The U.S. Government and Gavi, the Vaccine Alliance
Editorial Note: Originally published in June 2011, this resource is updated as needed to reflect the latest developments.
Key Facts
- Gavi, the Vaccine Alliance (Gavi) is an independent public-private partnership and multilateral funding mechanism that aims to expand global access to and use of vaccines, particularly among vulnerable children.
- Since its launch in 2000, Gavi has provided approximately $29 billion to support immunization efforts in low- and middle-income countries, not including funding for COVAX.
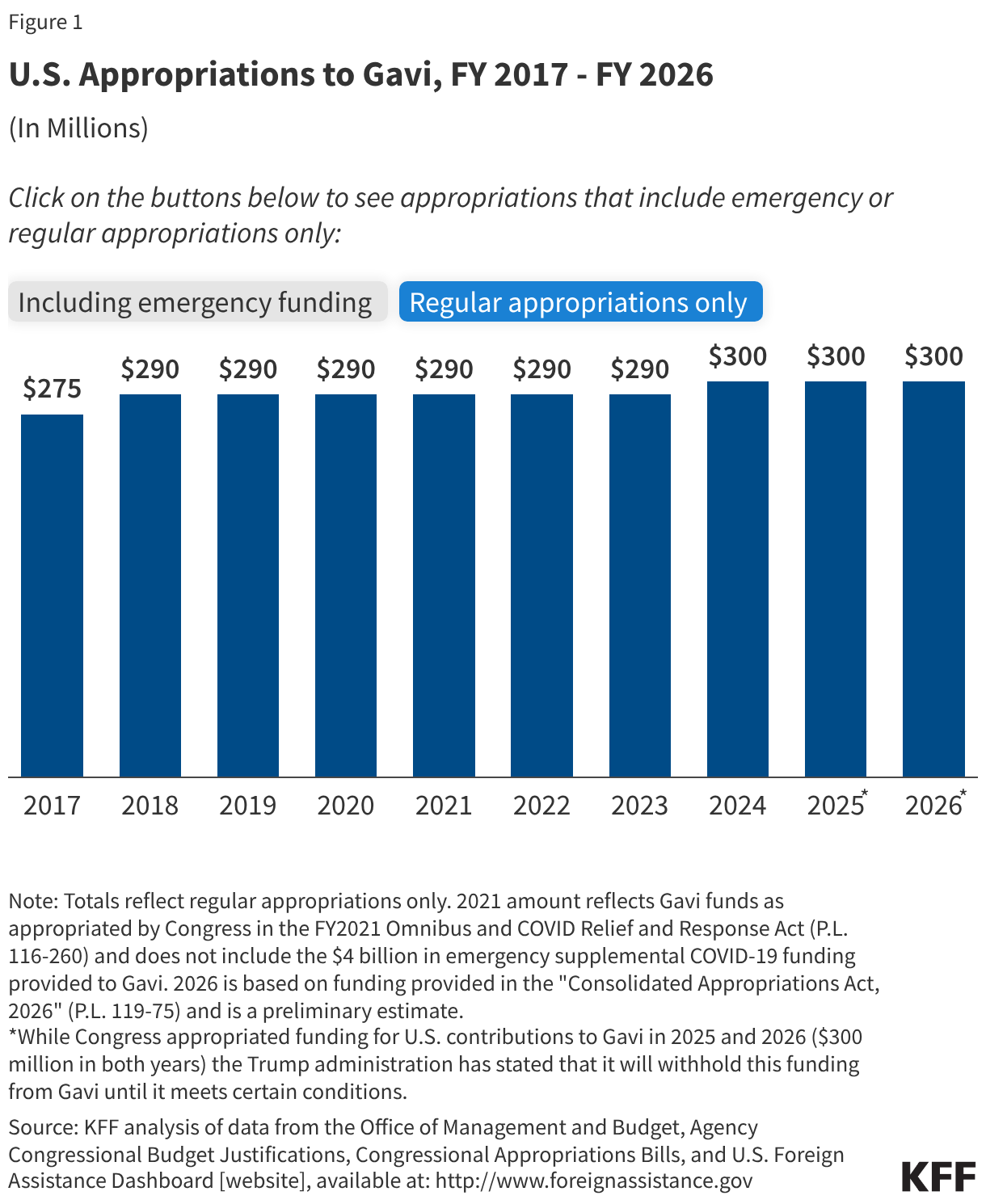
- The U.S. government (U.S.) has supported Gavi since its creation through direct financial contributions, participation in Gavi’s governance as a member of the Board, and technical assistance; it had been its third largest contributor in recent years, providing 13% of its funding since its inception (not including funding for COVAX, the global COVID-19 pandemic vaccine response), reaching $300 million in FY 2024. In addition, the Biden administration had pledged that the U.S. would provide approximately $1.6 billion to Gavi over its 2026-2030 funding period.
- While Congress again appropriated $300 million for Gavi in both FY 2025 and FY 2026, the Trump administration has not provided this funding to the organization, citing concerns about vaccine safety despite Gavi following globally recognized scientific standards and evidence.
- Gavi’s latest replenishment summit secured pledges of more than $9 billion, towards a target of $11.9 billion, for the 2026-2030 period, as well as additional financing from development finance institutions and manufacturers to support country recipients. Still, the loss of U.S. funding in the context of a constrained financing environment globally presents new challenges for reaching children in low- and middle- income countries with life-saving vaccines.
Gavi Overview
Created in 1999 and formally launched in January 2000, Gavi, the Vaccine Alliance (Gavi) is an independent public-private partnership and multilateral funding mechanism that “aims to save lives and protect people’s health by increasing coverage and equitable and sustainable use of vaccines.” Gavi’s main activities include supporting low- and middle-income countries’ access to new and underused vaccines for vulnerable children through financial support, technical expertise, and market-shaping efforts, such as negotiating with manufacturers, to help lower the cost of procuring vaccines. Gavi operates in five-year funding cycles, with a revised strategy and goals for each cycle. Each five-year strategy is accompanied by a vaccine investment strategy, which determines which vaccines will be made available to countries.
Gavi’s current five-year strategy, for the 2026-2030 period, which is its sixth strategy, includes four core goals:
- introduce and scale-up vaccines,
- strengthen health systems to increase equity in immunization,
- improve sustainability of immunization programs, and
- ensure healthy markets for vaccines and related products.
The current strategy emphasizes reducing the number of ‘zero-dose’ children with the goal of reaching no zero-dose children by 2030, in alignment with Immunization Agenda 2030; prioritizing programmatic and financial sustainability of country immunization programs; supporting targeted countries that have phased out of Gavi support or have never been eligible for Gavi support to maintain immunization progress; and providing more tailored approaches for Gavi countries to reach under-vaccinated populations, such as those living in remote or conflict settings, by encouraging countries to adopt strategies that reduce potential barriers to vaccination.
In addition to Gavi’s role in routine childhood immunizations, Gavi was one of the organizations leading COVAX, a multilateral effort that supported the equitable development, procurement, and delivery of COVID-19 vaccines globally that began in 2020 and ended in 2023. Gavi’s role in COVAX was to facilitate the procurement and delivery of COVID-19 vaccines, with particular emphasis on low- and middle-income countries. Provision of COVID-19 vaccines and funding support to countries was integrated into Gavi’s regular programming from 2024-2025 (COVID-19 vaccine support has been discontinued).
Organization
Gavi’s Secretariat, with its main headquarters in Geneva and an office in Washington, D.C., carries out the day-to-day operations of the partnership. Gavi does not have program offices or staff based in recipient countries but rather relies on country health ministries and World Health Organization (WHO) regional offices to implement programs. Gavi is led by a Chief Executive Officer (CEO), currently Sania Nishtar.
The 28-member Gavi Board sets Gavi’s funding policies and strategic direction, and monitors program implementation. It includes 18 “representative” seats, nine seats for independent individuals, and one ex-officio non-voting seat for Gavi’s CEO. The 18 representative seats, as specified in Gavi’s statutes, are as follows: donor country governments (5), implementing country governments (5), the WHO, the United Nations Children’s Fund (UNICEF), the World Bank, and the Gates Foundation, and one seat each for civil society groups, the vaccine industry in industrialized countries, the vaccine industry in developing countries, and technical health/research institutes. Additionally, several Board committees guide and advise the Board and the CEO on Gavi activities under their purview. The U.S. government was represented on Gavi’s Board as the Board member for the donor country government constituency until the end of 2025. With the suspension of U.S. support, the U.S. lost eligibility to hold a seat on the Gavi Board.
Funding
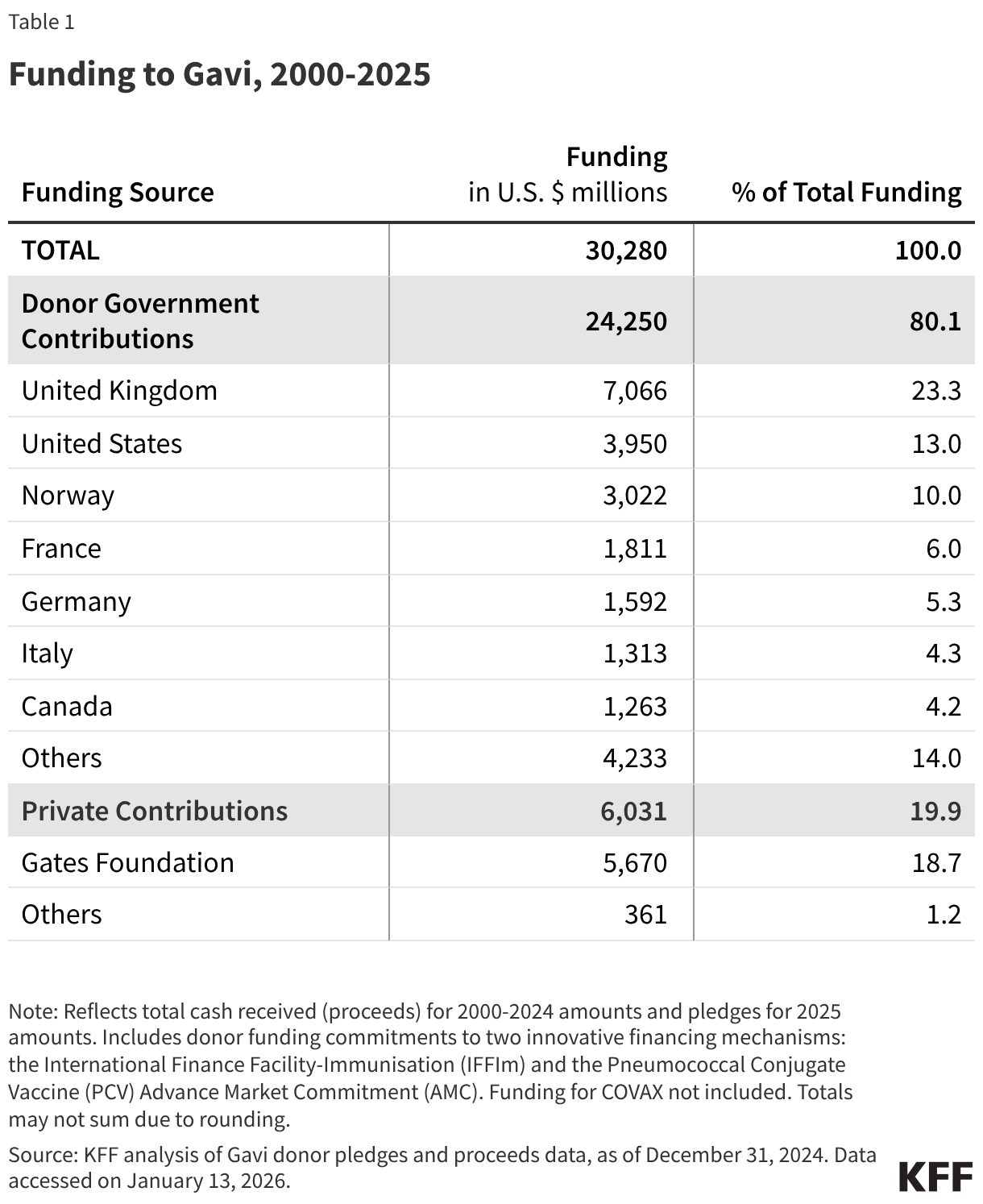
Since its 2000 launch, Gavi has received approximately $30 billion in financing, not including funding for COVAX (see Table 1).1 Approximately four-fifths (80%) of Gavi’s funding came from contributions provided by donor governments and private organizations and individuals. The top three government donors were the United Kingdom, the U.S. and Norway, while the largest private donor was the Gates Foundation.
Donors support Gavi through direct contributions as well as funding commitments to innovative financing mechanisms, the proceeds of which help support Gavi’s overall financing. These innovative financing mechanisms include the International Finance Facility-Immunisation (IFFIm) and the Pneumococcal Conjugate Vaccine (PCV) Advance Market Commitment (AMC). The IFFIm was created in 2006 and uses donor funding commitments to back the issuance of special bonds in capital markets, essentially providing “up-front” financing to Gavi. The PCV AMC began in 2010, and though it ended in 2020, it supported accelerated access to pneumococcal vaccines through up-front funding commitments from donors and continues to do so through contracts with manufacturers that extend until 2029. The U.S. does not provide support to either of these mechanisms.2
In addition to financing Gavi’s regular activities, donors pledged additional resources to support the Gavi COVAX Advance Market Commitment (COVAX AMC), a financial mechanism within COVAX that supported low- and middle-income countries through procurement and distribution of COVID-19 vaccines; through 2024, Gavi received $12.6 billion from donor governments, private philanthropy, and innovative financing mechanisms for the COVAX AMC for vaccine procurement, delivery, and logistics.3
Country Eligibility and Support
Eligibility
Only low- and middle-income countries with a Gross National Income (GNI) per capita below or equal to $1,8204 are eligible for Gavi support. In 2025, 54 countries were eligible for Gavi support.
Recipient countries’ governments are expected to share responsibility for funding their national immunization efforts through Gavi’s co-financing requirements (introduced in 2008), determined according to country income level and transition status. As countries develop economically, they are expected to contribute a greater share of the funding required for immunization programs. Countries classified as low-income by the World Bank are initial self-financing countries, while countries between the low-income threshold and Gavi eligibility threshold ($1,820 GNI per capita) are in preparatory transition. Initial self-financing countries are responsible for co-financing the equivalent of $0.20 per dose each year. Countries in preparatory transition gradually increase their co-financing contribution each year.5 When a country’s income rises above the GNI per capita threshold, it moves into an eight-year “accelerated transition” period of increasing domestic financing share, after which the country is expected to fully fund its own immunization programs.6 As of 2025, 19 countries have transitioned out of Gavi financial support.
Additionally, Gavi offers limited support for countries that have transitioned out of Gavi eligibility and for middle-income countries that have never been eligible for Gavi support.7 Recognizing that many formerly and never Gavi-eligible countries experience low coverage rates and have yet to make key vaccine introductions, eligible countries can apply for “catalytic” funding to support the introduction of key missing vaccines (HPV, PCV, or rotavirus) or mitigation of backsliding.8
Country Support
Gavi provides grant financing to country programs in the following two support types:
- Vaccine support, including for vaccine introduction, targeted and routine campaigns, and
- Health systems and immunization strengthening (HSIS) support,9 to improve country data systems and to support cold chain equipment, health workforce, and community engagement.
Country allocation formulas for HSIS support are based on the following metrics: number of zero-dose children, coverage of essential vaccines,10 GNI per capita, and if a country is considered fragile or conflict-affected.11 For vaccines, all countries are required to pay a share of the cost of their Gavi-supported vaccines.
Additionally, Gavi has provided country support through emergency response funding, including for Ebola vaccination during Ebola outbreaks12 and for COVID-19, allowing for up to $200 million in reprogrammed Gavi support for the COVID-19 response in Gavi-eligible countries, and other support for the COVID-19 response including through the creation of COVAX (which helped expand access to COVID-19 vaccines in lower-income countries) and the COVID-19 Vaccine Delivery Partnership (CoVDP, which aimed to improve COVID-19 vaccine coverage in certain COVAX countries, with a particular emphasis on countries that were below 10% coverage in January 2022).13 In 2022, Gavi supported 50 outbreak response vaccination campaigns. Gavi currently funds several emergency vaccine stockpiles, allowing for rapid deployment of vaccines during outbreaks, including for cholera, Ebola, meningitis, mpox,14 and yellow fever. In 2024, Gavi deployed vaccines from these emergency stockpiles to 20 countries.15
Since its launch in 2000, Gavi has provided approximately $29 billion to support country immunization programs (not including funding for COVAX).16 Over the past three years, 2023-2026, more than $8.1 billion has been disbursed, most of which has been for vaccine support (62%), followed by health systems strengthening (23%) (see Table 2).
Results
Gavi reports it has helped to immunize more than 1.2 billion children in supported countries, including more than 72 million in 2024 alone, and supported 58 different vaccine introductions and preventive campaigns and 50 outbreak response campaigns in 2024. Additionally, Gavi support has helped avert more than 20.6 million deaths and contributed to more than $280 billion in economic benefits, since its launch in 2000. Additionally, according to Gavi, its support has led to improved child health and immunization indicators across its supported countries. For example, the average vaccine coverage across multiple key Gavi-supported vaccines – including the human papillomavirus (HPV) vaccine, inactivated polio vaccine, and pentavalent vaccine (the vaccine providing protection against diphtheria, tetanus, pertussis, hepatitis B, and Hib),17 among others – was 63% in Gavi-supported countries in 2024, up from 48% in 2019.18 Lastly, Gavi’s work has contributed to vaccine market-shaping; for example, Gavi reports that its influence has helped lower the cost of the HPV vaccine from a price per dose of $4.50 in 2015 to $2.90 in 2022.19
U.S. Engagement with Gavi
The U.S. government has supported Gavi since its creation. President Clinton made the initial U.S. pledge to the newly formed partnership in 2000, and the U.S. provided its first contribution in 2001. Prior to the second Trump administration, the U.S. supported Gavi through financial contributions, participation in Gavi’s governance, and by providing technical assistance, but the current administration has not provided funding to the organization (see below).
Additionally, the U.S. had supported other global immunization activities that complemented Gavi’s efforts, providing bilateral (country-to-country) support for immunization through USAID (before its dissolution), CDC, and other agencies, focusing on strengthening routine immunization systems to deliver vaccines. However, the U.S. government is currently reorganizing how it supports global health programs, including immunization activities, under its “America First Global Health Strategy” which includes the development of bilateral agreements with countries. Given that Gavi was the mechanism through which the U.S. supported vaccine procurement, it is not yet clear how these agreements will support procurement going forward. See also the KFF fact sheet on the Trump administration’s foreign aid review and the proposed reorganization of U.S. global health programs.
Financial Support
The U.S. supported Gavi with direct contributions starting in 2001, with funding reaching $300 million in FY 2024, its highest level. Additionally, in response to the COVID-19 pandemic, the U.S. provided $4 billion in FY 2021 emergency funding to Gavi for COVID-19 vaccine procurement and delivery support under COVAX, making the U.S. the largest donor to COVAX (32% of $12.6 billion received overall).20In addition to its financial support for COVAX, the U.S. donated the largest number of COVID-19 vaccines to other countries. While Congress appropriated $300 million for U.S. contributions to Gavi in FY 2025 and FY 2026 (see Figure 1), the current administration has not provided funding to Gavi after citing concerns about vaccine safety despite Gavi following globally recognized scientific standards and evidence.21 See the KFF budget tracker and the KFF fact sheet on the U.S. Global Health Budget: Maternal & Child Health (MCH) for details on historical appropriations for Gavi, and also the KFF fact sheet on the Trump administration’s foreign aid review and the status of U.S. support for Gavi.
Governance Activities
The U.S. had historically played a role in Gavi’s governance, including as a Board and committee member, but with the suspension of U.S. funding by the Trump administration, the U.S. is no longer eligible to hold a Board seat.
Technical Support
The U.S. had historically provided Gavi with technical support and expertise in the design, implementation, and evaluation of its programs in the field through partnerships with several U.S. agencies. For example, Gavi’s accelerated vaccine introduction programs had been conducted with technical support from the Centers for Disease Control and Prevention (CDC) and the now-dissolved U.S Agency for International Development (USAID), along with other partners.
Endnotes
- This amount includes proceeds for 2000-2024 and pledges for 2025. ↩︎
- For further information about restrictions on U.S. support for these innovative financing mechanisms, see KFF, Innovative Financing Mechanisms for Global Health: Overview and Considerations for U.S. Government Participation, Sept. 2011. ↩︎
- KFF analysis of Gavi cash receipts data. Gavi, “Cash Receipts 31 December 2024,” https://www.gavi.org/news-resources/document-library/cash-receipts. ↩︎
- For countries to be eligible for Gavi support, their most recent GNI per capita must be at or below $1,820, or the country’s average GNI per capita over the last three years must be at or below $1,820. ↩︎
- Countries in the first year of the preparatory transition phase co-finance the equivalent of $0.20 per dose, the same as initial self-financing countries. For each subsequent year, countries in preparatory transition co-finance a 15% increase of the total fraction paid in the prior year. Gavi, “Co-financing policy,” https://www.gavi.org/sites/default/files/programmes-impact/our-impact/01_Gavi-Alliance-Co-financing-Policy-60.pdf. ↩︎
- Countries in the first year of the accelerated transition phase co-finance the equivalent of the prior year’s total fraction plus 15%, the same as countries in preparatory transition. For each year after, the amount per dose increases linearly until the country is fully financing each vaccine after the eighth year and end of Gavi support. Gavi, “Co-financing policy,” https://www.gavi.org/sites/default/files/programmes-impact/our-impact/01_Gavi-Alliance-Co-financing-Policy-60.pdf. ↩︎
- Countries eligible for this limited funding include those above the Gavi eligibility threshold ($1,820 GNI per capita) but below the World Bank lower-middle income threshold ($4,495 GNI per capita) or those that are eligible to borrow from the International Development Association. Gavi, “Annex D: Report to the Board, July 24-25 2025,” https://www.gavi.org/sites/default/files/%20/board/minutes/2025/24-25-july06%20-%20Annex%20D%20-%20Framework%20for%20Gavi%20Funding%20to%20Countries.pdf. ↩︎
- Support for backsliding mitigation is only available to former Gavi-eligible countries. Gavi, “Annex D: Report to the Board, July 24-25 2025,” https://www.gavi.org/sites/default/files/%20/board/minutes/2025/24-25-july06%20-%20Annex%20D%20-%20Framework%20for%20Gavi%20Funding%20to%20Countries.pdf. ↩︎
- HSIS support only available for Gavi-eligible countries. Gavi, “Annex D: Report to the Board, July 24-25 2025,” https://www.gavi.org/sites/default/files/%20/board/minutes/2025/24-25-july06%20-%20Annex%20D%20-%20Framework%20for%20Gavi%20Funding%20to%20Countries.pdf. ↩︎
- Includes coverage of first-dose diphtheria, tetanus, and pertussis containing vaccine (DPT1), coverage of DTP3, and coverage of second-dose measles containing vaccine (MCV2). Gavi, “Annex D: Report to the Board, July 24-25 2025,” https://www.gavi.org/sites/default/files/%20/board/minutes/2025/24-25-july06%20-%20Annex%20D%20-%20Framework%20for%20Gavi%20Funding%20to%20Countries.pdf. ↩︎
- Gavi, “Annex D: Report to the Board, July 24-25 2025,” https://www.gavi.org/sites/default/files/%20/board/minutes/2025/24-25-july06%20-%20Annex%20D%20-%20Framework%20for%20Gavi%20Funding%20to%20Countries.pdf. ↩︎
- Gavi, “500,000 doses of Ebola vaccine to be made available to countries for outbreak response,” webpage, https://www.gavi.org/news/media-room/500000-doses-ebola-vaccine-be-made-available-countries-outbreak-response. ↩︎
- CoVDP phased out its operations in June 2023 as the partnership was not set up to be a permanent structure. WHO, “COVID-19 Vaccine Delivery Partnership,” webpage, https://www.who.int/emergencies/diseases/novel-coronavirus-2019/covid-19-vaccines/covid-19-vaccine-delivery-partnership; Devex, “Exclusive: A COVID-19 initiative for vaccine delivery is winding down,” 11 January 2023, https://www.devex.com/news/exclusive-a-covid-19-initiative-for-vaccine-delivery-is-winding-down-104724. ↩︎
- Gavi, “Gavi 6.0 Funding Guidelines,” https://www.gavi.org/sites/default/files/support/guidelines-2026/gavi-60-funding-guidelines-annexes.pdf#page=43 ↩︎
- Gavi, “Vaccine stockpiles,” https://www.gavi.org/vaccineswork/vaccine-stockpiles-guide. ↩︎
- KFF analysis of data provided by Gavi on disbursements by program area and year. KFF personal communications with Gavi, March 19, 2026. ↩︎
- The vaccines included in Gavi’s breadth of protection measure include: the third dose of the pentavalent vaccine, third dose of the pneumococcal conjugate vaccine, first dose of the rubella-containing vaccine, last dose of the rotavirus vaccine, second dose of the measles-containing vaccine, yellow fever, meningococcal A, Japanese encephalitis, and last dose of the HPV vaccine. Gavi, “Gavi 2024 Annual Progress Report,” https://www.gavi.org/sites/default/files/programmes-impact/our-impact/apr/Gavi-2024-Annual-Progress-Report.pdf#page=17. ↩︎
- Gavi, “Gavi 2024 Annual Progress Report,” https://www.gavi.org/sites/default/files/programmes-impact/our-impact/apr/Gavi-2024-Annual-Progress-Report.pdf#page=17. ↩︎
- As another example of Gavi’s market-shaping influence, Gavi and UNICEF recently announced an agreement to make R21/Matrix-M malaria vaccines more affordable for Gavi countries through the IFFIm mechanism. UNICEF, “Gavi and UNICEF announce equitable pricing deal for malaria vaccine to protect 7 million more children by end of decade,” https://www.unicef.org/press-releases/gavi-and-unicef-announce-equitable-pricing-deal-malaria-vaccine-protect-7-million. ↩︎
- The U.S. announced it would donate 500 million Pfizer doses to COVAX at the G7 Summit in June 2021. However, a portion of these doses were purchased using funds appropriated to Gavi ($2 billion for 300 million Pfizer doses), while the remaining 200 million doses were purchased using $1.5 billion in other emergency funds from the American Rescue Plan Act. To avoid double-counting, Gavi counts the U.S. funding that was contributed to Gavi under its COVAX funding contributions, with only 200 million of the doses – those purchased directly by the U.S. – counted as COVAX vaccine dose donations. KFF personal communication with Gavi, Nov. 12, 2021; White House, “FACT SHEET: President Biden Announces Historic Vaccine Donation: Half a Billion Pfizer Vaccines to the World’s Lowest-Income Nations,” June 10, 2021; Gavi, “COVAX AMC Donors Table,” Apr. 7, 2022, https://www.gavi.org/sites/default/files/covid/covax/COVAX-AMC-Donors-Table.pdf; Gavi, “Cash Receipts 31 December 2024,” https://www.gavi.org/news-resources/document-library/cash-receipts. ↩︎
- Secretary Robert F. Kennedy Jr. (@SecKennedy), https://x.com/SecKennedy/status/1937986463510982869 [X post], June 25, 2025; Gavi, “Statement,” https://www.gavi.org/news/media-room/statement-response-25-june-2025; Reuters, “Exclusive: US conditions funding to global vaccine group on dropping mercury-based preservative from shots,” https://www.reuters.com/business/healthcare-pharmaceuticals/us-conditions-funding-global-vaccine-group-dropping-mercury-based-preservative-2026-01-28/. ↩︎