Implementing Coverage and Payment Initiatives: Results from a 50-State Medicaid Budget Survey for State Fiscal Years 2016 and 2017
Executive Summary
Medicaid plays a significant role in the U.S. health care system, now providing health insurance coverage to more than one in five Americans and accounting for one-sixth of all U.S. health care expenditures.1 The Medicaid program continues to evolve as state and federal policy makers respond to changes in the economy, the broader health system, state budgets, and policy priorities, and in recent years, to requirements and opportunities in the Affordable Care Act (ACA). This report provides an in-depth examination of the changes taking place in Medicaid programs across the country. The findings in this report are drawn from the 16th annual budget survey of Medicaid officials in all 50 states and the District of Columbia conducted by the Kaiser Commission on Medicaid and the Uninsured and Health Management Associates (HMA), in collaboration with the National Association of Medicaid Directors. This report highlights policy changes implemented in state Medicaid programs in FY 2016 and those implemented or planned for FY 2017 based on information provided by the nation’s state Medicaid directors. The District of Columbia is counted as a state for the purposes of this report. Key findings include the following:
Key Findings
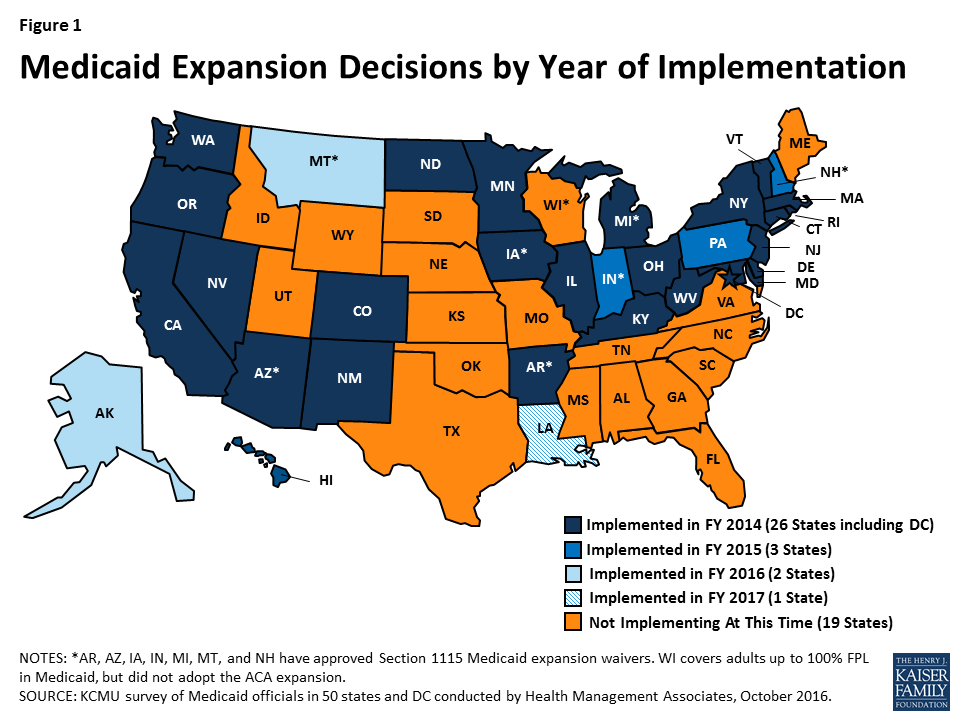
Eligibility and enrollment. As of October 2016, 32 states had adopted the ACA Medicaid expansion. Two states (Alaska and Montana) implemented the expansion FY 2016 and Louisiana implemented in FY 2017. Beyond the ACA, states made few major eligibility changes. Some states have approval or are seeking approval to impose premiums or monthly contributions under Medicaid expansion waivers. Many states have initiatives to expand coverage to the criminal justice involved population.
Managed care and delivery system reforms. In 28 of the 39 MCO states, at least 75 percent of all Medicaid beneficiaries were enrolled in MCOs. Many states are implementing quality initiatives such as pay for performance, reporting MCO quality metrics, or collecting adult and child quality measures. States are using MCO arrangements to promote value based payment and increase attention to the social determinants of health. Twenty-nine (29) states are also adopting or expanding other delivery system reforms in FY 2016 or FY 2017, such as patient-centered medical homes (PCMHs), Health Homes, Accountable Care Organizations (ACOs), Delivery System Reform Incentive Payment (DSRIP) programs, and other efforts to better manage the care of high-need populations.
Long-term services and supports (LTSS). Nearly every state reported actions to expand the number of persons served in community settings in FY 2016 and FY 2017, primarily through increased enrollment in HCBS waivers and implementing new HCBS SPAs. Twenty-three (23) states provided some or all LTSS through a managed care arrangement as of July 1, 2016, with 15 states offering MLTSS on a statewide basis for at least some LTSS populations.
Provider payment rates and taxes. In FY 2016, more states implemented provider rate increases than implemented restrictions; however, as economic conditions become more challenged, slightly fewer states are implementing rate increases (40 states) than restrictions (41 states) in FY 2017. All states (except Alaska) use at least one provider tax or fee to help finance Medicaid. Eight of the Medicaid expansion states reported plans to use provider taxes or fees to fund all or part of the costs of the ACA Medicaid expansion beginning in January 2017, when states must pay 5 percent of the costs of the expansion.
Benefits (including prescription drug policies). A total of 21 states expanded or enhanced covered benefits in FY 2016, and 20 states are planning expansions for FY 2017, most commonly for behavioral health and substance use disorder services. With rising drug costs, 31 states in FY 2016 and 23 in FY 2017 reported implementing or plans to implement pharmacy cost containment efforts, some targeted to high cost specialty drugs. As part of the battle to address the nation’s opioid crisis, a majority of states have adopted, and many are expanding, pharmacy management strategies specifically targeted at opioids.
Looking ahead. Many states report administrative challenges in implementing the ACA, major delivery system reforms, new federal regulations, and new systems due to limited resources in terms of staff and funding for administration. Despite the administrative and fiscal challenges, Medicaid directors listed priorities for FY 2017 and beyond that focus on payment and delivery system initiatives designed to control costs, improve access to care, and achieve better health outcomes.
Eligibility and Enrollment
Medicaid expansion under the ACA continues to affect state policies for FY 2016 and FY 2017. As of October 2016, 32 states had adopted the ACA Medicaid expansion. This includes 26 states that implemented the expansion in FY 2014, three states in FY 2015 (New Hampshire, Pennsylvania, and Indiana), two states in FY 2016 (Alaska and Montana), and Louisiana in FY 2017. Beyond eligibility changes tied to the ACA, in FY 2016 and FY 2017 states implemented or adopted only a few changes generally targeted to a limited number of beneficiaries. Medicaid policies related to beneficiary premiums and copayments changed little overall; most of the activity reported related to Medicaid waivers. In FY 2016, Montana implemented a Medicaid expansion waiver that included provisions to impose premiums or monthly contributions. At the time of the survey, Arkansas, Arizona, Kentucky, and Ohio had Medicaid waivers pending with premium provisions. HHS denied Ohio’s pending waiver on September 9, 2016 and the Arizona waiver was approved on September 30, 2016.
Many states have initiatives to expand coverage to criminal justice involved populations. In states that implemented the ACA Medicaid expansion, a greater proportion of this population is now eligible for Medicaid. Key initiatives include efforts to enroll individuals in Medicaid prior to their release and policies that maintain Medicaid eligibility during incarceration by suspending rather than terminating Medicaid coverage.
Managed Care and Other Delivery System Reforms
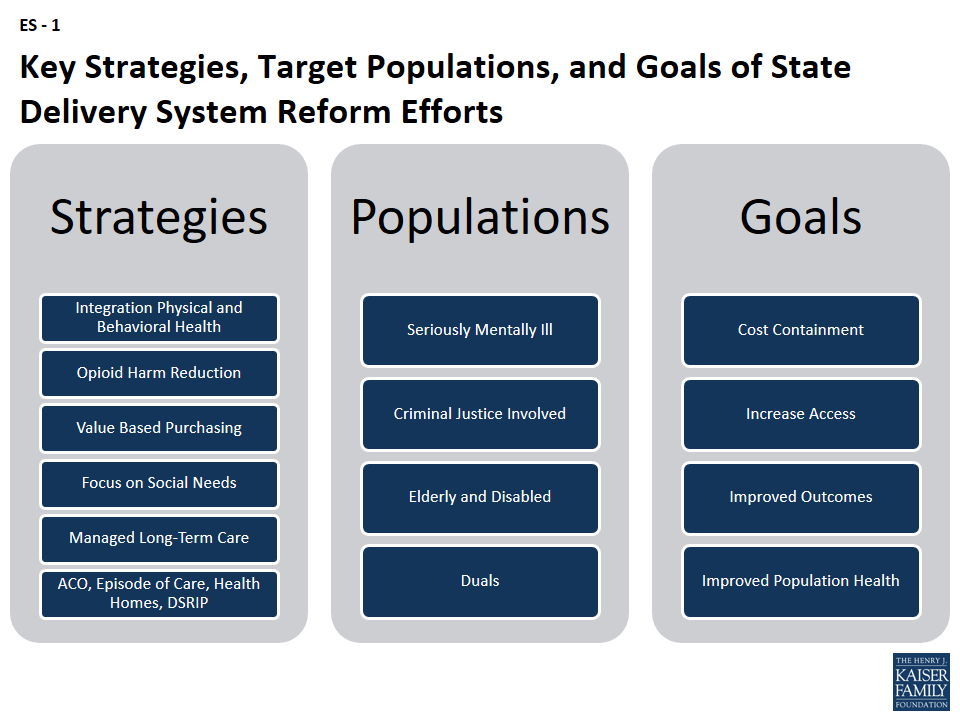
States continue to expand the use of MCOs and other delivery system reform efforts with goals to control costs, improve access to care and care outcomes, and ultimately improve population health (Figure ES-1). Many of these initiatives are targeted to high-need populations.
Reliance on risk-based managed care continues to grow as additional states use Managed Care Organizations (MCOs) to deliver care or enroll more populations. As of July 2016, 39 states contracted with risk-based managed care organizations (MCOs) to serve their Medicaid enrollees. (These states are referred to throughout the report as “MCO states.”) In 28 of the 39 MCO states, at least 75 percent of all Medicaid beneficiaries were enrolled in MCOs (an increase from 21 of 39 states in July 2015). Three states (Iowa, Rhode Island, and West Virginia) terminated their Primary Care Case Management (PCCM) programs in either FY 2016 or FY 2017 and shifted those populations into risk-based managed care. Alabama plans to implement a new MCO program in FY 2017 and Missouri plans to expand its MCO program statewide in FY 2017.
States sometimes exclude special populations and/or some behavioral health services from MCO contracts. This survey asked about populations with special needs that may be included or excluded from acute care MCO enrollment. States reported that of the special populations noted in the survey, pregnant women were most likely to be enrolled on a mandatory basis into acute care MCOs (28 states) while persons with intellectual or developmental disabilities (ID/DD) were least likely to be enrolled on mandatory basis (10 states) and also most likely to be excluded from MCO enrollment (7 states).
Many states are implementing quality initiatives encouraging or requiring MCOs to implement alternative payment models or screen for social needs. Thirty-six (36) of the 39 MCO states reported one or more select MCO quality initiatives in place in FY 2015 and 17 states in each of the survey years (FY 2016 and FY 2017) implemented or adopted new quality initiatives such as pay for performance, reporting MCO quality metrics or collecting adult and child quality measures. In FY 2016, five states identified targets in MCO contracts for the use of alternative provider payment models; 10 additional states intend to do so in FY 2017. States are also using MCO arrangements to increase attention to the social determinants of health. Twenty-six (26) states reported requiring or encouraging MCOs to screen for social needs and provide referrals to other services in FY 2016 and four states intend to do so in FY 2017. States commonly require MCOs to perform a health needs/risk assessment that includes information on social determinants as well as medical needs. In addition, five states reported that they have policies in place to encourage or require MCOs to provide care coordination services to enrollees prior to release from incarceration (Arizona, Iowa, Kentucky, New Mexico, and Ohio), and 10 states intend to add such requirements in FY 2017.
Over two-thirds of all states (36) have at least one delivery system or payment reform initiative in place and the majority of states are expanding current programs or adopting new initiatives. Twenty-nine (29) states in either FY 2016 or FY 2017 reported adopting or expanding one or more initiatives including patient-centered medical homes (PCMHs), Health Homes, Accountable Care Organizations (ACOs), and other initiatives to better manage the care of persons with multiple chronic conditions. Interest in Episode of Care initiatives also ticked upward for FY 2017 (7 states). These initiatives may be implemented through fee-for-service or managed care. Seven states had Delivery System Reform Incentive Payment (DSRIP) programs in place in FY 2015. Four states reported new or expanded DSRIP programs in FY 2016 and five states reported new or expanded DSRIP programs in FY 2017.
Long-Term Services and Supports
Nearly every state reported actions to expand the number of persons served in community settings in FY 2016 and FY 2017, primarily through increased enrollment in HCBS waivers and implementing new HCBS SPAs. New PACE sites or expanded enrollment in existing PACE sites as well as including specific rebalancing incentives into managed care contracts that cover long-term services and supports (LTSS) were also strategies to increase community-based care.
Twenty-three (23) states provided some or all LTSS through a managed care arrangement as of July 1, 2016. Fifteen (15) states offered managed LTSS (MLTSS) on a statewide basis for at least some LTSS populations. The most common model combines both acute care and LTSS in a single plan, providing a comprehensive approach to service integration. Five states offer a prepaid health plan that covers only Medicaid LTSS. In FY 2016, four states implemented MLTSS or expanded MLTSS to new parts of the state, and four states expanded MLTSS to new populations. In FY 2017, two states anticipate geographic expansion in MLTSS, while five states anticipate adding new populations to MLTSS. Enrollment into the MLTSS program is always mandatory for seniors in 13 of the 23 MLTSS states, for individuals who have full dual eligibility status in nine states, for nonelderly adults with physical disabilities in 12 states, and for individuals with I/DD in eight states. Thirteen (13) states with MCOs offering LTSS reported having LTSS quality measures in place in FY 2015. In FY 2016, a total of six states implemented new or expanded LTSS quality metrics; five states plan to expand quality measures for LTSS in FY 2017.
Provider Rates and Taxes
Changes in the economy continue to affect provider reimbursement rates, and states are also implementing reimbursement policies designed to promote quality. In FY 2016 more states implemented provider rate increases than implemented restrictions (45 and 38 states, respectively); however, as economic conditions become more challenged, slightly fewer states are implementing rate increases (40 states) than restrictions (41 states) in FY 2017. As part of efforts to improve the quality of health care and reduce costs, 21 states have or are adopting reimbursement policies in FY 2017 to reduce potentially preventable hospital readmissions in fee-for-service (FFS) and 11 of the 39 MCO states require or plan to require MCOs to adopt such incentives or penalties. Twenty (20) states have or are adopting reimbursement policies in FY 2017 designed to reduce the number of early elective deliveries in FFS and 14 states require or plan to require MCOs to adopt similar policies.
States continue to rely on provider taxes, with eight states using this financing mechanism to fund the state share of ACA expansion costs. All states except Alaska use at least one provider tax or fee to help finance Medicaid. In FY 2016 and FY 2017, 22 states increased or planned to increase one or more provider tax or fee and nine states are adding new provider taxes. Eight of the Medicaid expansion states (Arkansas, Arizona, Colorado, Illinois, Indiana, Louisiana, New Hampshire, and Ohio) reported plans to use provider taxes or fees to fund all or part of the costs of the ACA Medicaid expansion beginning in January 2017, when states must pay five percent of the costs of the expansion.
Benefits and Prescription Drugs
A total of 21 states expanded or enhanced covered benefits in FY 2016, and 20 states planned benefit expansions in FY 2017. The most common benefit enhancements reported were for behavioral health and substance use disorder services, telemedicine and tele-monitoring services, and dental services for adults. Far fewer states reported benefit restrictions.
With rising drug costs, many states are focused on pharmacy cost containment efforts. The vast majority of states identified high cost and specialty drugs as a significant cost driver for state Medicaid programs, most pointing specifically to hepatitis C antivirals. Many states are focused on refining and enhancing their pharmacy programs, including actions related to new and emerging specialty and high-cost drug therapies. A total of 31 states in FY 2016 and 23 in FY 2017 reported implementing or plans to implement pharmacy cost containment efforts. Thirty-three (33) of the 39 states with MCO contracts as of July 1, 2o16 reported that the pharmacy benefit was generally carved in, and another state reported plans to implement a full pharmacy carve-in in January 2017. States reported how they manage MCO pharmacy programs; in FY 2015, 13 states had uniform clinical protocols, 10 states had uniform prior authorization, and 10 states had uniform PDL requirements across fee-for-service and MCO programs. Many states reported expansions of these strategies in FY 2016 and FY 2017.
As part of the battle to address the nation’s opioid epidemic, a majority of states have adopted, and many are expanding, pharmacy management strategies specifically targeted at opioids. The CDC has developed and published recommendations for the prescribing of opioid pain medications for adults in primary care settings.2 Twenty-one (21) states reported adoption or plans for adoption in FY 2017 for their FFS programs. Of the 39 states with MCO contracts, 11 are requiring MCOs to adopt the CDC guidelines or are planning to do so in FY 2017. Many other states indicated these policies were under review for FFS and MCOs. States were also adopting strategies to expand access to naloxone, a prescription opioid overdose antidote that prevents or reverses the life-threatening effects of opioids. Almost all states reported specific opioid-focused pharmacy management policies. Imposing a quantity limit was the most common pharmacy management approach, used by almost all states (46) in FY 2015 for their FFS programs. Use of prior authorization (45 states), clinical criteria (42 states), and step therapy (32 states) were also widespread in FY 2015 for FFS. Significantly fewer states (12) reported having a requirement in place in FFS in FY 2015 for Medicaid prescribers to check their states’ Prescription Drug Monitoring Program before prescribing opioids to a Medicaid patient. Many states also reported MCO policies in place; however, it is unclear how many states require such policies.
Administration and Key Priorities for 2017 and Beyond
Medicaid provides health coverage for over one-fifth of all Americans and accounts for one-sixth of national health expenditures.3 Its administration involves complex systems, rules, and requirements. States indicated their most significant administrative challenges related to implementing the ACA, major delivery system reforms, new federal regulations, and new eligibility and IT systems. Medicaid directors noted that limited resources in terms of staff and funding for administration make it difficult to balance competing priorities and implement multiple significant initiatives. Despite the administrative and fiscal challenges, Medicaid directors listed an array of priorities for FY 2017 and beyond that focus on payment and delivery system initiatives designed to control costs and achieve better health outcomes.
Report: Introduction
Medicaid has become one of the nation’s most important health care programs, now providing health insurance coverage to more than one in five Americans, and accounting for over one-sixth of all U.S. health care expenditures.4 The Medicaid program continues to change, as policy makers in each state seek to improve their program, responding to changes in the economy, the broader health system, state budgets and policy priorities, and in recent years, to requirements and opportunities in the Affordable Care Act (ACA) as well as new guidance and regulations. In many ways, state Medicaid programs are national leaders in delivery and payment system initiatives designed to improve health care and outcomes, and to control health care spending.
This report examines the reforms, policy changes and initiatives that occurred in FY 2016 and those adopted for implementation for FY 2017 (which began for most states on July 1, 20165 ). The findings in this report are drawn from the annual budget survey of Medicaid officials in all 50 states and the District of Columbia conducted by the Kaiser Commission on Medicaid and the Uninsured (KCMU) and Health Management Associates (HMA), in collaboration with the National Association of Medicaid Directors (NAMD). This was the sixteenth annual survey, which has been conducted at the beginning of each state fiscal year from FY 2002 through FY 2017.6 (Copies of previous reports are archived here.)
The KCMU/HMA Medicaid survey on which this report is based was conducted from June through August 2016. The survey was sent to each state Medicaid director in June 2016. Directors and their staff provided data for this report in their written survey response and through a follow-up telephone interview. All 50 states and DC completed surveys and participated in telephone interview discussions between June and August 2016. The survey instrument is included as an appendix of this report.
The survey collects some data about Medicaid policies in place during a base year, but focuses on changes from year-to-year. For FY 2017, the survey includes policy changes implemented at the beginning of the year, or for which a definite decision has been made to implement during the fiscal year; it does not include policy changes under consideration but for which a definite decision on implementation has not been made. Medicaid policy makers know that policies adopted for the upcoming year are sometimes delayed or not implemented for reasons related to legal, fiscal, administrative, systems or political considerations, or due to delays in approval from CMS. The District of Columbia is counted as a state for the purposes of this report; the counts of state policies or policy actions that are interspersed throughout this report include survey responses from the 51 “states” (including DC). Key findings of this survey, along with state-by-state tables providing more detailed information, are described in the following sections of this report:
Report: Eligibility, Enrollment, Premiums, And Copayments
Key Section Findings
- As of October 2016, 32 states had adopted the ACA Medicaid expansion. This includes 26 states that implemented the expansion in FY 2014, three states in FY 2015 (Indiana, New Hampshire and Pennsylvania), two states in FY 2016 (Alaska and Montana), and Louisiana in FY 2017. Few states adopted or planned for eligibility changes in FY 2016 and FY 2017, and changes were targeted to a limited number of beneficiaries.
- As a result of new coverage pathways (including both expanded Medicaid coverage and the availability of Marketplace subsidies), some states are eliminating Medicaid coverage for beneficiaries with incomes above 138 percent federal poverty level (FPL) or more limited Medicaid eligibility pathways.
- All but three states have provisions for Medicaid coverage of inpatient care provided to incarcerated individuals in place or planned for FY 2017. Many state Medicaid agencies and their departments of corrections partners are working together to help ensure individuals have Medicaid coverage in place when they are released from jail or prison back to the community through a range of policies and procedures.
- Medicaid policies related to beneficiary premiums and copayments changed little for FY 2016 and FY 2017 except for Medicaid expansion waivers. In FY 2016, Montana implemented a Medicaid expansion waiver that included provisions to impose premiums/monthly contributions. At the time of the survey, Arkansas, Arizona, Kentucky, and Ohio had Medicaid waivers pending with premium provisions intended for FY 2017 implementation. HHS denied Ohio’s pending waiver on September 9, 2016 and Arizona received waiver approval on September 30, 2016.
Tables 1, 2, 3, and 4 at the end of this section include additional details on eligibility, premiums, and cost-sharing policy changes in FYs 2016 and 2017.
Changes to Eligibility Standards
The ACA Medicaid expansion was one of the most significant Medicaid eligibility changes in the history of the program. As of October 2016, 32 states had implemented the ACA Medicaid expansion: 26 states implemented the expansion in FY 2014; three states (Indiana, New Hampshire and Pennsylvania) in FY 2015; two states (Alaska and Montana) in FY 2016, and on July 1, 2016 (FY 2017), the expansion became effective in Louisiana (Figure 1). Beyond the Medicaid expansion, states implemented or adopted only a few eligibility changes generally targeted to a limited number of beneficiaries.
Coverage Transitions
As a result of new coverage pathways (including both expanded Medicaid coverage and the availability of Marketplace subsidies), some states eliminated Medicaid coverage for beneficiaries with incomes above 138 percent FPL (most of this activity occurred in FY 2014 and was covered in earlier surveys). In addition, some individuals who had qualified through more limited Medicaid eligibility pathways, such as those related to pregnancy, family planning, spend-down, and the Breast and Cervical Cancer Treatment (BCCT), could be eligible for more comprehensive coverage through income based pathways in states that adopted the ACA Medicaid expansion. Even with alternative coverage options, many states have maintained most limited coverage options, although enrollment through these pathways may have declined. Changes to coverage above 138 percent of the FPL or to limited pathway coverage in FY 2016 or FY 2017 are listed below. Because individuals have access to Medicaid through another eligibility pathway or to other coverage options, these changes are not counted as positive or negative eligibility changes, but as “no change” (Tables 1 and 2).
- In FY 2016, Connecticut reduced Medicaid parent eligibility levels from 201 percent FPL to 155 percent FPL; many parents previously eligible at the higher levels should be eligible for Marketplace subsidies.
- New Hampshire plans to phase out its BCCT pathway in FY 2017.
- Pennsylvania eliminated spend-down for parents and adults with disabilities over age 21 as part of its Healthy PA waiver, but reinstated this coverage in March 2016.
- Ohio eliminated its family planning waiver in FY 2016. Michigan closed its family planning waiver to new enrollment in April 2014 and officially ended the program June 30, 2016.
Other Eligibility Changes
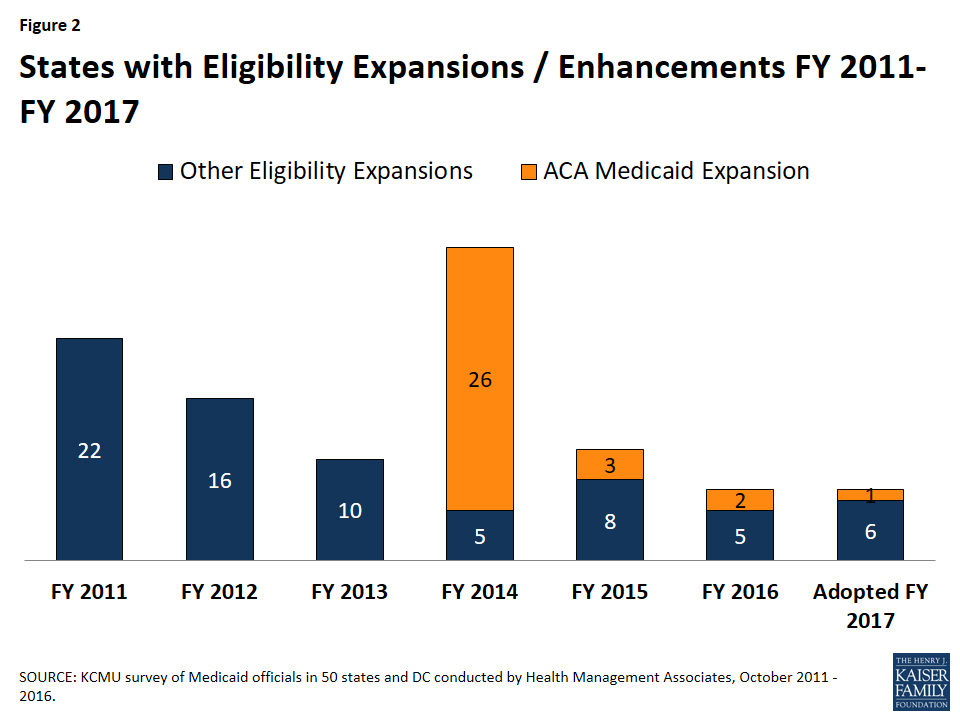
Other eligibility changes aside from the ACA expansion in FY 2016 and FY 2017 were limited and targeted to small numbers of beneficiaries (Tables 1 and 2). For FY 2016, a total of seven states made changes that expanded Medicaid eligibility and for FY 2017, seven states plan to implement Medicaid eligibility expansions (Figure 2). Key expansions include the following:
- Florida in FY 2016 and Utah in FY 2017 are implementing the option to eliminate the five-year bar on Medicaid eligibility for lawfully-residing immigrant children. Utah expects to cover an additional 750 children.
- Michigan implemented the Flint Water Group waiver, which extends Medicaid eligibility to children and pregnant women with incomes up to 400 percent FPL if they were exposed to tainted Flint water. (The waiver also expands benefits for existing eligible individuals by adding Targeted Case Management.)
- Maine will increase eligibility under its family planning pathway to 209 percent FPL in FY 2017.
Only two states in FY 2016 (Ohio and Virginia) and two states in FY 2017 (Arkansas and Missouri) made or plan to make eligibility restrictions. These are mostly targeted restrictions that would affect small groups of beneficiaries. Arkansas is seeking a modification to its “Private Option” waiver to, effective January 1, 2017, eliminate retroactive eligibility for expansion enrollees; the waiver is pending at CMS. Missouri plans to begin the process of suspending its family planning waiver in FY 2017 following legislative restrictions in the FY 2017 appropriations bill.7 The FY 2016 reduction in eligibility for waiver services in Virginia for seriously mentally ill individuals (GAP waiver program) was partially restored in FY 2017.
Coverage Initiatives for the Criminal Justice Population
With the ACA Medicaid expansion to low-income adults, many individuals involved with the justice system are now eligible for Medicaid. Connecting these individuals to health coverage can facilitate their integration back into the community by increasing their ability to address health needs, which may contribute to greater stability in their lives as well as broader benefits to the individual and society as a whole. An increasing number of states have efforts underway to enroll eligible individuals moving into and out of the justice system into Medicaid. In April 2016 guidance, the Centers for Medicare and Medicaid Services (CMS) clarified that incarcerated individuals may be determined eligible for Medicaid and that the state Medicaid agency must accept applications and process renewals for incarcerated individuals.8 Although individuals may be enrolled in Medicaid while they are incarcerated, Medicaid cannot cover the cost of their care, except for inpatient services. In its recent guidance, CMS clarified who is considered an inmate of a public institution and therefore only able to receive Medicaid coverage for inpatient care.9 This survey asked states about a number of initiatives to promote Medicaid coverage for individuals involved with the criminal justice system (Exhibit 1 and Table 3).
The vast majority of states (44) had policies in place as of FY 2015 to obtain Medicaid reimbursement for inpatient care provided to incarcerated individuals who are Medicaid eligible. Four additional states implemented these policies in FY 2016 or plan to in FY 2017.
Given that the Medicaid expansion has significantly increased Medicaid eligibility among individuals moving into and out of the criminal justice system, many states are newly adopting or expanding initiatives to connect this population to Medicaid coverage. States are adopting policies to suspend Medicaid eligibility (rather than terminate eligibility) during incarceration. In FY 2015, 25 states had these policies in place for at least some individuals entering jail or prison, and 41 states are expected to have suspension policies by the end of FY 2017.
Medicaid and corrections agencies are also working together to help connect individuals to coverage as they are released from jail or prison back to the community. Some of these approaches include providing outreach and enrollment assistance pre-release, expedited enrollment processes for individuals being released, and Medicaid eligibility staff dedicated to processing applications for this population. A number of states have such initiatives in place and some states reported expanding these enrollment activities in FY 2016 or 2017. The most common expansions involve increasing the geographic scope of jail initiatives or increasing the number of prisons where eligibility assistance is provided.
The survey did not ask states about initiatives specific to parolees and individuals residing in halfway houses. However, Colorado, Connecticut, and the District of Columbia specifically mentioned initiatives to cover individuals residing in halfway houses.
| Exhibit 1: Coverage Initiatives for the Criminal Justice Population(# of States) | ||||
| Select Medicaid Coverage Policies for the Criminal Justice Population | In Place in FY 2015 | New FY 16or 17 | Expanded FY 16 or 17 | In place/ planned for FY 2017 |
| Medicaid coverage for inpatient care provided to incarcerated individuals | 44 | 4 | 4 | 48 |
| Medicaid outreach/assistance strategies to facilitate enrollment prior to release | 31 | 11 | 13 | 42 |
| Medicaid eligibility suspended (rather than terminated) for enrollees who become incarcerated (jails OR prisons) | 25 | 16 | 3 | 41 |
Arizona Medicaid and Corrections Policies
Arizona has implemented a number of strategies to increase coverage and access to care for the criminal justice population. The Arizona Health Care Cost Containment System (AHCCCS), the state’s Medicaid agency, has agreements with most counties, including the two largest, and the Arizona Department of Corrections to allow for suspension of enrollment for jail and prison inmates. Arizona also provides support to counties to help them connect to the state’s eligibility system and facilitate enrollment and train community-based organizations, providers, and others on using the system for application assistance. The two most populous counties in the state have arrangements to provide enrollment assistance to persons on probation and throughout other areas of the system. The state also provides an expedited eligibility determination process for uninsured inmates with critical health needs who are scheduled to be released. Arizona has mandated that all Regional Behavioral Health Authorities have a designated liaison for coordinating care for persons with serious mental illness who are transitioning from the justice system. There is expedited review for individuals being discharged with a medical or behavioral health need so that care can be coordinated promptly. The state is also looking into ways to access data obtained through assessments as part of the probation process and other information that will help coordinate care.
Medicaid Financed Births
For over three decades, Medicaid has been a key source of financing of births for low- and modest-income families. Women who would not otherwise be eligible can qualify for Medicaid coverage for pregnancy, delivery, and postpartum care due to higher income eligibility thresholds for pregnant women.10 Medicaid directors were asked to provide the most recent available data on the share of all births in their states that were financed by Medicaid. About half of states were able to provide data for calendar 2015 or fiscal year 2015.11 Other states generally provided data from 2013 or 2014. On average,12 states reported that Medicaid pays for just over 47 percent of all births. Eight states (Arkansas,13 Louisiana, Mississippi, Nevada, New Mexico, Oklahoma, South Carolina, and West Virginia) reported that Medicaid pays for 60 percent or more of all births in their state, while nine states reported that Medicaid finances less than 40 percent of all births (Iowa, Kansas, Nebraska, New Hampshire, North Dakota, Pennsylvania, Utah, Virginia, and Wyoming).
Premiums and Copayments
States have flexibility to charge limited premiums and cost-sharing in Medicaid, subject to federal parameters. Premiums are generally prohibited for beneficiaries with income below 150 percent FPL. Cost-sharing for people with income below 100 percent FPL is limited to “nominal” amounts specified in federal regulations, with higher levels allowed for beneficiaries at higher income levels. However, certain groups are exempt from cost-sharing, including mandatory eligible children, pregnant women, most children and adults with disabilities, people residing in institutions, and people receiving hospice care. In addition, certain services are exempt from cost-sharing: emergency services, preventive services for children, pregnancy-related services, and family planning services. Total Medicaid premiums and cost-sharing for a family cannot exceed 5 percent of the family’s income on a quarterly or monthly basis.14
Details about state actions related to premiums and copayments can be found in Table 4.
Premiums
Medicaid generally is not allowed to charge premiums to Medicaid beneficiaries with incomes at or below 150 percent FPL, although in limited cases certain populations, generally with income above 100 percent FPL, may be charged premiums (sometimes referred to as “buy-in” programs). Forty-four (44) states have buy-in programs for working people with disabilities and most of these states impose premiums.15 States also have the option to implement buy-in programs for children with disabilities. States that reported implementing premium-based programs under the Family Opportunity Act (FOA) for children with disabilities in families with incomes that otherwise exceed Medicaid limits include Colorado, Louisiana, North Dakota, and Texas.16 More recently, some states have received approval or were seeking approval to impose premiums under a Medicaid expansion waiver.
Seven states reported that they implemented or plan to implement new or increased premiums in FY 2016 or FY 2017 (Table 4). Three of these premium changes are related to individuals with disabilities. In FY 2016, Iowa increased Medicaid premiums for working people with disabilities. Two other states (Michigan in FY 2016 and Colorado in FY 2017) implemented or plan to implement expanded coverage and premiums for individuals with disabilities.
Five states (Arkansas, Indiana, Iowa, Michigan, and Montana) have received federal waivers to require premiums/monthly contributions for Medicaid expansion enrollees.17 In some cases, monthly contributions may be imposed in lieu of point-of-services copayments.18
Other implemented or proposed changes in premiums or contributions are for the Medicaid expansion populations.
- Montana implemented the Medicaid expansion in January 2016 under a waiver that requires monthly premiums up to 2 percent of household income for newly eligible adults from 51-138 percent FPL.
- Arkansas has waiver amendments pending that would take effect in calendar year 2017. The proposed waiver would replace the current income-based monthly contributions to “Health Independence Accounts,” which are to be used to fund copayments instead of paying at the point of service, with a premium requirement of 2 percent of income for those with incomes above 100 percent of FPL.
- Arizona had a waiver pending that would impose monthly premiums of 2 percent of income or $25, whichever is less, on all Medicaid expansion adults from 0-138 percent FPL, paid into health savings accounts. The waiver was approved on September 30, 2016 and allows for premiums at 2 percent of income for non-medically frail 100-138 percent FPL; individuals that comply with a healthy behavior program could have premiums eliminated for six months.
- Ohio submitted a waiver request in June 2016 to change its traditional expansion to the Healthy Ohio program which would impose monthly contributions, equal to the lesser of 2 percent of annual income or $99 per year, as a condition of eligibility for all beneficiaries except pregnant women and those with zero income. On September 9, 2016 CMS denied the state’s waiver request.
Kentucky has a waiver pending with CMS that would add premiums to its traditional Medicaid expansion program, along with other changes, but the proposed effective date is not until July 1, 2017 (FY 2018) so it is not captured in this report.
Copayment Requirements
Most state Medicaid programs require beneficiary copayments, but to varying degrees. Twelve (12) states reported changes to copayment requirements in either FY 2016 or FY 2017 (Table 4). Key changes are described below:
- Five states reported new or increased copayment requirements for the Medicaid expansion population: Montana implemented new requirements in FY 2016, Louisiana plans to do so in FY 2017, Michigan plans to double copayments for Healthy Michigan Plan enrollees with incomes above 100 percent FPL in FY 2017, and New Hampshire planned copayment requirements for FY 2017 but these changes are included in waivers pending at CMS. The Ohio waiver that was denied would have increased copayments. The waiver approved in Arizona would allow for copayments (within state plan permissible levels) to be charged retrospectively for certain services such as non-emergency use of the emergency department, seeing a specialist without a referral, and use of brand name drugs when there is an available generic. Beneficiaries would get a quarterly invoice and be charged a monthly amount up to 3 percent of monthly income.
- In FY 2016, Indiana restored copayments for aged, blind, and disabled enrollees in managed care and Minnesota decreased copayment amounts for the Medical Assistance for Employed Persons with Disabilities (MA-EPD) group.
- In FY 2017, New Mexico plans to implement new copayments for non-emergency use of the emergency department for all Medicaid enrollees and new pharmacy copayments for all populations for brand name prescriptions when there is a less expensive generic equivalent available.19
- Four states are eliminating one or more copayment provisions in either FY 2016 or FY 2017 (North Dakota, New York, Oregon, and Vermont).
Table 1: Changes to Eligibility Standards in all 50 States and DC, FY 2016 and FY 2017
| Eligibility Standard Changes | ||||||
| States | FY 2016 | FY 2017 | ||||
| (+) | (-) | (#) | (+) | (-) | (#) | |
| Alabama | ||||||
| Alaska | X – Medicaid Expansion | |||||
| Arizona | ||||||
| Arkansas | X | |||||
| California | ||||||
| Colorado | X | X | ||||
| Connecticut | X | |||||
| Delaware | ||||||
| DC | X | |||||
| Florida | X | X | ||||
| Georgia | ||||||
| Hawaii | ||||||
| Idaho | ||||||
| Illinois | ||||||
| Indiana | ||||||
| Iowa | ||||||
| Kansas | ||||||
| Kentucky | ||||||
| Louisiana | X-Medicaid Expansion | X | ||||
| Maine | X | |||||
| Maryland | X | |||||
| Massachusetts | ||||||
| Michigan | X | X | ||||
| Minnesota | X | |||||
| Mississippi | ||||||
| Missouri | X | |||||
| Montana | X – Medicaid Expansion | |||||
| Nebraska | ||||||
| Nevada | ||||||
| New Hampshire | X | |||||
| New Jersey | ||||||
| New Mexico | ||||||
| New York | ||||||
| North Carolina | ||||||
| North Dakota | ||||||
| Ohio | X | X | X | |||
| Oklahoma | ||||||
| Oregon | ||||||
| Pennsylvania | X | |||||
| Rhode Island | ||||||
| South Carolina | ||||||
| South Dakota | ||||||
| Tennessee | ||||||
| Texas | ||||||
| Utah | X | X | X | |||
| Vermont | X | |||||
| Virginia | X | X | ||||
| Washington | ||||||
| West Virginia | ||||||
| Wisconsin | ||||||
| Wyoming | ||||||
| Totals | 7 | 2 | 7 | 7 | 2 | 3 |
| NOTES: Positive changes from the beneficiary’s perspective that were counted in this report are denoted with (+). Negative changes from the beneficiary’s perspective that were counted in this report are denoted with (-). Several states made reductions to Medicaid eligibility pathways in response to the availability of other coverage options (including Marketplace and/or Medicaid expansion coverage); these changes were denoted as (#) since most affected beneficiaries will have access to coverage through an alternative pathway.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||
Table 2: States Reporting Eligibility Changes in FY 2016 and FY 2017
| State | Fiscal Year | Eligibility Changes |
| Alaska | 2016 | Adults (+): Medicaid expansion on September 1, 2015 (estimated first year enrollment of 20,100). |
| Arkansas | 2017 | Adults (-): Pending waiver would eliminate retroactive eligibility for expansion population. |
| Colorado | 2016 | Children (+): Implement the CHIPRA option to eliminate the 5-year bar on Medicaid eligibility for legally-residing immigrant children (estimated to affect 1,699 children). |
| 2017 | Adults (+): Implementing annualized income for eligibility for other adults (affects 20,430 individuals). | |
| Connecticut | 2016 | Adults (#): Effective August 1, 2015 the income limits for HUSKY A parents and caretaker relatives were reduced from 201% FPL to 155% FPL. |
| DC | 2016 | Adults (#): Section 1115 Childless Adult waiver expired 12/31/2015. Adults with incomes from 133% to 210% FPL were transitioned from a Medicaid waiver to Medicaid state plan (8,500 individuals). |
| Florida | 2016 | Aged and Disabled (+): Increased the minimum monthly maintenance income allowance and excess standard for community spouses of institutionalized people. (The number of nursing facility residents eligible for Medicaid is also affected by 2016 cost of living adjustments and increases in the average private pay nursing facility used to set LTSS policy.) Children (+): Implement the CHIPRA option to eliminate the 5-year bar on Medicaid eligibility for legally-residing immigrant children. |
| 2017 | Aged and Disabled (+): Increased the minimum monthly maintenance income allowance and excess standard for community spouses of institutionalized people. (The number of nursing facility residents eligible for Medicaid is also affected by 2017 cost of living adjustments and increases in the average private pay nursing facility used to set LTSS policy.) | |
| Louisiana | 2017 | Adults (+): Implemented Medicaid expansion on July 1, 2016 (375,000 individuals). Adults (#): Effective July 1, 2016, 127,109 people covered in the Family Planning State Plan Amendment (SPA) were enrolled in the new Adult Group. The people remaining in the Family Planning SPA do not qualify for the Adult Group. |
| Maine | 2017 | Adults (+): Plan to increase eligibility under family planning pathway to 209% FPL in FY 2017. |
| Maryland | 2016 | Adults (#): Breast and Cervical Cancer Treatment Program continued only for enrollees in active treatment (400 individuals). |
| Michigan | 2016 | Adults (#): Family planning waiver ended 6/30/2016. Children & Pregnant Women (+): Flint Waiver Group Waiver extends Medicaid eligibility to 400% FPL for children and pregnant women exposed to tainted Flint water (up to 15,000 individuals). Aged & Disabled (+): Increased income and asset limits for working people with disabilities, effective 10/1/15. |
| Minnesota | 2017 | Aged & Disabled (+): Increased income standard for the medically needy from 75% FPL to 80% FPL on 7/1/2016. |
| Missouri | 2017 | Adults (-): Based on restrictions in the FY 2017 appropriation bill, Missouri will begin the process of suspending the Family Planning 1115 waiver. Expected transition 2/1/2017. |
| Montana | 2016 | Adults (+): Implemented ACA expansion via a waiver. Implemented 12-month continuous eligibility for newly eligible adults as part of the waiver. Effective 1/1/2016. |
| New Hampshire | 2017 | Adults (#): State legislation calls for ending the Breast and Cervical Cancer Treatment Program for new enrollees in FY 2017 while allowing current enrollees to continue treatment. |
| Ohio | 2016 | Adults (#): Ended Family Planning coverage group as of 1/1/16. Other (-): Change in transitional Medicaid for families from twelve-months eligibility to six-months eligibility with possible coverage for two reporting periods. |
| 2017 | Aged & Disabled (#): Conversion from 209(b) to 1634 for SSI related groups. | |
| Pennsylvania | 2016 | Adults (#): Medically Needy Spend-Down for Parents and People with Disabilities was restricted to individuals under the age of 21 as part of Healthy PA implementation. However, it was reinstated in March 2016 and is once again available to these adults. |
| Utah | 2016 | Children (+): Medically Complex Children’s Waiver (165 children). Children (#): Autism Waiver enrollment closed since autism services were added to the State Plan. |
| 2017 | Children (+): Implementing the CHIPRA option to eliminate the 5-year bar on Medicaid eligibility for legally-residing immigrant children (estimated to affect 750 children). Adults (+): Proposed limited adult expansion: Parents of dependent children with incomes 40% to 60% FPL; adults without dependent children with incomes up to 5% FPL meeting certain criteria (9,000 to 11,000 individuals). | |
| Vermont | 2016 | Aged & Disabled (+): Increased asset limits and income disregards for working people with disabilities (70 individuals). |
| Virginia | 2016 | Aged & Disabled (-): Reduced eligibility from 100% to 60% FPL for waiver services for people with serious mental illness (GAP waiver program). |
| 2017 | Aged & Disabled (+): Increased eligibility from 60% to 80% FPL for waiver services for people with serious mental illness (GAP waiver program). | |
| NOTE: Positive changes from the beneficiary’s perspective that were counted in this report are denoted with (+). Negative changes from the beneficiary’s perspective that were counted in this report are denoted with (-). Reductions to Medicaid eligibility pathways in response to the availability of other coverage options (including Marketplace or Medicaid expansion coverage) were denoted as (#). | ||
Table 3: Corrections-Related Enrollment Policies in all 50 States and DC, FY 2015-FY 2017
| States | Medicaid Coverage For Inpatient Care Provided to Incarcerated Individuals | Medicaid Outreach/Assistance Strategies to Facilitate Enrollment Prior to Release | Medicaid Eligibility Suspended Rather Than Terminated For Enrollees Who Become Incarcerated (Jails or Prisons) | |||||||||
| In place FY 2015 | NewFY 16/17 | Expanded FY16/17 | In place/planned for FY17 | In place FY 2015 | NewFY 16/17 | Expanded FY16/17 | In place/planned for FY17 | In place FY 2015 | NewFY 16/17 | Expanded FY16/17 | In place/planned for FY17 | |
| Alabama | X | X | X | X | X | X | X | X | ||||
| Alaska | X | X | X | X | X | X | X | |||||
| Arizona | X | X | X | X | X | X | ||||||
| Arkansas | X | X | X | X | X | X | ||||||
| California | X | X | X | X | X | X | ||||||
| Colorado | X | X | X | X | X | |||||||
| Connecticut | X | X | X | X | X | X | ||||||
| Delaware | X | X | X | X | X | X | ||||||
| DC | X | X | X | X | X | X | ||||||
| Florida | X | X | X | X | ||||||||
| Georgia | X | X | ||||||||||
| Hawaii | X | X | X | X | X | X | ||||||
| Idaho | X | X | X | X | ||||||||
| Illinois | X | X | X | X | X | X | ||||||
| Indiana | X | X | X | X | X | X | ||||||
| Iowa | X | X | X | X | X | X | ||||||
| Kansas | X | X | X | X | ||||||||
| Kentucky | X | X | X | X | X | X | X | |||||
| Louisiana | X | X | X | X | X | X | X | |||||
| Maine | X | X | X | X | ||||||||
| Maryland | X | X | X | X | X | X | X | |||||
| Massachusetts | X | X | X | X | X | X | ||||||
| Michigan | X | X | X | X | X | X | ||||||
| Minnesota | X | X | X | X | X | X | ||||||
| Mississippi | X | X | X | X | X | X | X | |||||
| Missouri | X | X | X | X | ||||||||
| Montana | X | X | X | X | X | X | X | X | ||||
| Nebraska | X | X | X | X | ||||||||
| Nevada | X | X | ||||||||||
| New Hampshire | X | X | X | X | X | X | X | |||||
| New Jersey | X | X | X | X | X | X | ||||||
| New Mexico | X | X | X | X | X | X | X | X | X | |||
| New York | X | X | X | X | X | X | X | |||||
| North Carolina | X | X | X | X | X | X | ||||||
| North Dakota | X | X | X | X | X | X | X | |||||
| Ohio | X | X | X | X | X | X | X | |||||
| Oklahoma | X | X | ||||||||||
| Oregon | X | X | X | X | X | X | ||||||
| Pennsylvania | X | X | X | X | X | X | ||||||
| Rhode Island | X | X | X | X | X | X | X | |||||
| South Carolina | X | X | X | X | X | X | X | X | ||||
| South Dakota | X | X | X | X | ||||||||
| Tennessee | X | X | X | X | ||||||||
| Texas | X | X | X | X | X | X | ||||||
| Utah | X | X | X | X | ||||||||
| Vermont | X | X | ||||||||||
| Virginia | X | X | X | X | X | X | ||||||
| Washington | X | X | X | X | X | X | ||||||
| West Virginia | X | X | X | X | X | X | ||||||
| Wisconsin | X | X | X | X | ||||||||
| Wyoming | ||||||||||||
| Totals | 44 | 4 | 4 | 48 | 31 | 11 | 13 | 42 | 25 | 16 | 3 | 41 |
| NOTES: States were asked to indicate if any of the above corrections-related policies were in effect in FY 2015 and if they were newly adopted or expanded in FY 2016 or FY 2017. The “in place/planned for FY 2017” columns indicate states that either had a given policy in place as of FY 2015, newly implemented the policy in FY 2016, or plan to newly implement the policy in FY 2017. States with “Medicaid outreach assistance strategies to facilitate enrollment prior to release” include those with Medicaid led/coordinated efforts on outreach/enrollment assistance prior to release, expedited enrollment prior to release (e.g. presumptive eligibility), and/or Medicaid eligibility staff devoted to processing determinations prior to release.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||||||||
Table 4: States Reporting Premium and Copayment Actions Taken in FY 2016 and FY 2017
| State | Fiscal Year | Premium and Copayment Changes |
| Arizona | 2017 | Premiums (New non-medically frail adults 100-138% FPL): Waiver approved September 30, 2016 would allow premiums of 2 percent of income for adults with incomes 100-133% FPL. Individuals that comply with a health behavior program could have premiums eliminated for six months. Copayments (New non-medically frail adults 100-138% FPL): The approved waiver would allow for copayments (within state plan permissible levels) to be charged retrospectively for certain services such as non-emergency use of the emergency department, seeing a specialist without a referral and use of brand name drugs when there is an available generic. Beneficiaries would get a quarterly invoice and be charged a monthly amount up to 3% of monthly income. Premiums and copayments together would be limited to the 5% cap of household income per quarter. |
| Arkansas | 2017 | Premiums (New for expansion population): Pending “Arkansas Works” waiver amendments would replace current required contributions to “Health Independence Accounts” in lieu of point-of-service copayments with required monthly premiums of 2% of household income for individuals between 100 and 138% FPL |
| Colorado | 2017 | Premiums (New option for LTSS populations): Implement a Medicaid Buy-In program for 3 HCBS waivers (7/1/16). |
| Indiana | 2016 | Copayments (New): Restore copayments for ABD enrollees in managed care (Jan 2016). |
| Iowa | 2016 | Premiums (Increased): The premium for working people with disabilities is based on state employee health insurance premium which increased in 2016. Unknown for 2017. |
| Louisiana | 2017 | Copayments (New for expansion population): New cost-sharing requirements for the expansion population are the same as those in place for the rest of the Medicaid population. |
| Michigan | 2016 | Premiums (Increased): Premiums for the Freedom to Work population are now calculated using a percent of a beneficiary’s MAGI income (10/1/2015). |
| 2017 | Copayments (Increase): Increase in prescription, hospital, and office visit copays for Healthy Michigan Plan enrollees with incomes above 100% FPL. | |
| Minnesota | 2016 | Premiums (Decreased): Minimum premium for Medical Assistance for Employed Persons with Disabilities (MA-EPD) reduced (Sep 2015). Copayments (Decreased): Decreased copayment amounts for MA-EPD group (Sep 2015). |
| Montana | 2016 | Premiums (New only for expansion population): Newly eligible adults between 51 and 138% FPL required to pay monthly premiums up to 2% of household income (1/1/2016). Copayments (New for expansion population): Childless adults with incomes below 138% FPL and parents with incomes between 51% and 138% FPL (1/1/2016). Copayments (Neutral): Cost sharing for adults with incomes up to 50% FPL was standardized with some amounts increased and some decreased (6/1/2016). |
| New Hampshire | 2016 | Copayments (Increased): Pharmacy copayments for the expansion population (those above 100% FPL) are being increased from $1/$4 (generic/brand) to $2/$8 (Jan 2016). |
| 2017 | Copayments (New only for expansion population): Pending waiver would subject expansion population to copayments on some medical services. | |
| New Mexico | 2017 | Copayments (New for all populations): Copays for non-emergency use of the emergency department (1/1/2017 target date). Copayments (New for all populations): Copays for brand-name prescriptions when there is a less expensive generic equivalent medicine available (1/1/2017 target date). |
| New York | 2016 | Copayments (Elimination): Exemption from Medicaid co-pays for members with incomes below 100% FPL, hospice patients, and American Indians/Alaskan Natives who have never received a service from IHS, tribal health programs, or under contract health services referral (10/1/15). |
| North Dakota | 2017 | Copayments (Elimination): Higher copayment for non-emergency use of the ER will be eliminated (1/1/2017). |
| Ohio | 2017 | Premiums (New and would apply to all Medicaid beneficiaries except pregnant women and individuals with zero income): Waiver request to impose monthly premiums (the lesser of 2% of income or $99 per year). CMS denied Ohio’s pending waiver in September 2016. Copayments (Increase): Healthy Ohio 1115 waiver would increase copayments for all beneficiaries covered by the waiver at the maximum amounts allowable under federal law and copayments would be paid into a Health Savings Account and paid from that account at point of service. CMS denied Ohio’s pending waiver in September 2016. |
| Oregon | 2017 | Copayments (Elimination): Copayments are being eliminated for preventive services for all Medicaid groups (1/1/2017). |
| Vermont | 2017 | Copayments (Elimination): Remove copays for sexual assault-related services for all Medicaid groups (10/1/2016). |
| NOTE: New premiums or copayments as well as new requirements such as making copayments enforceable are denoted as (New). Increases in existing premiums or copayments are denoted as (Increased), while decreases are denoted as (Decreased) and eliminations are denoted as (Eliminated). | ||
Report: Managed Care Initiatives
Key Section Findings
- As of July 2016, a total of 39 states had contracts with comprehensive risk-based managed care organizations (MCOs). Among these states, 28 states reported that 75 percent or more of their beneficiaries were enrolled in MCOs as of July 1, 2016 (up from 21 states in last year’s survey), including four of the five states with the largest total Medicaid enrollment across the country.20 (This section focuses on MCOs for acute care; managed long-term services and supports (MLTSS) is discussed in the long-term services and supports (LTSS) section.)
- Some states treat special populations differently in terms of mandatory versus voluntary MCO enrollment. Of the special populations the survey asked about, pregnant women were the group most likely to be enrolled into MCOs on a mandatory basis (28 states), while persons with intellectual or developmental disabilities (ID/DD) were least likely to be enrolled on mandatory basis (10 states) and also most likely to be entirely excluded from MCO enrollment (7 states).
- In both FY 2016 and in FY 2017, states continued to take actions to increase enrollment in risk-based managed care, most commonly by enrolling additional eligibility groups. In addition, three states (Iowa, Rhode Island and West Virginia) terminated Primary Care Case Management (PCCM) programs in either FY 2016 or FY 2017 and shifted those populations into risk-based managed care. Alabama plans to implement a new MCO program in FY 2017 and Missouri plans to expand its MCO program statewide in FY 2017.
- About half of the 39 MCO states reported that four behavioral health service types (specialty outpatient mental health services, inpatient mental health services, and outpatient and inpatient substance use disorder (SUD) services) were carved into their MCO contracts as of July 1, 2016, with specialty outpatient mental health services somewhat less likely to be carved in.
- States are using managed care to advance quality and alternative payment models and to help screen for social needs. In FY 2016, a total of 17 states implemented new or expanded quality initiatives, and 17 states plan to do so in FY 2017. In FY 2016, five states identified targets in MCO contracts for the use of alternative provider payment models; 10 additional states intend to do so in FY 2017. Twenty-six (26) states reported requiring or encouraging MCOs to screen for social needs and provide referrals to other services in FY 2016 and four states intend to do so in FY 2017.
- Five states with MCO contracts report that they encourage or require MCOs to provide care coordination services to enrollees prior to release from incarceration (Arizona, Iowa, Kentucky, New Mexico and Ohio), and 10 states intend to add such requirements in FY 2017.
- Twenty (20) states reported specifying a minimum MLR for all or some plans, most often equal to or greater than 85 percent. Thirteen (13) states always require some form of MCO remittance if the minimum MLR is not achieved.
Tables 5 through 9 include more detail on the populations covered under managed care (Tables 5 and 6), behavioral health services covered under MCOs (Table 7), managed care quality initiatives (Table 8), and MLR (Table 9).
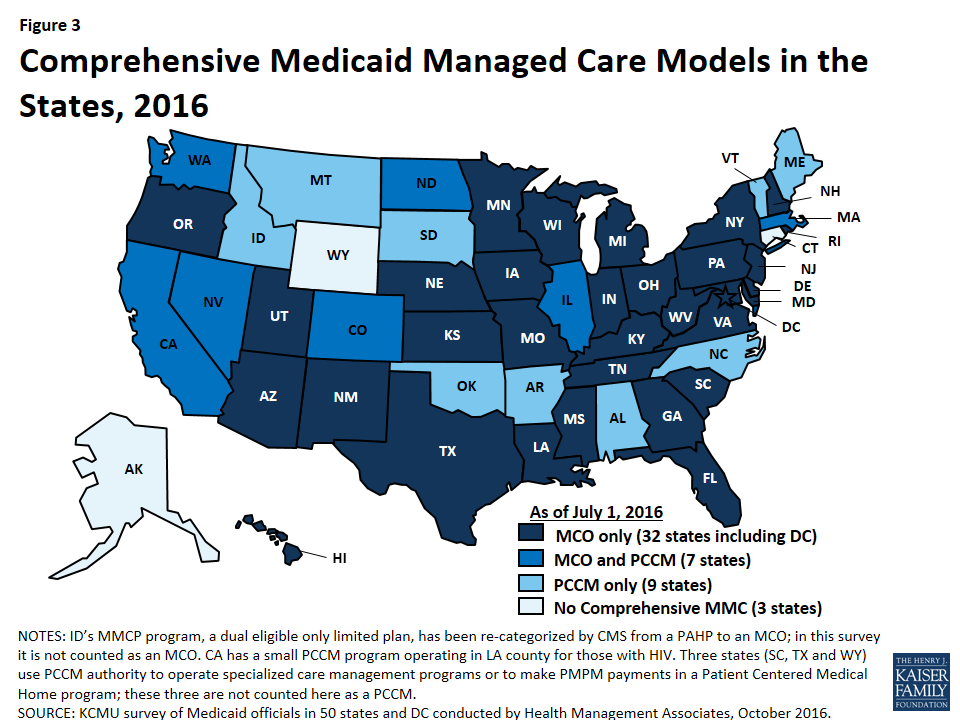
Managed care remains the predominant delivery system for Medicaid in most states. As of July 2016, all states except three – Alaska, Connecticut and Wyoming– had in place some form of managed care.21 Across the 48 states with some form of managed care, 39 had contracts with comprehensive risk-based managed care organizations (MCOs), unchanged from July 1, 2015. Three states (Iowa, Rhode Island, and West Virginia) reported ending their Primary Care Case Management (PCCM) programs leaving 16 states that administered a PCCM program as of July 1, 2016, down from 19 states a year earlier. PCCM is a managed fee-for-service (FFS) based system in which beneficiaries are enrolled with a primary care provider who is paid a small monthly fee to provide case management services in addition to primary care.
Of the 48 states that operate some form of managed care, seven operate both MCOs and a PCCM program while 32 states operate MCOs only and nine states operate PCCM programs only22 (Figure 3). Wyoming, one of the three states without any managed care (i.e., without either MCOs or a PCCM program), does operate a limited-benefit risk-based prepaid health plan (PHP). In total, 24 states (including Wyoming) contracted with one or more PHPs to provide selected Medicaid benefits, such as behavioral health care, dental care, maternity care, non-emergency medical transportation, LTSS, or other benefits.
Populations Covered by Risk-Based Managed Care
The share of Medicaid beneficiaries enrolled in MCOs or PCCM programs or remaining in FFS for their acute care varies widely by state. However, the share of Medicaid beneficiaries enrolled in MCOs has steadily increased as states have expanded their managed care programs to new regions and new populations and made MCO enrollment mandatory for additional eligibility groups. The survey asked states to indicate the approximate share of specific Medicaid populations who receive their acute care in MCOs, PCCM programs, and FFS. As shown in Figure 4, among the 39 states with MCOs, 28 states reported that 75 percent or more of their Medicaid beneficiaries were enrolled in MCOs as of July 1, 2016 (up from 21 states in last year’s survey), including four of the five states with the largest total Medicaid enrollment. These four states (California, New York, Texas, and Florida) account for nearly four out of every 10 Medicaid beneficiaries across the country (Figure 4 and Table 5).23
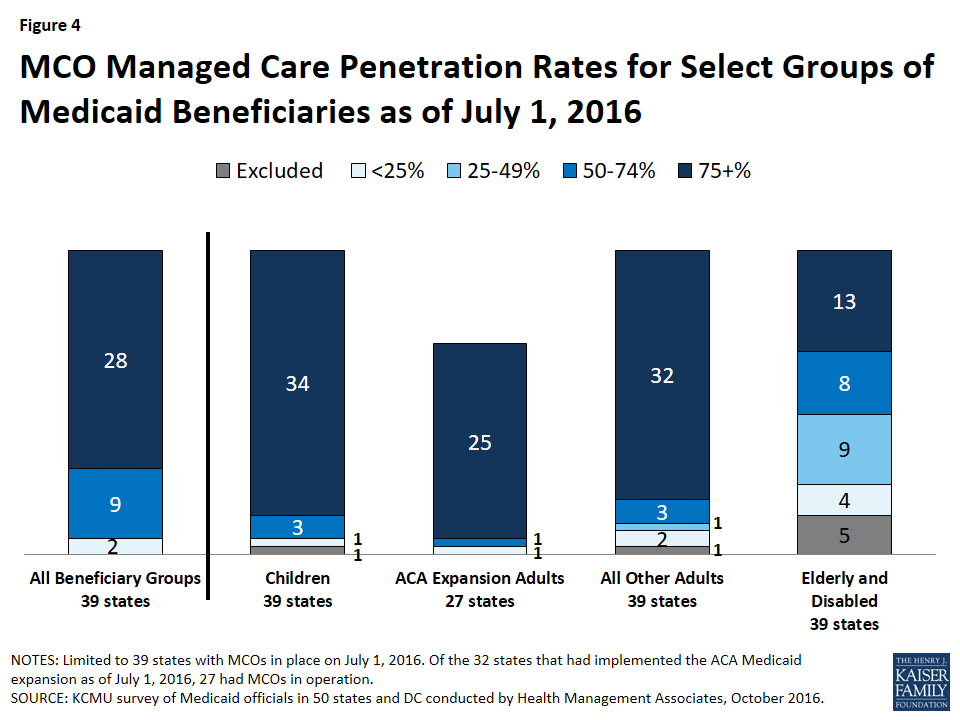
Children and adults (particularly those enrolled through the ACA Medicaid expansion) are much more likely to be enrolled in an MCO than elderly Medicaid beneficiaries or those with disabilities. Thirty-four (34) of the 39 MCO states covered 75 percent or more of all children through MCOs. Thirty-two (32) of the 39 MCO states covered 75 percent or more of low-income adults in pre-ACA expansion groups (e.g., parents, pregnant women) through MCOs. The elderly and people with disabilities were the group least likely to be covered through managed care contracts, with only 13 of the 39 MCO states covering 75 percent or more such enrollees through MCOs (Figure 4).
Of the 32 states that had implemented the ACA Medicaid expansion as of July 1, 2016, 27 were using MCOs to cover newly eligible adults. (The five Medicaid expansion states without risk-based managed care were Alaska, Arkansas, Connecticut, Montana, and Vermont.) The large majority (25) of these 27 states covered more than 75 percent of beneficiaries in this group through risk-based managed care. The remaining two states, which reported less than 75 percent MCO penetration for this group, were Colorado and Illinois.
Seven of the 16 states with PCCM programs also contract with MCOs. In most of these states, MCOs cover a larger share of beneficiaries than PCCM programs. However, Colorado and North Dakota are exceptions: as of July 1, 2016, a majority of Colorado’s enrollees were in the PCCM program, which is the foundation of the state’s Accountable Care Collaboratives, and approximately half (49 percent) of enrollees in North Dakota were enrolled in the PCCM program.
Populations with Special Needs
This year’s survey also asked states with MCOs whether, as of July 1, 2016, certain subpopulations with special needs were enrolled in MCOs for their acute care services on a mandatory or voluntary basis or were always excluded. On the survey, states selected from “always mandatory,” “always voluntary,” “varies (by geography or other factor),” or “always excluded” for the following populations: pregnant women, foster children, persons with intellectual and developmental disabilities (ID/DD), children with special health care needs (CSHCNs), adults with serious mental illness (SMI) and adults with physical disabilities. As shown in Exhibit 2 (and Table 6) below, pregnant women were the group most likely to be enrolled on a mandatory basis (28 states) while persons with ID/DD were least likely to be enrolled on mandatory basis (10 states) and also most likely to be excluded from MCO enrollment (7 states). Foster children were the group most likely to be enrolled on a voluntary basis (10 states) (although they were enrolled on a mandatory basis in a larger number of states).
Among states indicating that the enrollment approach for a given group or groups varied, geographic location and LTSS eligibility were the primary bases of variation. Six states (Colorado, Illinois, Missouri, Nevada, Utah, and Washington) specifically mentioned geographic variations and five states (Indiana, Kentucky, Louisiana, Ohio, and Texas) mentioned variations based on LTSS eligibility (or “level of care”).
| Exhibit 2: MCO Enrollment of Populations with Special Needs, July 1, 2016(# of States) | ||||||
| Pregnant women | Foster children | Persons with ID/DD | CSHCNs | SMI Adults | Adults w/ physical disabilities | |
| Always mandatory24 | 28 | 16 | 10 | 16 | 16 | 16 |
| Always voluntary | 1 | 10 | 7 | 3 | 3 | 3 |
| Varies (by geography or other factor) | 9 | 10 | 15 | 18 | 17 | 15 |
| Always excluded | 1 | 3 | 7 | 2 | 3 | 5 |
Acute Care Managed Care Population Changes
In both FY 2016 and FY 2017, states continued to take actions to increase enrollment in acute care managed care, although fewer states reported doing so than in the last two surveys (in 2014 and 2015) reflecting full or nearly full MCO saturation in a growing number of states. Of the 39 states with MCOs, a total of 16 states indicated that they made specific policy changes in either FY 2016 (11 states) or FY 2017 (11 states) to increase the number of enrollees in MCOs through geographic expansions, voluntary or mandatory enrollment of new groups into MCOs, or mandatory enrollment of specific eligibility groups that were formerly enrolled on a voluntary basis (Exhibit 3).
| Exhibit 3: Medicaid Acute Care Managed Care Population Expansions, FY 2016 and FY 2017 | ||
| FY 2016 | FY 2017 | |
| Geographic Expansions | IA, MS, UT | AL, CO, MS, MO |
| New Population Groups Added | CA, IA, LA, MS, NE, NY, WA, WV | AL, LA, NE, OH, RI, TX, UT, WV |
| Voluntary to Mandatory Enrollment | NH, RI, UT | |
Some of the notable acute care MCO expansions include:
- Three states (Iowa, Rhode Island, and West Virginia) terminated their PCCM programs in FY 2016 and shifted those populations into risk-based managed care. Iowa implemented statewide MCO coverage for almost all Medicaid enrollees on April 1, 2016 and ended its PCCM and behavioral health PHP programs.25 Rhode Island eliminated its PCCM program for adults with disabilities (Connect Care Choice) in FY 2016 and transitioned the enrollees to MCOs. West Virginia ended its small PCCM program and also transitioned its SSI population from FFS to mandatory MCO enrollment in July 2016.
- Alabama plans to implement mandatory MCO enrollment for nearly all Medicaid enrollees (currently served through PCCM and FFS) in FY 2017, although the state recently requested CMS approval to delay implementation until July 1, 2017.26 (Alabama’s fiscal year ends on September 30.)
- Missouri will extend its MCO program geographically statewide on May 1, 2017 for the populations eligible for managed care under current rules.
Geographic expansions of MCO service areas were reported in three states in FY 2016 (Iowa, Mississippi, and Utah), and in four states for FY 2017 (Alabama, Colorado, Mississippi, and Missouri).
In FY 2016 and FY 2017, states expanded MCO enrollment (either voluntary or mandatory) to additional groups. Some states added multiple groups. Some groups that states added or are planning to add include: foster care or adoption assistance children (Louisiana, Nebraska, Ohio, and Texas); persons eligible for LTSS (Nebraska, New York, and Washington); ACA expansion, newly eligible adult group (Louisiana and West Virginia); Breast and Cervical Cancer Treatment Program group (Ohio and Texas); children with special health care needs (Louisiana and Ohio); pregnant women (California); Native Americans (Louisiana); children (Mississippi); SSI population (West Virginia); persons with intellectual and developmental disabilities (Ohio).
Three states made enrollment mandatory in FY 2016 for specific eligibility groups that were formerly enrolled on a voluntary basis: New Hampshire (dual eligibles, disabled children, and foster care children), Rhode Island (SSI), and Utah (enrollees in nine new mandatory counties).
Although outside the period covered by this survey report, Oklahoma reported plans to implement risk-based managed care for the aged, blind, and disabled population after FY 2017.
Services Covered Under MCO Contracts
Behavioral Health Services Covered Under MCO Contracts
Although MCOs are at risk financially for providing a comprehensive set of acute care services, nearly all states exclude or “carve-out” certain services from their MCO contracts, most commonly behavioral health services. In this year’s survey, states with acute care MCOs were asked to indicate whether specialty outpatient mental health services, inpatient mental health services, and outpatient and inpatient substance use disorder (SUD) services are always carved-in (i.e., virtually all services are covered by the MCO), always carved-out (to PHP or FFS), or carve-in status varies by geographic or other factors.
For purposes of this survey, “specialty outpatient mental health” services mean services used by adults with Serious Mental Illness (SMI) and/or youth with serious emotional disturbance (SED), commonly provided by specialty providers such as community mental health centers. Depending on the service, about half of the 39 MCO states reported that specific behavioral health service types were carved into their MCO contracts, with specialty outpatient mental health services somewhat less likely to be carved in (Exhibit 4 and Table 7).
Of the nine states that indicated variation in the carve-in status of some behavioral health services, Texas and Washington cited geographic variation for all four service types; Ohio and Virginia indicated that outpatient mental health and SUD services were carved in only for dual eligibles in their Financial Alignment Demonstrations; Arizona reported that all four service types were carved out for children with a severe emotional disturbance; Missouri indicated variation based on diagnosis for children; New Jersey stated that inpatient medical detoxification services were always carved in while non-medical detoxification and short-term residential treatment for SUD were always carved out; South Carolina mentioned variation in specialty outpatient mental health services based on eligibility category, and also reported that psychiatric services in a freestanding hospital or dedicated unit are carved out, but psychiatric care during a hospital stay is carved in, and Wisconsin indicated that some specialty outpatient mental health services are carved in while others are carved out.
| Exhibit 4: MCO Coverage of Behavioral Health, July 1, 2016(# of States) | ||||
| Specialty Outpatient MH | Inpatient MH | Outpatient SUD | Inpatient SUD | |
| Always carved-in | 20 | 24 | 24 | 26 |
| Always carved-out | 12 | 10 | 9 | 8 |
| Varies (by geography or other factor) | 7 | 5 | 6 | 5 |
Seven states in both FY 2016 (Arizona, Louisiana, New Hampshire, New York, Rhode Island, Washington, and West Virginia) and FY 2017 (Alabama, Nebraska, New York, Rhode Island, South Carolina, Virginia and Washington) reported a new action to carve in, or plans to carve in, behavioral health services into their MCO contracts. Also, Wisconsin reported that, in an effort to promote care coordination, beginning in FY 2016, members receiving medication-assisted treatment for opioid addiction are no longer exempt from managed care enrollment, except for continuity of care reasons.
Institutions for Mental Diseases (IMD) Rule Change
The recently finalized Medicaid Managed Care final rule27 allows states (under the authority for health plans to cover services “in lieu of” those available under the Medicaid state plan), to receive federal matching funds for capitation payments on behalf of adults who receive inpatient psychiatric or substance use disorder treatment or crisis residential services in an IMD for no more than 15 days in a month.28
States were asked in the survey whether they planned to use this new authority. Of the 39 states with MCOs plus Alabama (which plans to implement MCOs in FY 2017), 16 states answered “yes,” six answered “no,” and 18 states said a decision had not yet been made. Maryland said “no” but indicated that the state had applied for an IMD waiver to offer residential services for persons with an SUD diagnosis.
Additional Services
States with MCO contracts reported that plans in their states may offer a range of services beyond those described in the state plan or waivers. Twelve (12) states reported that MCOs in their states provide limited or enhanced adult dental services beyond contractually required state plan benefits. Nine states reported enhanced vision services for adults. Vermont reported enhanced mental health and substance use disorder services and the District of Columbia reported telemedicine for behavioral health services. States also reported a wide range of other extra services, including car seats, wireless cell phones or smart phone applications, gym memberships, smoking cessation supports, nutrition education, transportation, adult vaccines, health and wellness outreach centers, equine therapy, and Native American healing benefits. Some states (including Arizona and California) reported that MCOs are not required to report non-covered services to the state, but have the discretion to offer them when the plan judges an additional service to be beneficial and cost-effective. New Mexico allows MCOs to provide additional services, subject to state approval.
Managed Care Quality, Contracts Requirements and Administration
Quality Initiatives
States procure MCO contracts using different approaches. States may set a capitation rate that meets the test of actuarial-soundness and contract with any MCO willing to meet state and federal requirements. Most states now competitively bid for Medicaid MCOs, in part because the dollar value is so large – in some cases the largest procurement ever undertaken by the state. In these procurements, states can specify requirements and criteria that go beyond price such as value-based payments, specific policy priorities such as improving birth outcomes, or strategies to address social determinants of health, as well as specific performance and quality criteria. In this year’s survey, states were asked if they used, or planned to use, National Committee for Quality Assurance’s (NCQA’s) Healthcare Effectiveness Data and Information Set (HEDIS®) scores as criteria for selecting MCOs to contract with. Of the 39 states with MCOs, 14 answered “yes.”
After contracts are procured, all states with MCO programs track one or more quality measures and require other health plan activities to improve health care outcomes and plan performance. States were asked to indicate whether they had selected quality strategies in place in FY 2015, to establish a baseline, and also to indicate newly added or expanded initiatives in FY 2016 or FY 2017. Thirty-six (36) of the 39 MCO states reported one or more select MCO quality initiatives in place in FY 2015. The most common strategies were the collection of adult and child quality measures and pay for performance (Figure 5 and Table 8).
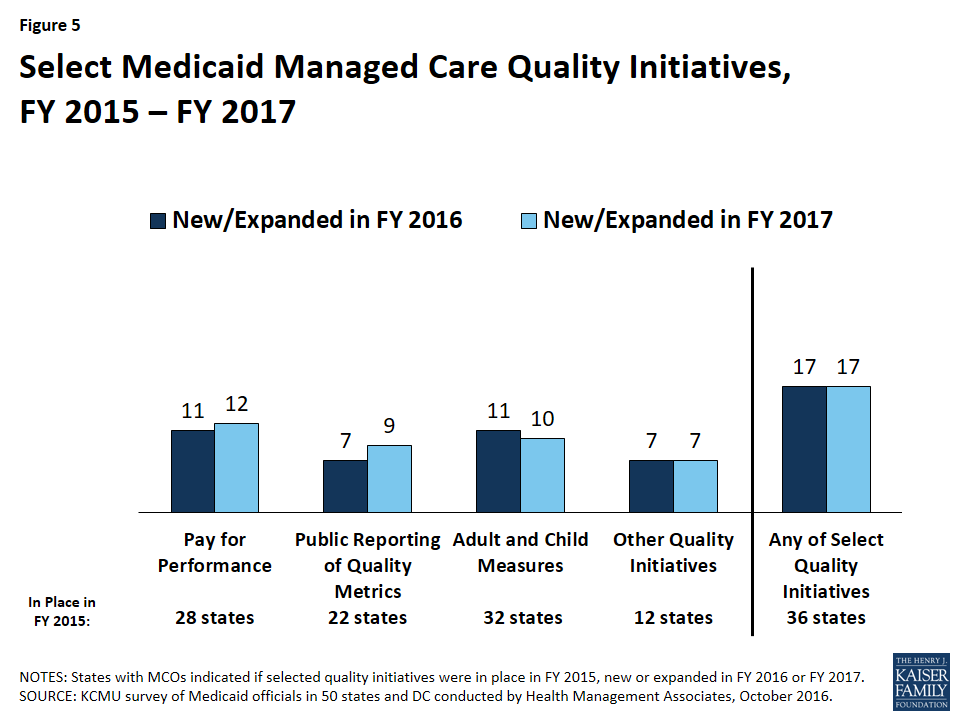
In FY 2016, 17 states implemented new or expanded quality initiatives and 17 states plan to do so in FY 2017 (Figure 5 and Table 8). Of the 39 MCO states, a total of 38 states in FY 2016 and all 39 states in FY 2017 will have at least one of these initiatives in place. The most common new quality initiatives were pay for performance and use of quality measures pulled from CMS’s core measure sets for adults and children (which are available but not mandatory for states to use).
States were also asked if capitation withholds in MCO contracts were in place in FY 2015, added in FY 2016, or planned for FY 2017. Twenty (20) states indicated withholds were in place as of FY 2015. States were also asked to specify what share of MCO capitation payments were withheld in FY 2016 and FY 2017. One state added a new MCO capitation payment withhold tied to quality performance in FY 2016 (Iowa) and three states intend to add a new MCO withhold in FY 2017 (Alabama, DC, and Oregon). Withhold amounts typically ranged from 1 percent (Massachusetts, Michigan, Texas, and Washington) to 5 percent (Georgia, West Virginia, and Minnesota). Tennessee reported using withholds in a range from 2.5 percent to 10 percent.
Contract Requirements
Alternative [Provider] Payment Models (APM) within MCO Contracts
Alternative provider payment models to advance value-based purchasing (VBP) strategies are a sharp focus of Medicaid programs, as states pursue improved quality and outcomes and reduced costs of care within Medicaid and across payers. Many states have included a focus on adopting and promoting alternative provider payment models as part of their State Innovation Models (SIM) projects, and some states have considered specifically how Medicaid MCOs can play a part in achieving improved accountability in the health care delivery system.29 The survey found that:
- Five states (Arizona, Delaware, Hawaii, Iowa, and South Carolina) identified a specific target in their MCO contracts for the percentage of provider payments, network providers, or plan members that plans must cover via alternative provider payment models in FY 2016; and
- Ten (10) additional states (California, Kansas, Louisiana, Michigan, Nebraska, New York, Oregon, Pennsylvania, Rhode Island, and Virginia) intend to include a target percentage in their contracts for FY 2017.
Further, 12 states had contracts that encouraged or required Medicaid MCOs to adopt alternative provider payment models in FY 2016, with eight additional states intending to encourage or require alternative provider payment arrangements within MCOs in FY 2017. The following box provides state examples of alternative provider payment targets.
Alternative Provider Payment Targets
- Arizona established an initial target of 5 percent for the share of each MCO’s total payments to providers made under alternative payment models. The state intends to raise this target to 50 percent by calendar year (CY) 2018 for acute care and by CY 2019 for LTSS.
- Iowa has a target of 40 percent for the share of an MCO’s membership to be covered by a VBP arrangement by FY 2018.
- Nebraska has a phased approach. Its target calls for 30 percent of a plan’s provider network to be subject to alternative payment models by year 3 of its contract, and 50 percent by year 5.
- New York has committed under its Section 1115 waiver to have between 80 and 90 percent of MCO payments to providers be under alternative payment models by year 5 of the waiver and plans to begin specifying the yearly targets in FY 2017.
- Pennsylvania also plans a phased approach, beginning with a target of 7.5 percent of a plan’s provider network in alternative payment models in CY 2016, 15 percent in CY 2017, and 30 percent in CY 2018.
Social Determinants of Health
In 2016, the CMS Center for Innovation announced a new Accountable Health Community model that represents the first CMS innovation model that focuses on social determinants of health. The goal of the five-year program is to raise awareness of and access to community-based services for Medicaid and Medicare beneficiaries.30 This development reflects growing awareness and interest on the part of CMS to seek improved health outcomes and reduced costs by linking beneficiaries to social services and supports to address issues such as housing and food insecurity, among others, that can impact the ability of individuals to achieve health goals. States have also been focused on addressing social determinants of health, so federal and state activity are occurring simultaneously.
The survey found that 26 of the 39 states that contract with MCOs required or encouraged plans to screen enrollees for social needs and provide referrals to other services in FY 2016. Several states required MCOs to perform a health needs/risk assessment that includes information on social needs as well as medical needs. The following box provides state examples.
Strategies to Address Social Determinants of Health
- Florida requires MCOs to have policies and procedures to identify available community support services and facilitate referrals for enrollees with identified needs for such services. Plans must document in the enrollee’s case record any referrals made for other services in the community and follow up on the enrollee’s receipt of services.
- Michigan requires that MCOs offer population health management interventions designed to address the social determinants of health, reduce disparities in health outcomes between different subpopulations, and ultimately achieve health equity. Population health management services can be provided in the enrollee’s home, place of employment or school, and at shelters for enrollees who are homeless.
- Rhode Island requires MCOs to coordinate with other assistance programs, such as SNAP, Special Education, WIC, and Rehabilitation Services.
Four states (Hawaii, Maryland, Nebraska, and New York) plan to require or encourage MCOs to screen and/or refer to social services and other programs in FY 2017. For example, Nebraska will require all MCO staff to be trained on how social determinants affect members’ health and wellness, including issues related to housing, education, food, physical and sexual abuse, violence, and risk and protective factors for behavioral health concerns.
Criminal Justice Involved Populations
Five of the 39 states with MCO contracts encourage or require MCOs to provide care coordination services to enrollees prior to release from incarceration (Arizona, Iowa, Kentucky, New Mexico, and Ohio), and 10 states intend to add such requirements in FY 2017. Ohio described a system in which pre-release care coordination is provided for enrollees with serious health conditions. An MCO care manager develops a care-focused transition plan to help facilitate access to needed services in the community, and a videoconference is conducted as a means to establish a relationship between the enrollee and the care manager. The care manager will follow up with the enrollee post-release to identify and remove barriers to care. Arizona noted that, in CY 2016, the requirement for pre-release care coordination services were limited to the behavioral health carve-out plan, but indicated that it would be extended to apply to all MCOs in CY 2017. In the letter approving the Arizona waiver on September 30, 2016, CMS said they would continue to work with Arizona on the delivery system reforms to integrate physical and behavioral health for Medicaid beneficiaries leaving the justice system.
While Florida does not require MCOs to provide pre-release care coordination, the state has a multi-agency project in place to implement pre-release care coordination to incarcerated Medicaid enrollees.
Administrative Policies
Minimum Medical Loss Ratios
The proportion of total capitation payments received by an MCO that is spent on clinical services and quality improvement is known as the Medical Loss Ratio (MLR). In 2016, CMS published a final rule that requires states to develop capitation rates for Medicaid so as to achieve an MLR of at least 85 percent in the rate year.31 This is consistent with the minimum MLR established in the ACA for commercial health plans in the Marketplace and for small group and individual plans in the private market and with the minimum MLR applied to Medicare Advantage plans. There is no federal requirement that states require Medicaid plans to remit payment if they fail to meet the MLR standard, but states have discretion to require remittances. The minimum MLR requirement for Medicaid takes effect for rating periods and contracts starting on or after July 1, 2017.32
As of July 1, 2016, 20 of the 39 states that contract with comprehensive risk-based MCOs already specified a minimum MLR. Eighteen (18) of these 20 states applied the MLR requirement to all MCO contracts, while two states applied it on a limited basis (in Virginia, for the Financial Alignment Demonstration (FAD) only; in Massachusetts, for the Senior Care Options (SCO) program only). Thirteen (13) of the 20 states with minimum MLR requirements always require remittance payments to the state if the minimum MLR is not achieved; three states require remittances under some circumstances.
Medicaid MLRs vary by state but are most commonly set at 85 percent. A few states noted that their minimum MLRs varied by type of plan or population. For example, in New Jersey, the MLR is calculated separately for each population covered. Fourteen (14) states count some or all care management costs as medical (rather than administrative) expenses in the calculation of the MLR. For example, New Mexico spells out a broad set of activities that can be counted as medical expenses, including face-to-face and telephonic interactions between a care coordinator and a member; comprehensive needs assessment, development of a care plan, case management, health education, disease management, and costs associated with Community Health Workers.
Table 9 provides state-specific information regarding the use of a minimum MLR.
Auto-Enrollment
Generally, beneficiaries who are required to enroll in MCOs must be offered a choice of at least two plans. Those who do not select a plan are auto-enrolled in a plan by the state. The proportion of MCO beneficiaries who are auto-enrolled, which may reflect the level of consumer understanding and engagement or design aspects of the managed care program, varies widely across states. Three of the 39 states with MCOs had auto-enrollment rates of 10 percent or less (Georgia, New York, and Pennsylvania) while seven states auto-enrolled 75 percent or more of new MCO enrollees.33 State auto-enrollment algorithms also vary, but they are usually designed to take into consideration previous plan or provider relationships, geographic location of the beneficiary, and/or plan enrollments of other family members. In addition, over half (23) of MCO states reported that their auto-enrollment algorithms were designed to balance enrollments among plans.
As of July 1, 2016, 10 states took plan quality rankings into consideration in the auto-enrollment algorithm, and Illinois plans to incorporate plan quality into its auto-enrollment algorithm during CY 2017 (Exhibit 5). This is an increase from eight states a year earlier. California noted that it makes auto-assignments based in part on MCO compliance with encounter data reporting requirements and plan inclusion of safety net providers in their provider networks. Michigan reported incorporating MCO performance on the Consumer Assessment of Healthcare Providers and Systems (CAHPS) survey.34 Exhibit 5 shows use of selected components in state auto-enrollment algorithms.
| Exhibit 5: Select Factors Used in State Auto-Enrollment Algorithms, as of July 2016 | ||
| # of States | States | |
| Balancing Enrollment | 23 | DC, DE, HI, IA, IL, KS, KY, MA, MD, MO, MS, NE, NM, NV, NY, PA, RI, SC, TN, TX, UT, WI, WV |
| Quality | 10 | CA, GA, LA, MI, MN, NM, NY, OH, SC, WA |
| Encouraging New Plan Entrants | 1 | HI |
PCCM and PHP Program Changes
Primary Care Case Management (PCCM) Program Changes
Of the 16 states with PCCM programs, three reported enacting policies to increase PCCM enrollment in FY 2016 or FY 2017: Colorado reported continued growth in its PCCM-based Accountable Care Collaboratives in both FY 2016 and FY 2017; Montana enrolled its ACA expansion population into its PCCM program in FY 2016, and Massachusetts allowed members in the CarePlus (ACA expansion) program, who were previously required to enroll in an MCO, to enroll in either the Primary Care Clinician plan (PCCM) or an MCO. Also, Alaska – one of only three states without either an MCO or PCCM program as of July 1, 2016 – reported plans to implement a PCCM program in FY 2017.
In contrast, five states (Alabama, Iowa, Rhode Island, Washington, and West Virginia) have taken actions to decrease enrollment in their PCCM programs in FY 2016 or FY 2017. Four of these states (Alabama, Iowa, Rhode Island, and West Virginia) ended or plan to end their PCCM programs and transition PCCM enrollees to risk-based managed care. Nevada stated that it was evaluating the cost-effectiveness of its PCCM program before making any policy changes and Oregon reported that it was working with nine federally recognized tribes to determine whether to pursue PCCM for tribal members and Tribal Health Centers.
Limited-Benefit Prepaid Health Plans (PHP) Changes
In this year’s survey, the 24 states contracting with at least one PHP as of July 1, 2016, were asked to indicate the services provided under these arrangements. As shown in Exhibit 6 below, the most frequently cited services provided were outpatient behavioral health services (13 states) and inpatient behavioral health services (12 states), followed by substance use disorder treatment and non-emergency medical transportation (NEMT) (10 states each). “Other” services reported included maternity care (Alabama), primary care (Colorado), behavioral health-related “diversionary” services (Massachusetts), incontinence supplies and vision care (Wisconsin), and mental health wrap-around services for children with emotional disturbances (Wyoming).
| Exhibit 6: Services Covered Under PHP Contracts, July 1, 2016 | ||
| # of States | States | |
| Outpatient Behavioral Health | 13 | AZ, CA, CO, HI, ID, MA, MI, NC, NE, PA, TX, UT, WA |
| Inpatient Behavioral Health | 12 | AZ, CA, CO, HI, MA, MI, NC, NE, PA, TX, UT, WA |
| Outpatient Substance Use Disorder Treatment | 10 | AZ, CO, ID, MA, NC, NE, PA, TX, UT, WA |
| Inpatient Substance Use Disorder Treatment | 10 | AZ, CO, MA, MI, NC, NE, PA, TX, UT, WA |
| Non-Emergency Medical Transportation (NEMT) | 10 | IA, KY, ME, MI, NJ, NV, RI, TX, UT, WI |
| Dental | 7 | IA, ID, LA, MI, RI, TX, UT |
| Long-Term Services and Supports | 5 | ID, MI, NC, NY, WI |
| Other | 5 | AL, CO, MA, WI, WY |
Four states reported implementing policies to increase PHP enrollment in FY 2016 or FY 2017. Michigan expanded or will expand its dental PHP program to additional counties in both FY 2016 and FY 2017. In FY 2017, California is implementing a waiver to provide substance use disorder services under a county-based PHP arrangement;35 Colorado implemented a primary care PHP in one region; Indiana is planning to implement an NEMT PHP. While not counted in this report as an expansion, Massachusetts noted that allowing newly eligible adults to enroll in the PCCM program could also result in higher enrollment in its behavioral health PHP. Also, Arkansas and Nevada are planning to implement dental PHPs in FY 2018.
Five states also reported actions to decrease PHP enrollment in FY 2016 or FY 2017. Four states reported ending a PHP and folding the covered services into MCO contracts – Iowa (behavioral health) in FY 2016 and Alabama (maternity care), Nebraska (behavioral health), and Texas (behavioral health) in FY 2017. Also, Washington reported that PHP enrollment decreased in FY 2016 and will decrease further in FY 2017 when the state converts behavioral health PHPs to fully integrated MCO contracts in two counties.
Table 5: Share of the Medicaid Population Covered Under Different Delivery Systems in all 50 States and DC, as of July 1, 2016
| States | Type(s) of Managed Care In Place | Share of Medicaid Population in Different Managed Care Systems | ||
| MCO | PCCM | FFS / Other | ||
| Alabama | PCCM | — | 65.7% | 34.4% |
| Alaska | FFS | — | — | 100.0% |
| Arizona | MCO | 92.8% | — | 7.2% |
| Arkansas | PCCM | — | NR | NR |
| California | MCO and PCCM* | 84.6% | — | 15.4% |
| Colorado | MCO and PCCM* | 8.6% | 77.2% | 14.2% |
| Connecticut | FFS* | — | — | 100.0% |
| Delaware | MCO | >90% | — | <10% |
| DC | MCO | 76.0% | — | 24.0% |
| Florida | MCO | 93.0% | — | 7.1% |
| Georgia | MCO | 69.0% | — | 31.0% |
| Hawaii | MCO | 99.9% | — | <0.1% |
| Idaho | PCCM* | — | 93.0% | 7.0% |
| Illinois | MCO and PCCM | 63.4% | 11.5% | 25.1% |
| Indiana | MCO | 79.0% | — | 21.0% |
| Iowa | MCO | 96.0% | — | 4.0% |
| Kansas | MCO | 95.0% | — | 5.0% |
| Kentucky | MCO | 91.0% | — | 9.0% |
| Louisiana | MCO | 70.0% | — | 30.0% |
| Maine | PCCM | — | NR | NR |
| Maryland | MCO | 80.0% | — | 20.0% |
| Massachusetts | MCO and PCCM | 53.5% | 26.0% | 20.5% |
| Michigan | MCO | 75.0% | — | 25.0% |
| Minnesota | MCO | 75.0% | — | 25.0% |
| Mississippi | MCO | 70.0% | — | 30.0% |
| Missouri | MCO | 51.2% | — | 48.8% |
| Montana | PCCM | — | 71.0% | 29.0% |
| Nebraska | MCO | 77.0% | — | 23.0% |
| Nevada | MCO and PCCM | 77.0% | 6.9% | 16.0% |
| New Hampshire | MCO | 95.7% | — | 4.3% |
| New Jersey | MCO | 94.6% | — | 5.4% |
| New Mexico | MCO | 88.2% | — | 11.8% |
| New York | MCO | 77.1% | — | 22.9% |
| North Carolina | PCCM | — | 80.3% | 19.7% |
| North Dakota | MCO and PCCM | 22.0% | 49.0% | 29.0% |
| Ohio | MCO | 88.0% | — | 12.0% |
| Oklahoma | PCCM | — | 74.8% | 25.2% |
| Oregon | MCO* | 85.7% | — | 14.3% |
| Pennsylvania | MCO | 82.8% | — | 17.2% |
| Rhode Island | MCO | 90.0% | — | 10.0% |
| South Carolina | MCO* | 72.7% | — | 27.3% |
| South Dakota | PCCM | — | 80.0% | 20.0% |
| Tennessee | MCO | 100.0% | — | — |
| Texas | MCO* | 88.0% | — | 12.2% |
| Utah | MCO | 81.5% | — | 18.5% |
| Vermont | PCCM | — | >90% | <10% |
| Virginia | MCO | 83.0% | — | 17.0% |
| Washington | MCO and PCCM | 83.0% | 2.0% | 15.0% |
| West Virginia | MCO | 63.0% | — | 36.0% |
| Wisconsin | MCO | 67.0% | — | 33.0% |
| Wyoming | FFS* | — | — | 100.0% |
| NOTES: NR – not reported. Share of Medicaid Population that is covered by different managed care systems. MCO refers to risk-based managed care; PCCM refers to Primary Care Case Management. FFS/Other refers to Medicaid beneficiaries who are not in MCOs or PCCM programs. *CA – PCCM program operates in LA county for those with HIV. *CO – PCCM enrollees are part of the state’s Accountable Care Collaboratives (ACCs). *CT – terminated its MCO contracts in 2012 and now operates its program on a fee-for-service basis using four Administrative Services Only entities. *ID – The Medicaid-Medicare Coordinated Plan (MMCP) has been recategorized by CMS as an MCO but is not counted here as such since it is secondary to Medicare. *OR – MCO enrollees include those enrolled in the state’s Coordinated Care Organizations. *SC – uses PCCM authority to provide care management services to approximately 200 medically complex children. *TX – Texas Medicaid Wellness program provides care management services for high-cost/high-risk enrollees (under PCCM authority).*WY – the state does not operate a traditional PCCM or MCO program, but does use PCCM authority to make PCMH payments.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||
Table 6: Enrollment of Special Populations Under Medicaid Managed Care Contracts for Acute Care in all 50 States and DC, as of July 1, 2016
| States | Pregnant Women | Foster Children | Persons with ID/DD | CSHCNs | SMI Adults | Adults w/ physical disabilities |
| Alabama | — | — | — | — | — | — |
| Alaska | — | — | — | — | — | — |
| Arizona | Varies | Varies | Varies | Varies | Varies | Varies |
| Arkansas | — | — | — | — | — | — |
| California* | Always Mandatory | Always Mandatory | Varies | Always Mandatory | Always Mandatory | Always Mandatory |
| Colorado | Varies | Varies | Varies | Varies | Varies | Varies |
| Connecticut | — | — | — | — | — | — |
| Delaware | Always Mandatory | Varies | Varies | Always Mandatory | Always Mandatory | Always Mandatory |
| DC | Always Mandatory | Varies | Always Excluded | Varies | Varies | Varies |
| Florida | Always Mandatory | Always Mandatory | Always Voluntary | Always Voluntary | Always Mandatory | Always Mandatory |
| Georgia | Always Mandatory | Always Mandatory | Always Excluded | Always Excluded | Always Excluded | Always Excluded |
| Hawaii | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| Idaho | — | — | — | — | — | — |
| Illinois | Varies | Always Excluded | Varies | Varies | Varies | Varies |
| Indiana | Always Mandatory | Always Voluntary | Varies | Varies | Varies | Varies |
| Iowa | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| Kansas | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| Kentucky | Varies | Varies | Varies | Varies | Varies | Varies |
| Louisiana | Always Mandatory | Always Mandatory | Varies | Always Mandatory | Varies | Varies |
| Maine | — | — | — | — | — | — |
| Maryland | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| Massachusetts | Always Voluntary | Always Voluntary | Always Voluntary | Always Voluntary | Always Voluntary | Always Voluntary |
| Michigan | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| Minnesota | Always Mandatory | Always Voluntary | Always Voluntary | Always Voluntary | Always Voluntary | Always Voluntary |
| Mississippi | Always Mandatory | Always Voluntary | Varies | Varies | Varies | Varies |
| Missouri | Always Mandatory | Always Mandatory | Always Excluded | Varies | Varies | Always Excluded |
| Montana | — | — | — | — | — | — |
| Nebraska | Always Mandatory | Always Mandatory | Always Excluded | Always Mandatory | Always Mandatory | Varies |
| Nevada | Varies | Varies | Always Excluded | Varies | Varies | Always Excluded |
| New Hampshire | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| New Jersey | Varies | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| New Mexico | Varies | Varies | Varies | Varies | Varies | Varies |
| New York | Always Mandatory | Varies | Always Voluntary | Varies | Always Mandatory | Always Mandatory |
| North Carolina | — | — | — | — | — | — |
| North Dakota | Always Excluded | Always Excluded | Always Excluded | Always Excluded | Always Excluded | Always Excluded |
| Ohio | Always Mandatory | Always Voluntary | Always Voluntary | Varies | Varies | Always Mandatory |
| Oklahoma | — | — | — | — | — | — |
| Oregon | Always Mandatory | Always Voluntary | Always Voluntary | Always Mandatory | Always Mandatory | Always Mandatory |
| Pennsylvania | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| Rhode Island | Always Mandatory | Always Voluntary | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| South Carolina | Always Mandatory | Always Voluntary | Varies | Varies | Varies | Varies |
| South Dakota | — | — | — | — | — | — |
| Tennessee | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory | Always Mandatory |
| Texas | Always Mandatory | Varies | Varies | Varies | Varies | Varies |
| Utah | Varies | Varies | Varies | Varies | Varies | Varies |
| Vermont | — | — | — | — | — | — |
| Virginia | Always Mandatory | Always Mandatory | Varies | Varies | Varies | Varies |
| Washington | Varies | Always Voluntary | Varies | Varies | Varies | Varies |
| West Virginia | Always Mandatory | Always Excluded | Always Excluded | Always Mandatory | Always Excluded | Always Excluded |
| Wisconsin | Always Mandatory | Always Voluntary | Always Voluntary | Varies | Always Voluntary | Always Voluntary |
| Wyoming | — | — | — | — | — | — |
| Always Mandatory | 28 | 16 | 10 | 16 | 16 | 16 |
| Always Voluntary | 1 | 10 | 7 | 3 | 3 | 3 |
| Varies | 9 | 10 | 15 | 18 | 17 | 15 |
| Always Excluded | 1 | 3 | 7 | 2 | 3 | 5 |
| NOTES: “–” indicates there were no MCOs operating in that state’s Medicaid program in July 2016. ID/DD – intellectual and developmental disabilities, CSHCN – Children with special health care needs, SMI – Serious Mental Illness. States were asked to indicate for each group if enrollment in MCOs is “always mandatory,” “always voluntary,” “varies (by geography or other factor),” or if the group is “always excluded” from MCOs as of July 1, 2016. *CA was re-categorized from “Varies” to “Always Mandatory” across all population groups (except for persons with ID/DD) as the state noted that enrollment is generally mandatory across the state with the exception of one, small rural county where managed care is voluntary because there is only one plan and it is not a COHS county. The ID/DD population is subject to mandatory enrollment only in COHS counties.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||
Table 7: Behavioral Health Services Covered under Acute Care MCO Contracts in all 50 States and DC, as of July 1, 2016
| States | Specialty OP Mental Health | Inpatient Mental Health | Outpatient SUD | Inpatient SUD |
| Alabama | — | — | — | — |
| Alaska | — | — | — | — |
| Arizona | Varies | Varies | Varies | Varies |
| Arkansas | — | — | — | — |
| California | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Colorado | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Connecticut | — | — | — | — |
| Delaware | Always Carved-out | Always Carved-in | Always Carved-in | Always Carved-in |
| DC | Always Carved-in | Always Carved-in | Always Carved-out | Always Carved-in |
| Florida | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Georgia | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Hawaii | Always Carved-out | Always Carved-out | Always Carved-in | Always Carved-in |
| Idaho | — | — | — | — |
| Illinois | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Indiana | Always Carved-out | Always Carved-in | Always Carved-in | Always Carved-in |
| Iowa | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Kansas | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Kentucky | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Louisiana | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Maine | — | — | — | — |
| Maryland | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Massachusetts | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Michigan | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Minnesota | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Mississippi | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Missouri | Always Carved-out | Varies | Varies | Varies |
| Montana | — | — | — | — |
| Nebraska | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Nevada | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| New Hampshire | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| New Jersey | Always Carved-out | Always Carved-out | Always Carved-out | Varies |
| New Mexico | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| New York | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| North Carolina | — | — | — | — |
| North Dakota | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Ohio | Varies | Always Carved-in | Varies | Always Carved-in |
| Oklahoma | — | — | — | — |
| Oregon | Always Carved-in | Always Carved-out | Always Carved-in | Always Carved-out |
| Pennsylvania | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Rhode Island | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| South Carolina | Varies | Varies | Always Carved-in | Always Carved-in |
| South Dakota | — | — | — | — |
| Tennessee | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Texas | Varies | Varies | Varies | Varies |
| Utah | Always Carved-out | Always Carved-out | Always Carved-out | Always Carved-out |
| Vermont | — | — | — | — |
| Virginia | Varies | Always Carved-in | Varies | Always Carved-in |
| Washington | Varies | Varies | Varies | Varies |
| West Virginia | Always Carved-in | Always Carved-in | Always Carved-in | Always Carved-in |
| Wisconsin | Varies | Always Carved-in | Always Carved-in | Always Carved-in |
| Wyoming | — | — | — | — |
| Always Carved-in | 20 | 24 | 24 | 26 |
| Always Carved-out | 12 | 10 | 9 | 8 |
| Varies | 7 | 5 | 6 | 5 |
| NOTES: OP – Outpatient. SUD – Substance Use Disorder. “–” indicates there were no MCOs operating in that state’s Medicaid program in July 2016. For beneficiaries enrolled in an MCO for acute care benefits, states were asked to indicate whether these benefits are always carved-in (meaning virtually all services are covered by the MCO), always carved-out (to PHP or FFS), or whether the carve-in varies (by geography or other factor). “Specialty outpatient mental health” refers to services utilized by adults with Serious Mental Illness (SMI) and/or youth with serious emotional disturbance (SED) commonly provided by specialty providers such as community mental health centers.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||
Table 8: Medicaid Managed Care Quality Initiatives in all 50 States and DC, FY 2015 – FY 2017
| States | Pay for Performance/ Performance Bonus or Penalties | Adult and Child Quality Measures | Publicly Report MCO Quality Metrics | Other Quality Initiatives | Any Quality Initiatives | ||||||||||
| In Place | New/Expanded | In Place | New/Expanded | In Place | New/Expanded | In Place | New/Expanded | In Place | New/Expanded | ||||||
| 2015 | 2016 | 2017 | 2015 | 2016 | 2017 | 2015 | 2016 | 2017 | 2015 | 2016 | 2017 | 2015 | 2016 | 2017 | |
| Alabama | X | X | X | X | |||||||||||
| Alaska | |||||||||||||||
| Arizona | X | X | X | X | X | X | X | X | X | X | X | ||||
| Arkansas | |||||||||||||||
| California | X | X | X | X | X | X | X | X | X | X | X | X | X | X | X |
| Colorado | X | X | X | X | |||||||||||
| Connecticut | |||||||||||||||
| Delaware | X | X | X | X | X | X | X | X | X | ||||||
| DC | X | X | X | X | X | X | X | X | X | X | |||||
| Florida | X | X | X | X | X | ||||||||||
| Georgia | X | X | X | X | |||||||||||
| Hawaii | X | X | X | X | |||||||||||
| Idaho | |||||||||||||||
| Illinois | X | X | X | X | |||||||||||
| Indiana | X | X | |||||||||||||
| Iowa | X | X | X | X | X | X | |||||||||
| Kansas | X | X | X | ||||||||||||
| Kentucky | X | X | X | X | X | ||||||||||
| Louisiana | X | X | X | X | |||||||||||
| Maine | |||||||||||||||
| Maryland | X | X | X | X | |||||||||||
| Massachusetts | X | X | X | X | X | ||||||||||
| Michigan | X | X | X | X | |||||||||||
| Minnesota | X | X | X | X | X | ||||||||||
| Mississippi | X | X | |||||||||||||
| Missouri | X | X | X | X | X | X | X | X | X | ||||||
| Montana | |||||||||||||||
| Nebraska | X | X | X | X | |||||||||||
| Nevada | X | X | |||||||||||||
| New Hampshire | X | X | X | X | X | X | X | X | |||||||
| New Jersey | X | X | X | X | X | X | |||||||||
| New Mexico | X | X | X | X | |||||||||||
| New York | X | X | X | X | |||||||||||
| North Carolina | |||||||||||||||
| North Dakota | X | X | |||||||||||||
| Ohio | X | X | X | X | X | ||||||||||
| Oklahoma | |||||||||||||||
| Oregon | X | X | X | X | |||||||||||
| Pennsylvania | X | X | X | X | X | X | X | X | X | X | X | X | X | X | |
| Rhode Island | X | X | X | ||||||||||||
| South Carolina | X | X | X | X | X | X | |||||||||
| South Dakota | |||||||||||||||
| Tennessee | X | X | X | X | X | X | X | X | |||||||
| Texas | X | X | X | X | X | X | X | X | X | X | |||||
| Utah | X | X | X | X | |||||||||||
| Vermont | |||||||||||||||
| Virginia | X | X | X | X | X | X | X | X | X | X | X | X | X | X | X |
| Washington | X | X | X | X | |||||||||||
| West Virginia | X | X | X | X | X | X | X | X | X | ||||||
| Wisconsin | X | X | X | X | X | X | X | ||||||||
| Wyoming | |||||||||||||||
| Totals | 28 | 11 | 12 | 32 | 11 | 10 | 22 | 7 | 9 | 12 | 7 | 7 | 36 | 17 | 17 |
| NOTES: States with MCO contracts were asked to report if select quality initiatives were included in contracts in FY 2015, new or expanded in FY 2016 or FY 2017. “Adult and Child Quality Measures” refers to CMS’s core measure sets for adults and children, which are available but not mandatory for states to use. The table above does not reflect all quality initiatives states have included as part of MCO contracts.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, 2016 | |||||||||||||||
Table 9: Minimum Medical Loss Ratio Policies for Medicaid MCOs in all 50 States and DC, as of July 1, 2016
| Minimum Medical Loss Ratio (MLR) | ||
| States | Require minimum MLR | % if required |
| Alabama | — | |
| Alaska | — | |
| Arizona | Yes — always | 85% |
| Arkansas | — | |
| California | No | |
| Colorado | Yes — always | 85% |
| Connecticut | — | |
| Delaware | No | |
| DC | Yes — always | 85% |
| Florida | Yes — always | 85% |
| Georgia | No | |
| Hawaii | No | |
| Idaho | — | |
| Illinois | Yes — always | 85%-88% * |
| Indiana | Yes — always | 85%-87%* |
| Iowa | Yes — always | 88% |
| Kansas | No | |
| Kentucky | Yes — always | 90% |
| Louisiana | Yes — always | 85% |
| Maine | — | |
| Maryland | Yes — always | 85% |
| Massachusetts | Yes — sometimes* | 80% |
| Michigan | No | |
| Minnesota | No | |
| Mississippi | Yes — always | 85% |
| Missouri | No | |
| Montana | — | |
| Nebraska | No | |
| Nevada | No | |
| New Hampshire | No | |
| New Jersey | Yes — always | 85% |
| New Mexico | Yes — always | 85% |
| New York | No | |
| North Carolina | — | |
| North Dakota | No | |
| Ohio | Yes — always | 85% |
| Oklahoma | — | |
| Oregon | Yes — always | 80% |
| Pennsylvania | No | |
| Rhode Island | No | |
| South Carolina | Yes — always | 86% |
| South Dakota | — | |
| Tennessee | No | |
| Texas | No* | |
| Utah | No* | |
| Vermont | — | |
| Virginia | Yes — sometimes | 85% |
| Washington | Yes — always | 85-87%* |
| West Virginia | Yes — always | 85% |
| Wisconsin | No* | |
| Wyoming | — | |
| Yes — always | 18 | |
| Yes — sometimes | 2 | |
| No | 19 | |
| N/A – No MCOs | 12 | |
| NOTES: “–” indicates states that do not have Medicaid MCOs. MLR refers to the proportion of total per member per month capitation payments that is spent on clinical services and for quality improvement. *MA reported that there is no minimum MLR for acute MCOs or the One Care (FAD) program; however, the SCO program has a minimum MLR of 80%. *UT and WI reported not requiring a minimum MLR but using a target MLR as part of their rate setting process. *TX has experience rebates on plans above a certain profit level. *IL, IN and WA indicated that the minimum MLR varies by population.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||
Report: Emerging Delivery System And Payment Reforms
Key Section Findings
- Over two-thirds of all state Medicaid programs (36) have at least one delivery system or payment reform initiative in place designed to improve health outcomes and constrain cost growth.
- Twenty-one (21) states in FY 2016 and 25 states in FY 2017 (29 states in either year) reported adopting or expanding one or more initiatives, including patient-centered medical homes (PCMHs), Health Homes, Accountable Care Organizations (ACOs), and other initiatives to better manage the care of persons with multiple chronic conditions. Interest in Episode of Care payments also ticked upward for FY 2017 (7 states).
- Seven states had Delivery System Reform Incentive Payment (DSRIP) programs in place in FY 2015. Four states reported new or expanded DSRIP programs in FY 2016 and five states reported new or expanded DSRIP programs in FY 2017.
Tables 10 and 11 contain more detailed information on emerging delivery system and payment reform initiatives in place in FY 2015, implemented in FY 2016, or planned for FY 2017.
This year’s survey asked states to identify which delivery system and payment reform models were in place in FY 2015, and whether they had adopted or were enhancing such models in FY 2016 or FY 2017. Over two-thirds of all state Medicaid programs, 36 states in FY 2015 and 39 states in FY 2016, currently have at least one delivery system or payment reform initiative in place designed to improve health outcomes and constrain cost growth (Figure 6 and Table 10). If all actions reported by states for FY 2017 are implemented as planned, that number will grow to 42 states by the end of FY 2017, demonstrating the continued widespread and growing interest in Medicaid transformation. A total of 21 states in FY 2016 and 25 states in FY 2017 reported adopting or expanding one or more initiatives that seek to reward quality and encourage integrated care. Key initiatives include patient-centered medical homes (PCMHs), Health Homes, and Accountable Care Organizations (ACOs). Interest in Episode of Care initiatives also ticked upward for FY 2017 (Figure 6 and Table 11).
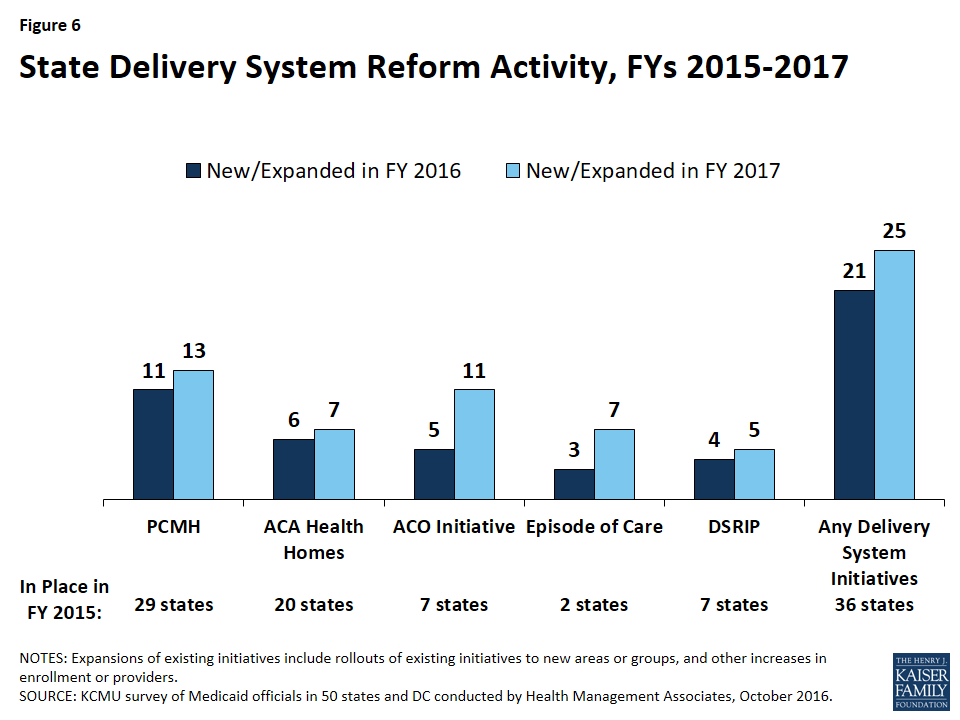
Patient-Centered Medicaid Homes (PCMHs)
PCMH initiatives operated in over half (29) of Medicaid programs in FY 2015 (Table 10). Under a PCMH model, a physician-led, multi-disciplinary care team holistically manages the patient’s ongoing care, including recommended preventive services, care for chronic conditions, and access to social services and supports. Generally, providers or provider organizations that operate as a PCMH seek recognition from organizations like the National Committee for Quality Assurance (NCQA).36 PCMHs are often paid (by state Medicaid agencies directly or through MCO contracts) a per member per month (PMPM) fee in addition to regular FFS payments for their Medicaid patients.37
In this year’s survey, 11 states reported having adopted or expanded PCMHs in FY 2016 and 13 states indicated plans to do so in FY 2017 (Table 11). A few of these states reported notable expansions. Wyoming, a state without MCO or PCCM programs, implemented PCMHs in FY 2015 and is expanding the number of practices participating in both FY 2016 and FY 2017. Idaho reported that its PCMH program expanded to its full PCCM network in FY 2016. In addition to the established PCMH programs administered by the three MCOs operating in Tennessee, a new, multi-payer PCMH initiative would begin in January 2017, starting with 20 to 30 practices. Alaska and Ohio are planning to implement new PCMH initiatives in FY 2017 and Pennsylvania’s new MCO contracts will encourage PCMHs beginning in FY 2017.
In contrast, Alabama expects a reduction in PCMHs in FY 2017 when the state begins contracting with MCOs. Maryland reported that an all-payer PCMH initiative sunsetted in December 2015; however, noted that Medicaid continued the program for an additional six months.
ACA Health Homes
Over one-third of states (20) had at least one Health Home initiative in place in FY 2015 (up from 16 states in FY 2014) (Table 10). This option, created under Section 2703 of the ACA, builds on the PCMH concept. By design, Health Homes must target beneficiaries who have at least two chronic conditions (or one and risk of a second, or a serious and persistent mental health condition), and provide a person-centered system of care that facilitates access to and coordination of the full array of primary and acute physical health services, behavioral health care, and social and long-term services and supports. This includes services such as comprehensive care management, referrals to community and social support services, and the use of health information technology (HIT) to link services, among others. States receive a 90 percent federal match rate for qualified Health Home service expenditures for the first eight quarters under each Health Home State Plan Amendment; states can (and have) created more than one Health Home program to target different populations.
In this survey, six states reported having adopted or expanded Health Homes in FY 2016 and seven states reported plans to do so in FY 2017 (Table 11). Of these six states, three reported new Health Home State Plan Amendments (SPAs) in FY 2016: two targeting persons with serious mental illness (SMI) (DC and New Mexico) and one targeting chronic conditions, implemented in primary care settings (Michigan). Four states plan to implement new Health Home SPAs in FY 2017: two targeting persons with SMI (Minnesota and Tennessee) and two targeting persons with multiple chronic conditions (California and DC). Also, Wyoming reported that Health Homes were under consideration for possible implementation in FY 2018.
Idaho reported ending their Health Home program in FY 2016. Two states (Alabama and Kansas) reported ending their Health Home programs in FY 2017. Alabama noted that its Health Home program would end in FY 2017 but would be encompassed in its Section 1115 waiver program establishing Regional Care Organizations.
Accountable Care Organizations (ACOs)
Seven states reported having ACOs in place for at least some of their Medicaid beneficiaries in FY 2015 (Table 10). While there is no uniform, commonly accepted federal definition of an ACO, an ACO generally refers to a group of health care providers or, in some cases, a regional entity that contracts with providers and/or health plans, that agrees to share responsibility for the health care delivery and outcomes for a defined population.38 An ACO that meets quality performance standards that have been set by the payer and achieves savings relative to a benchmark can share in the savings. States use different terminology in referring to their Medicaid ACO initiatives, such as Coordinated Care Organizations (CCOs) in Oregon and Regional Care Collaborative Organizations (RCCOs) in Colorado.39
In this survey, five states reported adopting or expanding ACOs in FY 2016 and 11 states reported plans to do so in FY 2017 (Table 11) – a significant increase over the three states in last year’s survey that reported new or expanded ACO initiatives in FY 2015. This includes seven states that have implemented or are planning to implement new ACO initiatives: Delaware, Massachusetts, Missouri, New Mexico, Pennsylvania, Rhode Island, and Washington. Three of these states (Missouri, Pennsylvania, and Rhode Island) reported that they were building provisions into their MCO contracts either encouraging or requiring their MCOs to contract with ACOs. Another three states with more mature ACO programs (Colorado, Maine, and Minnesota) reported expansions of those programs in both FY 2016 and FY 2017. Vermont, which currently has a shared savings program with two ACOs, reported plans to move to risk-based ACOs – that is, both shared savings and shared risk – in FY 2017, and also indicated that it was in negotiations for an all-payer ACO that would begin in January 2017. Washington reported that components of its ACO program for public employees may be offered to the Medicaid population in FY 2017. While not counted among the states implementing ACOs in this year’s report, Maryland reported that it is working on a stakeholder process to develop recommendations for ACOs.
One state (Iowa) reported eliminating its ACO program in FY 2016 when it was subsumed into the state’s recently launched Medicaid managed care program.
Massachusetts ACO Pilot
In response to a state law requiring MassHealth to adopt alternative payment methodologies to promote more coordinated and efficient care, MassHealth is working to restructure its delivery system and payments within the next few years to transition from FFS care to integrated ACOs. The state plans to launch an ACO pilot by the end of CY 2016, with a full ACO roll-out planned for FY 2018. Through the renewal and renegotiation of its Section 1115 waiver (which expires June 30, 2017), MassHealth is proposing to implement a $1.8 billion Delivery System Reform Incentive Payment (DSRIP) program that will be used to support ACOs, invest in behavioral health care and long-term services and supports community capacity, and address health-related social needs. Also, to encourage enrollment in either an ACO or MCO, the state’s waiver extension request proposes to eliminate or limit certain optional benefits available to PCCM enrollees (e.g., chiropractic services, orthotics, eye glasses, and hearing aids) and impose differential copayments (i.e., lower for ACO and MCO enrollees).40
Episode-of-Care Payment Initiatives
Unlike FFS reimbursement, where providers are paid separately for each service, or capitation, where a health plan receives a PMPM payment for each enrollee intended to cover the costs for all covered services, episode-of-care payment provides a set dollar amount for the care a patient receives in connection with a defined condition or health event (e.g., pregnancy and delivery, heart attack, or knee replacement). Episode-based payments usually involve payment for multiple services and providers, creating a financial incentive for physicians, hospitals and other providers to work together to improve patient care and manage costs. Two states (Arkansas and Tennessee) reported that they had episode-of-care payment initiatives in place in FY 2015 (Table 10). Both of these states also reported expansions of these initiatives in FY 2016 and planned for FY 2017, with Tennessee commenting that it had designed over 25 episodes of care since 2013.
One state (New Mexico) reported a new episode-of-care initiative in FY 2016 and four states (Ohio, Pennsylvania, Rhode Island, and Washington) reported plans for new initiatives in FY 2017 (Table 11). Ohio reported that, in CY 2017, it will make actual reward payments for three defined episodes of care. Pennsylvania indicated that its new MCO contracts encourage MCOs to adopt value-based purchasing arrangements, including episodes of care; Rhode Island reported that it was looking at a bundled payment rate for Maternity/NICU care; and Washington reported that public employee bundled payment programs may be expanded to the Medicaid population as well. While not counted in this year’s report, Michigan, South Carolina and Wyoming reported that episode-of-care payment was under consideration for future implementation.
Delivery System Reform Incentive Payment (DSRIP) Program
Delivery System Reform Incentive Payment (DSRIP) programs, which are part of broader Section 1115 demonstration waiver programs, provide states with significant funding to support hospitals and other providers in changing how they provide care to Medicaid beneficiaries. DSRIP waivers are not grant programs – they are performance-based incentive programs. Originally, DSRIP initiatives were more narrowly focused on funding for safety-net hospitals and often grew out of negotiations between states and HHS over the appropriate way to finance hospital care. Now, however, they are used to promote far more sweeping payment and delivery system reforms.
The first DSRIP initiatives were approved and implemented in California and Texas in 2010, followed by New Jersey, Kansas, New Mexico, Massachusetts, and New York (Table 10).41
- Four states reported new or expanded DSRIP programs in place in FY 2016: California’s new “PRIME” program began in January 2016 with the state’s Section 1115 waiver renewal; Massachusetts’s “Delivery System Transformation Initiatives” (DSTI) program was extended through FY 2017 and hospitals are adding new projects;42 New Hampshire implemented a new DSRIP program focused on mental health and substance use disorder services, and New Mexico expanded its existing program.
- For FY 2017, Alabama, Arizona, and Washington reported plans to implement new DSRIP programs. Arizona’s 1115 demonstration waiver was approved; however, CMS noted in the approval that they would continue to work with Arizona on the delivery system reforms to integrate physical and behavioral health for children and adults and Medicaid beneficiaries leaving the justice system. On September 30, 2016, CMS issued a letter approving core facets of Washington’s waiver proposal subject to final approval of the special terms and conditions. Washington has committed under the waiver that 90 percent of its provider payments under state-financed health care (Medicaid and public employees) will be linked to quality and value by 2021. Massachusetts and New Mexico reported planned expansions of their existing programs (Table 11). Looking further into the future, Massachusetts reported that it was currently applying for a new five-year DSRIP waiver starting in FY 2018. Under the proposal, the Commonwealth’s current DSTI program would be restructured substantially.
Other Initiatives
In addition to the initiatives discussed already, states reported on a variety of other delivery system and payment reform initiatives. For example, two states (California and Connecticut) reported plans to implement shared savings arrangements or alternative payment methodologies for federally qualified health centers (FQHCs), and two states reported implementing “Health Home-like” programs, one for addressing opioid issues “Centers of Excellence” (Pennsylvania) and the other for behavioral health (Virginia). Two states (Oklahoma and Oregon) reported participating in the CMS Innovation Center’s Comprehensive Primary Care Initiative.43 Also, California reported on the transformation of its traditional Disproportionate Share Hospital funding program to a global budget structure for services provided to the uninsured, and Louisiana indicated plans to modernize its hospital reimbursement methods by converting from cost report-based per diem payment to value-based payment.
All-payer claims database (APCD) systems are large-scale databases that systematically collect medical claims, pharmacy claims, dental claims (typically, but not always), and eligibility and provider files from both private and public payers. APCD can be used to help identify areas to focus reform efforts and for other purposes. Eleven states (Colorado, Connecticut, Massachusetts, Maine, Minnesota, New Hampshire, Oregon, Rhode Island, Tennessee, Virginia, and Wisconsin) reported having APCDs in place in FY 2015. An additional state (Maryland) reported implementing an APCD in FY 2016, and four states (Delaware, Hawaii, Vermont, and Washington) reported plans to implement an APCD in FY 2017. New Mexico reported that planning was underway for the future implementation of an APCD.
Table 10: Delivery System and Payment Reform Initiatives in Place in all 50 States and DC, FY 2015
| States | Patient-Centered Medical Homes(PCMH) | ACA Health Homes | Accountable Care Organizations (ACO) | Episode of Care Payments | Delivery System Reform Incentive Payment Program (DSRIP) | Other Initiatives | Any of these Initiatives in Place in FY 2015 |
| Alabama | X | X | X | ||||
| Alaska | |||||||
| Arizona | |||||||
| Arkansas | X | X | X | ||||
| California | X | X | |||||
| Colorado | X | X | X | ||||
| Connecticut | X | X | X | ||||
| Delaware | |||||||
| DC | |||||||
| Florida | X | X | |||||
| Georgia | |||||||
| Hawaii | |||||||
| Idaho | X | X | X | ||||
| Illinois | |||||||
| Indiana | |||||||
| Iowa | X | X | X | ||||
| Kansas | X | X | X | ||||
| Kentucky | |||||||
| Louisiana | X | X | |||||
| Maine | X | X | X | X | |||
| Maryland | X | X | X | ||||
| Massachusetts | X | X | X | ||||
| Michigan | X | X | X | ||||
| Minnesota | X | X | X | ||||
| Mississippi | |||||||
| Missouri | X | X | X | ||||
| Montana | X | X | |||||
| Nebraska | X | X | |||||
| Nevada | |||||||
| New Hampshire | |||||||
| New Jersey | X | X | X | X | X | ||
| New Mexico | X | X | X | ||||
| New York | X | X | X | X | |||
| North Carolina | X | X | X | ||||
| North Dakota | |||||||
| Ohio | X | X | |||||
| Oklahoma | X | X | X | X | |||
| Oregon | X | X | X | X | |||
| Pennsylvania | |||||||
| Rhode Island | X | X | X | ||||
| South Carolina | X | X | |||||
| South Dakota | X | X | |||||
| Tennessee | X | X | X | ||||
| Texas | X | X | X | ||||
| Utah | |||||||
| Vermont | X | X | X | X | |||
| Virginia | X | X | |||||
| Washington | X | X | |||||
| West Virginia | X | X | |||||
| Wisconsin | X | X | X | ||||
| Wyoming | X | X | |||||
| Totals | 29 | 20 | 7 | 2 | 7 | 2 | 36 |
| NOTES: “Other initiatives” – OK and OR reported participating in the CMS Innovation Center’s Comprehensive Primary Care Initiative. Oregon has a hospital quality incentive program that is “DSRIP-like” and is authorized under a Section 1115 waiver but is not counted here.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | |||||||
Table 11: Delivery System and Payment Reform Actions Taken in All 50 States and DC, FY 2016 and FY 2017
| States | Patient-Centered Medical Homes(PCMH) | ACA Health Homes | Accountable Care Organizations (ACO) | Episode of Care Payments | Delivery System Reform Incentive Payment Program(DSRIP) | Other Initiatives | Any New or Expanded Initiative | |||||||
| 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | |
| Alabama | X | X | ||||||||||||
| Alaska | X | X | ||||||||||||
| Arizona | X | X | ||||||||||||
| Arkansas | X | X | X | X | X | X | ||||||||
| California | X | X | X | X | X | X | ||||||||
| Colorado | X | X | X | X | X | X | ||||||||
| Connecticut | X | X | ||||||||||||
| Delaware | X | X | ||||||||||||
| DC | X | X | X | X | ||||||||||
| Florida | X | X | ||||||||||||
| Georgia | ||||||||||||||
| Hawaii | ||||||||||||||
| Idaho | X | X | ||||||||||||
| Illinois | ||||||||||||||
| Indiana | ||||||||||||||
| Iowa | ||||||||||||||
| Kansas | ||||||||||||||
| Kentucky | ||||||||||||||
| Louisiana | X | X | ||||||||||||
| Maine | X | X | X | X | ||||||||||
| Maryland | ||||||||||||||
| Massachusetts | X | X | X | X | X | X | X | |||||||
| Michigan | X | X | X | X | X | |||||||||
| Minnesota | X | X | X | X | X | |||||||||
| Mississippi | ||||||||||||||
| Missouri | X | X | X | X | X | |||||||||
| Montana | ||||||||||||||
| Nebraska | ||||||||||||||
| Nevada | ||||||||||||||
| New Hampshire | X | X | ||||||||||||
| New Jersey | X | X | X | X | X | X | ||||||||
| New Mexico | X | X | X | X | X | X | X | X | X | X | X | |||
| New York | X | X | X | X | X | X | ||||||||
| North Carolina | ||||||||||||||
| North Dakota | ||||||||||||||
| Ohio | X | X | X | |||||||||||
| Oklahoma | ||||||||||||||
| Oregon | ||||||||||||||
| Pennsylvania | X | X | X | X | X | X | ||||||||
| Rhode Island | X | X | X | X | X | X | X | |||||||
| South Carolina | ||||||||||||||
| South Dakota | ||||||||||||||
| Tennessee | X | X | X | X | X | X | X | |||||||
| Texas | ||||||||||||||
| Utah | ||||||||||||||
| Vermont | X | X | ||||||||||||
| Virginia | X | X | ||||||||||||
| Washington | X | X | X | X | X | X | X | |||||||
| West Virginia | ||||||||||||||
| Wisconsin | ||||||||||||||
| Wyoming | X | X | X | X | ||||||||||
| Totals | 11 | 13 | 6 | 7 | 5 | 11 | 3 | 7 | 4 | 5 | 4 | 5 | 21 | 25 |
| NOTES: Expansions of existing initiatives include rollouts of existing initiatives to new areas or groups and significant increases in enrollment or providers.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||||||||||
Report: Long-term Services And Supports Reforms
Key Section Findings
- Nearly every state reported actions to expand the number of persons served in community settings in both years (46 states in FY 2016 and 47 states in FY 2017). Forty-two (42) states in FY 2016 and 41 states in FY 2017 increased or plan to increase the number of people served in community-based settings through existing or expanded Section 1915(c) HCBS waivers, new Section 1915(i) HCBS State Plan Options, or Section 1915(k) Community First Choice State Plan Amendments. Twenty-three (23) states in FY 2016 and 18 states in FY 2017 reported expanding PACE programs through new enrollment and/or new sites. Nineteen (19) states in FY 2016 and 18 states in FY 2017 report they include/will include specific rebalancing incentives in managed care contracts that cover LTSS. Fourteen (14) states in FY 2016 and nine states in FY 2017 expect to close or downsize a state institution and transition residents into community settings.
- In June 2015, CMS issued an Informational Bulletin to clarify when and how Medicaid reimburses for certain housing-related activities. Sixteen (16) states reported that they have or will implement or expand housing-related services as outlined in the Informational Bulletin in FY 2016 or FY 2017.
- Twenty-three (23) states provided some or all LTSS through a managed care arrangement as of July 1, 2016; 15 states offered MLTSS on a statewide basis for at least some LTSS populations. Twenty-one (21) states offered at least one MCO arrangement that covers both Medicaid acute and Medicaid LTSS (including dual eligible demonstration models), while five states offer a prepaid health plan that covers only Medicaid LTSS. In FY 2016, four states implemented MLTSS or expanded MLTSS to new parts of the state, and four states expanded MLTSS to new populations. In FY 2017, two states anticipate geographic expansion in MLTSS, while five states anticipate adding new populations to MLTSS.
- Enrollment into the MLTSS program is mandatory statewide for seniors in 13 of the 23 MLTSS states, for individuals with ID/DD in eight states, for non-elderly adults with physical disabilities in 12 states, and for individuals who have full dual eligibility status in nine states.
Additional information on LTSS expansions implemented in FY 2016 or planned for FY 2017 as well as state-level details on capitated MLTSS models can be found in Tables 12 and 13.
LTSS System Rebalancing
Medicaid is the nation’s primary payer for long-term services and supports (LTSS), covering a continuum of services ranging from home and community-based services (HCBS) that allow persons to live independently in their own homes or in other community settings to institutional care provided in nursing facilities (NF) and intermediate care facilities for individuals with intellectual disabilities (ICF-ID). In 2013, spending on HCBS (51 percent of total LTSS expenditures) surpassed spending on institutional LTSS (49 percent of total LTSS expenditures) for the first time in the history of the program. The trend toward spending for services in the community continues, with the percentage of spending for HCBS in 2014 growing to 53 percent of total LTSS spending.44 This achievement represents a fundamental rebalancing of program expenditures over the last twenty years; in 1995, Medicaid reported that 82 percent of national expenditures on LTSS was in institutional settings, and that share is now less than half.45
This year’s survey shows, once again, that a large majority of states are employing a variety of tools and strategies to expand the number of people served in home and community-based settings for LTSS, including serving more people through existing or expanded Section 1915(c) HCBS waivers, new Section 1915(i) HCBS State Plan Option or Section 1915(k) Community First Choice State Plan Amendments, PACE program growth,46 and incentives to support system rebalancing through use of managed long-term services and supports (MLTSS). Expanding the numbers of individuals served through HCBS waivers and SPAs remains the most popular strategy, with 42 states in FY 2016 and 41 states in FY 2017 reporting they plan to increase the number of Section 1915(c) waiver slots, serve more individuals under existing Section 1915(c) waiver program caps, or are adding Section 1915(i) or Section 1915(k) state plan options to serve more individuals (Figure 7). Four states reported implementing a new Section 1915(i) state plan option for targeted populations in FY 2016, with five states intending to implement a Section 1915(i) state plan option for targeted populations in FY 2017. Three states implemented a new Section 1915(k) state plan option in FY 2016 (Connecticut, New York and Washington), while one state intends to implement Section 1915(k) in FY 2017 (Wyoming).
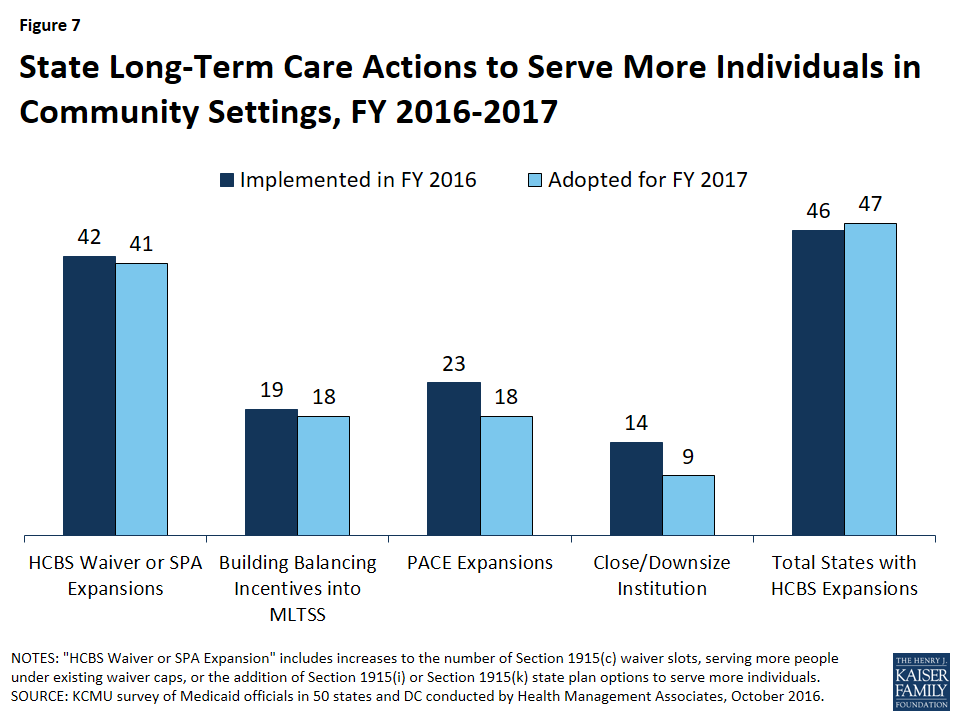
Several states are also using MLTSS strategies intended to serve more individuals in home and community-based settings. Nineteen (19) states in FY 2016 and 18 states in FY 2017 report they include or will include specific rebalancing incentives (performance targets and/or financial incentives) in managed care contracts to encourage MCOs that cover LTSS to expand access to HCBS. This includes a number of states that provide HCBS for some or all populations under Section 1115 waivers, in connection with MLTSS, instead of through Section 1915(c).47 For example, Tennessee, which transferred its Section 1915(c) waiver services for several LTSS populations to a Section 1115 waiver with its expansion to MLTSS in 2010 (TennCare CHOICES), reported that it expects to convert its remaining Section 1915(c) waiver for persons with intellectual and developmental disabilities to Section 1115 authority in FY 2017. The state anticipates significant expansion in HCBS capacity for this population under its managed care approach. See the MLTSS section below for more detail on the use of MLTSS in state Medicaid programs.
PACE continues to be reported as a rebalancing tool, with 23 states in FY 2016 and 18 states in FY 2017 expecting growth in these programs. For most of these states, growth will come within existing PACE sites; however, Florida, New Jersey, Rhode Island, Texas, and Maryland reported that they expect to add at least one new PACE site.
Further, 14 states in FY 2016 and nine states in FY 2017 expect to close or downsize a state institution and transition residents into community settings. This strategy is still an important tool of rebalancing. In addition, both Indiana and Massachusetts imposed a moratorium on new nursing facility beds in FY 2016.
States were also asked if they have adopted or plan to adopt new restrictions on the number of people served in the community in FY 2016 or FY 2017. Two states (Missouri and North Carolina) reported that they were acting to restrict PACE programs. Missouri terminated its PACE site in FY 2016, and North Carolina, citing concern over the rate of growth in the program, placed a limit on the number of individuals each PACE site can enroll each month. Virginia reported that it intends to cap enrollment in its Alzheimer’s Assisted Living Section 1915(c) waiver, which currently serves 56 people and cannot meet the new CMS regulatory standards for home and community-based settings; a stakeholder group is meeting to formulate plans for individuals who are currently served under the waiver. While New Jersey is expanding HCBS through other initiatives, the state also increased the number of institutional LTSS beds in FY 2016; 60 additional long-term care beds for individuals who have Huntington Disease were approved by the New Jersey Department of Health in FY 2016.
Table 12 shows state use of LTSS rebalancing tools in FY 2016 and FY 2017.
Housing Supports
In June 2015, CMS issued an Informational Bulletin to clarify when and how Medicaid reimburses for certain housing-related activities, including individual housing transition services, individual housing and tenancy sustaining services, and state-level housing related collaborative activities.48 CMS’s intent was to assist states in designing benefits that support community integration for seniors, individuals with disabilities, and individuals experiencing chronic homelessness.
Many of the services outlined in CMS’s Informational Bulletin were developed under the auspices of federal grant programs, including the Money Follows the Person (MFP) rebalancing demonstration. MFP is a federal grant program, enacted under the Deficit Reduction Act of 2005 and extended through September 2016 by the Affordable Care Act, which operates in 44 states. Enhanced federal funding under MFP has supported the transition of over 52,000 individuals from institutional to home and community-based settings of LTSS as of mid-2015.49 Under MFP, states identified the lack of affordable and accessible housing as a major barrier to assisting individuals to leave institutional settings of care. With MFP resources, many states have offered new housing related services, incorporated housing expertise within the Medicaid program to increase the likelihood of successful community living for persons who need supports, and engaged in strategic activities to assist in identifying and securing housing resources for individuals who choose HCBS.
After September 2016, states can continue to transition individuals under MFP through 2018 (with CMS approval) and have through 2020 to use their remaining funding.50 As of July 2016, 23 states reported that they currently offer housing-related services under a state plan, Section 1915(c), or 1115 waiver authority that the state intends to continue after the expiration of the MFP grant program. Most of these states are using current Section 1915(c) waivers that provide community transition services and environmental modifications for seniors, individuals with physical disabilities and/or individuals with intellectual or developmental disabilities. Other states, including Alabama, Kansas, Massachusetts, Michigan, and Ohio, plan to continue to offer housing coordinators or other search services to assist waiver beneficiaries. States have noted that some demonstration services and program supports will terminate when MFP funding expires.
Beyond MFP, 16 states reported that they have or will implement or expand housing-related services, as outlined in the Informational Bulletin, in FY 2016 or FY 2017; one state has done so in 2016, eight states plan to do so in 2017, and seven states plan to implement or expand housing-related services in both years (Exhibit 7). States report planning to use an array of authorities, in addition to Section 1915(c) waivers, for offering housing related services. For example, the District of Columbia proposes to offer health home services that support links to housing, and California proposes to offer housing-related services using a health home-based Whole Person Care Pilot under a Section 1115 waiver. New Jersey plans to use a Section 1115 waiver to offer a supported housing benefit to a wide range of Medicaid beneficiaries, including people who are homeless, at-risk of homelessness, residing in nursing facilities, are jail involved, or have a behavioral health diagnosis. Connecticut proposes to provide transition supports and tenancy sustaining supports using a Section 1915(i) State Plan Amendment. Under its Section 1115 waiver, Washington plans to provide supportive housing services (including individual housing transition services and individual tenancy sustaining services) to Medicaid beneficiaries age 18 or older who meet HUD’s definition of “chronically homeless” or have frequent or lengthy institutional contacts or adult residential care stays or have frequent in-home caregiver/provider turnover or meet specific risk criteria.51
| Exhibit 7: States Implementing or Expanding Housing-related Services Outlined in the CMS Informational Bulletin | ||
| FY 2016 only | FY 2017 only | both FY 2016 and FY 2017 |
| OH | CT, DC, DE, FL, HI, NJ, RI, VT | CA, LA, MA, NC, OK, TN, WA |
HCBS Benefit Changes
Ten (10) states in FY 2016 and 14 states in FY 2017 reported a wide variety of HCBS benefit additions or expansions. HCBS benefits include those in Section 1915(c) waivers, Section 1915(i) authority, Section 1915(k) authority (known as “Community First Choice” or “CFC”), and State Plan personal care services, home health services and private duty nursing (Exhibit 8).52 For example, three states in FY 2016 (Connecticut, New York, and Washington) and one in FY 2017 (Wyoming) reported implementing or planning to implement CFC and one state (Texas) reported enhancing its CFC benefit in FY 2016 to add transportation and respite services for persons with intellectual and developmental disabilities; one state in FY 2016 (Texas) and two in FY 2017 (Florida and South Carolina) reported behavioral health-related HCBS service additions; two states (Colorado and DC) reported increasing access to consumer directed service options in FY 2016; two states (Pennsylvania and Tennessee ) reported employment services expansions in FY 2017 and one state (Wisconsin) reported adding consultative and therapeutic services for caregivers and training services for unpaid caregivers in FY 2016.
HCBS benefit restrictions reflect the elimination of a covered benefit or the application of utilization controls for existing benefits. For FY 2016, West Virginia applied service limitations in its home and community-based services waiver serving persons with intellectual and developmental disabilities and eliminated a benefit in two other HCBS waivers. For FY 2017, Tennessee is limiting coverage for facility-based day services (Exhibit 8).
| Exhibit 8: HCBS Benefit Enhancements or Additions and Restrictions or Eliminations | ||
| Benefit | FY 2016 | FY 2017 |
| HCBS Enhancements or Additions | CA, CO, CT, DC, MS, MT, NY, TX, WA, WI | CT, DC, FL, ID, IN, KY, MA, MN, MT, PA, SC, SD, TN, WY |
| HCBS Restrictions or Eliminations | WV | TN |
Capitated Managed Long-Term Services and Supports (MLTSS)
As of July 1, 2016, almost half of states (23 states) covered LTSS through one or more of three types of capitated managed care arrangements: a Medicaid MCO (covering Medicaid acute care and LTSS), a PHP (covering only Medicaid LTSS), or an MCO arrangement for dually eligible beneficiaries (covering Medicaid and Medicare acute care and Medicaid LTSS services in a single contract, under the financial alignment demonstration for dual eligibles). Of the 23 states that reported using one or more of these MLTSS models, eight states reported using two models, and one state (New York) reported using all three. Among states with MLTSS arrangements, 18 states offer a Medicaid MCO that covers both Medicaid acute services and Medicaid LTSS. (Michigan, South Carolina, and Virginia are not among these 18 states; however, they use Medicaid MCOs that cover Medicaid acute and Medicaid LTSS (as well as Medicare acute care) in financial alignment demonstration (FAD) initiatives for duals.) Just five states reported offering a Medicaid LTSS PHP. Of the states with capitated MLTSS, 15 offered some form of managed care plan on a statewide basis for at least some LTSS populations as of July 1, 2016.
Nine states offered an MCO-based FAD (California, Illinois, Massachusetts, Michigan, New York, Ohio, South Carolina, Texas, and Virginia) as of July 1, 2016.53 The FAD model involves a three-way contract between an MCO, Medicare and the state Medicaid program.54 ,55 Two states reported new FAD initiatives: in FY 2016, New York launched a second FAD initiative, contracting with a managed care plan to serve dual eligibles with ID/DD, and Rhode Island is launching a FAD initiative in FY 2017. Massachusetts also operates an administrative alignment demonstration (without financial alignment) for dually eligible beneficiaries (Senior Care Options (SCO) program). Minnesota only operates an administrative alignment demonstration (without financial alignment) for dually eligible beneficiaries (Minnesota Senior Health Options program).
Other states not participating in a formal demonstration have taken action to encourage improved coordination and integration of services for the dually eligible population under MCO arrangements. Five states (Arizona, Hawaii, Minnesota, Tennessee, and Wisconsin) reported that they require Medicaid-contracting MCOs to also offer a Medicare Dual Eligible Special Needs Plan (D-SNP)56 to allow a beneficiary to choose to receive Medicare as well as Medicaid benefits from the same plan (though not through a single contract). Florida contracts for MLTSS with MCOs that are chronic disease SNPs57 for dually eligible beneficiaries. New Jersey and Idaho reported that at least one plan in each state is a Fully Integrated Dual Eligible (FIDE) SNP,58 which allows beneficiaries to choose a single MCO to offer both Medicare and Medicaid benefits, creating an opportunity for improved coordination and integration.
Table 13 provides state-level details on MLTSS models.
MLTSS Enrollment
This year’s survey also asked states with capitated MLTSS arrangements whether, as of July 1, 2016, certain populations were enrolled on a mandatory or voluntary basis or were always excluded. On the survey, states selected from “always mandatory,” “always voluntary,” “varies (by geography or other factor),” or “always excluded” for the following populations: seniors, persons with ID/DD, non-elderly adults with physical disabilities, and full-benefit dually eligible beneficiaries. As shown in Exhibit 9 below, seniors were most likely to be enrolled on a mandatory basis statewide (13 states), while persons with ID/DD were least likely to be enrolled on a mandatory basis (8 states) and also most likely to be excluded from MLTSS enrollment (4 states). No state with a MLTSS program always excludes individuals who have full dual eligibility status from enrollment.
| Exhibit 9: MLTSS Enrollment by Populations, July 1, 2016(# of States) | ||||
| Seniors | Persons with ID/DD | Nonelderly Adults with Physical Disabilities | Full Benefit Dual Eligibles | |
| Always mandatory | 13 | 8 | 12 | 9 |
| Always voluntary | 5 | 6 | 4 | 7 |
| Varies (by geography or other factor) | 4 | 5 | 4 | 7 |
| Always excluded | 1 | 4 | 3 | 0 |
MLTSS Benefits
Almost every MLTSS state (22 states) includes both institutional and HCBS in the same contractual arrangement, though this sometimes varies within a state across populations. For example, Minnesota offers both institutional and HCBS in the same MCO for seniors, but only institutional LTSS are included in an MCO for individuals with disabilities who are under the age of 65. One state (North Carolina) reported covering only HCBS in its MLTSS program for persons with intellectual and developmental disabilities.
MLTSS Population Changes
The growth in the use of MLTSS has continued since the prior survey reporting period. In FY 2015, six states (California, Michigan, New Jersey, New York, South Carolina, and Texas) implemented MCO arrangements for LTSS for at least some populations, some of these in conjunction with implementing a FAD. In FY 2016, four states implemented or expanded MLTSS to new parts of the state, while four states expanded MLTSS to new populations. South Carolina and Wisconsin anticipate geographic expansion in MLTSS in FY 2017, while five states anticipate adding populations to MLTSS in FY 2017 (Exhibit 10).
| Exhibit 10: MLTSS Population Expansions, FY 2016 and FY 2017 | ||
| FY 2016 | FY 2017 | |
| Geographic Expansions | IA, ID, SC, WI | SC, WI |
| New Population Groups Added | IA, NJ, NY, SC | IL, NY, SC, TN, TX |
Only two states reported any reduction in the use of MLTSS. Massachusetts imposed a temporary cap on enrollment in OneCare (its FAD model) in FY 2016, but that cap was subsequently lifted. Idaho noted that its one MLTSS PHP is expected to reduce its service plan area in FY 2017.
MLTSS Quality
Most states with MCO programs track state-identified quality measures and require other health plan quality-related activities to improve health care outcomes and plan performance under MLTSS. Thirteen (13) states with MCOs offering LTSS reported having LTSS quality measures in place in FY 2015. In FY 2016, a total of six states implemented new or expanded MLTSS quality measures, bringing the total to 15 states. Five of these 15 states plan to expand quality measures for LTSS in FY 2017. See Exhibit 11 for information on states with quality measures for MLTSS.
CMS has identified a gap in the national availability of tested, reliable and valid quality measures for HCBS. A variety of efforts are underway to address this gap. The US Department of Health and Human Services has contracted with the National Quality Forum to create a conceptual framework for HCBS quality measurement; to synthesize existing evidence, measures, and measure concepts; to identify gaps in HCBS measures based on the framework; and to make recommendations for HCBS measure development efforts.59 60
| Exhibit 11: MLTSS Quality Measures, FY 2015, FY 2016, and FY 2017 | ||
| In Place FY 2015 | New/Expanded FY 2016 | New/Expanded FY 2017 |
| AZ, CA, DE, FL, IL, KS, MA,MN, NM, OH, TN, TX, VA | AZ, CA, DE, IA, NJ, VA | AZ, CA, DE, TX, VA |
Table 12: Long-Term Care Expansions in All 50 States and DC, FY 2016 and FY 2017
| States | Waiver or SPA Expansions | Building Balancing Incentives in MLTSS | PACE Expansions | Downsize/Close Institution | Total States with HCBS Expansions | |||||
| 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | |
| Alabama | X | X | X | X | ||||||
| Alaska | ||||||||||
| Arizona | X | X | X | X | ||||||
| Arkansas | X | X | X | X | X | X | ||||
| California | X | X | X | X | X | X | X | X | X | X |
| Colorado | X | X | X | X | X | X | ||||
| Connecticut | X | X | X | X | X | X | ||||
| Delaware | X | X | X | X | X | X | X | X | X | |
| DC | X | X | X | X | ||||||
| Florida | X | X | X | X | X | X | X | X | ||
| Georgia | X | X | ||||||||
| Hawaii | X | X | X | X | ||||||
| Idaho | X | X | X | X | ||||||
| Illinois | X | X | X | X | X | X | ||||
| Indiana | X | X | X | X | X | X | ||||
| Iowa | X | X | X | X | X | X | X | |||
| Kansas | X | X | X | X | X | X | ||||
| Kentucky | X | X | ||||||||
| Louisiana | ||||||||||
| Maine | X | X | X | X | ||||||
| Maryland | X | X | X | X | ||||||
| Massachusetts | X | X | X | X | X | X | X | X | ||
| Michigan | X | X | X | X | X | X | X | X | ||
| Minnesota | X | X | X | X | ||||||
| Mississippi | X | X | X | X | X | X | X | |||
| Missouri | X | X | X | X | ||||||
| Montana | X | X | X | X | X | X | ||||
| Nebraska | X | X | X | X | ||||||
| Nevada | X | X | X | X | ||||||
| New Hampshire | X | X | X | X | ||||||
| New Jersey | X | X | X | X | X | |||||
| New Mexico | X | X | X | X | ||||||
| New York | X | X | X | X | X | X | X | X | X | |
| North Carolina | X | X | X | |||||||
| North Dakota | X | X | X | X | X | |||||
| Ohio | X | X | X | X | X | X | ||||
| Oklahoma | X | X | X | X | X | X | ||||
| Oregon | X | X | X | X | X | X | ||||
| Pennsylvania | X | X | X | X | X | X | X | X | X | |
| Rhode Island | X | X | X | X | X | X | X | X | ||
| South Carolina | X | X | X | X | X | X | X | |||
| South Dakota | X | X | X | X | ||||||
| Tennessee | X | X | X | X | X | X | X | X | ||
| Texas | X | X | X | X | X | X | X | X | X | X |
| Utah | X | X | X | X | ||||||
| Vermont | ||||||||||
| Virginia | X | X | X | X | X | X | X | X | X | X |
| Washington | X | X | X | X | X | X | ||||
| West Virginia | X | X | X | X | ||||||
| Wisconsin | X | X | X | X | X | X | ||||
| Wyoming | X | X | X | X | X | X | ||||
| Totals | 42 | 41 | 19 | 18 | 23 | 18 | 14 | 9 | 46 | 47 |
| NOTES: “HCBS Waiver or SPA Expansions” include increases to the number of Section 1915(c) waiver slots, serving more people under existing waiver caps, or the addition of Section 1915(i) or Section 1915(k) state plan options to serve more individuals. In addition to the actions reported here, two states (IN and MA) also reported imposing a moratorium on construction of new nursing facility beds in FY 2016.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||||||
Table 13: Capitated MLTSS Models in all 50 States and DC, as of July 1, 2016
| States | Medicaid MCO | PHP | Medicare + Medicaid Demonstration | Any MLTSS | Statewide |
| Alabama | |||||
| Alaska | |||||
| Arizona | X | X | X | ||
| Arkansas | |||||
| California | X | X | X | ||
| Colorado | |||||
| Connecticut | |||||
| Delaware | X | X | X | ||
| DC | |||||
| Florida | X | X | X | ||
| Georgia | |||||
| Hawaii | X | X | X | ||
| Idaho | X | X | |||
| Illinois | X | X | X | ||
| Indiana | |||||
| Iowa | X | X | X | ||
| Kansas | X | X | X | ||
| Kentucky | |||||
| Louisiana | |||||
| Maine | |||||
| Maryland | |||||
| Massachusetts | X | X* | X | ||
| Michigan | X | X | X | X | |
| Minnesota | X | X* | X | X | |
| Mississippi | |||||
| Missouri | |||||
| Montana | |||||
| Nebraska | |||||
| Nevada | |||||
| New Hampshire | |||||
| New Jersey | X | X | X | ||
| New Mexico | X | X | X | ||
| New York | X | X | X | X | X |
| North Carolina | X | X | X | ||
| North Dakota | |||||
| Ohio* | X | X | X | ||
| Oklahoma | |||||
| Oregon | |||||
| Pennsylvania | |||||
| Rhode Island* | X | X | X | ||
| South Carolina | X | X | |||
| South Dakota | |||||
| Tennessee | X | X | X | ||
| Texas | X | X | X | X | |
| Utah | |||||
| Vermont | |||||
| Virginia | X | X | |||
| Washington | |||||
| West Virginia | |||||
| Wisconsin | X | X | X | ||
| Wyoming | |||||
| Totals | 18 | 5 | 10 | 23 | 15 |
| NOTES: States were asked whether they cover long-term services supports through any of the following managed care (capitated) arrangements as of July 1, 2016: Medicaid MCO (MCO covers Medicaid acute + Medicaid LTSS); PHP (covers only Medicaid LTSS); or Medicare + Medicaid Demonstration (Medicaid MCO covers Medicaid and Medicare acute + Medicaid LTSS). “Medicare + Medicaid Demonstration” – these states use Medicaid MCOs in Financial Alignment Demonstration (FAD) initiatives which involve care coordination for dually eligible beneficiaries. States were also asked whether MLTSS plans were operating in all regions of the state as of July 1, 2015 (statewide). *MA operates a FAD and another administrative alignment demonstration for dually eligible beneficiaries. *MN operates an administrative alignment demonstration (without financial alignment) for dually eligible beneficiaries. *RI is launching a FAD initiative in FY 2017. *OH offers a Medicaid MCO (MCO offers Medicaid acute + Medicaid LTSS) only in those counties where the FAD is offered; dually eligible seniors who opt out of the FAD must enroll in this Medicaid MCO model for Medicaid services.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | |||||
Report: Provider Rates And Taxes
Key Section Findings
- Provider rate changes are often tied to the economy. In FY 2016, more states implemented rate increases (45 states) compared to rate restrictions (38 states). For FY 2017, fewer states adopted rate increases (40 states) than rate restrictions (41 states). States were more likely to increase rates for outpatient hospital, primary care physicians, specialist physicians, dentists, MCOs, and nursing facilities and more likely to restrict rates for inpatient hospitals. A growing number of states are adopting reimbursement policies to reduce potentially preventable readmissions and early elective deliveries.
- All states except Alaska rely on provider taxes and fees to provide a portion of the non-federal share of the costs of Medicaid. In recent years, states made very few changes to the number of provider taxes but increased the level of provider taxes. Fifteen (15) states increased one or more provider tax rates in FY 2016 and 13 states have made or plan to make increases to one or more provider taxes in FY 2017. Eight of the expansion states (Arkansas, Arizona, Colorado, Illinois, Indiana, Louisiana, New Hampshire, and Ohio) reported plans to use new or increased provider taxes or fees, including premium tax revenues, to fund all or part of the state costs of the ACA Medicaid expansion beginning in January 2017.
Tables 14 through 16 provide complete listings of Medicaid provider rate changes and provider taxes and fees in place in FY 2016 and FY 2017.
Provider Rates
Provider rate changes are often tied to the economy. During economic downturns and budget shortfalls, states often turn to rate restrictions to contain costs, while during periods of recovery and revenue growth, states are more likely to increase rates. This report examines rate changes across major provider categories (hospital inpatient, nursing facilities, MCOs, outpatient hospital, primary care physicians, specialists, and dentists). States were asked to report aggregate changes for each major provider category. In FY 2016, more states implemented rate increases (45 states) compared to rate restrictions (38 states). For FY 2017, the number of states with planned or implemented rate restrictions (41 states) is one greater than the number of states with planned rate increases (40 states). FY 2017 is the first year since FY 2012 with a greater number of states planning or implementing rate restrictions than rate increases (Figure 8).
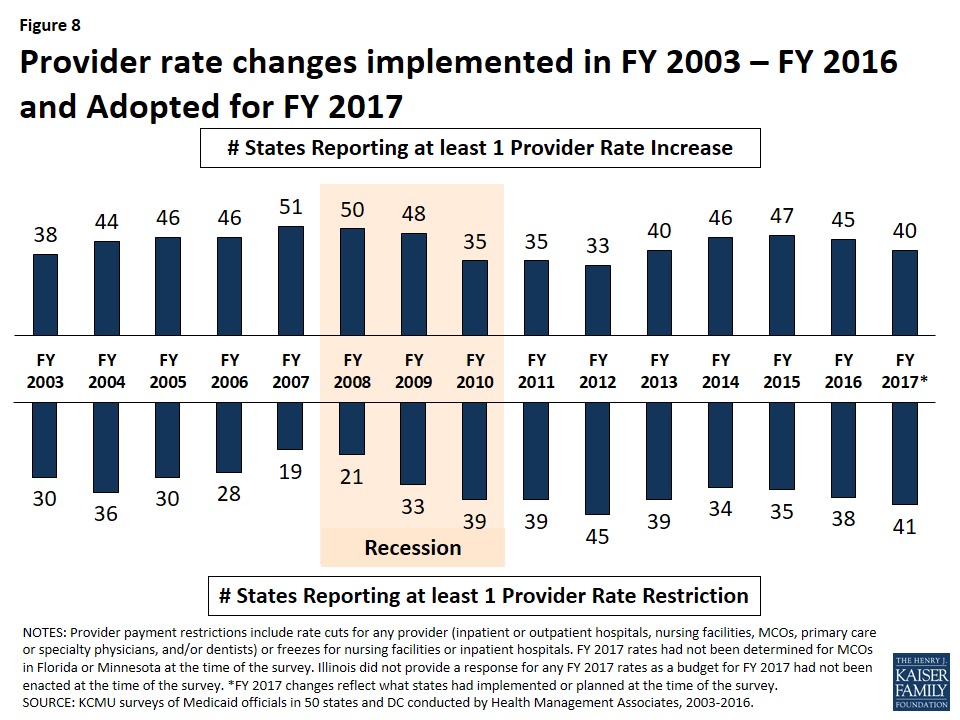
The number of states with rate increases exceeded the number of states with restrictions in FY 2016 and FY 2017 across all major categories of providers (physicians, MCOs, and nursing facilities) with the exception of rates for inpatient hospital services61 (Figure 9).
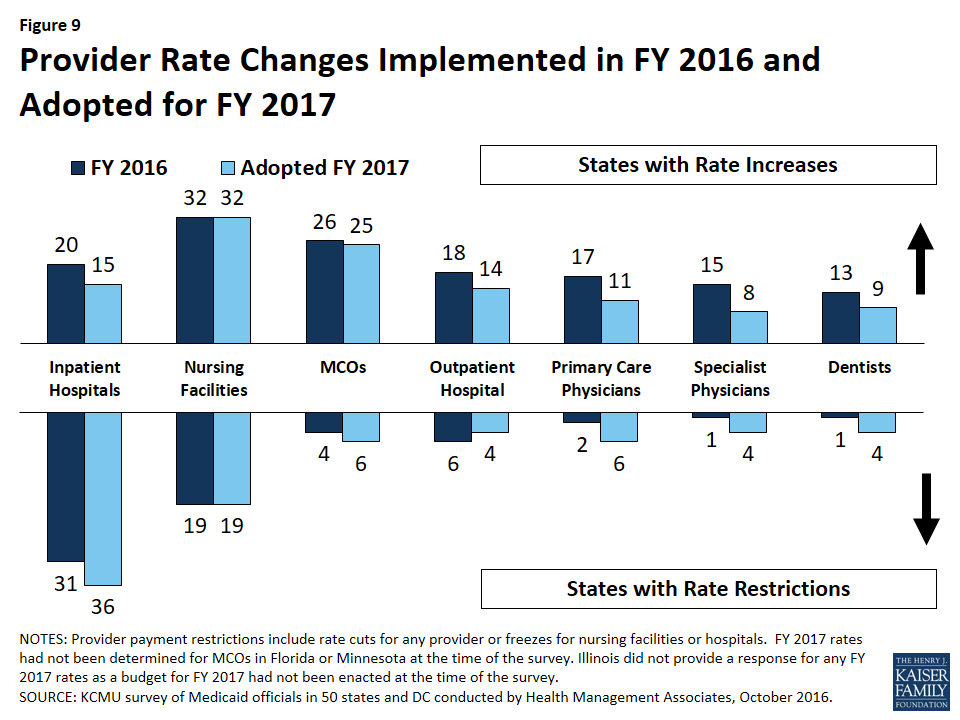
For the purposes of this report, cuts or freezes in rates for inpatient hospitals and nursing facilities are counted as restrictions. Only three states in FY 2016 and five states in FY 2017 had implemented or planned inpatient hospital rate reductions; the vast majority of hospital rate restrictions were freezes in rates. The number of states increasing nursing facility rates dropped in FY 2016 compared to previous years. While four states cut nursing facility rates in FY 2016, only one state indicated a plan to cut nursing facility rates in FY 2017. The other nursing facility rate restrictions are rate freezes.
Capitation payments for Medicaid Managed Care Organizations (MCOs) are generally bolstered by the federal requirement that states pay actuarially sound rates. In FY 2016 and FY 2017, the majority of the 39 states with Medicaid MCOs implemented or planned increases in MCO rates. Four states reported MCO rate cuts in FY 2016, and six states plan to cut MCO rates in FY 2017. Several of these states also reported provider rate reductions in their FFS programs. Three states could not report MCO rate changes for FY 2017 because rate development was not complete. States are increasingly moving to calendar year MCO contracts. The effect of FFS rate restrictions for hospitals, physicians, and nursing facilities rates may have less of a direct impact on providers in states that rely heavily on managed care; however, even states with small FFS programs may use FFS rates as the base for setting MCO rates.
For calendar years 2013 and 2014, the ACA provided funding to increase Medicaid primary care physician (PCP) rates in all states to Medicare rates, with 100 percent federal funding of the rate differential. As a result, recent surveys did not ask about state-initiated Medicaid rates for primary care physicians. This year’s survey included FY 2016 and FY 2017 information about PCPs, specialist physicians, dentists, and outpatient hospital services. Rate increases are more prevalent than rate reductions for FY 2016 through FY 2017 for ambulatory Medicaid providers; however, fewer states adopted rate increases in FY 2017 compared to prior years.
Tables 14 and 15 provide state level details on provider rate changes in FY 2016 and FY 2017.
Potentially Preventable Readmissions
As state Medicaid programs work to improve the quality of health care, increase beneficiary wellness, and reduce costs, one area of focus is a reduction in admissions and readmissions to hospitals that could have been prevented by the provision of appropriate care. States were asked if they had, or planned to implement, an inpatient hospital reimbursement incentive or penalty policy for potentially preventable readmissions in fee-for-service and managed care.
- Eighteen (18) states indicated that they had such policies in place in FY 2015 and two more states implemented such policies in FY 2016. One state indicated that they have plans to implement in FY 2017 in FFS.
- Of the 39 states that use MCOs for part or all of their Medicaid delivery system, eight indicated that they had state directed policies in place in FY 2015. Another three states plan to implement such a policy in FY 2017 and one state plans to implement such a policy after FY 2017. Some MCOs may have their own policies related to potentially preventable readmissions.
Some states reported specific policies related to MCO rates. For example, California adjusts MCO capitation rates to account for incidence of potentially preventable admissions compared to a benchmark for all MCOs. Ohio and Washington reported that capitation rates are based on reductions in potentially preventable readmissions similar to the FFS experience. New York indicated that their Value Based Purchasing model will adjust rates to plans who then can adjust rates to providers.
Early Elective Deliveries
States were asked about reimbursement policies designed to reduce the number of early elective deliveries (EEDs). Nineteen (19) states indicated that they had a FFS policy in place in FY 2016 and one additional state plans to adopt such a policy in FY 2017. For example, states may not pay for any delivery prior to 39-weeks gestation that is the result of either a Cesarean section or induction, unless there is a documented medical reason for the early delivery. Other states pay for these services, but at a reduced rate, or include EEDs as a component of their hospital pay for performance metrics.
Twelve (12) states indicated that as of FY 2016 they required MCOs to have similar policies on EEDs. Two additional states will be implementing such policies in FY 2017. Absent a requirement by the state agency, states report that some MCOs have developed their own policies on EEDs or are following state fee-for-service policy. States are also implementing incentive programs that reward physicians for reducing the rate of early elective deliveries. Some MCOs may elect to have policies that are not directed by the state.
Provider Taxes and Fees
States reported a continued and increasing reliance on provider taxes and fees to provide a portion of the non-federal share of Medicaid costs continued or increased in FY 2016 and FY 2017. At the beginning of FY 2003, a total of 21 states had at least one provider tax in place. Over the next decade, a majority of states imposed new taxes or fees and increased existing tax rates and fees to raise revenue to support Medicaid. By FY 2013, all but one state (Alaska) had at least one provider tax or fee in place.62 In FY 2016, 34 states had three or more provider taxes in place (Figure 10).

The most common provider taxes in place in FY 2016 were taxes on nursing facilities (44 states), followed by taxes on hospitals (40 states) and intermediate care facilities for the intellectually disabled (36 states) (Table 16). Five states in FY 2016 and four states in FY 2017 added new taxes.
- For FY 2016, five states added new provider taxes. DC has a new hospital tax, Connecticut added a tax on ambulatory surgery centers, and Utah added a tax on ambulance providers. Both California and Pennsylvania implemented new MCO taxes, replacing prior MCO taxes that did not meet new federal guidelines for Medicaid MCO taxes.
- For FY 2017, four states are adding new taxes. Louisiana and Wyoming are adding hospital taxes, and Louisiana, Michigan, and Vermont are adding taxes on ambulance providers.
Some states reported changes to existing taxes in FY 2016 and FY 2017. In total there were 15 states that increased one or more provider tax rates in FY 2016 and 13 states have made or plan to make increases in one or more provider taxes in FY 2017. Most notable were rate increases for hospital taxes and fees (six states in FY 2016 and seven states in FY 2017) as well as increases to rates for nursing facility taxes and fees (eight states in FY 2016 and six states in FY 2017). Some states also reported reducing tax rates, again mostly for hospitals (four states in FY 2016 and three states in FY 2017) and nursing facility taxes and fees (one state in FY 2016 and two states in FY 2017).
States were asked whether in the future they planned to use increased provider taxes or fees to fund all or part of the costs of the ACA Medicaid expansion that will occur in calendar year 2017 and beyond when the 100 percent federal match rate for expansion costs starts to decline. Eight of the expansion states (Arkansas, Arizona, Colorado, Illinois, Indiana, Louisiana, New Hampshire, and Ohio) responded that part or all of the non-federal share would be funded with new or increased provider taxes or fees, or with insurance premium taxes.
In addition to the “Medicaid provider taxes” included in this report, several states have more general health care taxes that are used to fund their Medicaid programs. For instance, some states have taxes on insurance premiums or health care claims that apply to all payers. California, Michigan, Ohio, and Pennsylvania are examples of states that had taxes on MCOs that were deemed by the states to be non-Medicaid taxes, but were found by CMS to be non-permissible Medicaid provider taxes. As noted above, California and Pennsylvania have replaced their MCO taxes with ones that meet federal guidelines. The Michigan “use tax,” which applies to MCOs among other entities, is not included in the tables in this report and will end on December 31, 2016. (The Ohio MCO tax is also scheduled to sunset, but not until June 30, 2017.)
Table 14: Provider Rate Changes in all 50 States and DC, FY 2016
| States | Inpatient Hospital | Outpatient Hospital | Primary Care Physicians | Specialists | Dentists | Managed Care Organizations | Nursing Facilities | Total | ||||||||
| Rate Change | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – |
| Alabama | X | X | — | — | X | X | X | |||||||||
| Alaska | X | — | — | X | X | |||||||||||
| Arizona | X | X | X | X | X | |||||||||||
| Arkansas | X | — | — | X | X | X | ||||||||||
| California | X | X | X | X | X | X | ||||||||||
| Colorado | X | X | X | X | X | X | ||||||||||
| Connecticut | X | — | — | X | X | X | ||||||||||
| Delaware | X | X | X | X | X | X | X | X | X | |||||||
| DC | X | X | X | X | X | X | X | |||||||||
| Florida | X | X | X | X | X | |||||||||||
| Georgia | X | X | X | X | X | X | X | |||||||||
| Hawaii | X | X | X | X | X | X | X | X | ||||||||
| Idaho | X | X | X | — | — | X | X | X | ||||||||
| Illinois | X | X | X | X | X | |||||||||||
| Indiana | X | X | X | X | X | |||||||||||
| Iowa | X | X | X | X | ||||||||||||
| Kansas | X | X | X | |||||||||||||
| Kentucky | X | X | X | X | ||||||||||||
| Louisiana | X | X | X | X | ||||||||||||
| Maine | X | — | — | X | X | X | ||||||||||
| Maryland | X | X | X | X | X | X | X | X | X | |||||||
| Massachusetts | X | X | X | X | X | |||||||||||
| Michigan | X | X | X | X | X | |||||||||||
| Minnesota | X | X | X | X | X | X | X | |||||||||
| Mississippi | X | X | X | X | X | X | ||||||||||
| Missouri | X | X | X | X | X | X | X | X | X | |||||||
| Montana | X | X | X | X | X | — | — | X | X | |||||||
| Nebraska | X | X | X | X | X | X | X | |||||||||
| Nevada | X | X | X | X | X | X | X | |||||||||
| New Hampshire | X | X | X | X | X | |||||||||||
| New Jersey | X | X | X | X | X | X | ||||||||||
| New Mexico | X | X | X | X | X | |||||||||||
| New York | X | X | X | X | X | X | ||||||||||
| North Carolina | X | — | — | X | X | |||||||||||
| North Dakota | X | X | X | X | X | X | X | X | ||||||||
| Ohio | X | X | X | X | X | X | X | X | ||||||||
| Oklahoma | X | X | X | X | X | — | — | X | X | |||||||
| Oregon | X | X | X | X | ||||||||||||
| Pennsylvania | X | X | X | X | X | |||||||||||
| Rhode Island | X | X | X | X | X | X | ||||||||||
| South Carolina | X | X | X | X | X | X | ||||||||||
| South Dakota | X | X | X | X | X | — | — | X | X | |||||||
| Tennessee | X | X | X | |||||||||||||
| Texas | X | X | X | X | X | X | ||||||||||
| Utah | X | X | X | X | X | X | X | X | ||||||||
| Vermont | X | X | — | — | X | X | X | |||||||||
| Virginia | X | X | X | X | X | X | ||||||||||
| Washington | X | X | X | X | X | |||||||||||
| West Virginia | X | X | X | X | ||||||||||||
| Wisconsin | X | X | X | X | X | |||||||||||
| Wyoming | X | — | — | X | X | X | ||||||||||
| Totals | 20 | 31 | 18 | 6 | 17 | 2 | 15 | 1 | 13 | 1 | 26 | 4 | 32 | 19 | 45 | 38 |
| NOTES: “+” refers to provider rate increases and “-” refers to provider rate restrictions. For the purposes of this report, provider rate restrictions include cuts to rates for physicians, dentists, outpatient hospitals, and managed care organizations as well as both cuts or freezes in rates for inpatient hospitals and nursing facilities. There are 12 states that did not have Medicaid MCOs in operation in FY 2016; they are denoted as “–” in the MCO column.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||||||||||||
Table 15: Provider Rate Changes in all 50 States and DC, FY 2017
| States | Inpatient Hospital | Outpatient Hospital | Primary Care Physicians | Specialists | Dentists | Managed Care Organizations | Nursing Facilities | Total | ||||||||
| Rate Change | + | – | + | – | + | – | + | – | + | – | + | – | + | – | + | – |
| Alabama | X | — | — | X | X | |||||||||||
| Alaska | X | — | — | X | X | |||||||||||
| Arizona | X | X | X | X | X | |||||||||||
| Arkansas | X | — | — | X | X | X | ||||||||||
| California | X | X | X | X | X | |||||||||||
| Colorado | X | X | X | X | X | |||||||||||
| Connecticut | X | X | — | — | X | X | ||||||||||
| Delaware | X | X | X | X | X | X | X | X | X | |||||||
| DC | X | X | X | X | X | |||||||||||
| Florida | X | X | TBD | X | X | X | ||||||||||
| Georgia | X | X | X | X | X | X | X | |||||||||
| Hawaii | X | X | X | X | X | X | X | X | ||||||||
| Idaho | X | X | X | X | — | — | X | X | X | |||||||
| Illinois | X | TBD | X | X | ||||||||||||
| Indiana | X | X | X | X | X | |||||||||||
| Iowa | X | X | X | |||||||||||||
| Kansas | X | X | X | X | X | X | X | X | X | |||||||
| Kentucky | X | X | X | X | X | X | ||||||||||
| Louisiana | X | X | X | X | X | X | ||||||||||
| Maine | X | — | — | X | X | X | ||||||||||
| Maryland | X | X | X | X | X | |||||||||||
| Massachusetts | X | X | X | X | X | |||||||||||
| Michigan | X | X | X | X | X | |||||||||||
| Minnesota | X | X | TBD | X | X | X | ||||||||||
| Mississippi | X | X | X | X | X | X | X | X | ||||||||
| Missouri | X | X | X | X | X | X | X | X | X | |||||||
| Montana | X | X | X | X | — | — | X | X | X | |||||||
| Nebraska | X | X | X | X | X | X | X | |||||||||
| Nevada | X | X | X | X | X | |||||||||||
| New Hampshire | X | X | X | X | X | |||||||||||
| New Jersey | X | X | X | X | X | |||||||||||
| New Mexico | X | X | X | X | X | X | X | X | ||||||||
| New York | X | X | X | X | X | |||||||||||
| North Carolina | X | — | — | X | X | |||||||||||
| North Dakota | X | X | X | X | X | X | ||||||||||
| Ohio | X | X | X | X | X | |||||||||||
| Oklahoma | X | — | — | X | X | |||||||||||
| Oregon | X | X | X | X | ||||||||||||
| Pennsylvania | X | X | X | X | X | |||||||||||
| Rhode Island | X | X | X | X | X | |||||||||||
| South Carolina | X | X | X | X | X | |||||||||||
| South Dakota | X | X | X | X | X | — | — | X | X | |||||||
| Tennessee | X | X | X | |||||||||||||
| Texas | X | X | X | X | X | |||||||||||
| Utah | X | X | X | X | X | X | X | |||||||||
| Vermont | X | X | — | — | X | X | X | |||||||||
| Virginia | X | X | X | X | X | |||||||||||
| Washington | X | X | X | X | X | |||||||||||
| West Virginia | X | X | X | X | ||||||||||||
| Wisconsin | X | X | X | X | X | |||||||||||
| Wyoming | X | X | X | X | X | — | — | X | X | |||||||
| Totals | 15 | 36 | 14 | 4 | 11 | 6 | 8 | 4 | 9 | 4 | 25 | 6 | 32 | 19 | 40 | 41 |
| NOTES: “+” refers to provider rate increases and “-” refers to provider rate restrictions. For the purposes of this report, provider rate restrictions include cuts to rates for physicians, dentists, outpatient hospitals, and managed care organizations as well as both cuts or freezes in rates for inpatient hospitals and nursing facilities. Wisconsin is moving to APR-DRGs in January 2017, which could impact inpatient and outpatient rates. There are 12 states that did not have Medicaid MCOs in operation in FY 2017; they are denoted as ‘–‘ in the MCO column. TBD – At the time of the survey, calendar year 2017 rates had not been determined for MCOs in Florida, Illinois, or Minnesota. In addition, Illinois only has a budget for the first six months of FY 2017.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||||||||||||
Table 16: Provider Taxes in Place in all 50 States and DC, FY 2016 and FY 2017
| States | Hospitals | Intermediate Care Facilities | Nursing Facilities | Other | Any Provider Tax | |||||
| 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | 2016 | 2017 | |
| Alabama | X | X | X | X | X | X | X | X | ||
| Alaska | ||||||||||
| Arizona | X | X | X | X | X | X | ||||
| Arkansas | X | X | X | X | X | X | X | X | ||
| California | X | X | X | X | X | X | X | X | X | X |
| Colorado | X | X | X | X | X | X | X | X | ||
| Connecticut | X | X | X | X | X | X | X | X | X | X |
| Delaware | X | X | X | X | ||||||
| DC | X | X | X | X | X | X | X | X | X | X |
| Florida | X | X | X | X | X | X | X | X | ||
| Georgia | X | X | X | X | X | X | ||||
| Hawaii | X | X | X | X | X | X | ||||
| Idaho | X | X | X | X | X | X | X | X | ||
| Illinois | X | X | X | X | X | X | X | X | ||
| Indiana | X | X | X | X | X | X | X | X | ||
| Iowa | X | X | X | X | X | X | X | X | ||
| Kansas | X | X | X | X | X | X | ||||
| Kentucky | X | X | X | X | X | X | X* | X* | X | X |
| Louisiana | X | X | X | X | X | X | X* | X | X | |
| Maine | X | X | X | X | X | X | X | X | X | X |
| Maryland | X | X | X | X | X | X | X | X | X | X |
| Massachusetts | X | X | X | X | X | X | X | X | ||
| Michigan | X | X | X | X | X | X | X | |||
| Minnesota | X | X | X | X | X | X | X | X | X | X |
| Mississippi | X | X | X | X | X | X | X | X | X | X |
| Missouri | X | X | X | X | X | X | X* | X* | X | X |
| Montana | X | X | X | X | X | X | X | X | ||
| Nebraska | X | X | X | X | X | X | ||||
| Nevada | X | X | X | X | ||||||
| New Hampshire | X | X | X | X | X | X | ||||
| New Jersey | X | X | X | X | X | X | X* | X* | X | X |
| New Mexico | X* | X* | X | X | ||||||
| New York | X | X | X | X | X | X | X* | X* | X | X |
| North Carolina | X | X | X | X | X | X | X | X | ||
| North Dakota | X | X | X | X | ||||||
| Ohio | X | X | X | X | X | X | X | X | X | X |
| Oklahoma | X | X | X | X | X | X | X | X | ||
| Oregon | X | X | X | X | X | X | ||||
| Pennsylvania | X | X | X | X | X | X | X* | X* | X | X |
| Rhode Island | X | X | X | X | X | X | X | X | ||
| South Carolina | X | X | X | X | X | X | ||||
| South Dakota | X | X | X | X | ||||||
| Tennessee | X | X | X | X | X | X | X | X | X | X |
| Texas | X | X | X | X | X | X | ||||
| Utah | X | X | X | X | X | X | X | X | X | X |
| Vermont | X | X | X | X | X | X | X | X* | X | X |
| Virginia | X | X | X | X | ||||||
| Washington | X | X | X | X | X | X | X | X | ||
| West Virginia | X | X | X | X | X | X | X* | X* | X | X |
| Wisconsin | X | X | X | X | X | X | X | X | X | X |
| Wyoming | X | X | X | X | X | |||||
| Totals | 40 | 42 | 36 | 36 | 44 | 44 | 24 | 21 | 50 | 50 |
| NOTES: This table includes Medicaid provider taxes as reported by states. Some states also have premium or claims taxes that apply to managed care organizations and other insurers. Since this type of tax is not considered a provider tax by CMS, these taxes are not counted as provider taxes in this report. (*) has been used to denote states with multiple “other” provider taxes.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||||||||
Report: Benefits And Pharmacy
Key Section Findings
- A total of 21 states expanded or enhanced covered benefits in FY 2016 and 20 states plan to expand benefits in FY 2017. The most common benefit enhancements reported were for behavioral health and substance use disorder services, telemedicine and tele-monitoring services, and dental services for adults.
- The vast majority of states identified high cost and specialty drugs as a significant cost driver for state Medicaid programs, with most states pointing specifically to hepatitis C antivirals. Thirty-one (31) states in FY 2016 and 23 in FY 2017 reported actions to refine and enhance their pharmacy programs, including policies focused on addressing costs for emerging specialty and high-cost drug therapies.
- Most states have also adopted various pharmacy management strategies targeted at opioid harm reduction including quantity limits (46 states), prior authorization (45 states), clinical criteria (42 states), and step therapy (32 states). Fewer states (12 states) reported requirements in place in FY 2015 for Medicaid prescribers to check their states’ Prescription Drug Monitoring Program before prescribing opioids to a Medicaid patient. These policies refer to FFS programs. Many states also reported policies in place for MCOs. Many states have taken steps to expand access to naloxone to enable family members and first responders to administer the antidote to save lives; for example, half of the states (26) reported making naloxone (in at least one formulation) available without prior authorization or adding naloxone to their PDL.
Tables 17 and 18 provide complete listings of Medicaid benefit changes for FY 2016 and FY 2017. Table 19 provides additional details on Medicaid pharmacy benefit management strategies for opioids in FFS programs in FY 2015-FY 2017.
In this year’s survey, the number of states reporting benefit cuts or restrictions – three in FY 2016 and four in FY 2017 – is comparable to the number reporting cuts in last year’s survey, but remains far below the number seen during the economic downturn. A far larger number of states, 21 states in FY 2016 and 20 in FY 2017, reported enhancing or adding new benefits (Figure 11).
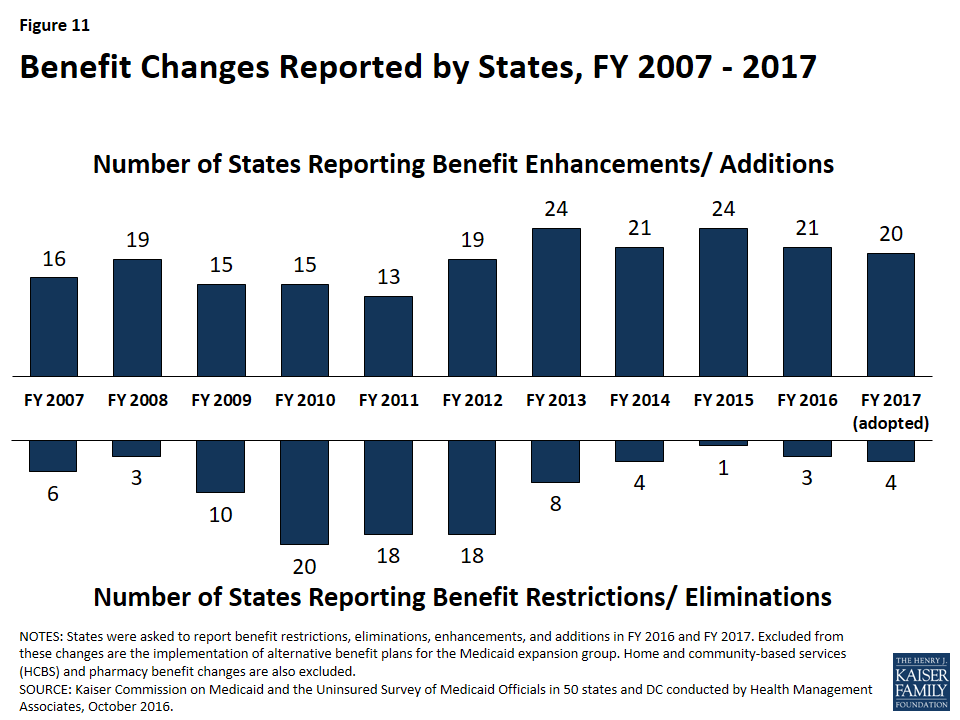
The most common benefit enhancements or additions reported were for behavioral health and/or substance use disorder services, telemedicine and tele-monitoring services, and dental services (Exhibit 12).
| Exhibit 12: Benefit Enhancements or Additions | ||
| Benefit | FY 2016 | FY 2017 |
| Behavioral Health/Substance Use Disorder | MT, NH, NY, SC, TX, VT, WY | DC, HI, NE, NJ, RI, TX, VA |
| Telemedicine / Tele-monitoring | DC, GA, NV, NY, OK, VT | NE, RI, TX |
| Dental Services | MT, NY | AZ, MD, OR, VT |
California and Michigan implemented other notable benefit expansions in FY 2016. California expanded benefits to provide the full Medicaid benefit package to pregnant women between 60 and 138 percent FPL in place of the former, more limited pregnancy-related benefit package. As part of its Section 1115 waiver to expand coverage to additional children and pregnant women with lead exposure from tainted water in Flint, Michigan implemented Targeted Case Management services for waiver enrollees. For FY 2017, other notable benefit expansions include Oregon’s expanded coverage for alternative back therapies including acupuncture, chiropractic manipulation and yoga (to reduce reliance on medications and surgeries) and Rhode Island’s new “STOP” (Sobering Treatment Opportunity Program) pilot. This ER diversion pilot in Providence will cover an overnight stay and referral to appropriate counseling for certain homeless individuals.
Benefit restrictions reflect the elimination of a covered benefit or the application of utilization controls for existing benefits. Most benefit restrictions in FY 2016 and FY 2017 were narrowly targeted; however, in FY 2017, Wyoming reported plans to adopt several benefit reductions and eliminations including: eliminating non-emergency adult dental and vision coverage, reducing nursing facility bed-hold days, and applying soft service caps for behavioral health, therapy, and home health services.
Tables 17 and 18 provide state-level information on benefit changes in FY 2016 and FY 2017.
Autism Services
On July 7, 2014, CMS issued an Informational Bulletin63 describing approaches and Medicaid authorities available to cover Autism Spectrum Disorder (ASD) services. The bulletin also clarified state obligations under the Early and Periodic Screening, Diagnostic and Treatment (EPSDT) benefit to cover all medically necessary services for children, including ASD services. A number of states reporting adding coverage for ASD services, but because these policy changes were required they were not counted as positive or negative.
Table 17: Benefit Changes in the 50 States and DC, FY 2016 and FY 2017
| States | FY 2016 | FY 2017 | ||
| Enhancements/ Additions | Restrictions/ Eliminations | Enhancements/ Additions | Restrictions/ Eliminations | |
| Alabama | ||||
| Alaska | X | |||
| Arizona | X | X | ||
| Arkansas | X | |||
| California | X | X | ||
| Colorado | X | |||
| Connecticut | X | |||
| Delaware | ||||
| DC | X | X | ||
| Florida | ||||
| Georgia | X | |||
| Hawaii | X | |||
| Idaho | ||||
| Illinois | ||||
| Indiana | ||||
| Iowa | ||||
| Kansas | X | |||
| Kentucky | ||||
| Louisiana | X | |||
| Maine | ||||
| Maryland | X | X | ||
| Massachusetts | X | |||
| Michigan | X | |||
| Minnesota | X | |||
| Mississippi | ||||
| Missouri | X | |||
| Montana | X | |||
| Nebraska | X | |||
| Nevada | X | X | ||
| New Hampshire | X | X | ||
| New Jersey | X | |||
| New Mexico | ||||
| New York | X | X | ||
| North Carolina | ||||
| North Dakota | ||||
| Ohio | ||||
| Oklahoma | X | X | X | X |
| Oregon | X | |||
| Pennsylvania | ||||
| Rhode Island | X | |||
| South Carolina | X | |||
| South Dakota | X | |||
| Tennessee | X | |||
| Texas | X | X | ||
| Utah | X | |||
| Vermont | X | X | ||
| Virginia | X | |||
| Washington | X | |||
| West Virginia | ||||
| Wisconsin | ||||
| Wyoming | X | X | X | |
| Totals | 21 | 3 | 20 | 4 |
| NOTES: States were asked to report benefit restrictions, eliminations, enhancements, and additions in FY 2016 and FY 2017. Excluded from these changes are the implementation of alternative benefit plans for the Medicaid expansion group. Home and community-based services (HCBS) and pharmacy benefit changes are also excluded.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | ||||
Table 18: States Reporting Benefit Actions Taken in FY 2016 and FY 2017
| State | Fiscal Year | Benefit Changes |
| Alaska | 2017 | Children (+): Will expand availability of Applied Behavioral Analysis services by developing new ABA provider certification requirements. |
| Arizona | 2016 | Adults (+): Remove limits on coverage for certain orthotic devices (October 1, 2015). |
| 2017 | Adults (+): Add coverage for podiatry services (August 6, 2016). LTSS Adults (+): Add a $1,000 per year dental benefit for MLTSS enrollees (October 1, 2016). | |
| Arkansas | 2017 | Expansion Adults (-): Eliminating non-emergency medical transportation coverage for expansion adults participating in Employer Sponsored Insurance feature of the Section 1115 waiver renewal. |
| California | 2016 | Pregnant Women (+): Expansion to full-scope coverage to pregnant women 60-133% FPL. |
| 2017 | All (+): Restored acupuncture services (eliminated in 2009 for most populations excluding children, pregnant women, and nursing facility residents) (July 1, 2016). Pregnant Women (+): Added Licensed midwives to the Comprehensive Perinatal Services Program (July 1, 2016). All (+): Adding pulmonary and cardiac rehabilitation in outpatient settings (January 1, 2017). (Currently only available in inpatient settings.) | |
| Colorado | 2016 | Children (nc): Added coverage for Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (July 1, 2015). Adults (+): Added coverage for iPads as augmented communication devices (ACDs) (July 1, 2015). |
| Connecticut | 2016 | Adults (+): Added coverage of select over the counter drugs (July 1, 2015). Pregnant Women (+): Added coverage of low-dose aspirin (July 1, 2015). |
| District of Columbia | 2016 | All (+): Expanded coverage for telemedicine services. |
| 2017 | Children (+): Adding reimbursement for adolescent substance abuse treatment. | |
| Florida | 2017 | Persons with SMI or SUD (nc): Delivery of service changes for behavioral health – housing supports as part of the 1115 waiver. |
| Georgia | 2016 | Adults (+): Added coverage for medically necessary emergency transportation by rotary wing air ambulance. All (+): Added coverage for Emergency Ambulances to serve as Telemedicine Origination Sites (April 22, 2016). |
| Hawaii | 2017 | Aged and Disabled (+): Expanding mental health and substance abuse benefits including addition of intensive case management and tenancy supports as part of chronic homelessness initiative (upon CMS approval). |
| Indiana | 2016 | Children (nc): Adding coverage for Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (February 6, 2016). |
| Kansas | 2016 | Children (+): Expanded coverage for developmental therapy (OT/PT speech). |
| 2017 | Children (nc): Moving autism services from HCBS waiver coverage to State Plan coverage. | |
| Louisiana | 2016 | Pregnant Women (nc): Added coverage for free standing birthing centers (an ACA requirement) (December 20, 2015). All (+): Removed limits on physician visits (December 20, 2015). |
| Maryland | 2016 | All (+): Added Physician Assistants as a new provider type (July 1, 2015). |
| 2017 | Children (nc): Adding coverage for Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (January 1, 2017). Former foster youth (+): Extending dental coverage for former youth up to age 26 (January 1, 2017). | |
| Massachusetts | 2016 | Children (nc): Added coverage of Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (July 1, 2015). |
| 2017 | All (+): Adding coverage of American Society of Addiction Medicaid Level 3.1 Residential Rehabilitation Services and Transitional Support Services (January 1, 2017). | |
| Michigan | 2016 | Children and Pregnant Women (+): Targeted Case Management services added for pregnant women and children covered under the Flint Michigan Section 1115 waiver (for persons served by the Flint water system) (May 9, 2016). Children (nc): Expanded autism services from age 6 to age 21 (January 1, 2016). |
| Minnesota | 2016 | Children (nc): Added coverage for treatment of autism spectrum disorder to meet federal requirements (July 1, 2015). |
| 2017 | All (+): Adding coverage for community emergency medical technician services (January 1, 2017). | |
| Missouri | 2016 | Children (+): Adding coverage for asthma education and environmental assessment services. (upon CMS approval). Children (nc): Added coverage for Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (October 2015). |
| Montana | 2016 | Non-Disabled Adults (+): Added dental benefits with a limit of $1,125 per benefit year (July 1-June 30). Diagnostic, preventive, denture, and anesthesia services are excluded from the financial cap (January 1, 2016). All (+): Removed limits on mental health therapy and occupational, speech and physical therapy (January 1, 2016). All (+): Age limits removed for Substance Use Disorder treatment services (January 1, 2016). |
| Nebraska | 2016 | Children (nc): Added coverage for Behavior Modification/Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (October 1, 2015). |
| 2017 | Children (+): Adding coverage for Multisytemic Therapy/Family Functional Therapy (July 1, 2016). All (+): Adding coverage for MH/SUD peer support services (January 1, 2017). All (+): Adding coverage for telehealth and tele-monitoring services (January 1, 2017). | |
| Nevada | 2016 | All (+): Expanding coverage for telemedicine services to additional provider types and eliminating requirement for an origination site thereby allowing beneficiaries to access telemedicine services from home (December 1, 2015). Children (nc): Added coverage for Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (January 1, 2016). |
| 2017 | All (+): Added coverage for paramedicine services (July 1, 2016). | |
| New Hampshire | 2016 | Non Expansion Population (+): Enhanced the Substance Use Disorder benefit (to align with ABP) (July 1, 2016). Expansion Adults (-): Eliminated coverage of non-emergent use of the ER (January 1, 2016). |
| New Jersey | 2017 | Non-Expansion Adults (+): Substance Use Disorder benefit from the state’s Alternative Benefit Package for expansion adults added for all other Medicaid enrollees (July 1, 2016). |
| New Mexico | 2017 | Pregnant Women (nc): Implementing coverage for Birthing Centers. |
| New York | 2016 | All (-): Discontinued coverage for viscosupplementation of the knee for an enrollee with a diagnosis of osteoarthritis of the knee (April 1, 2015 for FFS and July 1, 2015 for managed care). All (-): Limited coverage of DEXA Scans for Screening to one time every 2 years for Women Over Age 65 and Men Over Age 70 (April 1, 2015 for FFS and July 1, 2015 for managed care). All (+): Expanded smoking cessation counseling providers to include dental practitioners (April 1, 2015 for FFS and July 1, 2015 for managed care). All (+): Expanded Telehealth services. All (+): Expanded Dental Hygienist services. Aged & Disabled (+): Added services for adults with serious mental illness services under 1915(i) authority as part of the state’s Health and Recovery Plans (HARP) managed care program. |
| Oklahoma | 2016 | Adults (-): Eliminated coverage for sleep studies (July 1, 2015). All (+): Added coverage for virtual visits with annual limits (January 2016). All (+): Telemedicine policy rules around origination sites were removed. Patients no longer have to be at a specified “origination site” (e.g. they can now be in their homes). |
| 2017 | Children (+): Mandated polycarbonate lenses for children (September 1, 2016). Pregnant Women (-): Reducing number of covered high risk OB visits based on utilization data (September 1, 2016). | |
| Oregon | 2017 | Adults (+): Restoring previously cut adult restorative dental benefits (relaxed limitation criteria for dentures; coverage for crowns; scaling and planning) (July 1, 2016). Adults (+): Expanding coverage for alternative back pain therapies including acupuncture, chiropractic manipulation and yoga (July 1, 2016). Children (nc): Added coverage for Applied Behavioral Analysis services for children with autism spectrum disorder to meet federal requirements (July 1, 2016). |
| Rhode Island | 2017 | All (+): Add coverage for home stabilization services. All (+): Initiating coverage for Telehealth services in new MCO contracts. Aged and Disabled (+): Implementing the Sobering Treatment Opportunity Program (STOP), an ER diversion pilot in Providence that will cover an overnight stay and referral to appropriate counseling for homeless chronic inebriates. |
| South Carolina | 2016 | Children (+): Expanded coverage for treatment of eating disorders ages 0-21. |
| 2017 | Children (nc): Adding autism spectrum disorder treatment State Plan services to meet federal requirement; will replace existing HCBS waiver coverage that will sunset (January 2017). | |
| South Dakota | 2017 | Adults (+): Added coverage for BRCA gene testing (July 1, 2016). |
| Tennessee | 2017 | Adults (-): Limiting Allergy Immunotherapy to practice guidelines (July 1, 2016). |
| Texas | 2016 | Children (+): Added coverage for Prescribed Pediatric Extended Care Centers. Children (+): Texas Health Steps Preventive Care Medical Checkups added mental health screening with separate reimbursement and screening for critical congenital heart disease (CCHD); updated laboratory screening policy for anemia, dyslipidemia and HIV screenings (11/1/2015). All (+): Added coverage for Magneto Encephalography (MEG) (November 1, 2015). Aged and Disabled (+): Expanded coverage for Screening, Brief Intervention, and Referral to Treatment (SBIRT) services to include more providers in outpatient settings (July 1, 2016). All (+): Updated gynecological and reproductive health services coverage and reimbursement policy regarding IUD reimbursement and implantable contraceptive capsules (January 1, 2016). |
| 2017 | Children (+): Adding coverage for family therapy without the patient present as a benefit for children under 21. Pre-doctoral psychology interns and post-doctoral psychology fellows will be added as a recognized service provider when under delegation by a licensed psychologist. All (+): Expanding coverage of tele-monitoring services to include congestive heart failure (CHF) and diagnoses related to high-risk pregnancy. | |
| Utah | 2016 | Children (nc): Added autism spectrum disorder treatment to meet federal requirement (July 2015). |
| 2017 | Children (+): Eliminating the state’s Section 1115 EPSDT waiver which enables 19 and 20 year-old parents to be able to receive EPSDT services, which are not part of current 1115 waiver. | |
| Vermont | 2016 | All (+): Added coverage for Licensed Alcohol and Drug Counselors (July 1, 2015). All (+): Added coverage for primary care telemedicine outside of a facility (October 1, 2015). Children (nc): Added coverage for Applied Behavior Analysis for treatment of autism spectrum disorder to meet federal requirements (July 1, 2015). |
| 2017 | All (+): Allowing Licensed Dental Hygienists to bill Medicaid directly (July 1, 2016). | |
| Virginia | 2017 | All (+): Under Section 1115 waiver authority, expanding Substance Use Disorder (SUD) services to add coverage of peer supports, inpatient residential for adults, and up to 15 days in an IMD in facilities with more than 16 beds (upon CMS approval). All (+): Removing prior authorization requirements for low-dose computed tomography (LDCT) lung cancer screenings (July 1, 2016). |
| Washington | 2016 | All (+): Added coverage for gender reassignment surgery (August 6, 2015). |
| Wisconsin | 2016 | Children (nc): Added State Plan coverage (to replace HCBS waiver coverage) for behavioral health services for treatment of autism spectrum disorder to meet federal requirements (January 1, 2016). |
| Wyoming | 2016 | All (+): Added chiropractic benefit (July 1, 2015). All (+): Added coverage for additional provisionally licensed MH provider types (July 1, 2015). |
| 2017 | All (+): Adding coverage for dietician services (July 1, 2016). Adults (-): Eliminating dental and vision coverage (except emergency services) (October 1, 2016). LTSS Adults (-): Reducing Nursing facility bed-hold days (October 1, 2016). Adults (-): Adding soft service caps for behavioral health, therapy, and home health services (January 1, 2017). | |
| NOTE: Positive changes counted in this report are denoted with (+). Negative changes counted in this report are denoted with (-). Changes that were not counted as positive or negative in this report, but were mentioned by states in their responses, are denoted with (nc). | ||
Prescription Drug Utilization and Cost Control Initiatives
Prior to the passage of the Medicare drug benefit, most states had implemented aggressive strategies to slow Medicaid spending growth for prescription drugs, including preferred drug lists (PDLs), supplemental rebate programs, and state maximum allowable cost (SMAC) programs. State focus on pharmacy cost containment diminished after nearly half of Medicaid drug spending shifted to the Medicare drug benefit in 2006. Since 2014, however, a disproportionate increase in prescription drug costs relative to overall spending has refocused state attention on pharmacy reimbursement and coverage policies. In this year’s survey, states reported a variety of actions in FY 2016 and FY 2017 to refine and enhance their pharmacy programs, including actions to react to new and emerging specialty and high-cost drug therapies.
Pharmacy Cost Drivers
This year’s survey asked states to identify the three biggest cost drivers that affected growth in total pharmacy spending (federal and state) in FY 2016 and projected for FY 2017. Consistent with the results from last year’s survey, the vast majority of states identified specialty and high cost drugs as the most significant cost driver.
Most states pointed specifically to hepatitis C antivirals as driving prescription drug costs; high costs are attributable to the high per prescription cost as well as increased utilization. In November 2015, CMS issued guidance to states regarding coverage policies for hepatitis C drugs. In that guidance, CMS expressed concern that some states were restricting access to these drugs contrary to statutory requirements and directed states to “examine their drug benefits to ensure that limitations do not unreasonably restrict coverage of effective treatment using the new direct-acting antiviral (DAA) hepatitis C drugs.”64 65 In May 2016, a federal court issued a preliminary injunction ordering Washington state to provide hepatitis C treatment to all Medicaid beneficiaries.66 This represents a turning point, as it was the first time a court declared restrictions to hepatitis C drugs based on disease severity illegal. A handful of states have eased restrictions in part due to an acknowledgement of the implications of the decision in Washington, as well other lawsuits and new guidance.
Other specialty drugs and behavioral health and/or substance use disorder drugs were cited as cost drivers, and some specific drug classes (such as hemophilia factor, oncology drugs, diabetes products, cystic fibrosis agents, and HIV drugs) were also identified as major cost drivers. In addition, states noted large price increases for existing generics and higher than expected prices for new generics entering the market as cost drivers.
Pharmacy Cost Containment Actions in FY 2016 and FY 2017
A majority of states had prescription drug cost containment policies (including prior authorization requirements and preferred drug lists (PDLs)) in place prior to FY 2016, and states are constantly refining and updating these policies. Although states may not have reported every refinement or routine change in this year’s survey, 31 states in FY 2016 and 23 states in FY 2017 reported implementing or making changes to a wide variety of cost containment initiatives in the area of prescription drugs, comparable to the number of states taking such actions in recent years. The most frequently cited actions were:
- New prior authorization requirements (12 states in FY 2016 and 6 in FY 2017),
- Updates or expansions of a PDL (10 states in FY 2016 and 4 in FY 2017), and
- Increased rebate collections (6 states in FY 2016 and 4 in FY 2017).
Multiple states also reported new or expanded Medication Therapy Management programs, imposing new quantity or dosage limits, implementing additional clinical claims system edits, specific drug carve-outs (e.g., hepatitis C antivirals), and updates or additions to State Maximum Allowable Cost programs. Also, two states (New Mexico and New York) described pharmacy “efficiency adjustments” that are applied during the MCO rate setting process to incentivize efficient pharmacy management by the MCOs.
Medicaid Covered Outpatient Drug Final Rule
State Medicaid programs historically reimbursed pharmacies for the “ingredient cost” of each prescription using an Estimated Acquisition Cost (EAC), plus a dispensing fee.67 On January 21, 2016, CMS released the Covered Outpatient Drug final rule68 which, among other changes, replaces the term EAC with the term “Actual Acquisition Cost” (AAC) and also requires states to provide a “professional dispensing fee” that reflects the pharmacist’s professional services and costs to dispense a drug to a Medicaid beneficiary. States can define their own AAC prices or use the pricing files published and updated weekly by CMS – the “National Average Drug Acquisition Costs” (NADACs) – which are derived from outpatient drug acquisition cost surveys of retail community pharmacies.69 Some states had already transitioned to an AAC methodology prior to the issuance of the final rule. While this year’s survey did not ask specifically about state implementation of the Covered Outpatient Drug Rule, three states in FY 2016 and 16 in FY 2017 referenced implementation of the rule as a pharmacy cost containment action, suggesting that these states expected net savings from the AAC methodology change. One state referenced a cost neutral implementation of the rule, and one state listed the rule implementation as a cost driver for FY 2017. For purposes of this report, however, implementation of the Covered Outpatient Drug final rule is not counted as a cost containment action because it is an implementation of a federal regulatory requirement.
Managed Care’s Role in Delivering Pharmacy Benefits
Since the passage of the ACA, states have been able to collect rebates on prescriptions purchased by managed care organizations (MCOs) operating under capitated arrangements. As a result, many states have chosen to “carve-in” the pharmacy benefit to their managed care benefits. As more states have enrolled additional Medicaid populations into managed care arrangements over time, and as Medicaid enrollment has increased due to ACA coverage expansions, MCOs have played an increasingly large role in administering the Medicaid pharmacy benefit. In this year’s survey, states with MCO contracts were asked whether pharmacy benefits were covered under those contracts as of July 1, 2016.
Thirty-three (33) of the 39 MCO states reported that the pharmacy benefit was “generally carved in.” Among the states that carved drugs in to MCOs, several reported carve-outs for selected drug classes. Behavioral health drugs (Maryland, Michigan, Oregon, and Utah), HIV drugs (Maryland and Michigan), hemophilia clotting factor (Michigan, New Hampshire, New York, Utah, and Washington), and hepatitis C antivirals (Michigan, New Hampshire, South Carolina, Washington, and West Virginia) were among the most common drugs carved out of MCOs. California referred generally to a select list of carved out drugs.
Four states (Colorado, Missouri, Nebraska, and Tennessee) reported that the pharmacy benefit was “generally carved out.” Nebraska noted that injectables were carved in and that a full carve in would be implemented in January 2017.
Two states reported variations by MCO program: Indiana reported that pharmacy was carved in for “HIP 2.0” (ACA expansion program) and “Hoosier Care Connect” (aged, blind and disabled program), but was currently carved out for Hoosier Healthwise (program for low-income pregnant women and children) until January 2017 when pharmacy would be carved in for this program too. Wisconsin reported that pharmacy was generally carved out except for its Family Care Partnership program (an integrated health and long-term care program for frail elderly people and people with disabilities) where it was carved in.
Prior reports show that nearly all states use prior authorization and PDLs in FFS programs. The survey asked about whether MCO contract requirements for uniform clinical protocols, a uniform PDL, or uniform prior authorization requirements were in place in FY 2015, added or expanded in FY 2016, or would be added or expanded in FY 2017 (Exhibit 13). This means that to the extent states impose or change these policies in FFS, the same policies would apply in managed care.
| Exhibit 13: Managed Care Pharmacy Policies | ||||||
| Policy | In Place in FY 2015 | New or Expanded | ||||
| FY 2016 | FY 2017 | |||||
| Uniform Clinical Protocols | 12 States | AZ, CA, GA, HI, IL, IN, KS, MA, NJ, PA, TX, WV | 6 States | DC, IA, KY, MA, MI, NY | 5 States | KY, MA, NE, NJ, NY |
| Uniform Prior Authorization Requirements | 9 States | AZ, GA, KS, MA, MS, NM, PA, TX, WV | 7 States | DC, IA, KY, MA, MI, NM, UT | 7 States | GA, KY, MA, NE, NM, UT, VA |
| Uniform PDL | 9 States | CA, DE, FL, KS, MA, MS, NH, TX, WV | 3 States | AZ, CA, IA | 3 States | MA, NE, UT |
Uniform Clinical Protocols
Twelve (12) states reported having uniform clinical protocol requirements in place in FY 2015, while six states in FY 2016 and five states in FY 2017 reported new or expanded clinical protocol requirements. These requirements were usually limited to specific drug classes. For example, several states mentioned particular drugs or drug classes including hepatitis C antivirals (Arizona, District of Columbia, Georgia, Hawaii, Illinois, Massachusetts, New Jersey, and Pennsylvania). A few states provided some additional details. In Iowa, MCOs are required to impose the same clinical edits70 as FFS; in Delaware, New Jersey and Texas, MCO clinical edits must be approved by the state; and Florida reported there were no specific protocols, but that MCO protocols may be no more restrictive than the FFS program policies.
Uniform Prior Authorization Requirements
Nine states reported having uniform prior authorization (PA) requirements in place in FY 2015, while seven states in FY 2016 and seven states in FY 2017 reported new or expanded uniform PA requirements. These requirements were usually limited to specified drug classes and in some cases overlap with the uniform clinical criteria responses described above (as states may use the PA process as a tool to enforce adherence to the states’ clinical criteria). For example, the District of Columbia and Virginia noted uniform PA requirements for substance use disorder drugs and New Mexico, Utah, and Virginia cited hepatitis C antivirals. Delaware and New Jersey noted that MCO PA requirements must be approved by the state.
Uniform PDL
Nine states reported having a uniform PDL requirement in place in FY 2015, while three states in FY 2016 and three states in FY 2017 reported new or expanded uniform PDL requirements. California reported that as its FFS formulary expanded over time, so have the MCO formularies. Massachusetts reported that the uniform PDL applied to a limited number of therapeutic classes. Louisiana reported that its five MCOs created a common PDL for selected drug classes which could be expanded by the MCOs in the future. One state (New Hampshire) reported eliminating its uniform PDL requirement in FY 2o16.
Other Managed Care Pharmacy Policies
Several states reported other managed care pharmacy policies. Kentucky reported that in FY 2016 FFS and the MCOs began to develop uniform PA forms (to make PA processes more manageable for providers) and align clinical criteria for high profile pharmaceutical products and disease states. In FY 2017, a Uniform Pharmacy Policy Committee in Kentucky will tackle topics such as access to hepatitis C treatment, opioid prescribing and limitations on utilization, and insect repellant coverage for Zika virus. Some states had strategies to mitigate the risk for certain drugs. For example, Hawaii, New Mexico, Ohio, Pennsylvania, Rhode Island, and Texas use risk corridor, risk pool, stop-loss arrangement and expense reimbursement for hepatitis C drugs; Pennsylvania reported risk sharing for cystic fibrosis drugs; Virginia reported a stop loss policy for any drug spending greater than $150,000 per member per year; and Washington reported an MCO PDL for antipsychotics.
Opioid Harm Reduction Strategies
According to the Centers for Disease Control and Prevention (CDC), overdose deaths from prescription opioid pain medications in the United States have more than quadrupled from 1999 to 2011.71 In addition to drug-related deaths, inappropriate opioid use causes other medical complications and suffering and has a disproportionate impact on Medicaid beneficiaries who are “prescribed painkillers at twice the rate of non-Medicaid patients and are at three-to-six times the risk of prescription painkillers overdose.”72 In a January 2016 Information Bulletin, CMS highlighted the important role state Medicaid programs can play to help address the opioid epidemic in their states by encouraging safer opioid alternatives for pain relief, working with other state agencies to educate Medicaid providers on best practices for opioid prescribing, employing pharmacy management practices (e.g., PDL placement, clinical criteria, prior authorization, quantity limits, etc.), and working to increase access to naloxone, an overdose antidote. In this year’s survey, states were asked about their opioid harm reduction strategies in place in FY 2015, implemented in FY 2016, and planned for FY 2017.
CDC Opioid Prescribing Guidelines
The CDC has developed and published recommendations for the prescribing of opioid pain medications for adults in primary care settings.73 This year’s survey asked states if their Medicaid program has adopted or is planning to adopt these guidelines in their FFS programs or as a requirement for MCOs to adopt. As shown in Exhibit 14 below, 21 states reported adopting the guidelines or plans to adopt in FY 2017 for their FFS programs. Of the 39 states with MCO contracts, 11 states reported requiring MCOs to adopt the CDC guidelines or plans to do so in FY 2017. Many states indicated these policies were under review for FFS and MCOs.
| Exhibit 14: Number of States Adopting CDC Opioid Prescribing Guidelines | ||||
| Status | For FFS | As a requirement for MCOs to adopt | ||
| Yes, have adopted | 7 States | AR, ID, MA, NE, NY, VA, VT | 2 States | MA, NY |
| Plan to adopt in FY 2017 | 14 States | AK, CT, DC, IA, LA, ME, MI, MS, NC, NH, OR, TN, WA, WV | 9 States | DC, IA, MS, NE, NH, OR, VA, WA, WV |
States were also asked to describe any implementation challenges related to the CDC guidelines. Some of the commonly reported challenges included system challenges; obtaining stakeholder consensus and support (including providers); titrating dosages downward for patients who have been stabilized on higher dosages; and the need for more provider education. A few states had state guidelines already in place that were aligned with the CDC guidelines.
Increasing Access to Naloxone
Naloxone is a prescription opioid overdose antidote that prevents or reverses the life-threatening effects of opioids including respiratory depression, sedation, and hypotension. Many states have taken steps to expand access to naloxone to enable family members and first responders to administer the antidote to save lives, including, for example, allowing “standing orders” or issuance of a statewide standing order that allows naloxone to be distributed by designated people, such as pharmacists or others meeting criteria established in the order. In this year’s survey, states were asked if their Medicaid program had implemented, or planned to implement, any initiatives to increase access to naloxone.
Half of the states (26) reported making naloxone (in at least one formulation) available without prior authorization or adding naloxone to their PDL. Some states (including Colorado and Michigan) reported expanding coverage of naloxone products beyond vials and syringes to include nasal spray and auto-injectors. Two states reported Medicaid coverage for naloxone prescribed to a family member or friend, and another state reported increasing access to naloxone by issuing a letter of direction to its MCOs.
Several states reported broader initiatives that are not specific or limited to Medicaid, including issuance or authorization of standing orders (6 states), allowing pharmacists to prescribe naloxone (3 states), third party prescribing laws that allow prescriptions to family members or friends (5 states), Good Samaritan laws that protect non-clinicians that administer naloxone (2 states); initiatives to educate and raise awareness (4 states), and making naloxone available without a prescription (3 states). According to a recent National Safety Council Report, however, a total of 35 states allow naloxone to be prescribed with a standing order and 35 states have enacted Good Samaritan provisions.74
Medicaid Pharmacy Benefit Management Strategies
The January 2016 CMS Informational Bulletin highlighted Medicaid pharmacy benefit management strategies for preventing opioid-related harms.75 The survey asked states to indicate whether one or more of these strategies was in place in FY 2015 for FFS and whether any changes to these strategies were made in FY 2016 or planned for FY 2017. Many states also have policies in place with regard to MCOs; however, it is unclear how many states require such policies to be in place.
Almost all states (44) took at least one action in FY 2016 or plan to take one action in FY 2017 to adopt or expand an opioid-focused pharmacy management policy in FFS. In FY 2015, 46 states imposed opioid quantity limits,76 45 states imposed prior authorization, 42 states had clinical criteria, and 32 states had step-therapy. (In some cases, the prior authorization actions reported may overlap with the responses regarding changes in opioid step therapy and/or clinical criteria as states may use the PA process as a tool to enforce adherence to the states’ clinical criteria and step therapy requirements.) Twelve states had a requirement that prescribers check the state’s Prescription Drug Monitoring Program before prescribing opioids. Prescription Drug Monitoring Programs (PDMPs) are state-run electronic databases that are valuable tools for addressing prescription drug diversion and abuse. Currently, with the exception of Missouri, every state operates a PDMP.77 78 Many states were newly implementing or expanding these programs in FY 2016 and FY 2017 (Exhibit 15 and Table 19).
| Exhibit 15: States Implementing Opioid-Focused Pharmacy Benefit Management Strategies in FFS | |||
| Strategy | In Place in FY 2015(# of states) | New or Expanded (# of states) | |
| FY 2016 | FY 2017 | ||
| Opioid Quantity Limits | 46 | 22 | 30 |
| Prior Authorization for Opioids | 45 | 18 | 27 |
| Opioid Clinical Criteria | 42 | 20 | 27 |
| Opioid Step Therapy Requirements | 32 | 13 | 20 |
| Required use of Prescription Drug Monitoring Programs | 12 | 10 | 11 |
Other Pharmacy Management Strategies. A few states mentioned other pharmacy management strategies in use or planned, including the following:
- Maryland and Mississippi reported provider and/or patient education efforts.
- New Jersey indicated that the medication assistance treatment (MAT) benefit already available to ACA expansion enrollees was expanded to all Medicaid enrollees July 1, 2016; Vermont cited its comprehensive “Hub and Spoke” MAT medical home program that provides broad access to anyone seeking treatment for substance use issues;
- Washington indicated that state law now requires consultation with a pain specialist for certain high dosage patients (greater than 120 morphine equivalent dose (MED));
- Alaska and Mississippi reported expanded Drug Utilization Review (DUR) activities that rely on PDMP access. Mississippi noted that its DUR program entered into a contract with the PDMP in 2016 to receive controlled substance claims for which Medicaid beneficiaries paid cash and those paid by Medicaid for monitoring purposes.
- In July 2016, Oregon Medicaid began covering various alternative treatment modalities (e.g., chiropractic, physical therapy, acupuncture, massage, yoga, and cognitive behavioral therapy), and has restrictions on the prescribing of opiates for back pain, neck pain, migraines, and fibromyalgia.
Table 19: Medicaid Pharmacy Benefit Management Strategies for Opioids in Fee-For-Service in All 50 States and DC, FY 2015 – FY 2017
| States | Opioid Quantity Limits | Prior Authorization for Opioids | Opioid Clinical Criteria | Opioid Step Therapy Requirements | Required use of Prescription Drug Monitoring Programs | ||||||||||
| In place FY 2015 | 2016 | 2017 | In place FY 2015 | 2016 | 2017 | In place FY 2015 | 2016 | 2017 | In place FY 2015 | 2016 | 2017 | In place FY 2015 | 2016 | 2017 | |
| Alabama | X | X | X | X | X | ||||||||||
| Alaska | X | X | X | X | X | X | X | X | |||||||
| Arizona | X | X | X | ||||||||||||
| Arkansas | X | X | X | X | X | X | X | X | |||||||
| California | X | X | |||||||||||||
| Colorado | X | X | X | X | X | X | X | X | X | X | |||||
| Connecticut | X | X | X | ||||||||||||
| Delaware | X | X | X | X | X | X | X | X | |||||||
| DC | X | X | X | X | X | X | X | ||||||||
| Florida | X | X | X | X | |||||||||||
| Georgia | X | X | X | X | X | X | X | X | X | X | |||||
| Hawaii | |||||||||||||||
| Idaho | X | X | X | X | X | X | X | X | X | X | X | X | |||
| Illinois | X | X | X | X | X | X | X | ||||||||
| Indiana | X | X | X | X | X | X | X | X | X | X | X | X | X | ||
| Iowa | X | X | X | X | X | X | X | X | X | ||||||
| Kansas | X | X | X | X | X | ||||||||||
| Kentucky | X | X | X | X | X | X | X | X | X | X | X | X | X | X | X |
| Louisiana | X | X | X | X | X | X | X | ||||||||
| Maine | X | X | X | X | |||||||||||
| Maryland | X | X | |||||||||||||
| Massachusetts | X | X | X | X | X | X | |||||||||
| Michigan | X | X | X | X | X | X | X | X | X | X | X | X | X | ||
| Minnesota | X | X | X | X | X | ||||||||||
| Mississippi | X | X | X | X | |||||||||||
| Missouri | X | X | X | X | X | X | X | ||||||||
| Montana | X | X | X | X | X | X | X | X | X | X | X | X | |||
| Nebraska | X | X | X | X | X | X | X | X | |||||||
| Nevada | X | X | X | X | X | X | X | X | X | X | X | X | X | ||
| New Hampshire | X | X | X | X | X | X | X | X | X | ||||||
| New Jersey | X | X | X | X | X | X | X | X | X | ||||||
| New Mexico | |||||||||||||||
| New York | X | X | X | X | |||||||||||
| North Carolina | X | X | X | X | X | X | X | X | X | ||||||
| North Dakota | X | X | X | X | X | X | X | X | X | X | |||||
| Ohio | X | X | X | X | X | X | X | ||||||||
| Oklahoma | X | X | X | X | X | X | X | X | X | X | |||||
| Oregon | X | X | X | X | X | X | X | X | X | X | X | ||||
| Pennsylvania | X | X | X | X | X | X | X | X | |||||||
| Rhode Island | X | X | X | X | X | X | X | X | |||||||
| South Carolina | X | X | X | X | X | ||||||||||
| South Dakota | X | X | X | X | X | ||||||||||
| Tennessee | X | X | X | X | X | X | X | X | X | X | X | X | X | X | X |
| Texas | X | X | X | X | X | X | X | X | |||||||
| Utah | X | X | X | X | X | X | X | ||||||||
| Vermont | X | X | X | X | X | ||||||||||
| Virginia | X | X | X | X | X | X | X | X | X | X | X | X | |||
| Washington | X | X | X | X | X | X | |||||||||
| West Virginia | X | X | X | X | X | X | X | ||||||||
| Wisconsin | X | X | X | X | |||||||||||
| Wyoming | X | X | X | X | X | X | |||||||||
| Totals | 46 | 22 | 30 | 45 | 18 | 27 | 42 | 20 | 27 | 32 | 13 | 20 | 12 | 10 | 11 |
| NOTES: States were asked to report whether they had select pharmacy benefit management strategies in place in their FFS programs in FY 2015, had adopted or expanded these strategies in FY 2016, or had plans to adopt or expand these strategies in FY 2017.SOURCE: Kaiser Commission on Medicaid and the Uninsured Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2016. | |||||||||||||||
Report: Administrative Challenges, Priorities, And Conclusion
Administrative Challenges
States noted a number of administrative challenges related to implementing the ACA, major delivery system reforms, new federal regulations, and new systems. While Medicaid directors noted that these were all high priorities, limited staff and resources in terms of staff and funding for administration make balancing across sometimes competing priorities a challenge. More specifically, some states are still implementing eligibility and enrollment systems related to the ACA while others are implementing complex ACA Medicaid expansion waivers. Around delivery system reforms, states noted challenges with data and systems and provider capacity as well as obtaining buy-in from advocates and other stakeholders, achieving multi-payer alignment, incentivizing MCOs to maintain improvement, and receiving timely approvals from CMS. States expressed concerns about the capacity to implement and comply with the magnitude of federal regulations in number and scope, most notably the final managed care regulations issued in May 2016 in addition to other major regulations. The cumulative effect of simultaneously implementing multiple regulations was more of a concern than any one specific regulation, and different regulations also have varying implications across individual states. In addition to implementing the ACA, delivery system reforms, and new regulations, many Medicaid directors also mentioned significant systems initiatives, including Medicaid Management Information Systems (MMIS).
Challenges and Priorities in FY 2017 and Beyond Reported by Medicaid Directors
As noted above, Medicaid directors note several administrative challenges.
“Staffing…the sheer difficulty of retaining staff with program expertise and recruiting staff with the skill sets to meet current demands. This is particularly true for managed care but also for systems.”
“Our most significant administrative challenge is to keep pace with implementing multiple payment reform initiatives concurrently with sharpening fiscal and staffing restraints and increasing Federal regulatory and oversight activity.”
Despite the administrative and fiscal challenges, Medicaid directors listed an array of priorities for FY 2017 and beyond. While the ACA has fundamentally changed Medicaid programs since 2014, the main priority looking ahead was not focused on the ACA but more on cost control, payment and delivery system reforms, and infrastructure development.
Controlling Medicaid costs. Controlling costs is a perennial priority for Medicaid. Even in relatively good economic times, the cost of Medicaid in state budgets is so significant that the program is under constant pressure to control spending and to achieve greater value for every dollar in its budget. Medicaid administrators and policy makers traditionally have focused on the components of Medicaid spending, including provider payment rates and the amounts paid for specific services, limits on covered benefits, eligibility policies, prior authorization, and other controls on the utilization of services. For FY 2017, cost control and cost containment was specifically mentioned as one of the top three priorities by a large share of states, either specifically as a budget control issue or as part of a value based purchasing strategy. In some cases, this refers to specific measures, such as those to control spending of prescription drugs. However, the major focus of cost control has shifted to delivery system and payment reforms that incentivize high quality care, better outcomes, and lower costs.
In the process of this transformation, Medicaid programs have become national leaders in delivery system and payment reforms that are now becoming operational. In many cases, the new systems build on managed care systems; recent Medicaid MCO procurements illustrate the trend, with selection of health plans and MCO payments now commonly based on the quality of care delivered and the achievement of specific quality metrics. A few recent MCO procurements have specifically required MCOs to address social determinants of health and to undertake population health strategies.
Payment and delivery system reform initiatives. Medicaid programs are developing and implementing significant initiatives that restructure delivery systems and payment structures with the goal of improving the quality of care and patient outcomes. Over half of states mentioned these initiatives as a top priority for FY 2017, including “value-based purchasing” approaches and other strategies, described by one state as “changes to the delivery system to improve efficiency and care outcomes,” in other states as “integration of physical and behavioral health,” “continuing to transform the system through managed care,” and in others as “system transformation, clinical management, and population health.”
Significantly, a number of the delivery and payment reform initiatives go beyond traditional medical care delivery, addressing goals related to social determinants of health and population health. One state described their priority for 2017 to be “coordination between the Department and Housing Division to develop housing opportunities for individuals with severe mental illness.” Another state said a priority for this year was to “get beyond health care to quality of life, employment, and community inclusion.” A third state said “use our leverage as a payer to support prevention efforts that address the social determinants of health and population health activities.”
Medicaid infrastructure development. Medicaid programs universally have undertaken major system development projects in recent years, most notably for new eligibility systems and for new MMIS systems. Several states listed the development and operationalization of these projects as a major priority in FY 2017, either as a priority in themselves or as necessary for the success of other initiatives. These Medicaid infrastructure initiatives are critically important for the success of the major delivery system and payment reforms that are often being implemented concurrently. Medicaid programs also need the systems capability to implement quality improvement, provider and MCO monitoring, data analytics, and cost control strategies. A major Medicaid issue and priority is the staffing and other resources for systems and IT development, and the infrastructure necessary for Medicaid to implement its major initiatives.
Priorities for FY 2017, in the words of the Medicaid Directors, include:
“To take health system transformation to the next level, continue to bend the cost curve, and increase the focus on behavioral health and social determinants of health.”
“Maintaining budget control, integrating physical and behavioral health in a way that achieves goals of cost, quality and effectiveness, and managing concurrent high-risk IT projects – eligibility system and MMIS replacements.”
Medicaid Directors also were able to reflect on key areas of success:
“Our MCO initiative, getting people in a managed care system that improves care and reduces costs. We are no longer just a payer of claims. We are a leader in improving health and health care in this state.”
“Growing the program in a responsible manner – leading the state towards new value based purchasing models and patient centered medical homes, modernizing and improving the program, and coming in under budget 5 years in a row.”
Conclusion
This report has described Medicaid policy changes in eligibility, payment rates, benefits and pharmacy, long-term services and supports, managed care, and payment and delivery system initiatives. Medicaid programs now play a significant leadership role in the health care systems in every state. Consistent with this role, state Medicaid officials and policy makers now focus on innovative delivery system and payment initiatives designed to improve health care and health outcomes, resulting in better health status and lower costs. It is an approach that incorporates value based payments and purchasing, and includes strategies to address the social determinants of health and improve overall population health. The impact of Medicaid’s role is seen not only in the lives of those who are served by the program, but in a higher functioning health system that benefits all citizens in each state.
Methods
The Kaiser Commission on Medicaid and the Uninsured (KCMU) commissioned Health Management Associates (HMA) to survey Medicaid directors in all 50 states and the District of Columbia to identify and track trends in Medicaid spending, enrollment, and policy making. This is the 16th annual survey, each conducted at the beginning of the state fiscal year from FY 2002 through FY 2017. Additionally, eight mid-fiscal year surveys were conducted during state fiscal years 2002-2004 and 2009-2013, when a large share of states were considering mid-year Medicaid policy changes due to state budget and revenue shortfalls. Findings from previous surveys are referenced in this report when they help to highlight current trends. Archived copies of past reports are available on the following page.
The KCMU/HMA Medicaid survey on which this report is based was conducted from June through August 2016. The survey instrument (in the Appendix) was designed to document policy actions states implemented in FY 2016 and adopted for FY 2017 (which began for most states on July 1, 2016).79 Each survey is designed to capture information consistent with previous surveys, particularly for eligibility, provider payment rates, benefits, long-term care, and managed care. Each year, questions are added to address current issues, such as state actions to address the opioid epidemic.
Medicaid directors and staff provided data for this report in response to a written survey and a follow-up telephone interview. The survey was sent to each Medicaid director in June 2016. All 50 states and DC completed surveys and participated in telephone interview discussions in July and August 2016. The telephone discussions are an integral part of the survey to ensure complete and accurate responses and to record the complexities of state actions. FY 2017 information was incomplete for Illinois as the budget for FY 2017 had not been adopted at the time the survey and telephone discussions were completed.
The survey does not attempt to catalog all Medicaid policies in place for each state. The focus is on changes in Medicaid policy and new initiatives that are implemented in FY 2016 and those adopted and planned for implementation in FY 2017. Experience has shown that adopted policies are sometimes delayed or not implemented, for reasons related to legal, fiscal, administrative, systems or political considerations, or due to delays in approval from CMS. Policy changes under consideration without a definite decision to implement are not included in the survey. The District of Columbia is counted as a state for the purposes of this report; the counts of state policies or policy actions that are interspersed throughout this report include survey responses from the 51 “states” (including DC).
Appendix
Additional Resources
Kaiser Commission on Medicaid and the Uninsured Resources
- 50-State Medicaid Budget Survey Archiveshttps://www.kff.org/medicaid/report/medicaid-budget-survey-archives/
- Michigan’s Medicaid Section 1115 Waiver to Address Effects of Lead Exposure in Flinthttps://www.kff.org/medicaid/fact-sheet/michigans-medicaid-section-1115-waiver-to-address-effects-of-lead-exposure-in-flint/
- Connecting the Justice-Involved Population to Medicaid Coverage and Care: Findings from Three Stateshttps://www.kff.org/medicaid/issue-brief/connecting-the-justice-involved-population-to-medicaid-coverage-and-care-findings-from-three-states/
- Medicaid Financial Eligibility for Seniors and People with Disabilities in 2015, Appendixhttps://www.kff.org/report-section/medicaid-financial-eligibility-for-seniors-and-people-with-disabilities-in-2015-appendix/
- Medicaid Expansion in Indianahttps://www.kff.org/medicaid/fact-sheet/medicaid-expansion-in-indiana/
- Medicaid Expansion in Iowahttps://www.kff.org/medicaid/fact-sheet/medicaid-expansion-in-iowa/
- Medicaid Expansion in Michiganhttps://www.kff.org/medicaid/fact-sheet/medicaid-expansion-in-michigan/
- Medicaid Expansion in Montanahttps://www.kff.org/medicaid/fact-sheet/medicaid-expansion-in-montana/
- Proposed Changes to Medicaid Expansion in Arizonahttps://www.kff.org/medicaid/fact-sheet/proposed-changes-to-medicaid-expansion-in-arizona/
- CMS’s Denial of Proposed Changes to Medicaid Expansion in Ohiohttps://www.kff.org/medicaid/fact-sheet/proposed-changes-to-medicaid-expansion-in-ohio/
- Proposed Changes to Medicaid Expansion in Kentuckyhttps://www.kff.org/medicaid/fact-sheet/proposed-changes-to-medicaid-expansion-in-kentucky/
- Findings from the Field: Medicaid Delivery Systems and Access to Care in Four States in Year Three of the ACAhttps://www.kff.org/report-section/findings-from-the-field-medicaid-delivery-systems-and-access-to-care-in-four-states-in-year-three-of-the-aca-issue-brief/
- Key Themes From Delivery System Reform Incentive Payment (DSRIP) Waivers in 4 Stateshttps://www.kff.org/medicaid/issue-brief/key-themes-from-delivery-system-reform-incentive-payment-dsrip-waivers-in-4-states/
- Money Follows the Person: A 2015 State Survey of Transitions, Services, and Costshttps://www.kff.org/medicaid/report/money-follows-the-person-a-2015-state-survey-of-transitions-services-and-costs/
- Medicaid’s Most Costly Outpatient Drugshttps://www.kff.org/medicaid/issue-brief/medicaids-most-costly-outpatient-drugs/
CMS Guidance
- State Health Official Letter: To Facilitate Successful Re-Entry for Individuals Transitioning from Incarceration to their Communitieshttps://www.medicaid.gov/federal-policy-guidance/downloads/sho16007.pdf
- CMCS Informational Bulletin: Coverage of Housing-Related Activities and Services for Individuals with Disabilitieshttps://www.medicaid.gov/federal-policy-guidance/downloads/CIB-06-26-2015.pdf
- Medicaid Drug Rebate Program Notice: Assuring Medicaid Beneficiaries Access to Hepatitis C Drugs https://www.medicaid.gov/Medicaid-CHIP-Program-Information/By-Topics/Benefits/Prescription-Drugs/Downloads/Rx-Releases/State-Releases/state-rel-172.pdf
- CMCS Informational Bulletin: Best Practices for Addressing Prescription Opioid Overdoses, Misuse, and Addictionhttps://www.medicaid.gov/federal-policy-guidance/downloads/CIB-02-02-16.pdf
Endnotes
- Centers for Medicare and Medicaid Services. National Health Expenditures (Washington, DC: Centers for Medicare and Medicaid Services, December 2015). http://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-andReports/NationalHealthExpendData/NationalHealthAccountsHistorical.html. ↩︎
- Deborah Dowell, Tamara Haegerich, and Roger Chou, “CDC Guideline for Prescribing Opioids for Chronic Pain — United States, 2016,” Centers for Disease Control and Prevention, Morbidity and Mortality Weekly Report, 65, no.1 (March 2016): 1-49, http://dx.doi.org/10.15585/mmwr.rr6501e1. ↩︎
- Centers for Medicare and Medicaid Services. National Health Expenditures (Washington, DC: Centers for Medicare and Medicaid Services, December 2015). http://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-andReports/NationalHealthExpendData/NationalHealthAccountsHistorical.html. ↩︎
- Ibid. ↩︎
- State fiscal years begin on July 1 except for these states: NY on April 1; TX on September 1; AL, MI and DC on October 1. ↩︎
- An archive of previous survey reports is available at: 50-State Medicaid Budget Survey Archives, Kaiser Commission on Medicaid and the Uninsured, accessed October 1, 2016, https://modern.kff.org/medicaid/report/medicaid-budget-survey-archives/. ↩︎
- Missouri plans to replace its family planning waiver with a state-funded family planning coverage program that will not cover or pay for services provided by organizations that also provide abortion services. Women who are eligible for the federally-funded program will continue to be eligible for the state-funded program, without change. The available services will also remain the same but the provider qualifications will be changed. Missouri Department of Social Services, Public Notice of Suspension of Federal Expenditure Authority for Section 1115 Family Planning Demonstration, entitled “Missouri Woman’s Health Services Program,” (Missouri Department of Social Services, July 2016), https://dss.mo.gov/mhd/waivers/1115-demonstration-waivers/files/missouri-women-health-services-waiver-suspension-notice-phase-out-plan.pdf. ↩︎
- Centers for Medicare and Medicaid Services, To facilitate successful re-entry for individuals transitioning from incarceration to their communities, State Health Official Letter SHO #16-007, (Baltimore, MD: Centers for Medicare and Medicaid Services, April 2016), https://www.medicaid.gov/federal-policy-guidance/downloads/sho16007.pdf. ↩︎
- Ibid. ↩︎
- The Omnibus Budget Reconciliation Act (OBRA) of 1986 established the option for states to cover pregnant women and infants (up to 1 year of age) up to 100 percent of federal poverty level (FPL). OBRA of 1989 mandated coverage for pregnant women and children under age 6 in families with incomes at or below 133 percent of FPL ↩︎
- A key challenge for most states is that there is no common data source for both the total number of births and the number financed by Medicaid. Data on total births generally comes from vital records data maintained by state public health agencies, although at least two states provided information from an all-payer hospital data base. ↩︎
- State results are weighted based on the June 2016 Medicaid and CHIP enrollment in each state. ↩︎
- Arkansas notes that Medicaid has historically funded sixty-six percent of all births in that state. With the implementation of Medicaid expansion, most Medicaid-funded births now occur through the Private Option plans. As a result, the Arkansas the Medicaid agency does not have data on the number of these Medicaid-funded births. ↩︎
- Julia Paradise, Medicaid Moving Forward (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, March 2015), https://modern.kff.org/health-reform/issue-brief/medicaid-moving-forward/. ↩︎
- Out of those 44 states, seven states (DC, Idaho, Kentucky, Louisiana, New Mexico, Vermont, and Virginia) report not charging premiums to enrollees in their buy-in programs and four states (Arkansas, Nebraska, New Jersey, and South Dakota) did not respond to the question about premiums. ↩︎
- Iowa has a FOA, but does not charge premiums. ↩︎
- Indiana requires premiums for some non-expansion enrollees. ↩︎
- Robin Rudowitz and MaryBeth Musumeci, The ACA and Medicaid Expansion Waivers (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, November 2015), https://modern.kff.org/medicaid/issue-brief/the-aca-and-medicaid-expansion-waivers/ . ↩︎
- New Hampshire has a waiver pending to impose copayments for non-emergency use of the emergency department, but this benefit is not covered in the QHP benefit package. ↩︎
- Centers for Medicare and Medicaid Services, Medicaid & CHIP Monthly Application, Eligibility Determinations, and Enrollment Reports. (Washington, DC: Centers for Medicare and Medicaid Services, June 2016), http://www.medicaid.gov/medicaid-chip-program-information/program-information/medicaid-and-chip-enrollment-data/medicaid-and-chip-application-eligibility-determination-and-enrollment-data.html. ↩︎
- Connecticut does not have capitated managed care arrangements, but does carry out many managed care functions, including ASO arrangements, payment incentives based on performance, intensive care management, community workers, educators, and linkages with primary care practices. ↩︎
- Idaho’s MMCP program, which is secondary to Medicare, has been re-categorized by CMS from a PAHP to an MCO by CMS but is not counted here as such. California has a small PCCM program operating in LA County for those with HIV. Three states use PCCM authority to operate specialized programs that are not counted here as PCCM programs: South Carolina uses PCCM authority to provide care management services to approximately 200 medically complex children; the Texas Medicaid Wellness program provides care management services for high-cost/high-risk enrollees, and Wyoming’s Patient Centered Medical Home program uses PCCM authority to make PMPM payments. ↩︎
- Centers for Medicare and Medicaid Services, Medicaid & CHIP Monthly Application, Eligibility Determinations, and Enrollment Reports. (Washington, DC: Centers for Medicare and Medicaid Services, June 2016), http://www.medicaid.gov/medicaid-chip-program-information/program-information/medicaid-and-chip-enrollment-data/medicaid-and-chip-application-eligibility-determination-and-enrollment-data.html. ↩︎
- California was re-categorized from “Varies” to “Always Mandatory” across all population groups (except for persons with ID/DD) as the state noted that enrollment is generally mandatory across the state with the exception of one, small rural county where managed care is voluntary because there is only 1 plan and it is not a COHS county. The ID/DD population is subject to mandatory enrollment only in COHS counties. ↩︎
- The state had planned to start implementation on January 1, 2016 but implementation was delayed due to delayed approval from CMS to allow the state additional time to complete readiness activities. See Letter from Vikki Wachino, Director Center for Medicaid & CHIP Services to Mikki Stier, Iowa Medicaid Director, February 23, 2016: https://governor.iowa.gov/sites/default/files/documents/CMS%20Letter%20to%20Branstad%20Administration.pdf. ↩︎
- “Medicaid to request July 1, 2017 start for Regional Care Organizations,” Alabama Medicaid Agency, September 14, 2016, http://medicaid.alabama.gov/news_detail.aspx?ID=11768. ↩︎
- 81 FR 27497, available at: https://www.gpo.gov/fdsys/granule/FR-2016-05-06/2016-09581. ↩︎
- In the rule, CMS formalizes its policy around “in lieu of,” which is an authority that a number of states were using to cover stays in IMDs prior to this rule. Some of these states must now adapt policies to meet the 15-day requirement, which may have fiscal and programmatic implications for these states. ↩︎
- For more information on the State Innovation Models (SIM) initiative, see: https://innovation.cms.gov/initiatives/state-innovations/. ↩︎
- “Accountable Communities of Health,” CMS, accessed September 5, 2016, https://innovation.cms.gov/initiatives/AHCM. ↩︎
- 81 FR 27497, available at: https://www.gpo.gov/fdsys/granule/FR-2016-05-06/2016-09581. ↩︎
- “Medicaid and CHIP Managed Care Final Rule (CMS 2390-F) Implementation Dates,” CMS, April 25, 2016, https://www.medicaid.gov/medicaid-chip-program-information/by-topics/delivery-systems/managed-care/downloads/implementation-dates.pdf. ↩︎
- Hawaii, North Dakota and Tennessee auto-assign all new members to a health plan. Hawaii and Tennessee then offer beneficiaries a choice, while North Dakota has only one plan. ↩︎
- Consumer Assessment of Healthcare Providers and Systems survey (CAHPS) was developed by the Agency for Health Research and Quality (AHRQ), http://www.ahrq.gov/cahps/about-cahps/index.html. ↩︎
- California notes that the delivery of substance abuse services is moving to an “Organized Delivery System operated by counties” in FY 2016. For purposes of this report, this new arrangement is treated as a PHP as it is recognized at the federal level. ↩︎
- “Patient-Centered Medical Home Recognition,” National Committee on Quality Assurance, accessed October 1, 2015, http://www.ncqa.org/Programs/Recognition/Practices/PatientCenteredMedicalHomePCMH.aspx. ↩︎
- Kaiser Commission on Medicaid and the Uninsured, Medicaid Delivery System and Payment Reform: A Guide to Key Terms and Concept (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, June 2015), https://modern.kff.org/medicaid/fact-sheet/medicaid-delivery-system-and-payment-reform-a-guide-to-key-terms-and-concepts/. ↩︎
- Kaiser Commission on Medicaid and the Uninsured, Medicaid Delivery System and Payment Reform: A Guide to Key Terms and Concept (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, June 2015), https://modern.kff.org/medicaid/fact-sheet/medicaid-delivery-system-and-payment-reform-a-guide-to-key-terms-and-concepts/. ↩︎
- Ibid. ↩︎
- Massachusetts Executive Office of Health and Human Services, Office of Medicaid, Section 1115 Demonstration Project Amendment and Extension Request (Massachusetts Executive Office of Health and Human Services, Office of Medicaid, July 22, 2016), https://www.medicaid.gov/Medicaid-CHIP-Program-Information/By-Topics/Waivers/1115/downloads/ma/ma-masshealth-pa.pdf. ↩︎
- Oregon also reported having “DSRIP-like” quality incentive programs in place in FY 2015. ↩︎
- Massachusetts Executive Office of Health and Human Services, Office of Medicaid, Delivery System Transformation Initiatives Trust Fund Legislative Report (Massachusetts Executive Office of Health and Human Services, Office of Medicaid, March 16, 2016), http://www.mass.gov/eohhs/docs/masshealth/research/legislature-reports/dsti-delivery-system-transformation-initiatives-status-report-03-16-16.pdf. ↩︎
- Comprehensive Primary Care Plus (CPC+) is a national advanced primary care medical home model. It is a five-year model that will begin in January 2017. Other states that include Medicaid as a partner but were not reported on this survey include: AR, CO, MT, RI, and TN. For more information see: https://innovation.cms.gov/initiatives/comprehensive-primary-care-plus. ↩︎
- Steve Eiken, Kate Sredl, Brian Burwell, and Paul Saucier, Medicaid Expenditures for Long-Term Services and Supports (LTSS) in FY 2014: Managed LTSS Reached 15 Percent of LTSS Spending (Baltimore, MD: CMS, April 15, 2016) https://www.medicaid.gov/medicaid-chip-program-information/by-topics/long-term-services-and-supports/downloads/ltss-expenditures-2014.pdf. ↩︎
- Ibid. ↩︎
- The “Program of all All-Inclusive Care for the Elderly” (PACE) is a capitated managed care benefit for frail seniors age 55 and older provided by a not-for-profit or public entity that features a comprehensive medical and social service delivery system. It uses a multidisciplinary team approach in an adult day health center supplemented by in-home and referral services in accordance with participants’ needs. ↩︎
- There are 11 states with no Section 1915(c) waivers for some or all populations (using Section 1115 instead): AZ, CA, DE, HI, NJ, NM, NY, RI, TN, TX, VT. See: MaryBeth Musumeci, Key Themes in Capitated Medicaid Managed Long-Term Services and Supports Waivers (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, November 2014), https://modern.kff.org/medicaid/issue-brief/key-themes-in-capitated-medicaid-managed-long-term-services-and-supports-waivers/. ↩︎
- CMCS Informational Bulletin, Coverage of Housing-Related Activities and Services for Individuals with Disabilities (Baltimore, MD: CMCS, June 26, 2015), https://www.medicaid.gov/federal-policy-guidance/downloads/CIB-06-26-2015.pdf. ↩︎
- Molly O’Malley Watts, Erica L. Reaves, and MaryBeth Musumeci, Money Follows the Person: A 2015 State Survey of Transitions, Services, and Costs (Washington, DC: Kaiser Commission on Medicaid and the Uninsured), https://modern.kff.org/medicaid/report/money-follows-the-person-a-2015-state-survey-of-transitions-services-and-costs/. ↩︎
- “Money Follows the Person (MFP),” CMS, accessed September 23, 2016, https://www.medicaid.gov/Medicaid-CHIP-Program-Information/By-Topics/Long-Term-Services-and-Supports/Balancing/Money-Follows-the-Person.html. ↩︎
- CMS issued approval of the core facets of the Washington’s proposal on September 30, 2016 as they work with the state to finalize special terms and conditions (STC)s. ↩︎
- HCBS benefit expansions reported in this section may include new HCBS waiver or SPA initiatives which may have also been reported/counted as expansions in persons served under HCBS through waivers or SPAs. ↩︎
- This count does not include two states (Colorado and Washington) that have managed FFS FADs. For more information see: https://www.cms.gov/Medicare-Medicaid-Coordination/Medicare-and-Medicaid-Coordination/Medicare-Medicaid-Coordination-Office/FinancialAlignmentInitiative/ManagedFeeforServiceModel.html. ↩︎
- The Affordable Care Act (ACA) authorized the Secretary of Health and Human Services to implement the Financial Alignment Initiative to allow state-administered demonstration projects to improve the integration and coordination of services for individuals who are covered under both Medicare and Medicaid. This population, as a group, experiences high rates of hospitalization and use of LTSS and is, on average, a high need, high cost population. See: https://www.cms.gov/Medicare-Medicaid-Coordination/Medicare-and-Medicaid-Coordination/Medicare-Medicaid-Coordination-Office/FinancialAlignmentInitiative/FinancialModelstoSupportStatesEffortsinCareCoordination.html.. ↩︎
- Kaiser Commission on Medicaid and the Uninsured, Health Plan Enrollment in the Capitated Financial Alignment Demonstrations for Dual Eligible Beneficiaries (Washington, DC: Kaiser Commission on Medicaid and the Uninsured, August 2016), https://modern.kff.org/medicaid/fact-sheet/health-plan-enrollment-in-the-capitated-financial-alignment-demonstrations-for-dual-eligible-beneficiaries/. ↩︎
- Dual Eligible Special Needs Plans (D-SNPs) enroll beneficiaries who are entitled to both Medicare and Medicaid, and offer the opportunity to better coordinate benefits among Medicare and Medicaid. For more information see: https://www.cms.gov/Medicare/Health-Plans/SpecialNeedsPlans/DualEligibleSNP.html. ↩︎
- Chronic Condition Special Needs Plans must offer specially-designed plan benefit packages that provide supplemental health benefits and specialized provider networks specific to designated chronic conditions. For more information see: https://www.cms.gov/Medicare/Health-Plans/SpecialNeedsPlans/Chronic-Condition-Special-Need-Plans-C-SNP.html. ↩︎
- Fully Integrated Dual Eligible SNPs were created by Congress in Section 3205 of the Affordable Care Act to promote full integration and coordination of Medicaid and Medicare benefits for dual eligible beneficiaries by a single managed care organization. They must have a MIPPA compliant contract with a State Medicaid Agency that includes coverage of specified primary, acute and long-term care benefits and services under risk-based financing. For more information see: https://www.cms.gov/Medicare/Health-Plans/SpecialNeedsPlans/DualEligibleSNP.html#s3. ↩︎
- “Home and Community-Based Services Quality,” National Quality Forum, accessed September 21, 2016, http://www.qualityforum.org/ProjectDescription.aspx?projectID=77692. ↩︎
- National Quality Forum, Quality in Home and Community-Based Services to Support Community Living: Addressing Gaps in Performance Measurement (Washington, DC: National Quality Forum, September 2016), http://www.qualityforum.org/Publications/2016/09/Quality_in_Home_and_Community-Based_Services_to_Support_Community_Living__Addressing_Gaps_in_Performance_Measurement.aspx. ↩︎
- Rates for calendar 2017 not yet determined at the time of the survey included MCO rates for Florida, Illinois, Maryland, and Minnesota. While some states with calendar year contracts provided the budgeted level of MCO rate increases, these four states indicate that they are waiting for work by their actuaries. Wisconsin is implementing APR-DRGs in in January 2017 which potentially could move funds between inpatient and outpatient hospital rates. ↩︎
- Some states also have premium or claims taxes that apply to managed care organizations and other insurers. Since this type of tax is not considered a provider tax by CMS, these taxes are not counted as provider taxes in this report. ↩︎
- Centers for Medicare and Medicaid Services, CMCS Informational Bulletin: Clarification of Medicaid Coverage of Services to Children with Autism (Washington, DC: Centers for Medicare and Medicaid Services, July 2014), http://www.medicaid.gov/Federal-Policy-Guidance/Downloads/CIB-07-07-14.pdf. ↩︎
- Centers for Medicare and Medicaid Services, Medicaid Drug Rebate Program Notice, Release No. 172 (Centers for Medicare and Medicaid Services, November 2015), https://www.medicaid.gov/Medicaid-CHIP-Program-Information/By-Topics/Benefits/Prescription-Drugs/Downloads/Rx-Releases/State-Releases/state-rel-172.pdf. ↩︎
- Interferon-free Direct Acting Antivirals (DAAs) used to treat hepatitis C entered the market in 2013. They have very high cure rates and minimal side-effects, but have been priced expensively. ↩︎
- B.E. and A.R. v. Teeter, No. C16-227-JCC (W.D. Wa. May 27, 2016), https://today.law.harvard.edu/wpcontent/uploads/2016/06/40-5-27-16-Order-Granting-Preliminary-Injunction.pdf. ↩︎
- In accordance with federal and state law, states pay the lower of (a) the ingredient cost rate plus a dispensing fee; (b) the Federal Upper Limit (FUL) or State Maximum Allowable Cost rate, if applicable, plus a dispensing fee; or (c) the pharmacy’s Usual and Customary Charge. ↩︎
- 81 Fed. Reg. 5170. ↩︎
- Centers for Medicare and Medicaid Services, CMCS Informational Bulletin: Medicaid Pharmacy – Survey of Retail Prices (Washington, DC: Centers for Medicare and Medicaid Services, May 2012), http://www.medicaid.gov/Federal-Policy-Guidance/Downloads/CIB-05-31-12.pdf. ↩︎
- “Clinical edits” are clinically-based claims adjudication rules that a claims system will follow when processing a pharmacy claim. ↩︎
- Li Hui Chen, Holly Hedegaard, and Margaret Warner, Drug-poisoning deaths involving opioid analgesics: United States, 1999–2011, (National Center for Health Statistics, no. 166, September 2014), http://www.cdc.gov/nchs/products/databriefs/db166.htm ↩︎
- Centers for Medicare and Medicaid Services, CMCS Informational Bulletin: Best Practices for Addressing Prescription Opioid Overdoses, Misuse and Addiction, (Baltimore, MD: Centers for Medicare and Medicaid Services, January 2016), https://www.medicaid.gov/federal-policy-guidance/downloads/CIB-02-02-16.pdf. ↩︎
- Deborah Dowell, Tamara Haegerich, and Roger Chou, “CDC Guideline for Prescribing Opioids for Chronic Pain — United States, 2016,” Centers for Disease Control and Prevention, Morbidity and Mortality Weekly Report, 65, no.1 (March 2016): 1-49, http://dx.doi.org/10.15585/mmwr.rr6501e1. ↩︎
- National Safety Council, Prescription Nation 2016, Addressing America’s Drug Epidemic (National Safety Council, 2016), http://www.nsc.org/RxDrugOverdoseDocuments/Prescription-Nation-2016-American-Drug-Epidemic.pdf. ↩︎
- Centers for Medicare and Medicaid Services, CMCS Informational Bulletin: Best Practices for Addressing Prescription Opioid Overdoses, Misuse and Addiction, (Baltimore, MD: Centers for Medicare and Medicaid Services, January 2016), https://www.medicaid.gov/federal-policy-guidance/downloads/CIB-02-02-16.pdf. ↩︎
- Several states mentioned plans to implement quantity limits based on a “morphine equivalent dose” which is the amount of opioid prescription drugs, converted to a common “standard” unit (milligrams of morphine). For example, both 60 mg of oxycodone (approximately 2 tablets of oxycodone sustained-release 30 mg) and approximately 20 mg of methadone (4 tablets of methadone 5 mg) are equal to 90 MMEs (morphine milligram equivalents). ↩︎
- Prescription Drug Monitoring Program Training and Technical Assistance Center, Status of Prescription Drug Monitoring Programs (PDMPs), (Prescription Drug Monitoring Program Training and Technical Assistance Center, August 2015), http://www.pdmpassist.org/pdf/PDMPProgramStatus2015_v5.pdf. ↩︎
- Several states with requirements noted that they were statutory (rather than contractual) including two states (Arizona and Maryland) reporting a legislative requirement that would take effect in FY 2018. One state commented that its Medicaid agency did not have access to the PDMP and therefore was unable to mandate its use by Medicaid prescribers. ↩︎
- State fiscal years begin July 1 except for these states: NY on April 1; TX on September 1; AL, MI and DC on October 1. ↩︎