Rapid Home Tests for COVID-19: Issues with Availability and Access in the U.S.
Executive Summary
While rapid home COVID-19 tests have been identified as an important component of controlling the spread of SARS-CoV-2, the virus that causes COVID-19, there have been ongoing supply challenges in the U.S. Challenges in accessing rapid home tests became more acute as the Delta variant took hold, vaccination rates stagnated, and cases, hospitalizations, and deaths surged. In addition, some workplaces began reopening, millions of school age children returned to in-person school and colleges and universities began the fall semester. Home test supply and access challenges in the U.S. contrast with the experience in several peer countries which have made rapid home tests widely available and at little or no cost. The Biden administration has taken recent steps to address these challenges, including a new announcement that it would work to identify manufacturers of high quality tests to encourage them to bring them to the U.S. market and to streamline the regulatory review pathway for authorization of over-the-counter at-home tests, though it is unclear if or how quickly these measures will fully resolve the issues.
In this brief, we examine the various interrelated factors that have contributed to rapid home test scarcity in the U.S. The five factors we identify include:
- The regulatory framework: The FDA regulatory framework and process are central to the availability of COVID-19 testing, including for home testing. However, some argued the framework for evaluating rapid home tests was initially too stringent, creating challenges for manufacturers seeking authorization and hampering test availability. While the FDA took steps to modify the process, some suggested that the regulatory environment was still too strict. The new announcement that the FDA aims to further streamline the process is likely to address some of these issues.
- Supply chain and the unpredictability of demand: The COVID-19 pandemic has led to supply chain disruptions worldwide, including for tests and their components, negatively impacting test supply. This, coupled with the unpredictability of the pandemic, have made it difficult for manufacturers to gauge demand and at times limited their willingness to scale up production due to perceived business risk, again impacting test availability. Recent steps taken by the administration are designed to increase supply.
- Federal investment: While this may be beginning to shift, the lack of up-front federal investment in testing, including pre-purchasing of tests, is another factor that appears to have impacted testing availability and affordability, and contrasts with the federal government’s approach to vaccines.
- Federal messaging and guidance: Evolving federal messaging and guidance related to the role of testing over the course of the pandemic and limited messaging about home testing may have led to confusion about its importance and impacted public understanding of home testing specifically.
- Cost and coverage: While COVID-19 tests are generally covered by insurance, as required by emergency COVID-19 legislation passed by Congress, and are also available free of charge at many locations in the U.S., this is not the case for over-the-counter rapid home tests. Instead, consumers must pay out of pocket for these tests which can be costly, limiting access and potentially contributing to existing pandemic disparities. Recently, the federal government accelerated purchasing of rapid tests for use in certain community-based settings and negotiating with retailers to lower store prices, albeit for a time-limited period.
Taken together, we identify a number of barriers that have contributed to the scarcity of rapid home COVID-19 tests in the U.S. Recent actions taken by the Biden administration, including steps expected to result in an increase in home test production and to further streamline the authorization process, may alleviate some of these barriers, but others may persist.
Issue Brief
Introduction
While diagnostic testing is an important tool for helping to reduce the spread of COVID-19, there have been challenges in scaling up testing in the U.S. throughout the pandemic, and these have been particularly acute in the case of rapid home tests. Rapid home tests, some of which can provide results in as little as 15 minutes, have been identified as an important public health intervention for controlling COVID-19 transmission because of their ability to be used at home by an individual, provide results quickly, and to identify infection when someone is “likely to be most contagious.” Challenges in accessing rapid home tests became more acute as the Delta variant took hold, vaccination rates stagnated, and cases, hospitalizations, and deaths surged. In addition, some workplaces began reopening, millions of school age children returned to in-person school, and colleges and universities began the fall semester. These challenges contrast with the experiences of several other peer countries which have made rapid home tests widely available and at little or no cost. The UK government, for example, provides up to seven tests per day to those who cannot get tests from work or school and recommends each individual screen themselves twice weekly. Providing up to 7 tests per person allow one individual to collect tests for a whole household. Germany, until recently, made rapid antigen tests freely available as well (and tests can still be purchased for a few dollars in grocery stores).
President Biden announced early support for rapid tests. The Biden campaign said that if elected it would “Invest in next-generation testing, including at home tests and instant tests, so we can scale up our testing capacity by orders of magnitude.” The White House National Strategy for COVID-19, released on January 22, 2021, underscored the importance of rapid testing particularly to help “Safely reopen schools, businesses, and travel, while protecting workers” and funding was awarded in February to increase production of at-home tests. But ongoing supply shortages, particularly in the context of Delta, prompted more recent actions. On September 9, the President released a COVID-19 action plan that included, among other elements, the intent to use the Defense Production Act to increase the production of rapid tests; an investment of $2 billion to purchase 280 million rapid point-of-care and OTC at-home COVID-19 tests to be made available in a variety of community settings and long-term care facilities; and an agreement with several top retailers to sell rapid home tests at cost for a three month period. An additional $1 billion investment was announced on October 6 to further mobilize testing production, as well as agreements from some manufacturers to increase production. As a result, the administration expects to double rapid test capacity by the end of the year, from 100 million tests per month to 200 million, rising to 300 million by February 2022. As part of this effort, in late October, the administration announced additional measures, including a new program which aims to identify manufacturers of high quality tests to encourage them to bring them to the U.S. market, as well as a streamlining of the FDA regulatory pathway. It is not yet clear, however, if these recent actions will fully resolve rapid home test supply and access issues, particularly if there is another surge in cases at some point.
In this brief, we examine the various interrelated factors that have contributed to rapid home test scarcity in the U.S.
Background
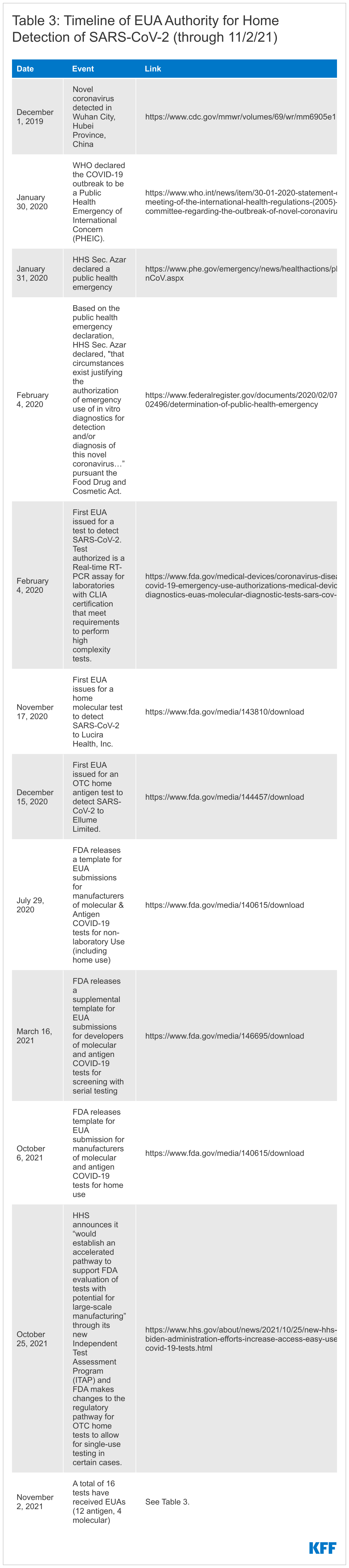
The declaration of a public health emergency due to COVID-19, first on January 20, 2020 and renewed every three months since (six times as of October 11, 2021), paved the way for the Food and Drug Administration (FDA) to exercise its expedited emergency use authorization (EUA) process for medical devices, including COVID-19 diagnostics.
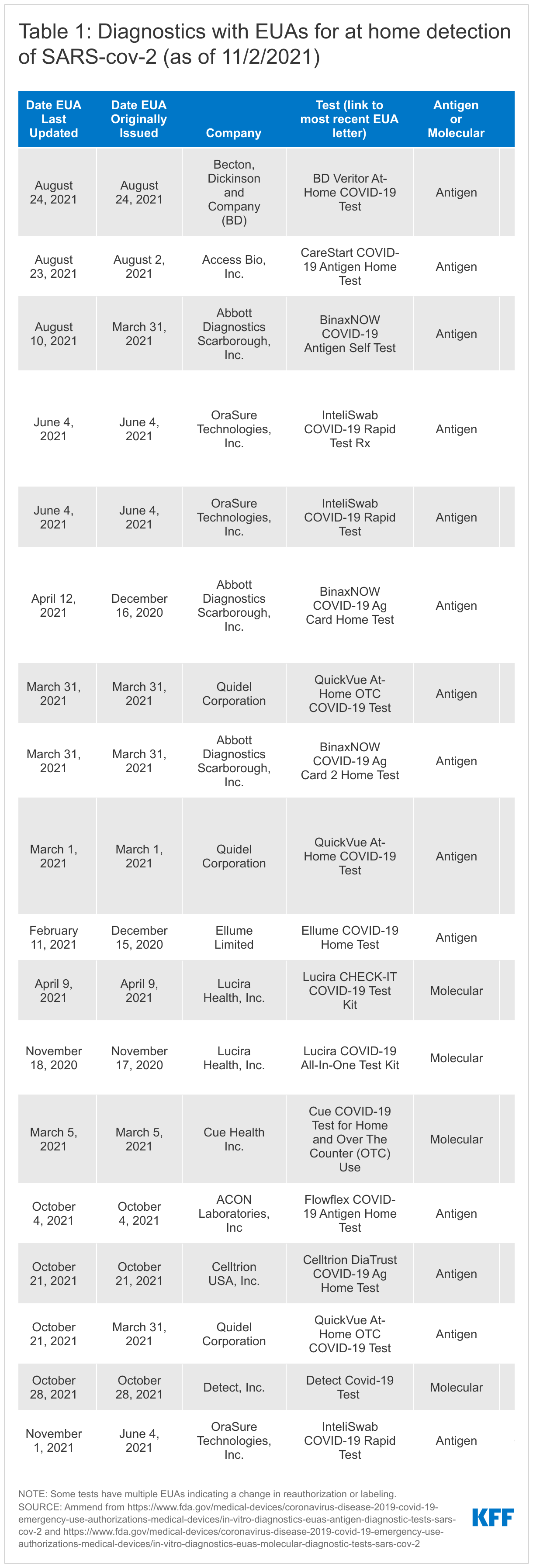
To date, the FDA has authorized 16 home tests (several receiving revised EUAs over time) to 11 companies (see Table1). In several cases, one manufacturer has received multiple EUAs for essentially the same product but has different indications in terms of prescription vs. over-the-counter use or with revisions to labeling. Of the 16 tests with EUAs, 12 are OTC while four require a prescription.
The 16 authorized home diagnostics include two types of tests: antigen tests (12 of the 16) and molecular tests (4 of the 16). Antigen tests detect specific viral antigens indicative of SARS-CoV-2, the virus that causes COVID-19. Molecular tests, including polymerase chain reaction (PCR) tests, detect the virus' genetic material (see Table 2). Antigen tests tend to be highly specific (the ability to accurately diagnose negative cases) but are typically less sensitive (the ability to accurately diagnose positive cases) compared with molecular tests. However, antigen tests demonstrate “comparable performance to” molecular tests in “symptomatic persons and/or if culturable virus present, when the person is presumed to be infectious.” They have advantages as well; they have a more simple design and are less expensive than molecular tests, potentially reducing both manufacturing and access barriers, and are available at the point-of-care, including in the home. Of the 12 authorized antigen tests, 9 appear to have been marketed directly to consumers, while none of the molecular tests have.
| Table 2. Differences in Home COVID-19 Testing Options |
| Test Type |
| Antigen – Antigen tests detect specific viral antigens indicative of SARS-CoV-2, the infection that causes COVID-19. Antigen tests tend to be highly specific but are typically less sensitive than molecular tests. |
| Molecular or Nucleic acid amplification tests (NAAT) - Detects the virus' genetic material (RNA), highly sensitive and highly specific. Polymerase chain reaction (RT-PCR or PCR) is one type of NAAT. |
| Prescription or Over-the-counter |
| Prescription - Some home tests with an EUA require a prescription from a medical provider. In certain cases, tests can be prescribed and ordered online (e.g. http://www.emed.com) |
| Over-the-counter (OTC) - Some tests have been authorized for OTC use, meaning they can be purchased in a store or online without a prescription. |
| Collection, Results, and Reporting |
| Home - The test is conducted at home and the results are returned to the user within a short window, often 15 minutes. The individual is responsible for reporting results to a public health authority/provider or an app is used during the home test process and the app/manufacturer reports results to the appropriate public health authority. |
| Self-collection - The user collects a sample and places it into a home collection kit which is the sent to a lab for results interpretation. The lab reports results to appropriate public health authorities. |
The first EUA for a COVID-19 diagnostic test, a laboratory-based PCR, was issued on February 4, 2020. Nine months later, on November 17, 2020, the first EUA for a home COVID-19 test, in this case a molecular test, was issued and the first EUA for a home antigen test was issued December 15, 2020. (See Table 3 for a more detailed timeline of authorization events).
Factors impacting availability of home COVID-19 tests
There are several interrelated factors that impact the availability of rapid home COVID-19 tests. These range from the regulatory environment to the supply chain to federal investment and messaging to affordability. Each is explored below.
The Regulatory Framework
The FDA regulatory framework and process are central to the availability of COVID-19 testing, be that in the home, community, clinic, or laboratory. All tests for COVID-19, including home tests, must go through regulatory review. Recognizing the importance of making safe and effective COVID-19 tests available and to facilitate and streamline the EUA process, the FDA provides manufactures with application templates. Though manufacturers are not required to use these templates, they can expedite the process and they provide agency recommendations for test performance and other elements.
There have been several templates and updates released since the beginning of the pandemic, targeting different types of tests, settings, and strategies. The first template was released in January 2020 for laboratory-based tests, with a template for non-laboratory-based tests, including for home use, coming in July 2020, and one for screening with serial testing (e.g. testing multiple times in a short window to improve chances of detecting infection) in March 2021. Most recently, the first template specifically for home tests was released in early October and then revised at the end of the month in 2021. (See Table 2 for a more detailed timeline.)
The recommendations for test performance continue to evolve over time. Earlier templates for use for rapid home tests set a high bar for efficacy, with FDA generally recommending efficacy quite similar to an “authorized high sensitivity molecular test.” Over time, FDA has changed its recommended benchmarks or created new pathways (e.g. for serial testing for use with asymptomatic individuals) and these changes have coincided with increased home test approval.
Despite these changes in the regulatory framework, however, some have argued that the comparison of antigen test performance to PCR test performance still sets the benchmark too high, making it overly difficult for developers to meet. One rationale behind this critique is that the focus on antigen test efficacy should be on its ability to detect infectious individuals to quickly interrupt onward transmission. Others disagree and suggest that while antigen tests perform well at detecting infectious individuals, they should not be used for assessing infectiousness on their own. CDC has found that Abbott’s antigen, “BinaxNow [test] performs better at identifying rRT-PCR–positive specimens with lower Ct (suggestive of higher viral loads) and positive viral cultures” but it warns “these factors are not precise proxies for infectiousness.”
Indeed, the U.S. recommendations are higher compared to standards in some peer countries, such as the UK. To the extent that FDA regulatory requirements are a barrier for companies to submit requests for EUAs, fewer tests with authorization could lead to decreased competition in the marketplace, potentially impacting both cost and availability. On the other hand, whether a lower standard would negatively impact test quality, and by association the ability to control the pandemic, is unknown.
While the FDA has not suggested it will lower the recommended threshold for tests, on October 25, 2021, HHS announced that the NIH “would establish an accelerated pathway to support FDA evaluation of tests with potential for large-scale manufacturing” through its new Independent Test Assessment Program (ITAP), an extension of its Rapid Acceleration of Diagnostics (RADx) initiative. HHS states the program, which will be a collaboration of NIH, FDA, and CDC and other HHS experts will “identify manufacturers of high quality tests and encourage them to bring those tests to the U.S. market, increasing options for people and overall supply and potentially lowering costs.” Companies will receive support to “help ensure they are providing the best submissions possible for FDA’s regulatory review” and scalable OTC tests will be prioritized. In addition, FDA made changes to the regulatory pathway for OTC home tests currently authorized only for serial testing by revising recommendations to allow for single-use testing for symptomatic individuals. This reduces the amount of data developers would need to submit and allows them to sell certain tests currently sold as two-packs as singles, potentially leading to more individual tests being available for sale at a lower price.
Finally, just because a test has an EUA does not mean that it will circulate widely in the consumer market, as we have seen with some of the antigen and all of the molecular tests with EUAs for home use, suggesting that regulatory issues alone are not the only factors in test availability.
Supply Chain and the Unpredictability of Demand
The COVID-19 pandemic has led to supply chain disruptions across the world, and this has, in some cases, included the raw materials needed to manufacture COVID-19 diagnostics. Certainly that both the Trump and Biden Administrations have leveraged the Defense Production Act (DPA) to support testing, including Biden’s use of the DPA for home tests, and address other supply shortages, such as for personal protective equipment, is suggestive of challenges in the production and availability of these essential goods.
Additionally, a review of testing manufacturer Securities and Exchange Commission (SEC) filings reveals concerns about supply chain with respect to raw materials and diagnostic components:
“As a result of the COVID-19 pandemic, we have seen delays in receipts for certain raw materials and components for our products. Such delays can result in disruption to our business operations….We cannot currently predict the frequency, duration or scope of these government actions and any supply disruptions, and the availability of various products is dependent on our suppliers, their location and the extent to which they are impacted by the COVID-19 pandemic, among other factors. …Our inventory levels may fluctuate due to supply chain variability in conjunction with larger and more frequent customer orders.” - Quidel Q2 2021 10-Q SEC filing
“Due to the significant uncertainty that exists relative to the duration and overall impact of the COVID-19 pandemic, our future operating performance, particularly in the short-term, may be subject to volatility. In this regard, we continue to see challenges posed by the pandemic to global transportation channels and other aspects of our supply chain, including the cost and availability of raw materials. As noted above, the pandemic continues to impact demand for certain of our products. The U.S. and other governments may enact or use laws and regulations, such as the Defense Production Act or export restrictions, to ensure availability of needed COVID-19 testing and vaccination delivery devices. Any such action may impact our global supply chain network.” - Becton, Dickinson and Company (BD) Q2 2021 10-Q SEC filing
Further, as manufacturers began securing EUAs for their at-home diagnostics, the pandemic appeared to be shifting, at least domestically. Vaccines were becoming more widely available, soon places of business and entertainment would start opening up, and the Delta variant had not yet taken hold in the United States. However, the landscape quickly reversed as vaccination rates stagnated and the more infectious Delta variant become the dominant strain in the U.S., resulting in increased cases, hospitalizations, and deaths. Investor calls and SEC filings by manufactures regarding testing overall, as well as home testing specifically, discussed this uncertainty, noting how an inability to predict the arc of the pandemic made assessing future testing demand difficult.
“Our financial performance and results of operations will depend on future developments and other factors that are highly uncertain, continuously evolving and cannot be predicted, including the duration of the COVID-19 outbreak, the severity and continuation of outbreak surges, actions to contain the spread of the virus such as mask wearing, social distancing and vaccination efforts globally, and the impact of these and other factors on testing demand.” – Quidel Q2 2021 10-Q SEC filing
“The real… factor here becomes COVID testing...that’s really the question here. How will testing play itself out in the second half here whether its variants, whether its vaccination rates, etc so that’s just something we are paying attention to…that’s why our guidance rage was pretty wide, to really account for that.” – Abbott Q2 2021 earning call
Indeed, Abbott’s decision to pull back on COVID-19 test production leading into the summer has been widely publicized and SEC filings indicate that this was related, at least in part, to a belief that the course of the pandemic had shifted and there was a reduced need for tests.
“On May 27, 2021, Abbott management approved a restructuring plan related to its Diagnostic Products segment to align its manufacturing network for COVID-19 diagnostic tests with changes in projected testing demand driven by several factors, including significant reductions in cases in the U.S. and other major developed countries, the accelerated rollout of COVID-19 vaccines globally and the U.S. health authority’s updated guidance on testing for fully vaccinated individuals.” - Abbott Q2 2021 10-Q SEC filing
As a result, by the time the demand for rapid home tests began to increase, due to rising cases, hospitalizations and deaths, re-opening workplaces, and the return to in-person schooling for millions of elementary school students, the supply was not there. The new federal ITAP effort is designed to help increase supply by seeking additional manufacturers to bring tests to the U.S, market and could help to address some of these barriers.
Federal Investment
The lack of up-front federal investment in testing, including advance purchasing of tests, is another factor that appears to have impacted testing availability and affordability. Whereas the federal government channeled billions of dollars into accelerating vaccine development and advance purchasing of millions of doses, which helped to mitigate manufacturer risk and allowed for vaccines to be provided at no cost to all in the United States, no similar strategy was undertaken for testing. Without such up-front investment, the risk and cost of scale up was largely placed on manufacturers, who might be reticent to do so in the face of uncertainty given their fiduciary responsibility to shareholders. As noted, this differs from the experience in the UK and Germany where governments simultaneously supported vaccination and home testing efforts.
The Biden Administration’s use of the DPA in September to spur more domestic production of rapid tests and its procurement of additional rapid tests injected new investments in this area. Since then, HHS announced contracts with Abbott, Celltrion, OraSure, Quidel, and Acon Laboratories. And, on October 6, 2021, the White House committed another $1 billion towards rapid tests to “further mobilize testing manufacturers…to expand production of tests…based on the United States government’s commitment to procure an additional 180 million rapid tests over the course of the next year.” Much of these new procurements will provide free and increased rapid testing to community sites and long-term care facilities, designed to reach high needs populations in specific settings, but not necessarily increase supply for consumers.
As mentioned above, this additional investment is expected to result in a supply of 200 million rapid antigen tests per month by December, and 300 million by February 2022. Some experts recommend, however, that rapid antigen testing be conducted once twice or even three times per week to optimally help to identify those who are infectious and interrupt onward transmission. In response to a recent study, an NIH official stated that “Rapid antigen testing at home, two to three times per week, is a powerful and convenient way for individuals to screen for COVID-19 infection,” further noting that “With schools and businesses reopening, an individual’s risk of infection can change from day to day. Serial antigen testing can help people manage this risk and quickly take action to prevent spread of the virus.” Indeed, many colleges and universities have adopted this approach to curb spread. Yet, the new U.S. investment, even if resulting in 300 million rapid tests per month, would be less than one test per month per person in the U.S. For two times per week testing, it would cover less than 40 million individuals. If everyone older than 11 were tested twice per week per the UK’s recommendation, we would need 2.3 billion tests per month. If just half the U.S. population tested weekly, that would translate to a need for more than 150 million tests per week, around 600 million per month. The recent announcement of the new federal ITAP effort invests an additional $70 million and could help to increase supply in the U.S. market, though how quickly is uncertain.
Federal Guidance and Messaging
One other challenge has been federal messaging and changing guidance related to the role of testing as part of the U.S. COVID-19 response over the course of the pandemic. This included mixed messages and changing recommendations during the Trump administration, including down-playing of the importance of testing and shifting guidance from the CDC during vaccine roll-out.
Before vaccine roll-out, in late 2020 CDC’s testing guidance was that “people who have had close contact (within 6 feet for a total of 15 minutes or more) with someone with confirmed COVID-19” should get tested. Then, as of March 17th 2021, when vaccines were starting to become more widely available to all adults in the United States, case rates were down, and Delta had not yet taken hold, CDC stated that “fully vaccinated people with no COVID-19 symptoms do not need to be tested following an exposure to someone with COVID-19.” Just a few months later, however, in early August 2021, the CDC again changed its guidance to recommend testing for fully vaccinated people following exposure: “fully vaccinated people should be tested 3-5 days following a known exposure to someone with suspected or confirmed COVID-19.” While this shift in messaging was responding to a rapidly changing pandemic and new evidence on an emerging and evolving disease, industry communications indicate that it had an impact on manufacturing:
“While we expect to close more accounts with employers and have several promising partnerships in the pipeline, they involve a good deal of blocking and tackling and are very hard to predict or value especially when guidance from CDC and the landscape of COVID testing seem to evolve daily. Recently the warning from public health officials that vaccinated people can become infected and spread the highly contagious delta variant of COVID-19 appears to be a near term driver for more masking and testing especially as schools and office look to reopen in the fall.” – Quidel Q2 2021 investor call
And as noted above:
“…changes in projected testing demand [are] driven by several factors, including significant reductions in cases in the U.S. and other major developed countries, the accelerated rollout of COVID-19 vaccines globally and the U.S. health authority’s updated guidance on testing for fully vaccinated individuals.” - Abbott Q2 2021 10-Q SEC filing
Beyond impact on industry, shifting messaging could have affected public understanding of testing and confidence in agency recommendations. More broadly, the CDC has provided limited guidance on the role of home or routine testing, as a public health tool in addressing the pandemic and how it may differ from testing in other settings. Guidance on preventing COVID-19 has focused on vaccination and the CDC page on COVID-19 testing is aimed at those who are at risk for infection, symptomatic or are traveling with no information on testing routinely or as a public health practice. In the How to Protect Yourself and Others factsheet (last updated in August 2021), there is no mention of how testing may be a tool in the pandemic response. Similarly, the home testing page focuses on testing among those who may be at risk for having COVID-19, and suggests that home testing may be something to pursue if you cannot get tested by a provider: “If you need to be tested for COVID-19 and can’t get tested by a healthcare provider, you can consider using either a self-collection kit or a self-test that can be performed at home or anywhere else.” CDC guidance on testing in schools and workplaces is more comprehensive in its discussion of tests as a public health or screening tools but is limited to those settings. The limited information on home tests and little explanation of their role as a public health tool for home use may impact public understanding of home testing and willingness to use rapid test kits. However, a recent FDA press release for a newly authorized home antigen test states that the agency “considers at-home COVID-19 diagnostic tests to be a high priority and we have continued to prioritize their review given their public health importance.”
Cost and Insurance Coverage Considerations
Cost may also be a barrier to accessing OTC rapid home tests. Domestically available OTC tests typically range from $14 for two tests, or $7 per test, to $38.99 per test.1 If a consumer wanted to test regularly, even the least expensive test ($14 for two tests) used twice a week would amount to $728 per year, assuming they could get tests in this quantity. The more typical (non-discounted) price would cost an individual more than $1,000 per year, and the most expensive test used twice a week would amount to over $4,000 per year. Elsewhere, outside the U.S., home tests are about a dollar and in some cases, like in the UK, Israel, and Singapore, free to all or many people.
While federal COVID-19 legislation ensures that a range of tests are available free of charge through insurance, federal guidance indicates that home tests are only required to be covered if “ordered by an attending health care provider who has determined that the test is medically appropriate for the individual based on current accepted standards of medical practice and the test otherwise meets the statutory criteria.” Further, if OTC home tests were covered by commercial insurance, it could be logistically cumbersome for an individual testing regularly to seek reimbursement each time.
The new ITAP aims to bring more tests to market which could increase competition and decrease costs. In addition, the revisions to the FDA pathway to allow for the sale of certain single use, rather than serial, tests could decrease the purchase price for some consumers. However, the real impact of these changes is yet to be realized.
Finally, even if supply problems are solved, cost could remain a barrier for some. Unless there is widespread scale up with multiple access points and free or truly inexpensive tests, disparities in terms of diagnosis and community transmission that already exist could be exacerbated. Some of these issues may be alleviated by recent federal investments to provide free rapid testing to certain community sites but that has yet to be proven.
Looking Ahead
Taken together, there are multiple interrelated factors that have contributed to the lack of availability of rapid home tests in the U.S., including the regulatory framework, supply chain and demand issues, federal investment, federal messaging, and affordability. Recent actions by the Biden administration are likely to help alleviate some of these challenges though it is not yet clear if they can fully resolve the issues. Even if scale-up hits the projected increase in supply to 300 million rapid tests per month by early next year, this would translate into less than one test per individual per month, not enough to test most people weekly as some recommend. Additionally, even with scale up and more widespread retail availability, cost will likely persist as a barrier for many people, and could run the risk of exacerbating existing inequities in the pandemic. It will also be important to monitor whether the growing vaccination or testing requirements from employers and other places of business/entertainment will generate more demand for tests and stress the already limited supply. As has been the case since the early days of the pandemic, the future of COVID-19 in the United States, from case load to vaccination to testing is hard to predict. More widespread use of testing, and home testing in particular, could help to address the pandemic in the face of ongoing uncertainty.
Endnotes
- Prices come from searches for OTC COVID tests conducted twice a day over a week period (between September 21 and 27, 2021) on Amazon, Walgreens, CVS, Kroger, Target, and Walmart websites. ↩︎