VOLUME 44
Abortion Pill’s Safety Called into Question in Congressional Actions Based on Misleading Data
Highlights
False claims about the safety of mifepristone, the abortion pill used in roughly two-thirds of U.S. abortions, are driving legislative and investigative action in Congress, even as major medical organizations and decades of clinical evidence support the drug’s safety.
The Monitor also examines how competing interpretations of what censorship and free speech mean for policy and law are contributing to recent developments, including a Supreme Court ruling, a federal lawsuit settlement, and new legal challenges with implications for how health misinformation is managed, moderated, and allowed to spread.
What We’re Watching
Calls to Investigate and Ban Abortion Drug Mifepristone Cite Unsupported Safety Claims
Misleading claims about the safety of mifepristone, the abortion pill used in roughly two-thirds of U.S. abortions, are driving new legislative and investigative action in Congress. Senator Josh Hawley has cited a widely criticized report from the faith-centric Ethics and Public Policy Center to support legislation that would revoke the drug’s Food and Drug Administration (FDA) approval and an investigation of the practices of the companies that manufacture and distribute mifepristone, claiming they ignored safety information about the drug. The rate of adverse events cited in the EPPC report is far higher than what existing evidence supports and contradicts decades of clinical data, FDA review findings, and the assessments of major medical organizations including the American College of Obstetricians and Gynecologists (ACOG), the American Medical Association (AMA), and the World Health Organization (WHO) regarding the drug’s long safety record. State-level efforts to limit access to mifepristone are also continuing in response to data on abortion volume that shows that patients in states that ban abortion are obtaining abortions through telehealth. For example, state lawsuits led by Louisiana, Missouri, and Florida are challenging either the FDA’s original approval of mifepristone or subsequent modifications that allow clinicians to dispense the abortion pills by mail. False claims about mifepristone’s safety may be contributing to public perception. KFF’s November 2025 Health Tracking Poll found that while more than twice as many adults say mifepristone is ‘safe’ (42%) than say it is ‘unsafe’ (18%) when taken as directed by a doctor, public confidence in the drug’s safety has declined since 2023 when just over half of the public (55%) viewed the abortion pill as safe.
What To Watch Out For: Will continued false claims about mifepristone’s safety contribute to further legislative and regulatory action, even as major medical organizations and decades of evidence support the drug’s safety record? As misleading claims about mifepristone circulate more widely, will public confidence in the drug’s safety continue to decline?
Claims of Censorship Continue to Shape the Policy and Legal Landscape Around Health Misinformation
Ongoing debates about the limits of free speech and what constitutes censorship are driving policy actions and legal challenges with implications for how false or misleading health claims are addressed and how health-related speech is regulated.
- Settlement in Federal COVID-19 Social Media Lawsuit: The Trump administration recently settled a high-profile lawsuit, originally brought during the Biden administration, that alleged that federal officials had censored protected speech and violated the First Amendment by pressuring social media companies to remove false content related to the COVID-19 pandemic. Under the settlement, the Surgeon General’s office, the Centers for Disease Control and Prevention (CDC), and the Cybersecurity and Infrastructure Security Agency (CISA) are prohibited from threatening social media companies with legal or regulatory consequences to compel the removal of online content. The case had previously reached the Supreme Court, which ruled in 2024 that the plaintiffs lacked standing without determining whether the content removals had violated free speech protections. The administration and its allies have framed the settlement as a victory against what they call government censorship. In 2023, KFF polling found that most of the public expressed a desire for a greater government role in limiting the spread of false health information, with at least two-thirds of adults saying at the time that Congress and President Biden were “not doing enough” to limit the spread of false and inaccurate health information.
- Researchers Challenge Immigration Policy Targeting Misinformation Researchers: A nonprofit coalition of academic researchers has filed a new lawsuit against the administration, arguing that the current administration is itself engaged in censorship through its policy of excluding and deporting noncitizens whose work involves combating misinformation, fact-checking, or content moderation. The coalition argues that using immigration enforcement to penalize researchers who study misinformation is itself a restriction of free speech, the same principle the administration has raised to justify its policy, arguing that fact-checking and content moderation amount to censorship. These competing claims reflect ongoing disagreement about whether independent content moderation and fact-checking amounts to censorship, a framing that may have already contributed to widespread platform policy changes.
- Ruling Blurs Medical Regulation and Viewpoint Discrimination: The Supreme Court ruled that a Colorado law prohibiting licensed therapists from promoting “conversion therapy” for minors—practices that attempt to change or suppress an LGBTQ person’s sexual orientation or gender identity—may have violated the First Amendment by restricting health providers’ speech based on viewpoint. The ruling centers on the tension between states’ authority to regulate harmful medical practices and therapists’ free speech rights. The case has implications beyond Colorado, as 23 states and D.C. have passed similar laws. Major medical organizations maintain that conversion therapy is ineffective and associated with harm, including increased rates of depression and suicidality. Physician organizations have warned that the ruling may lead to more widespread adoption of harmful practices. As such, removing state protections prohibiting conversion therapy could reinforce false narratives that these practices are generally accepted and/or that being LGBTQ+ is a mental health condition in need of treatment.
What To Watch Out For: Will the settlement barring government agencies from pressuring social media companies affect how these platforms respond to health misinformation? Will courts find that immigration enforcement against misinformation researchers constitutes an unconstitutional restriction of speech? Will the Supreme Court’s conversion therapy ruling impact similar state restrictions around the country, and will the case be cited more broadly to challenge other regulations governing health providers’ speech and practices?
FDA Expected to Lift Restrictions on Peptides as Unproven Claims Reach Large Audiences
Recent reporting indicates that the Food and Drug Administration (FDA) plans to lift restrictions on roughly 14 peptides that in 2023 the agency had removed from a list of products that compounding pharmacies could use due to potential safety risk. Peptides are injectable substances popular in some wellness communities for their purported effects on muscle recovery, injury healing, and anti-aging properties. Many of the claims about their health benefits are unproven, yet they are reaching large audiences and may be influencing federal policy. Peptide-related Google searches reached 10.1 million in January, according to an analysis cited by CBS News, with searches for peptides marketed for anti-aging and longevity up nearly 300% year-over-year. Many of these substances lack robust clinical evidence for the uses being promoted and are currently sold through an online gray market labeled “for research use only.” Still, imports of hormone and peptide compounds from China roughly doubled to $328 million in the first three quarters of 2025, according to U.S. customs data. Sellers of unregulated peptides regularly promote unsupported claims about both benefits and safety, despite a lack of evidence behind those claims. Their marketing can blur the line between research chemicals and legitimate medical treatments, leaving buyers with little sense of the uncertainties involved.
The move is reported to have surprised some current and former FDA staff amid concerns that the shift could heighten criticism that the agency is basing decisions on politics rather than science. Some career scientists have warned that the supposed benefits of these substances have not been proven in clinical trials and that expanding access without an established evidence base may pose risks to patients. Supporters of the move, including Health and Human Services (HHS) Secretary Robert F. Kennedy Jr., who has called himself a “big fan” of these treatments and said he has personally used them, have argued that expanding access through licensed pharmacies would help ensure safety standards that cannot be enforced through the current gray market. When unproven claims about a treatment’s benefits spread widely through social media and are amplified by senior officials, it can be difficult for the public to distinguish between evidence-based decisions and those motivated by personal beliefs.
What To Watch Out For: KFF polling finds fewer than half of the public (38%) and partisans have at least “some” confidence in federal health agencies like the CDC and FDA to make decisions based on science rather than the personal views of agency officials. Will this decision be interpreted as evidence-based, or will it reinforce existing doubts about whether regulatory decisions reflect science rather than personal views?
While Most Users of AI for Health Information Cite Quick Access, Cost Concerns Also Drive Some to These Tools
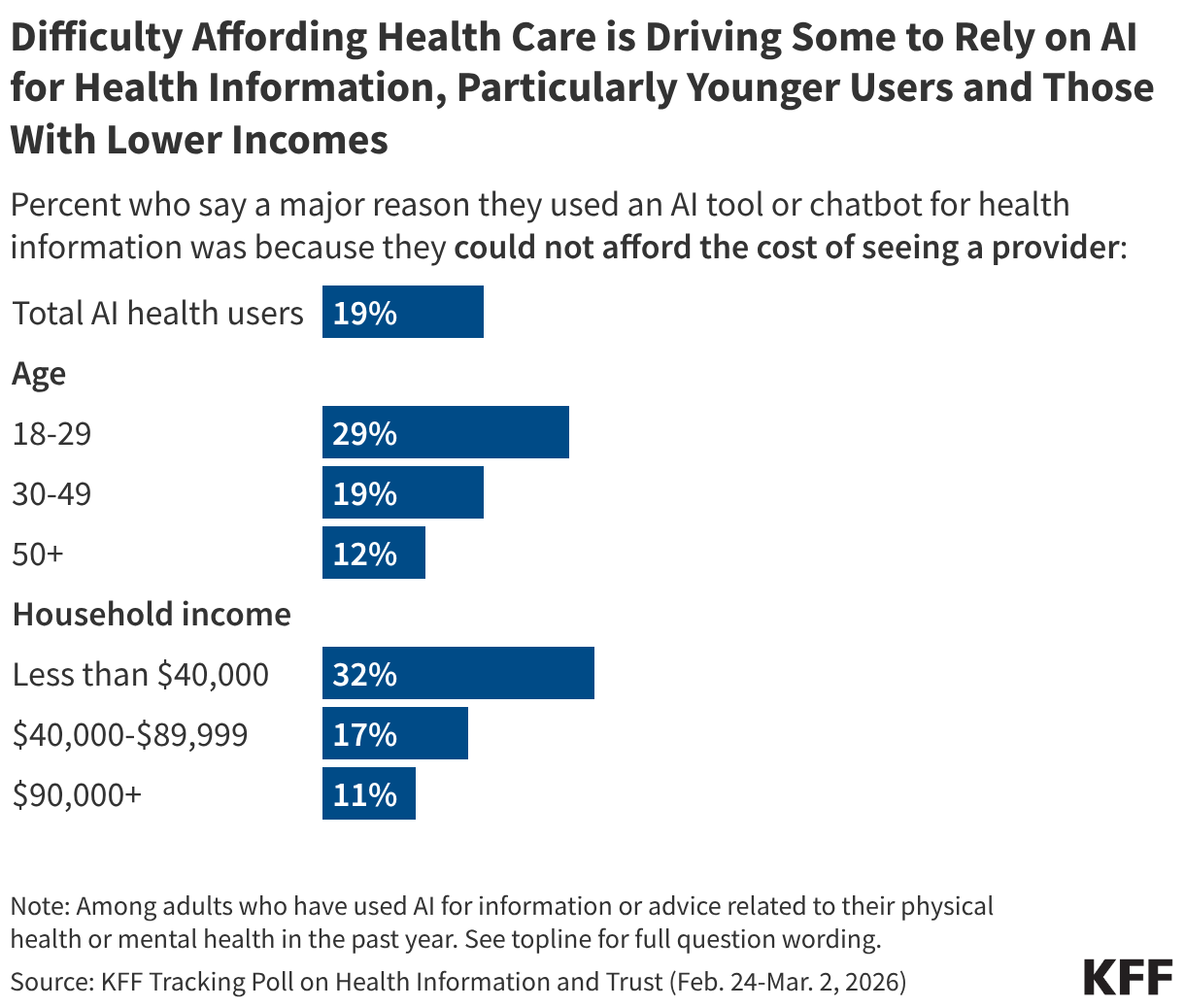
Recent KFF polling has found that difficulty affording health care is driving some adults to rely on AI for health advice at a time when many people are reporting increasing health care costs. Overall, about one-third of the public (32%) has turned to AI for health information and advice in the past year, according to KFF’s March Tracking Poll on Health Information and Trust. While most users say a desire for quick and immediate information drove them to these tools, one in five (19%) say a “major reason” they turned to AI was because they couldn’t afford the cost of seeing a provider. The share who report turning to AI because of costs rise to three in ten (29%) among younger users (under 30 years old) and one-third (32%) of users with incomes below $40,000.
These findings come as KFF’s January 2026 Health Tracking Poll found more than half (55%) of adults said their health care costs had increased in the past year, including two-thirds of people with employer-based health insurance (64%) and those who purchase their own coverage (66%). The cost of health care can also lead some to forego needed care. One-third (36%) of adults say they skipped or delayed needed health care in the past year because of the cost, rising to just under half (45%) among adults under 30 and three quarters of uninsured adults, according to KFF’s May 2025 Health Tracking Poll. Notably, KFF’s latest poll on AI use found that younger adults were more likely to say they used AI for health information and then did not follow up with a health care provider.
For those already facing barriers to accessing care, the risk of acting on incomplete or unreliable information provided by AI without clinical follow-up may be greatest.
What To Watch Out For: Will rising costs widen this information gap and increase reliance on unvetted sources among people facing the greatest barriers to care?
More From KFF
- KFF Tracker: The Status of Abortion-related State Ballot Initiatives Since Dobbs
- KFF Tracker: LGBT People Experience Widespread Concerns and Challenges When it Comes to Health Care Affordability
- KFF Event (Recording): Costs, Coverage, and Enrollment Changes: Exploring Current Public Opinion and Policy on the ACA Marketplaces
- KFF Health News: CDC’s Acting Chief Promises a Return to Stability in a Tumultuous Moment
Support for the Health Information and Trust initiative is provided by the Robert Wood Johnson Foundation (RWJF). The views expressed do not necessarily reflect the views of RWJF and KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities. The data shared in the Monitor is sourced through media monitoring research conducted by KFF.