cost to be over $8 billion dollars.
GAO evaluated the extent to which this demonstration conformed to principles of budget neutrality, relative to other Medicare demonstrations, and recommended canceling the demonstration due in large part to its high cost. The GAO concluded that this demonstration “dwarfs all other Medicare demonstrations—both mandatory and discretionary—conducted since 1995 in its estimated budgetary impact and is larger in size and scope than many of them.”
How Could the Biden Administration Use Executive Authority to Take Action on Prescription Drug Costs?
Having narrow Democratic majorities in both the House and the Senate may give the Biden Administration somewhat more room to implement pieces of its health policy agenda through legislation. But the Biden Administration may also consider executive action, potentially relying on the Innovation Center or Section 402 demonstration authority to pursue health care changes, including changes related to prescription drugs.
Several proposals supported by President Biden during his campaign would lower drug costs for Medicare and private insurers. The Innovation Center has authority and has tested multi-payer or all-payer models that include private insurers in the past (e.g., delivering primary care or providing coordinated oncology treatments). However, it may be somewhat easier to test models that apply exclusively to Medicare in the case of prescription drugs, because the Innovation Center has less leverage outside of the Medicare program to enforce lower payments. For example, the negotiation provision under H.R. 3, the drug price legislation passed by the House of Representatives in December 2019, applied to both Medicare and private insurance, enforced by financial penalties on drug manufacturers that did not agree to participate in the negotiation process. However, an Innovation Center model would not have the same leverage to reach all private insurers, and in particular, would not be able to compel drug manufacturers to offer the lower, negotiated Medicare drug price to private insurers. The Innovation Center would need to identify a new mechanism to apply lower drug prices more broadly to private insurers.
During his campaign, President Biden supported allowing the federal government to negotiate drug prices in Medicare Part D and for other payers as well as “directing the Medicare program to target excessively priced prescription drugs that face little or no competition.” To pursue this goal in the absence of Congressional action, the Biden Administration could, for example, consider initiating a model designed to test whether the federal government could achieve savings, without adversely affecting quality, by negotiating prices under Medicare for high-priced drugs that lack competition. According to CBO’s analyses of previous drug negotiation legislative proposals, CBO expects that Medicare savings would be achieved from negotiations over high-priced drugs that lack competition, which would lead to lower beneficiary premiums and cost sharing. CBO also noted that this new authority would lower revenues for some drug manufacturers, and (depending on design details) could lead to higher drug prices in other countries and a reduction of about 8 drugs coming to market from the 2020 to 2029 period and about 30 fewer drugs over the subsequent decade, due to the loss in revenue for drug manufacturers. However, CBO did not analyze the potential effects of this foregone innovation on public health.
The Biden campaign supported a proposal to “empower the HHS Secretary to negotiate prices that are capped to a level associated with average OECD median prices.” Depending on whether or not the Biden Administration moves forward in some fashion with the Trump Administration’s Most Favored Nation Model that is currently blocked in the courts, the Administration could design a new model to test the use of international reference prices for setting prices for certain Medicare-covered drugs. If similar to the Trump Administration’s model, Medicare’s actuaries estimated $85.5 billion in savings over the course of the 7-year demonstration. However, the pharmaceutical industry and others have raised concerns about the potential for this approach to adversely affect patients’ access to medications and to lead to higher prices in other countries.
During the campaign, President Biden also supported an approach that would limit drug price increases to the rate of inflation. In the absence of legislation, the Administration could consider initiating such a model through the Innovation Center, designed to test whether limiting price increases for certain Medicare-covered drugs to the rate of inflation, such as drugs that have historically high increases year over year, reduces spending for the Medicare program. CBO analyses of similar proposals lawmakers introduced during the 116th Congress for Medicare Part B and Part D drugs indicate the potential for significant savings if drug manufacturers limited price increases to the rate of inflation or paid a rebate to the federal government. Given concerns that inflation caps could result in higher launch prices that would affect Medicare spending and have spillover effects on costs incurred by other payers, the Innovation Center would need to assess in advance of launching the new model whether this model would be expected to achieve net savings for the program (without adversely affecting quality).
This is by no means an exhaustive list of ideas to address prescription drug costs, and Innovation Center and Section 402 demonstration authority are not the only regulatory vehicles for implementing changes to prescription drug policies. Previous administrations have also implemented changes to prescription drug policies through the annual rulemaking process, such as changes to formulary requirements, as in a recent final rule that allows Medicare Part D plans to establish a second specialty tier, and proposed (but not finalized) modifications to the treatment of protected classes in Part D. While the Administration can use annual payment and policy rules to implement programmatic changes, these changes typically are not major shifts in policy as they are not allowed to waive existing Medicare payment rules and statutory requirements using these mechanisms (unlike under demonstration and Innovation Center authority).
KFF polling from December 2020 shows that there is strong bipartisan support for some of President Biden’s health care proposals, including 89% who favor allowing the federal government to negotiate with drug companies to get a lower price on medications that would apply to both Medicare and private insurance – a proposal that would require legislation, such as the bill (H.R. 3) that passed the House of Representatives in the last Congress. While addressing the COVID-19 crisis is clearly the first order of business for the Biden Administration, there will likely be continued pressure to address high prescription drug prices and rising drug costs, and some of these policies could potentially be carried out via the Innovation Center or Section 402 demonstration authority.
This work was supported in part by Arnold Ventures. We value our funders. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.