Key Questions About Nursing Home Regulation and Oversight in the Wake of COVID-19
Issue Brief
Key Takeaways
The COVID-19 pandemic has led to renewed interest among policymakers, the media, residents, and their families in nursing home regulation and oversight, as residents and staff are at increased risk of infection due to the highly transmissible nature of the coronavirus, the congregate nature of facility settings, and the close contact that many workers have with patients. Certification of nursing home compliance with federal Medicare and/or Medicaid requirements generally is performed by states through regular inspections known as surveys. Federal regulations issued in 2016 require facilities to have an infection control and prevention program and a written emergency preparedness plan. This issue brief answers key questions about nursing home oversight and explains how federal policy has changed in light of COVID-19. Key findings include:
- Infection control deficiencies were widespread and persistent in nursing homes prior to the COVID-19 pandemic, according to a May 2020 GAO report.
- Despite widespread issues found in more comprehensive surveys before the pandemic, preliminary reports of targeted nursing home surveys focused on infection control and immediate jeopardy since March 4, 2020 reveal that only a small share have deficiencies. However, the pandemic has brought renewed attention to nursing home quality issues.
- While the pandemic has led to new federal guidance, funding, and reporting requirements, state survey agencies and nursing homes each may face issues related to funding, capacity, and data as the pandemic continues.
Introduction
There has been sustained attention on nursing homes in the wake of COVID-19, from the initial widespread outbreak at a facility in Washington State to the disproportionate number of cases and deaths among residents and staff nationally throughout the pandemic. Nursing home residents include 1.2 million seniors and nonelderly people with disabilities living in over 15,000 facilities. These residents and the 3 million people who work in skilled nursing or residential care facilities are at increased risk of infection due to the highly transmissible nature of the coronavirus, combined with the congregate nature of facility settings and the close contact that many workers have with patients. If infected, many residents are at increased risk of adverse health outcomes and death from COVID-19, due to old age and/or underlying chronic health conditions. Over 40% of all COVID-19 deaths have been residents or staff of long-term care facilities, with even higher numbers in some states. At the pandemic’s outset, nursing home oversight and response was concentrated at the state level, which led to different protocols across states and facilities, with little federal involvement and substantial shortages of personal protective equipment and coronavirus tests.
As a result of the pandemic’s impact, policymakers, residents, staff, and others have raised questions about whether there has been sufficient federal and state oversight of and guidance for nursing homes, and CMS has released a series of guidance and other policy actions. The Social Security Act authorizes the Health and Human Services (HHS) Secretary to establish requirements relating to nursing home residents’ health, safety and well-being as conditions for facilities to receive payment from the Medicare and Medicaid programs. Nursing homes can be certified as Medicare skilled nursing facilities (SNFs)1 and/or Medicaid nursing facilities (NFs).2 Medicare is the primary payer for about 12 percent of nursing home residents, with coverage limited to short-term stays for skilled nursing care or rehabilitation. Medicaid is the primary payer for 62 percent of nursing home residents, covering both short-term skilled nursing care and rehabilitation, as well as long-term care. This brief answers key questions about nursing home oversight and explains how federal policy has changed in light of COVID-19.
1. How are the federal requirements for nursing home oversight enforced?
Certification of nursing home compliance with federal Medicare and/or Medicaid requirements generally is performed by states through regular inspections known as surveys.3 States receive 75% federal matching funds for Medicaid nursing facility survey and certification activities,4 while Medicare SNF survey and certification activities are funded by a discretionary appropriation. Appendix 1 explains the survey process. States also must investigate complaints of facility violations of federal requirements5 and allegations of abuse, neglect, and misappropriation of resident property by nurse aides or other facility service providers6 and conduct periodic educational programs for facility staff and residents about current regulations, policies, and procedures.7
The penalties for facilities found to be out of compliance with federal requirements vary depending on whether the deficiency is determined to immediately jeopardize residents’ health or safety.8 For deficiencies that result in immediate jeopardy, a facility is subject to the appointment of temporary management to oversee operations while deficiencies are corrected or termination from the Medicare and/or Medicaid programs with the safe and orderly transition of residents to another facility or community setting.9 However, a facility may continue to receive Medicare and/or Medicaid payments for up to six months after a deficiency finding, if the state finds that this alternative is more appropriate than program termination. In these instances, the facility must agree to repay Medicare funds, and the state must agree to repay federal Medicaid funds, if corrective action is not taken according to a Secretary-approved plan and timetable. For deficiencies that do not result in immediate jeopardy, a facility may be allowed up to six months to correct deficiencies. A facility that does not come into substantial compliance within three months is subject to denial of Medicare and/or Medicaid payment for all individuals admitted after the deficiency finding date. A facility that is not in substantial compliance within six months is subject to Medicare program termination and discontinuance of Medicaid federal financial participation.
Civil money penalties (CMPs) can be imposed for the number of days a facility is not in substantial compliance or for each instance that a facility is not in substantial compliance.10 CMPs can range from $6,525 to $21,393 for deficiencies constituting immediate jeopardy, and from $107 to $6,417 for deficiencies that do not constitute immediate jeopardy but either caused actual harm or did not cause actual harm but have the potential for more than minimal harm. Per instance CMPs range from $2,140 to $21,393. A portion of CMP funds collected are returned to states and can be used for activities that protect or improve care quality or resident quality of life as approved by CMS, such as supporting and protecting residents during facility closures, relocating residents, supporting resident and family councils and other consumer involvement, facility improvement initiatives such as staff and surveyor training or technical assistance for quality assurance and performance improvement programs, or developing and maintaining temporary manager capability such as recruitment, training, or system infrastructure expenses.
Additional remedies apply to facilities with patterns of deficiencies over time. A facility that is found to provide substandard care quality on three consecutive standard surveys is subject to denial of Medicare and Medicaid payments for all new admissions or entirely and is subject to state monitoring until the facility demonstrates that it has regained and will remain in substantial compliance.11 CMS and/or the state also may direct staff in-service training for facilities with patterns of deficiencies.
2. How have the federal nursing home requirements evolved over time?
Prior to the late 1960s, nursing homes were “essentially unregulated in most states,” and care quality was generally considered to be poor.12 The creation of Medicare and Medicaid in 1965 led to greater federal involvement in nursing home regulation with the establishment of federal criteria to certify facilities. However, concern about care quality and inadequate enforcement continued through the 1970s and 1980s, leading to the appointment of an Institute of Medicine (IOM) committee to recommend changes. The IOM recommendations included incorporating resident assessment, care quality, quality of life, and residents’ rights in federal conditions of participation and making both regulatory requirements and the survey and certification process more “resident-centered and outcome-oriented,” with a shift in emphasis “from facility capability to facility performance.”13 The IOM report also recommended increased federal funding and oversight of state survey agencies and establishing intermediate sanctions short of program termination and facility closure to enforce compliance. Appendix 2 contains additional background about the evolution of federal nursing home oversight.
The IOM recommendations led to changes adopted in the 1987 Nursing Home Reform Act, which established Medicare SNF and Medicaid NF requirements in three main areas: service provision, residents’ rights, and administration and other matters (Appendix Table 1). The Nursing Home Reform Act strengthened federal standards, inspections, and enforcement provisions; merged Medicare and Medicaid standards; required comprehensive resident assessments; set minimal licensed nursing staff requirements; and required inspections to focus on care outcomes.14 Service provision requirements are related to quality of life, care plans, resident assessment, services and activities, nurse aide training, physician supervision and clinical records, social services, and nurse staffing information.15 Facilities must protect and promote specific residents’ rights including free choice, freedom from restraints, privacy, confidentiality, accommodation of needs, grievances, participation in resident and family groups and other activities, examination of survey results, and refusal of certain transfers. Additional requirements govern the use of psychopharmacologic drugs, advance directives, access and visitation, equal access to quality care, admissions, and protection of resident funds. Administration requirements include licensing and life safety code, sanitary and infection control and physical environment standards.
To help address continuing quality concerns, the 2010 Affordable Care Act (ACA) included some additional reforms. The Nursing Home Transparency and Improvement Act, adopted as part of the ACA, sought to address complex ownership, management, and financing structures that inhibited regulators’ ability to hold providers accountable for compliance with federal requirements. The ACA also incorporated the Elder Justice Act and the Patient Safety and Abuse Prevention Act, which include provisions to protect long-term care recipients from abuse and other crimes.
The 2016 nursing home regulations issued by the Obama Administration were the first comprehensive update in 25 years. The original consolidated Medicare and Medicaid facility participation requirements were issued in 1989, following the Nursing Home Reform Act, and revised in 1991. The 2016 regulations sought to account for ensuing innovations in resident care and quality assessment and an increasingly diverse and clinically complex resident population.16 New requirements added by the 2016 regulations most relevant to issues raised by COVID-19 include those related to infection control, facility assessment, and emergency preparedness (Box 1). The 2016 regulations also revised provisions related to resident rights, adopting a greater emphasis on person-centered care; reporting of abuse and neglect; and transfer and discharge rights. Additionally, the 2016 regulations added a new section on behavioral health services, adopted a competency requirement for determining staffing sufficiency and new staff training program requirements, and implemented ACA requirements for facility quality assurance and performance improvement programs17 and compliance and ethics programs.18 The regulations were implemented in three phases from 2016 through 2019.
Box 1: 2016 Nursing Facility Regulations Relevant to COVID-19 Pandemic
The 2016 regulations require facilities to establish an infection prevention and control program to provide a safe, sanitary, and comfortable environment and help prevent the development and transmission of communicable diseases. The program must include a system for preventing, identifying, reporting, investigating, and controlling infections and communicable diseases for all residents, staff, volunteers, visitors, and other individuals providing services under a facility contract. The program is based on the facility assessment (described below) and must follow “accepted national standards.” The program must include written standards, policies, and procedures for a surveillance system to identify possible communicable diseases before they can spread to others in the facility; when and to whom possible incidents should be reported; standard and transmission-based precautions to prevent infection spread; when and how isolation should be used for a resident; the circumstances under which the facility must prohibit employees with a communicable disease from direct contact with residents or their food; and hand hygiene procedures. The facility also must designate at least one infection preventionist responsible for the program. The program must be reviewed annually, and the facility must provide staff training on the program.
Facilities must conduct an assessment to determine what resources are necessary to care competently for residents during both regular day-to-day operations and emergencies. The facility assessment must consider the resident population; necessary staff competencies; the physical environment, equipment and services necessary to provide care; any ethnic cultural or religious factors that may affect care; facility resources such as buildings and equipment; services such as physical therapy, pharmacy, and specialized rehabilitation; personnel; contracts with third parties; and health information technology. The assessment must be updated as needed and at least annually.
Facilities must have a written emergency preparedness plan. This provision was issued in a separate 2016 provider emergency preparedness regulation.19 The plan must be reviewed and updated at least annually. Facilities must train all employees in emergency procedures when they begin to work in facility and periodically review procedures with existing staff. Facilities also must have an emergency preparedness communication plan including a means of providing information about the general condition and location of residents under the facility’s care and a method for sharing appropriate information from the emergency plan with residents and their families.
In July 2019, the Trump Administration proposed a number of changes to the 2016 regulations, which it said would increase provider flexibility and reduce regulatory burden. The proposed changes, which are still pending, include:
- Removing the existing requirement that the infection preventionist work at the facility at least part-time or have frequent contact with infection prevention and control program staff and instead requiring that the infection preventionist has “sufficient time” at facility to meet program objectives; and
- Modifying current provisions governing the use of psychotropic medications, the grievance process, the timeframe for retaining staffing data, the quality assurance and performance improvement program, and the compliance and ethics program, among other areas.
Also in July 2019, the Trump Administration issued a final regulation that eliminated the existing ban on pre-dispute arbitration agreements.
3. What was the state of nursing home quality before and during COVID-19?
Infection control deficiencies were widespread and persistent in nursing homes prior to the COVID-19 pandemic, according to a May 2020 GAO report.20 The GAO found that “infection prevention and control deficiencies were the most common type of deficiency cited in surveyed nursing homes, with most nursing homes having an infection prevention and control deficiency cited in one or more years from 2013 through 2017 (13,299 nursing homes, or 82 percent of all surveyed homes).” All states had facilities with infection prevention and control deficiencies cited in multiple consecutive years, indicating “persistent problems.” These deficiencies include staff failing to regularly use proper hand hygiene or failing to implement preventive measures to control infection spread during an outbreak, such as isolating sick residents and using masks and other personal protective equipment (PPE). GAO also found that nearly all infection prevention and control deficiencies were classified by surveyors as not severe, meaning the surveyor determined that residents were not harmed, and implemented enforcement actions for these deficiencies were typically rare. GAO plans future reports to examine CMS guidance and oversight of infection control more broadly as well as CMS’s response to COVID-19.
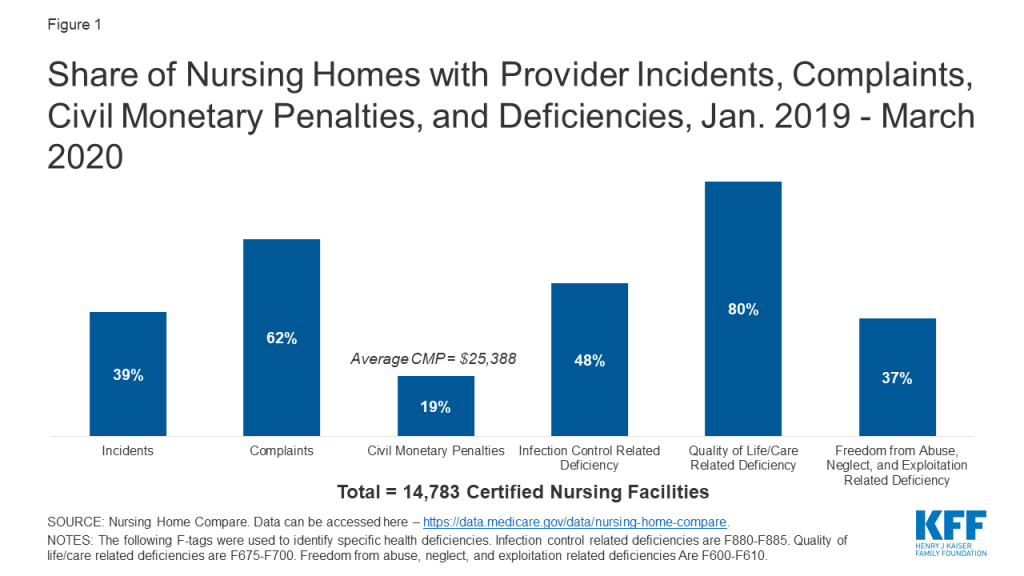
In regular surveys conducted from January 2019 through March 2020, nearly half of facilities received an infection control deficiency (Figure 1). Most facilities (80%) received a deficiency related to resident quality of life or care, and 37% received an abuse/neglect/exploitation deficiency (Figure 1). Just under 20% of facilities received a CMP, with the average amount just over $25,000 (Figure 1). During this same time period, 62% of facilities had substantiated complaints regarding violations of federal requirements, and 39% of facilities had incidents with alleged or suspected resident abuse, neglect, or misappropriation of property (Figure 1).
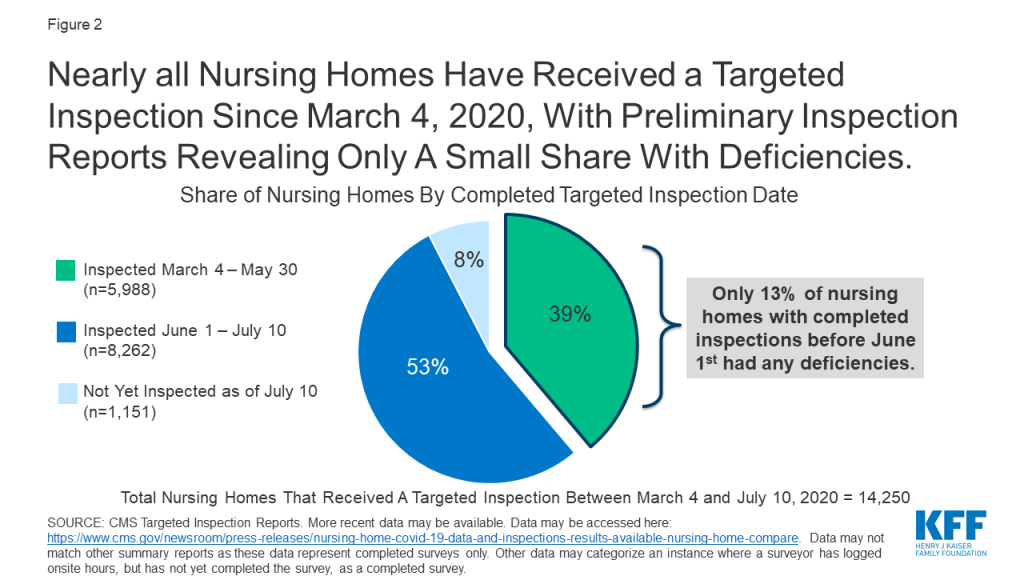
In response to COVID-19, CMS suspended state survey activities in March 2020, except for those related to infection control and immediate jeopardy. Nearly all nursing homes have received these targeted surveys since March 4, 2020, with preliminary inspection reports revealing only a small share with deficiencies (Figure 2). Just 13% of the nearly 6,000 facilities surveyed between March 4 and May 30, 2020 were cited as deficient in meeting any federal requirements. Though nursing homes across the country have experienced high rates of COVID-19 cases and deaths, the data does not point to quality deficiencies as a reason for this occurrence.
4. How has nursing home oversight changed in light of COVID-19?
As the number of COVID-19 cases and deaths in nursing homes increased, CMS has issued guidance about how facilities should respond to the pandemic (Figure 3). A February 2020 informational bulletin advised health care facilities to review the Centers for Disease Control (CDC) COVID-19 advisory and recommendations as well as their own infection control policies. As noted above, CMS suspended state survey activities in March 2020, except for those related to infection control and immediate jeopardy. That same month, CMS infection control and prevention guidance advised facilities to screen visitors and staff and about when to transfer residents to and accept those discharged from hospitals. CMS also required facilities to restrict all visitors except for compassionate care circumstances and cancel all communal dining and group activities, released guidance allowing facilities to perform COVID-19 tests, and issued a number of Section 1135 blanket waivers to help facilities’ emergency response. In April 2020, CMS issued guidance directing facilities to screen all staff, residents, and visitors for symptoms, ensure staff use PPE “to the extent available,” and designate separate staff and facilities or units for COVID-19 patients.
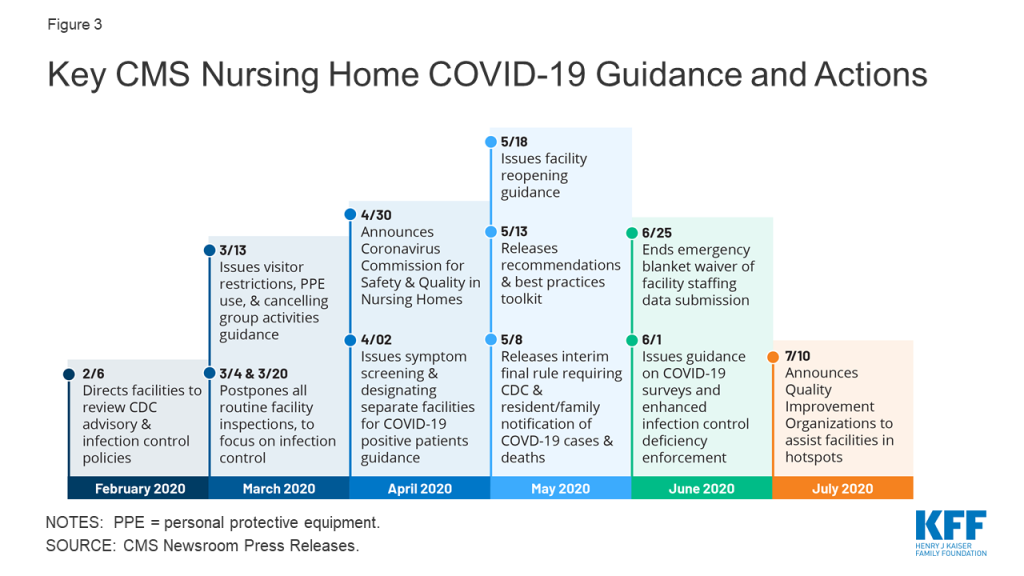
In April 2020, CMS announced the formation of an independent commission to conduct a comprehensive assessment of facility response to COVID-19. The commission is expected to make recommendations to protect residents from COVID-19 and improve care delivery responsiveness; strengthen efforts to rapidly identify and mitigate infectious disease transmission in nursing homes; and enhance strategies to improve compliance with infection control policies. The commission also is charged with identifying approaches to better use data to enable federal, state, and local entities to address the current spread of COVID-19 within facilities, analyze the impact of efforts to stop or contain the virus within facilities, and identify best practices to address COVID-19 that CMS or states could adopt. Commission members are to include residents, families, resident/patient advocates, industry experts, clinicians, medical ethicists, administrators, academics, infection control and prevention professionals, state and local authorities, and other experts. Twenty-five Commission members were announced in June 2020, and a final report is expected in fall 2020.
As of May 2020, facilities must report COVID-19 data to the CDC and provide information to residents and their families. Prior to this interim final rule, facilities were not required to report infectious disease information to the CDC, though they typically report to state and/or local health departments. The lack of centralized data contributed to difficulty in tracking disease spread and coordinating dissemination of personal protective equipment and testing supplies. The new rule requires weekly reporting of suspected and confirmed infections among residents and staff including residents previously treated for COVID-19; total deaths and COVID-19 deaths among residents and staff; PPE and hand hygiene supplies in the facility; ventilator capacity and supplies in the facility; resident beds and census; access to COVID-19 testing while a resident is in the facility; staffing shortages; and other information specified by the Secretary.
CMS is publicly reporting the data, and recently announced additional actions based on the data. In July 2020, CMS sent “Task Force Strike Teams” of clinicians and public health officials to 18 nursing homes in six states experiencing an increase in COVID-19 cases. The Teams are focused on determining immediate actions and needed resources to reduce virus spread. In late July 2020, CMS also announced that it would send a weekly list of nursing homes with an increase in cases to states.
The new rule also requires facilities to inform residents, their representatives, and families of confirmed or suspected COVID-19 cases among residents and staff by 5 pm the next calendar day following a single confirmed case or three or more residents or staff with new onset of respiratory symptoms within 72 hours of each other. Facilities also must provide cumulative updates at least weekly by 5 pm the next calendar day following each subsequent occurrence of confirmed infection or whenever three or more residents or staff with new onset of respiratory symptoms occur within 72 hours of each other. Facilities must include information on mitigating actions implemented to prevent or reduce the risk of transmission including whether normal operations will be altered such as restrictions or limitations on visits or group activities. Facilities cannot release personally identifiable information and can communicate through paper notices, a listserv, a website post or recorded phone messages.
In May 2020, CMS issued nursing home reopening recommendations and an informational toolkit with best practices for states to mitigate COVID-19 in nursing homes. The reopening guidance sets out criteria for relaxing restrictions using a phased approach and mitigating the risk of resurgence, including case status in the community, case status in the facility, adequate staffing, access to adequate testing, universal source control (masks, social distancing, and hand washing for visitors), adequate access to PPE for staff, and local hospital capacity. The guidance also includes considerations for allowing visitors and services and for restarting routine state survey activities in each phase. Additionally, quality improvement organizations (QIOs) under contract with CMS are providing technical assistance with a focus on about 3,000 low performing facilities with a history of infection control issues to help identify problems, create and implement an action plan, and monitor compliance. For example, QIOs train staff on proper PPE use, appropriate resident cohorting, and safe resident transfers. States also can request QIO technical assistance targeted to facilities that have experienced an outbreak.
In June 2020, CMS issued additional guidance to states on COVID-19 survey activities and enhanced enforcement for infection control deficiencies. CMS noted that focused infection control surveys pursuant to the March 2020 guidance had been completed in 53 percent of facilities, with wide state variation. As a result, states that have not completed all of these surveys by July 31, 2020 must submit a corrective action plan outlining their strategy to complete these surveys within 30 days. If all surveys still are not complete after the 30-day period, states’ CARES Act FY 2021 allocation may be reduced by up to 10%. Subsequent 30-day extensions could result in additional reductions up to 5%, with funding redistributed to states that have completed their surveys. The CARES Act provided $100 million for nursing home inspections focused on those with COVID-19 community spread from FY 2021 through FY 2023, $81 million of which will be available to state survey agencies. This represents a 6% annual increase in the nursing home survey and certification budget, which had remained at $397 million annually since October 2014. CARES funds may be used for state surveys, strike teams, enhanced surveillance or monitoring of nursing homes, or other state-specific interventions. The June 2020 guidance also establishes enhanced enforcement remedies for infection control deficiencies including directed plans of correction, discretionary denial of payment for new admissions, and CMPs.
The June 2020 guidance also announced three additional survey requirements: (1) states must perform on-site surveys of nursing homes with previous COVID-19 outbreaks by July 1, 2020; (2) states must perform on-site surveys within three to five days of identification of any nursing homes with three or more new suspected or confirmed cases since the last CDC COVID-19 report or one confirmed resident case in a facility that previously was COVID-free; and (3) starting in FY 2021, states must perform annual focused infection control surveys of 20 percent of facilities. States could forfeit 5% of their annual CARES Act allocation for failing to perform these activities.
The June 2020 guidance also authorizes states to expand survey activities to include more routine surveys once a state has entered phase 3 of the nursing home reopening guidance or earlier at state discretion. The expanded activities include complaint investigations that are not immediate jeopardy, revisit surveys to any facility with a removed immediate jeopardy finding but that is still out of compliance, special focus facility and special focus facility candidate recertification surveys, and nursing home and intermediate care facility for individuals with intellectual and developmental disabilities recertification surveys greater than 15 months. When expanding survey activities, states should prioritize facilities with a history or allegations of noncompliance regarding abuse or neglect, infection control, violations of transfer or discharge requirements, insufficient staffing or competency, or other care quality issues such as falls or pressure ulcers.
In late July 2020, CMS began requiring, rather than recommending, that all staff be tested weekly in nursing homes in states with a 5% or greater positivity rate. HHS also is distributing rapid diagnostic tests to nursing homes in COVID-19 hotspots through a one-time procurement to facilitate on-site testing of residents and staff. CMS and the CDC are offering COVID-19 training to nursing homes, which includes cohorting strategies and using telehealth to mitigate virus spread.
5. What are the key challenges for nursing homes as the pandemic continues?
As the pandemic continues, state survey agencies may face issues related to funding, capacity, and data. CMS will withhold CARES funds from state survey agencies that do not timely complete inspections, but these penalties may be too blunt for agencies whose lack of compliance stems from insufficient funding in the first place. As noted above, prior to the new CARES Act funds, the survey and certification budget had remained flat since 2014. It remains to be seen whether these new funds will be sufficient for state agencies to perform regular surveys as well as increased oversight in the foreseeable future resulting from the pandemic. While facilities are now reporting COVID-19 cases and deaths to CMS, these data are not cumulative prior to May 8, 2020, and it will be important for both state survey agencies and CMS to continue to monitor data and adjust policy guidance and facility oversight as needed.
Nursing facilities also face a number of challenges in their continued pandemic response. Nearly $5 billion in federal provider relief funds has been allocated to nursing facilities to cover health care related expenses or lost revenues attributed to coronavirus. On July 22, 2020, HHS announced that an additional $5 billion from the provider relief fund is being allocated to Medicare-certified long-term care facilities and state veterans homes. These funds may be used to hire additional staff, implement infection control “mentorship” programs with subject matter experts, increase testing, and provide additional services, such as technology so residents can connect with their families if they are not able to visit. As of June 28, 2020, nearly one in three nursing homes nationally report a shortage of staff and/or PPE. Despite federal legislation generally requiring insurers to cover coronavirus testing without cost-sharing, recent federal guidance concludes that insurers do not have to cover coronavirus “testing conducted to screen for general workplace health and safety (such as employee “return to work” programs).” This leaves open the question about who will pay for regular tests needed for facility staff to safely work during the pandemic. It also remains to be seen whether facilities will be able to maintain adequate staffing levels as the pandemic continues. CMS has lifted its emergency waiver of the staffing data submission requirement, and facilities must submit regular staffing data for April through June 2020 by August 14, 2020. Facilities also may be under financial strain due to lower occupancy levels as a result of the pandemic.
Conclusion
While the data and experience to date do not show a direct link between nursing home quality and COVID-19 cases, the pandemic has brought renewed attention to issues of nursing home quality and oversight at the federal and state levels. Nursing home quality concerns have existed for decades. With the current focus on challenges facing nursing homes and state survey agencies as they respond to the pandemic, policymakers may revisit whether federal Medicare and Medicaid requirements should be adjusted to improve oversight and whether additional funding is needed to support providers and agencies to ensure sufficient capacity and resources. These issues are likely to continue to be the subject of policy discussion and debate as long-term care coronavirus cases, particularly in “hotspot states” with wider community transmission continue to rise. While most nursing facilities are Medicare-certified, a small number (315 or 2%) are only Medicaid-certified and therefore appear ineligible for a share of the additional provider relief funds announced on July 22.
More broadly, seniors and people with disabilities who receive long-term services and supports in other settings, such as assisted living facilities, intermediate care facilities for people with intellectual/developmental disabilities, institutions for “mental disease,” and group homes, also are at increased risk of serious illness if infected with coronavirus based on older age and/or chronic health conditions. Unlike nursing homes, other long-term care congregate settings are primarily regulated by states, leading to greater variation in quality protections and lack of standardized reporting about coronavirus cases, deaths, staffing, supplies, and other data necessary to understand and respond to the pandemic and understand its full impact on all people who receive long-term care services and the staff who provide them.
Appendix
Appendix 1: Nursing Home Survey and Certification Process
A facility is subject to a standard survey, without advance notice, at least once every 15 months.21 If found to have provided substandard care quality,22 a facility is subject to an extended survey within two weeks of the standard survey.23 In addition, states may conduct special surveys within two months of any change in facility ownership, administration, management, or nursing director to determine whether care quality has declined as a result of the change.24 The Secretary also can conduct special surveys when the Secretary has reason to question facility compliance with federal requirements.25 Standard surveys include a case-mix stratified sample of residents.26 Extended surveys review and identify facility policies and procedures that produced substandard care quality, expand the sample size of resident assessments reviewed, and review staffing, in-service training, and if appropriate, consultant contracts.27
Facility surveys are conducted by a multi-disciplinary team using a protocol developed by the Secretary.28 The survey team must include a registered nurse and may include physicians, physician assistants, nurse practitioners, physical, speech, or occupational therapists, dieticians, sanitarians, engineers, licensed practical nurses, social workers, or other professionals.29 The survey team cannot include anyone who has served as staff or a consultant to the facility in the prior two years or who has a personal or familial financial interest in the facility.30
Survey and certification information, including statements of deficiencies, must be made publicly available by states and the Secretary within 14 days after the facility is notified.31 States submit this information, including any enforcement actions, to the Secretary on the same day that the facility is notified, and the Secretary uses this information to update the federal Nursing Home Compare website at least quarterly.32 The website must include facility staffing data, including resident census data and hours of care provided per resident per day and information on staffing turnover and tenure; links to state survey and certification programs, inspection reports, and facility plans of correction or report responses; information on how to file a complaint with the state survey and certification program and the state long-term-care ombuds program; summary information on the number, type, severity, and outcome of substantiated complaints; the number of adjudicated instances of criminal violations by the facility or its employees committed within the facility regarding abuse, neglect, exploitation, criminal sexual abuse or other violations resulting in serious bodily injury; and CMPs levied against the facility, its employees, contractors, and other agents. States also must notify the state long-term care ombudsman of any findings of noncompliance or adverse actions taken against facilities.
Appendix 2: History of Federal Nursing Home Requirements
After the creation of the Medicare and Medicaid programs, it soon became clear that “[s]trict enforcement of federal standards would have barred most nursing homes from participating in the Medicare program”33 in the mid-1960s, and certification of nursing homes to participate in Medicaid was left to the states.34 After increased congressional and media attention to substandard facility conditions and lax government oversight, revised federal regulations certifying facilities to participate in Medicare and Medicaid were established in 1974. Continued concern about care quality and inadequate enforcement led to new proposed federal regulations issued in 1980 by the Carter Administration. However, the nursing home industry objected to the new regulations’ projected costs, and the regulations were rescinded by the Reagan Administration. In turn, the Reagan Administration’s subsequent proposal, which would have deleted or relaxed many existing standards, was set aside after opposition from consumer advocates, states, Congress, and providers. The impasse led to the appointment of an Institute of Medicine (IOM) committee to recommend changes.
The 1986 IOM committee report contained numerous recommendations to enhance nursing home care quality and resident quality of life by improving nursing home regulation.35 While the IOM committee credited the 1974 regulations with contributing to better care quality, it noted “substantial room for improvement” due to weaknesses in the regulations and uneven administration and enforcement by states.36 At the time of the IOM study, “there [was] broad consensus that government regulation of nursing homes, as it now functions, is not satisfactory because it allows too many substandard nursing homes to continue in operation.” Concerns included “neglect and abuse leading to premature death, permanent injury, increased disability, and unnecessary fear and suffering on the part of residents,” poor quality of life and lack of autonomy, and poor quality medical and nursing care.37 The IOM committee emphasized that government regulation of nursing homes is warranted due to residents’ “large array of physical, functional, and mental disabilities,” limited opportunities for transfer among facilities, and inability to pay for care out-of-pocket.
Although the IOM recommendations led to changes adopted in the 1987 Nursing Home Reform Act, concerns about poor care quality and inadequate enforcement of federal standards continued. Between 1997 and 2010, the Government Accountability Office (GAO) issued more than 20 reports finding substandard care in many nursing homes; understatement of serious deficiencies by state surveyors; unenforced sanctions for harming residents; facilities cycling in and out of compliance; and inconsistent and ineffective federal oversight.38 During the same period, the HHS Office of Inspector General issued reports critical of facility compliance and state and federal enforcement.39 Recurring concerns include staffing levels, abuse and neglect, unmet resident needs, care quality, staff training and competency, and lack of integration between medical care and other services.40
| Table 1: Summary of Federal Requirements for Medicare and Medicaid Certified Nursing Homes | ||
| Area | Description | Citation |
| Requirements related to provision of services | ||
| Quality of Life | Care for residents in a manner and environment that promotes, maintains, or enhances each resident’s quality of life. Maintain a quality assessment and assurance committee to identify issues and develop and implement plans of action to correct identified deficiencies. | 42 U.S.C. § 1395i-3 (b)(1); 42 U.S.C. § 1396r (b)(1); 42 C.F.R. § 483.24. |
| Scope of services and activities under care plan | Provide services to attain or maintain the highest practicable physical, mental, and psychosocial well-being of each resident in accordance with a written care plan that describes the resident’s medical, nursing, and psychosocial needs and how these needs will be met. Care plan is initially prepared, with the participation to extent practicable of the resident or resident’s family or legal representative, by a team that includes the resident’s attending physician and a registered professional nurse with responsibility for the resident; and is periodically reviewed and revised. | 42 U.S.C. § 1395i-3 (b)(2); 42 U.S.C. § 1396r (b)(2); 42 C.F.R. § 483.21. |
| Residents’ assessments | Conduct a comprehensive, accurate, standardized, reproducible assessment of each resident’s functional capacity, which describes the resident’s capability to perform daily life functions and significant impairments in functional capacity. Assessment must be based on the uniform minimum data set specified by the Secretary, use an instrument specified by the states, include the identification of medical problems, and be conducted or coordinated by a registered professional nurse. Assessments must be conducted promptly and no later than 14 days after admission, promptly after any change in the resident’s physical or mental condition, and at least once a year. Examine each resident at least once every 3 months and revise the assessment as appropriate. | 42 U.S.C. § 1395i-3 (b)(3); 42 U.S.C. § 1396r (b)(3); 42 C.F.R. § 483.20. |
| Provision of services and activities | To the extent needed to fulfill care plans, provide or arrange for nursing and specialized rehabilitative services and medically related social services to attain or maintain each resident’s highest practicable physical, mental and psychosocial well-being; pharmaceutical services; dietary services that meet each resident’s daily nutritional and special dietary needs; an ongoing activity program to meet each resident’s interests and physical, mental and psychosocial well-being; routine dental services and emergency dental services; and treatment and services required by residents who have mental illness or intellectual disabilities not otherwise provided or arranged for by the state. Provide 24-hour licensed nursing services sufficient to meet resident needs, including registered professional nurse services at least 8 consecutive hours a day, 7 days a week, except that the Secretary may waive this requirement for certain rural SNFs, and states may waive this requirement for NFs that demonstrate their inability, despite diligent efforts (including offering wage at the community prevailing rate), to recruit appropriate personnel and if the state determines resident health and safety will not be endangered and a registered professional nurse or physician is obligated to respond immediately to phone calls from facility. | 42 U.S.C. § 1395i-3 (b)(4); 42 U.S.C. § 1396r (b)(4); 42 C.F.R. § § 483.25, 483.30, 483.35, 483.40, 483.45, 483.50, 483.55, 483.60, 483.65. |
| Required training of nurse aides | After 4 months, full-time nurse aides must have completed a training and competency evaluation program. Provide regular performance reviews and in-service education to assure nurse aide competency to provide services, including services to individuals with cognitive impairments. | 42 U.S.C. § 1395i-3 (b)(5); 42 U.S. C. § 1396r (b)(5). |
| Physician supervision and clinical records | Require that every resident’s medical care be provided under physician supervision (or for NFs, supervision of nurse practitioner, clinical nurse specialist, or physician assistant), provide for having a physician available to furnish necessary medical care in case of emergency and maintain clinical records on all residents which include care plans and resident assessments. | 42 U.S.C. § 1395i-3 (b)(6); 42 U.S.C. § 1396r (b)(6). |
| Required social services | Facilities with more than 120 beds must have at least one full-time social worker. | 42 U.S.C. § 1395i-3 (b)(7); 42 U.S.C. § 1396r (b)(7). |
| Information on nurse staffing | Post daily and make available to the public on request the current number of licensed and unlicensed nursing staff directly responsible for resident care in the facility for each shift. | 42 U.S.C. § 1395i-3 (b)(8); 42 U.S.C. § 1396r (b)(8). |
| Requirements related to residents’ rights | ||
| General rights | Protect and promote each resident’s rights including the rights to free choice (including choice of personal attending physician, to be fully informed in advance about care and treatment and any changes that may affect resident well-being and to participate in care planning and treatment); freedom from restraints (including physical or mental abuse, corporal punishment, involuntary seclusion, and any physical or chemical restraints imposed for discipline or convenience and not required to treat medical symptoms; restraints only may be imposed to ensure physical safety of resident or other residents and only upon written physician order that specifies duration and circumstances used, except in emergency circumstances specified by Secretary until such order could be reasonably obtained); privacy; confidentiality; reasonable accommodation of individual needs and preferences; grievances; participation in resident and family groups and social, religious, and community activities; to examine survey results, and to refuse transfer to another room if the purpose is to relocate from a part of the facility that is a SNF to a part of the facility that is not a SNF. Provide notice of these rights. Psychopharmacologic drugs may be administered only on physician orders and only as part of care plan designed to eliminate or modify symptoms for which drugs are prescribed and only if at least annually an independent external consultant reviews the appropriateness of the drug plan of each resident receiving such drugs. | 42 U.S.C. § 1395i-3 (c)(1); 42 U.S.C. § 1396r (c)(1); 42 C.F.R. § §483.10, 483.12. |
| Transfer and discharge rights | Cannot transfer or discharge resident unless necessary to meet the resident’s welfare, appropriate because resident’s health has improved, safety or health of other individuals in the facility would endangered, resident has failed to pay for stay, or facility ceases to operate. | 42 U.S.C. § 1395i-3 (c)(2); 42 U.S.C. § 1396r (c)(2); 42 C.F.R. § 483.15. |
| Access and visitation rights | Permit immediate access to any resident by any representative of the Secretary or the state, ombuds or resident’s individual physician; permit immediate access subject to resident’s right to deny or withdraw consent at any time to immediate family or other relatives; permit immediate access subject to reasonable restrictions and resident’s right to deny or withdraw consent at any time to others who are visiting with resident’s consent; permit reasonable access by any entity or individual that provides health, social, legal or other services to resident subject to resident’s right to deny or withdraw consent at any time; and permit state ombuds, with resident’s permission to examine resident’s clinical records. | 42 U.S.C. § 1395i-3 (c)(3); 42 U.S.C. § 1396r (c)(3). |
| Equal access to quality care | Establish and maintain identical policies and practices regarding transfer, discharge and covered services under Medicare and Medicaid for all individuals regardless of source of payment. | 42 U.S.C. § 1395i-3 (c)(4); 42 U.S.C. § 1396r (c)(4) |
| Admissions policy | Not require applicants or residents to waive their rights to Medicare or Medicaid benefits; not require oral or written assurance that such individuals are not eligible for or will not apply for Medicare or Medicaid; prominently display in facility and provide to such individuals written information about how to apply for and use Medicare and Medicaid and how to receive refunds for previous payments covered by such benefits; and not required third party guarantee of payment to facility as condition of admission or expedited admission to or continued stay in facility. | 42 U.S.C. § 1395i-3 (c)(5); 42 U.S.C. § 1396r (c)(5). |
| Protection of resident funds | Not require residents to deposit their personal funds with the facility and upon resident’s written authorization must hold, safeguard and account for such personal funds under a system established and maintained by the facility. Deposit personal funds in excess of $100 ($50 for NFs) in an interest bearing account separate from any of facility’s operating accounts. Any other personal funds must be maintained in a non-interest bearing account or petty cash fund. Maintain written financial records and pursue a surety bond to secure resident personal funds on deposit. | 42 U.S.C. § 1395i-3 (c)(6); 42 U.S.C. § 1396r (c)(6). |
| Requirements relating to administration and other matters | ||
| Administration | Be administered in a manner that enables facility to use its resources efficiently and effectively to attain or maintain each resident’s highest practicable physical, mental and psychosocial well-being and must have reports for any surveys, certifications, and complaint investigations during the preceding 3 years available for review. | 42 U.S.C. § 1395i-3 (d)(1); 42 U.S.C. § 1396r (d)(1); 42 C.F.R. § 483.70. |
| Licensing and life and safety code | Be licensed under state and local law and meet the life safety code requirements of the National Fire Protection Association Life Safety Code or state law. | 42 U.S.C. § 1395i-3 (d)(2); 42 U.S.C. § 1396r (d)(2); 42 C.F.R. § 483.90. |
| Sanitary and infection control program and physical environment | Establish and maintain an infection control program designed to provide a safe, sanitary, and comfortable environment in which residents reside and to help prevent the development and transmission of disease and infection and be designed, constructed, equipped and maintained in manner to protect health and safety of residents, personnel, and general public | 42 U.S.C. § 1395i-3 (d)(3); 42 U.S.C. § 1396r (d)(3); 42 C.F.R. § 483.80. |
| Miscellaneous | Comply with all applicable federal, state and local laws and accepted professional standards. | 42 U.S.C. § 1395i-3 (d)(4); 42 U.S.C. § 1396r (d)(4). |
Endnotes
- A Medicare skilled nursing facility (SNF) is an institution or distinct part that is primarily engaged in providing skilled nursing care and related services or rehabilitation services. 42 U.S.C § 1395i-3 (a). ↩︎
- A Medicaid nursing facility (NF) is an institution or distinct part that is primarily engaged in providing skilled nursing care and related services, rehabilitation services, or health-related care and services on a regular basis to individuals who because of their mental or physical condition require institutional care and services above the level of room and board. 42 U.S.C. § 1396r (a). In addition, SNFs and NFs cannot be primarily for the care or treatment of mental illnesses and must have a transfer agreement in effect with one or more hospitals. Id.; 42 U.S.C. § 1395i-3 (a). ↩︎
- The exception is state facilities, which are certified by the Secretary. 42 U.S.C § § 1395i-3 (g); 1396r (g). ↩︎
- 42 U.S.C. § 1396b (a)(2)(D). ↩︎
- 42 U.S.C. §§ 1395i-3 (g)(4); 1396r (g)(4). ↩︎
- States must maintain a registry of abuse, neglect, and misappropriation findings. 42 U.S.C. §§ 1395i-3 (g)(1)(C); 1396r (g)(1)(C). ↩︎
- 42 U.S.C. §§ 1395i-3 (g)(1)(B); 1396r (g)(1)(B). ↩︎
- For Medicare SNFs, states recommend penalties to be imposed by the Secretary based on survey results, and the Secretary also can independently impose penalties based on survey results or other findings. 42 U.S.C. § 1395i-3 (h). For Medicaid NFs, state law must include the specified penalties, which are imposed by states based on survey results or other findings. 42 U.S.C. § 1396r (h). ↩︎
- The facility must pay the temporary manager’s salary. If the immediate jeopardy is also substandard care quality, the state must notify each resident’s attending physician and the state facility administrator licensing board. 42 U.S.C. § § 1395i-3 (g)(5)(B), (C); 1396r (g)(5)(B),(C). ↩︎
- A CMP can be reduced in two ways, though only one penalty reduction can be applied. A CMP is reduced by 35 percent if a facility waives its right to a hearing to dispute the penalty. Or, a CMP may be reduced by 50 percent if a facility self reports and promptly corrects a deficiency, except that CMPs for repeat deficiencies within the prior year and for deficiencies found to result in a pattern of harm or widespread harm, immediately jeopardy to resident health or safety or resident death may not be reduced. 42 U.S.C. § § 1395i-3 (h)(2)(B)(ii); 1396r (h)(3)(C)(ii). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(4); 1396r (g)(4). ↩︎
- Institute of Medicine 1986. Improving the Quality of Care in Nursing Homes. Washington, DC: The National Academies Press at 5. https://doi.org/10.17226/646. ↩︎
- Id. at 79. ↩︎
- KFF, Key Issues in Long-Term Services and Supports Quality (Oct 2017), https://modern.kff.org/medicaid/issue-brief/key-issues-in-long-term-services-and-supports-quality/. ↩︎
- Medicaid NFs are subject to additional preadmission screening and resident review (PASRR) requirements to ensure that individuals with serious mental illness or intellectual disabilities require a nursing facility level of services and whether any specialized services are required. PASRR reviews are conducted by the state mental health or developmental disability authorities upon admission and when there is a significant change in a resident’s condition. 42 U.S.C. § 1396r (e)(7). ↩︎
- 81 Fed. Reg. 68688 (Oct. 4, 2016), https://www.govinfo.gov/content/pkg/FR-2016-10-04/pdf/2016-23503.pdf. ↩︎
- 42 C.F.R. § 483.75. ↩︎
- 42 C.F.R. § 483.85. ↩︎
- 42 C.F.R. § 483.73. ↩︎
- U.S. Gov’t Accountability Office, Infection Control Deficiencies Were Widespread and Persistent in Nursing Homes Prior to COVID-19 Pandemic (May 20, 2020), https://www.gao.gov/assets/710/707069.pdf. ↩︎
- There must be a 12-month statewide average interval between standard surveys 42 U.S.C. § § 1395i-3 (g)(2)(A)(i), (iii)(I); 1396r (g)(2)(A)(i), (iii)(l). The Secretary conducts surveys of a representative sample of facilities in each state, within two months of state surveys, to validate state findings. If the Secretary finds that a state’s surveys were inadequate, the state is subject to sanctions, including a correction plan, training, and/or technical assistance for SNF survey inadequacies, and a reduction in federal matching funds for Medicaid administrative costs for a pattern of failure to identify deficiencies and training for NF survey deficiencies. 42 U.S.C. § § 1396r (g)(3)(C) 1395i-3 (g)(3)(C); 42 C.F.R. § 488.320. ↩︎
- Substandard care quality includes one or more deficiencies in the following areas: treating residents with dignity and respect and in a manner that promotes quality of life; providing equal access to quality care regardless of condition severity or payment source; ensuring residents can freely exercise rights; not imposing physical or chemical restraints for discipline or convenience; reasonably accommodating resident needs and preferences in receiving services; allowing married residents to share a room with spouse; allowing residents to have roommate of their choice; providing written notice of change of room or roommate; allowing residents to choose activities, schedules, health care and providers; allowing residents to make choices about aspects of facility life significant to the resident; allowing residents to participate in community activities; allowing resident and family groups; allowing family or representatives of the resident to meet in the facility with other residents’ family or representatives; and providing a safe, clean, comfortable, and homelike environment. 42 C.F.R. § 488.301. ↩︎
- 42 U.S.C. § § 1395i-3 (g)(2)(B); 1396r (g)(2)(B). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(2)(A)(iii)(II); 1396r (g)(2)(A)(iii)(ll). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(3); 1396r (g)(3). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(2)(A)(ii); 1396r (g)(2)(A)(ii). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(2)(B)(iii); 1396r (g)(2)(B)(iii). ↩︎
- The Secretary also is responsible for providing survey team training. 42 U.S.C. § § 1395i-3 (g)(2)(C), (E)(iii); 1396r (g)(2)(C) (E)(iii). ↩︎
- 42 C.F.R. § 488.314 (a)(2). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(2)(E)(i), (ii); 1396r (g)(2)(E)(i), (ii). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(5)(A); 1396r (g)(5)(A). ↩︎
- 42 U.S.C. § § 1395i-3 (g)(5)(E); 1396r (g)(5)(E). ↩︎
- Institute of Medicine 1986. Improving the Quality of Care in Nursing Homes. Washington, DC: The National Academies Press at 147. https://doi.org/10.17226/646. ↩︎
- Id. at 242. ↩︎
- See generally Id. ↩︎
- Id. at 6. ↩︎
- Id. at 3-4. ↩︎
- KFF, Implementation of Affordable Care Act Provisions to Improve Nursing Home Transparency, Care Quality, and Abuse Prevention (Jan. 2013), https://modern.kff.org/medicaid/report/implementation-of-affordable-care-act-provisions-to-improve-nursing-home-transparency-care-quality-and-abuse-prevention/. ↩︎
- Id. ↩︎
- KFF, Key Issues in Long-Term Services and Supports Quality (Oct 2017), https://modern.kff.org/medicaid/issue-brief/key-issues-in-long-term-services-and-supports-quality/. ↩︎