What to Know About the BALANCE Model for GLP-1s in Medicare and Medicaid
GLP-1s, a class of drugs used to treat type 2 diabetes, obesity, cardiovascular disease, and other conditions, have exploded in popularity in recent years due to their demonstrated effectiveness, but are often not covered by insurance, particularly for the treatment of obesity. According to KFF polling, about half (56%) of GLP-1 users say these drugs were difficult to afford, including one in four who say they were “very difficult” to afford. The Trump administration is pursuing various approaches to lowering the cost and expanding coverage of these medications. These approaches include striking “most-favored nation” deals with GLP-1 manufacturers Novo Nordisk and Eli Lilly, providing access to discounted prices for GLP-1s through TrumpRx, and implementing a new demonstration program called the BALANCE (Better Approaches to Lifestyle and Nutrition for Comprehensive hEalth) Model to expand Medicare and Medicaid coverage of GLPs for obesity, which is currently subject to statutory limitations (prohibited in Medicare, permissible but not required in Medicaid). In addition, the GLP-1 drug semaglutide (branded as Ozempic, Wegovy, and Rybelsus) was selected for Medicare drug price negotiation in 2025, with a negotiated price set to take effect in 2027.
This brief describes current coverage of GLP-1s in Medicare and Medicaid, the Centers for Medicare & Medicaid Services’ (CMS) efforts to expand access and lower costs for GLP-1s through temporary demonstration programs including the BALANCE Model, and potential impacts on beneficiaries and program budgets.
Current law prohibits Medicare from covering obesity drugs and gives states flexibility to cover weight loss drugs under Medicaid
Limitations on coverage for obesity drugs in Medicare and Medicaid mean that millions of people who have obesity and might benefit from taking GLP-1s may be unable to access them unless they are able to pay the full cash price out of their own pockets, which would likely be prohibitive for people with Medicaid who must have low incomes to qualify for the program, as well as for many people on Medicare with low and modest incomes. Under the Medicare Part D outpatient prescription drug benefit program, Part D plans are required to cover a minimum of two drugs in each therapeutic category and class, but from the outset, Medicare has been prohibited by law from covering medications when used specifically for weight loss. People on Medicare can get GLP-1s covered by Part D plans only if they are used for a medically accepted FDA-approved indication other than obesity, like type 2 diabetes, cardiovascular disease risk reduction, or sleep apnea.
Under the Medicaid Drug Rebate Program (MDRP), state Medicaid programs must cover nearly all of a participating manufacturer’s FDA-approved drugs for medically accepted indications. However, federal law gives states the option whether to cover drugs used for weight loss. As a result, GLP-1 coverage for medically accepted FDA-approved indications other than obesity is required while access to GLP-1s to treat obesity under Medicaid is currently limited. Only 13 states provided coverage as of January 2026, down from 16 states in 2025, likely reflecting the significant costs of coverage and recent state budget challenges and federal funding cuts.
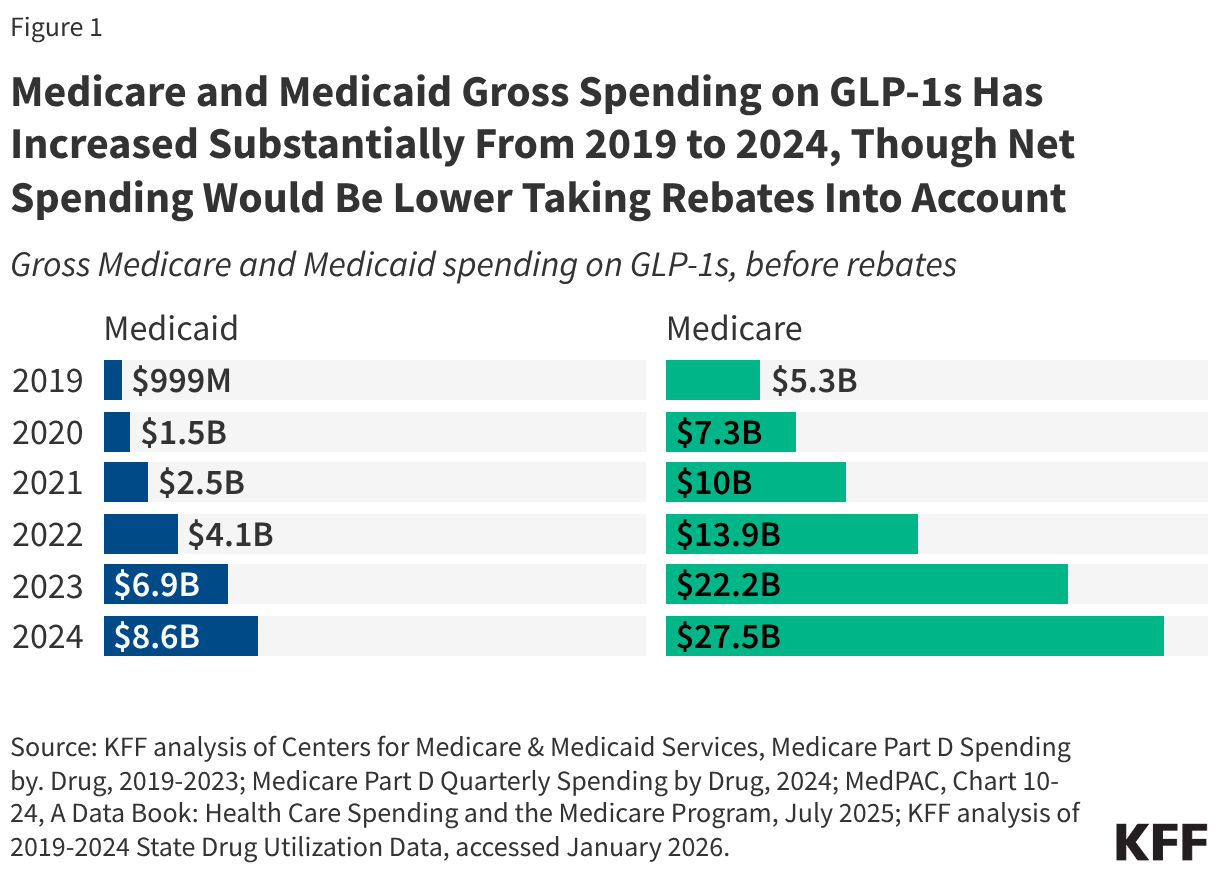
Even with these coverage limits on obesity drugs in place, utilization and gross spending on GLP-1 drugs for approved uses in Medicare and Medicaid have increased considerably in recent years. In 2024, there were 8.4 million prescriptions and $8.6 billion in spending on GLP-1s in Medicaid, and 21.8 million claims and $27.5 billion in gross spending (not excluding rebates) on GLP-1s in Medicare (Figure 1).
CMS is proposing temporary expansions of Medicare and Medicaid coverage of GLP-1s for obesity through demonstration programs
CMS has proposed a two-step approach to expanding coverage of GLP-1s for obesity in Medicare – a temporary payment demonstration for 2026, known as the Medicare GLP-1 Bridge, and a new Center for Medicare and Medicaid Innovation (CMMI) model, known as the BALANCE Model, beginning January 2027. For Medicaid, GLP-1 coverage will be expanded through the BALANCE Model beginning in May 2026. The BALANCE Model ends for both state Medicaid agencies and Medicare Part D plans in December 2031. (See Figure 2 for a timeline of key activities associated with the GLP-1 coverage demonstrations.)
Medicare coverage of GLP-1s for obesity begins in July 2026 through the Medicare GLP-1 Bridge, prior to implementation of the BALANCE Model
Until implementation of the BALANCE Model for Part D plans begins in January 2027, CMS will provide Part D beneficiaries with coverage of select GLP-1s for obesity from July 1, 2026 to December 31, 2026 through the Medicare GLP-1 Bridge, which is a separate short-term demonstration established using Section 402 demonstration authority. The Medicare GLP-1 Bridge is a nationwide demonstration program that will be separate from Part D coverage, meaning Part D sponsors will not have to opt into the demonstration for eligible beneficiaries to gain access, nor will Part D sponsors bear any financial risk for costs incurred by their enrollees associated with the demonstration.
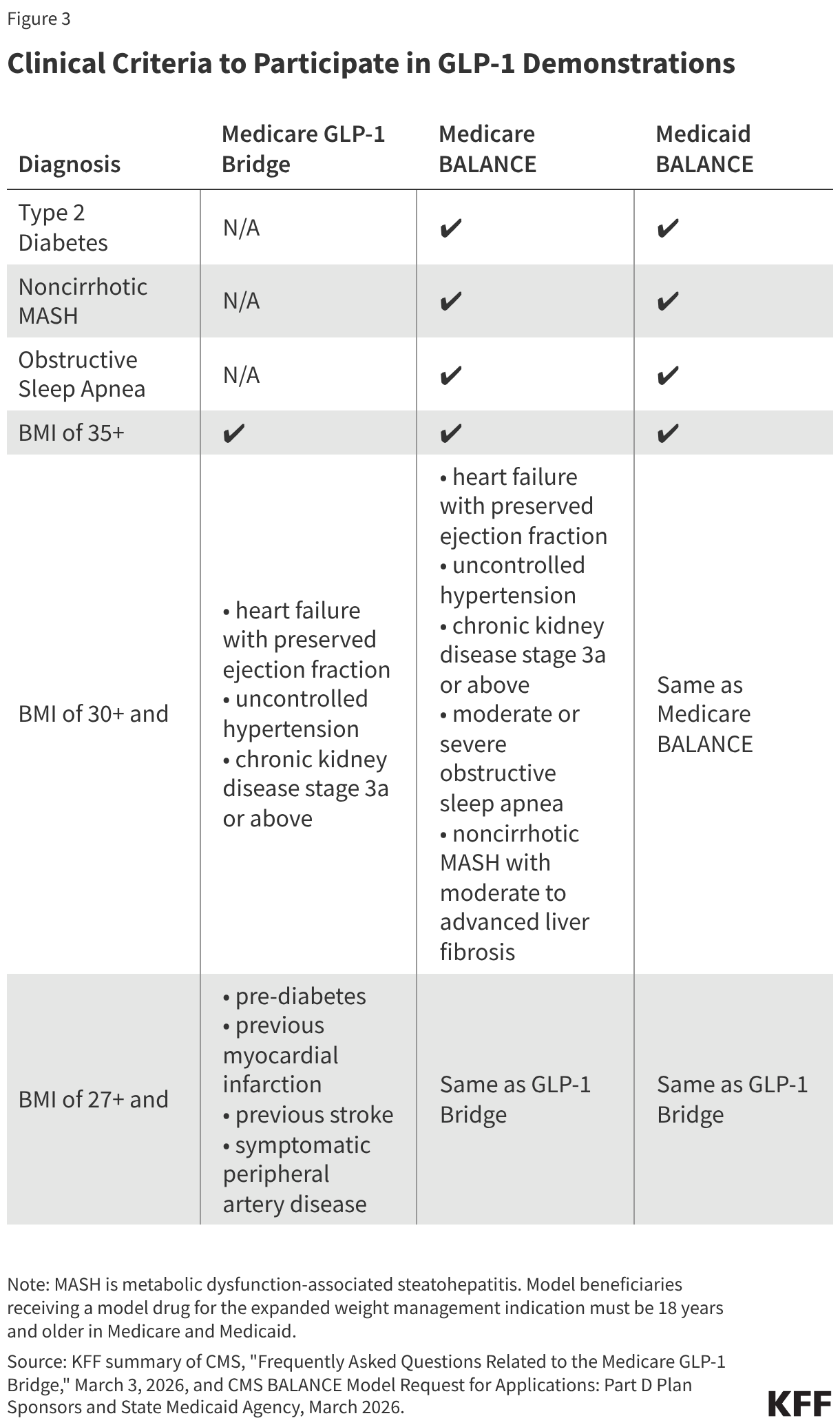
According to FAQs released by CMS in March 2026, Medicare beneficiaries enrolled in Part D plans who meet the eligibility criteria will have access to GLP-1 medications approved for weight reduction (Wegovy and Zepbound) at a copayment of $50 per month. For a beneficiary to qualify, their provider must submit a prior authorization request that attests the beneficiary is being prescribed the drug to reduce excess body weight and ongoing maintenance of weight reduction and that they fall into one of three categories related to BMI and other clinical diagnostic criteria (Figure 3).
Manufacturers have agreed to provide Wegovy and Zepbound at a net price of $245 per month supply. When participating beneficiaries fill a prescription for one of these drugs, pharmacies will collect the $50 copayment and submit claims to a central processor for reimbursement. Pharmacies will be reimbursed by CMS at no lower than the wholesale acquisition cost (WAC) of a drug, less the beneficiary copay, plus a dispensing fee and, as applicable, sales tax. Manufacturers will then owe money back to CMS for the difference between the WAC and the negotiated $245 net price.
Because this payment demonstration operates outside coverage under a Part D plan, the $50 copayment toward these medications will not count toward a participating beneficiary’s Part D deductible or the $2,100 out-of-pocket maximum, and copayments will stay consistent at $50 per month, regardless of the Part D benefit phase a beneficiary is in when they fill the prescription. Medicare beneficiaries who are already receiving coverage from their Part D plan for a GLP-1 for a Medicare-covered use, such as type 2 diabetes, cardiovascular disease risk reduction, or sleep apnea, will continue to access the drug through their Part D plan and not through the Medicare GLP-1 Bridge, which will only provide coverage of GLP-1s when used for obesity.
For beneficiaries enrolled in the Low-Income Subsidy (LIS) program, the LIS cost-sharing subsidies will not apply in the Medicare GLP-1 Bridge. This may make it more difficult for low- and modest-income beneficiaries who are otherwise eligible to participate to take advantage of coverage under the short-term demonstration in 2026 if the $50 monthly copayment is unaffordable.
For participating beneficiaries to maintain Medicare coverage of their GLP-1 medication for obesity after the Medicare GLP-1 Bridge ends at the end of 2026, they will need to be enrolled in a Part D plan that chooses to participate in the BALANCE Model in 2027. This could mean having to switch Part D plans for the coming year, with potential cost and coverage implications for other medications beneficiaries use.
The BALANCE Model expands coverage of GLP-1s for obesity in Medicaid beginning in 2026 and under Medicare beginning in 2027
According to CMS, the BALANCE Model aims to increase access to GLP-1 medications and healthy lifestyle interventions to help people on Medicare and Medicaid improve their overall health. Under this model, CMS will negotiate with manufacturers of GLP-1s to provide lower prices to the state Medicaid programs and Medicare Part D plans that choose to participate in the model. For beneficiaries to be eligible under the BALANCE Model in either Medicaid or Medicare Part D (including Part D LIS enrollees), providers must attest that they meet certain clinical criteria, including qualifying for a GLP-1 for a currently covered use (such as type 2 diabetes or sleep apnea) or for use to treat obesity based on similar clinical criteria as applied in the Medicare GLP-1 Bridge (Figure 3).
In addition to offering lower prices for GLP-1s, this model will provide patients with access to lifestyle support programs at no cost, which are intended to support medication adherence as well as increase GLP-1 effectiveness. (Details about these programs are not yet available.) These lifestyle support programs will be provided by participating manufacturers. As part of their agreements with CMS, the manufacturers must demonstrate how these lifestyle support programs will meet the program requirements, including encouraging healthy eating and increasing physical activity, supporting medication adherence, ensuring engagement with the program on a regular basis, and ensuring availability of this program to all patients receiving these medications, either online or offline for those who have limited digital access. However, prescribing providers will not be required to document that patients are actively participating in these lifestyle support programs as part of attesting to their eligibility for the model based on other clinical criteria.
Participation in the BALANCE Model is voluntary for drug manufacturers, state Medicaid programs, and Medicare Part D plans
Participation in BALANCE is voluntary for drug manufacturers, state Medicaid agencies, and Medicare Part D plans. The date for manufacturers to notify CMS of their interest in participating was January 8, 2026; for Medicare Part D plans is April 20, 2026; and for state Medicaid programs is July 31, 2026 (Figure 2).
Drug Manufacturers
During the model pre-implementation period in early 2026, CMS negotiated with Novo Nordisk and Eli Lilly to come to agreement on the key parameters of the model, including details about pricing of the model drugs, cost sharing, rebate calculations, access policies (i.e., coverage criteria and prior authorization policies), the length of the agreement, data sharing arrangements, and agreement on lifestyle supports that will be offered. Both manufacturers have agreed to participate in the model, and the following medications will be included: all formulations of Mounjaro, Ozempic, Rybelsus, and Wegovy, the KwikPen formulation of Zepbound, and, if approved by the FDA, the tablet formulation of Orforglipron. The manufacturers have agreed to a $245 net price per 30-day supply for all model drugs in 2027 for the Medicare program though the net price for state Medicaid programs is confidential to the public. CMS and manufacturers may renegotiate terms in the future depending on certain circumstances such as changes in the FDA labeling, new clinical evidence, or new products launched.
State Medicaid Programs
Participation. For state Medicaid agencies that opt to participate in the model, implementation will be on a rolling basis from May 1, 2026 through January 1, 2027. To participate in the model, state Medicaid programs must sign a State Agreement with CMS and then adopt supplemental rebate agreements (SRAs) with each participating manufacturer that reflects the standard key terms that the participating manufacturers and CMS have agreed to. While states typically develop their own utilization management strategies, the model key terms establish standardized coverage criteria (Figure 3). States may offer broader coverage but cannot make coverage criteria more restrictive, and the Medicaid key terms must apply equally in both fee-for-service and Medicaid managed care. Although the Medicaid component of the BALANCE Model will launch May 1, the deadline for the state Medicaid agency Request for Applications (RFA) is July 31, 2026.
Cost. The discounted GLP-1 net price for state Medicaid programs will be available through additional supplemental rebates (on top of statutory rebates through the MDRP). To participate in the model, state Medicaid programs must terminate or update any existing SRAs with participating manufacturers. While the original announcement of the MFN deals with Eli Lilly and Novo Nordisk noted Medicaid programs would also have access to the $245 price available to Medicare, the final negotiated discounted price available to state Medicaid programs mentioned in the state Medicaid RFA is not available to the public, only participating states. The model will not affect out-of-pocket costs for Medicaid enrollees, which are limited to nominal amounts under federal law.
Medicare Part D Plans
Participation. Part D plan sponsors must apply to participate in the model by April 20, 2026, and after CMS confirms eligibility, plan sponsors will indicate whether they will participate in the model by June 1, 2026. Participants can include sponsors of Part D stand-alone prescription drug plans (PDPs) and Medicare Advantage prescription drug plans (MA-PDs), including Special Needs Plans (SNPs), and employer plans that offer Part D. Eligible plan types exclude Defined Standard benefit plans, which charge a standard 25% coinsurance amount for all covered drugs and do not vary cost sharing by drug type or formulary tier, although plan sponsors can indicate if they wish to convert a Defined Standard benefit plan to another basic benefit type in order to become eligible. Participation is at the plan sponsor level, and plan sponsors can choose which of their specific plan benefit packages will be part of the model. However, CMS will require plan sponsor participants to include all of their enhanced alternative plans and 90% of their enrollment in basic plans.
Cost sharing. Part D plans must adopt specific cost-sharing limits under the basic benefit plan structure as agreed upon by CMS and manufacturers. Cost sharing for model drugs will be limited to $245 for a 30-day supply in the deductible phase and a lower amount in the initial coverage phase: $50 per month for enhanced plans and employer group plans and $125 per month for basic plans (though plan sponsors can choose to apply lower cost-sharing amounts to model drugs as long as they do so uniformly across all model drugs). Once beneficiaries reach their out-of-pocket maximum (set at $2,400 in 2027), they will pay $0 for their medications, including for model drugs. Part D plans participating in the model are required to place all model drugs on the same formulary tier, cover all medically accepted indications for model drugs, and apply the same cost sharing to all indications.
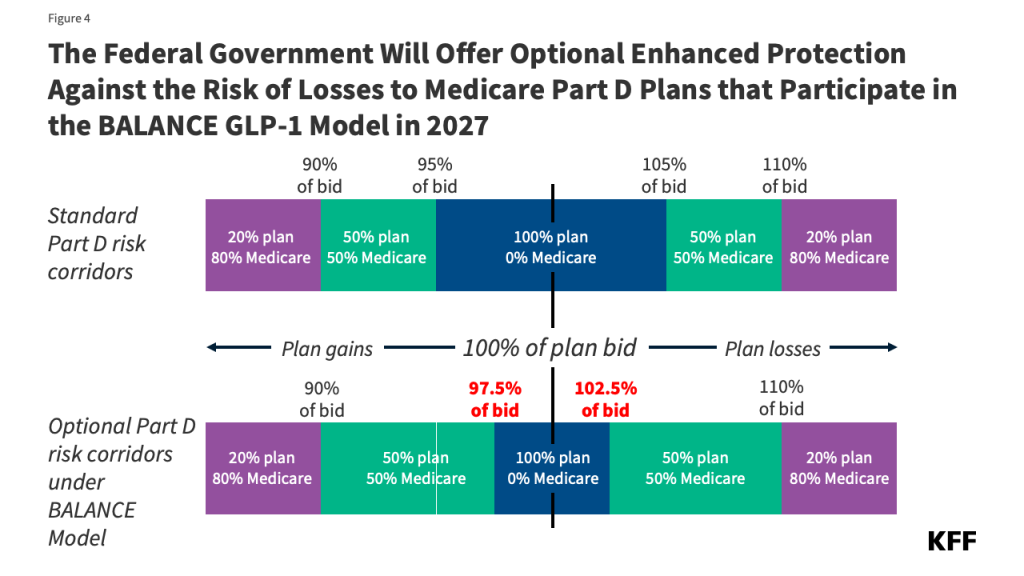
Participation incentives. Because the model is voluntary for Part D plan sponsors, CMS has designed financial incentives to encourage plan participation. The primary approach to be taken in 2027 involves an optional narrowing of the risk corridor thresholds to reduce the range of spending where PDPs bear full risk for actual costs higher than their bids (Figure 4). Model participants will qualify for this additional financial protection if they opt in and have higher-than-average utilization of model drugs relative to other similar plan participants. In future model years, CMS is considering additional payment incentives, including higher direct subsidy payments based on an adjustment factor to the beneficiary risk score in participating plan bids.
CMS is aiming for a “critical mass” of Part D sponsors to participate and has established a threshold participation rate of 80% for 2027. This participation rate will be calculated as the number of beneficiaries enrolled in Part D plans applying to participate in the model divided by the total number of beneficiaries in all Part D plans (excluding enrollment in special needs plans and employer plans from these calculations, even though they are eligible to participate in the model, but including enrollment in Defined Standard benefit plans, even though they are ineligible to participate). (This calculation method could produce a different participation rate than it would be if the calculation was based on enrollment in all eligible plans that apply to participate divided by enrollment in all eligible plan types overall. CMS has not explained the rationale for their chosen method.) If the 80% threshold is not met, CMS will not move forward with the BALANCE Model in Medicare in 2027. CMS will notify Part D plan sponsor applicants whether this threshold is met by April 30, 2026. If the participation threshold isn’t met for BALANCE, which will be known by the end of April, CMS has not indicated if or how it would affect the implementation of the GLP-1 Bridge.
Millions of people with Medicare and Medicaid could benefit from expanded coverage of GLP-1s for obesity
Overall, the BALANCE Model could greatly expand access to GLP-1s depending on the level of participation by drug manufacturers, state Medicaid agencies and Part D plans. In addition to providing coverage of these drugs for obesity, which would be a new indication in Medicare and in most states under Medicaid, this model would also allow beneficiaries in participating states and Part D plans to access GLP-1s for obesity at a lower out-of-pocket cost than if they were to purchase them with cash at the direct-to-consumer prices offered by the manufacturers. For Medicare Part D enrollees who already have coverage of a GLP-1 due to a currently covered indication, they might face lower out-of-pocket costs if their current cost-sharing amounts are higher than the cost-sharing limits that have been established for the BALANCE Model ($50 for enhanced plans, $125 for basic plans). Additionally, this model will provide access to a lifestyle support program to promote healthy behaviors and increase the effectiveness of GLP-1s, which could provide improvements in health along with sustained weight reduction.
However, there are factors that could limit the reach of this model for both the Medicaid and Medicare populations. For example, if the level of participation by state Medicaid agencies is relatively low, the impact of this model for Medicaid recipients may not be very far reaching. In addition, state Medicaid agencies can choose to participate in the model initially but then decide to drop out of the model in later years, which could result in disruptions in coverage.
Within Medicare, if participation by Part D plan sponsors is less than the 80% threshold, CMS has stated that it will not launch the model in Medicare in 2027. This sets up the possibility that Medicare beneficiaries could have coverage of GLP-1s for obesity under the Medicare GLP-1 Bridge in the latter half of 2026 but then lose access in 2027 if the BALANCE model is not implemented. Similarly, beneficiaries will need to be enrolled in a participating plan in order to maintain coverage of GLP-1s for obesity in 2027, which could mean switching plans and disrupting current coverage.
Assuming the BALANCE Model launches in 2027 with at least 80% participation, Part D plans could drop out of the model in future years, which could interrupt treatment, or require frequent plan changes for Medicare Part D enrollees to maintain access. It is unclear from CMS documentation whether they will reassess the 80% participation threshold in each model year for the model to continue. It is also unclear whether Part D plan sponsors will be able continue to cover GLP-1 drugs for the treatment of obesity once the model ends if Medicare’s statutory exclusion on weight loss drugs is not lifted.
Another uncertainty for Part D enrollees in terms of their out-of-pocket costs is the impact of the model on Part D premiums. Participating plan sponsors will factor the cost of the model into their bids for contract year 2027, and assuming the participation threshold is met, plans may incur higher costs, which would be incorporated into the bids of plans that most beneficiaries are currently enrolled in. That could have the effect of increasing Part D premiums across the board, although Medicare Advantage plans can use rebates to buy down Part D premiums, but this is not an option for stand-alone PDPs. It is possible that CMS could modify the parameters of the Part D PDP premium stabilization demonstration to provide greater premium subsidies for PDP plan sponsors that participate in the BALANCE Model. The ultimate effect on Part D plan bids and premiums is unknown at this time.
The potential federal and state budgetary impacts of expanded coverage of GLP-1s for obesity in Medicare and Medicaid are unknown
CMS documentation does not include potential federal or state budgetary impacts from either the BALANCE Model or the Medicare GLP-1 Bridge. Spending on GLP-1 drugs for currently covered uses under Medicare and Medicaid has increased substantially in a relatively short period of time and could increase further with expanded coverage of GLP-1s for obesity, even at the lower net prices for these medications under both demonstrations.
The law requires Innovation Center models to either maintain or reduce program expenditures. The model will test whether lower negotiated prices as applied to currently covered indications will lower program spending. It is unclear how the new negotiated prices under the model for state Medicaid programs (which remain confidential) compare to the net prices state Medicaid programs are currently paying for these drugs, but Medicaid already typically pays lower prices than other payers for prescription drugs. In general, without knowing what Medicaid and Medicare generally are currently paying for these drugs on net, it is uncertain whether the lower prices that will be made available to state Medicaid programs and Part D plans under the BALANCE Model for currently covered uses of GLP-1s will generate enough savings to offset the additional costs from expanded use of these medications for obesity. If state Medicaid programs estimate the costs from expanded use will outweigh the savings, initial state Medicaid participation may be limited, especially given recent state budget challenges and federal funding cuts.
Evaluations will also determine whether improvements in health related to the use of these drugs and associated reductions in healthcare utilization are significant enough to maintain or reduce health care costs in the Medicaid and Medicare programs, after taking into account expanded use and coverage of GLP-1s for the treatment of obesity. Even with lower prices, there is little evidence to date to suggest that the expanded use of GLP-1s will be offset by lower spending on other health care services in the short term, even though the drugs do provide significant health benefits to users.
The cost of the Medicare GLP-1 Bridge is not discussed in CMS documentation about this demonstration. The federal government is likely to incur additional spending under this demonstration due to paying for GLP-1s for obesity, which is not currently covered under Medicare. In addition, because the demonstration will operate outside the Part D benefit, the manufacturers won’t be responsible for providing the manufacturer price discount on eligible GLP-1s (10% in the Part D initial benefit phase and 20% in the catastrophic phase). While many prior section 402 demonstrations have had to conform to budget neutrality rules by the Office of Management and Budget (OMB), some demonstrations have been able to move forward without this requirement. Given the short timeframe of the GLP-1 demonstration, it is unlikely that there will be cost offsets from improved health due to increased GLP-1 use for obesity that can be documented.
This work was supported in part by Arnold Ventures. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.